Inamed Corporations Mc Ghan SiliconeFilled Breast Implants October

Inamed Corporation’s Mc. Ghan Silicone-Filled Breast Implants October 14 -15, 2003

FDA Presenters CDR Samie Allen, USPHS n Sam Arepalli, Ph. D. n David Berkowitz, Ph. D. , V. M. D. n Sahar Dawisha, M. D. n Telba Irony, Ph. D. n S. Lori Brown, Ph. D. , M. P. H. n 2

Device Description, Mechanical Testing, Retrieval Study, & Shelf Life Overview Samie Allen

Device Description Styles 10, 20, 45, 110, 120 & 153 n Round & shaped n Standard, moderate, high, & full profiles n Smooth & textured (Biocell) surfaces n Single lumen except Style 153 n Components: shell, patch, filler, & silicone adhesive n 4

Mechanical Testing n Gel Cohesion n Gel Bleed n Fatigue 5

Gel Cohesion Testing n Gel Cohesion Testing of Final Gel ¨ASTM F 703 (<4. 5 cm & no gel separation) ¨Results: passed n Penetrometer Testing of In-Process Gel ¨No standard (internal specification) ¨Results: 49. 2 (39. 5 -56. 0) 6

Gel Bleed Testing n Gel Bleed Testing ¨ASTM F 703 ¨Results: 0. 0152 g/cm 2 for Style 40 0. 0048 g/cm 2 for Style 110 7
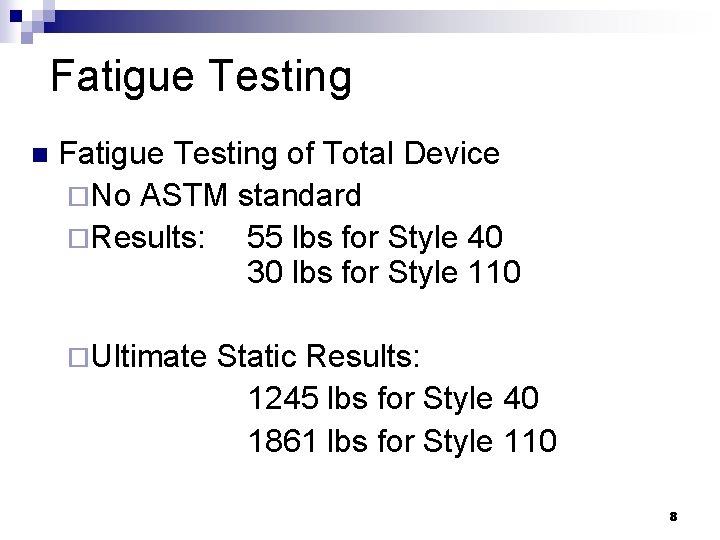
Fatigue Testing n Fatigue Testing of Total Device ¨No ASTM standard ¨Results: 55 lbs for Style 40 30 lbs for Style 110 ¨Ultimate Static Results: 1245 lbs for Style 40 1861 lbs for Style 110 8

Retrieval Study n From 7/31/00 to 10/1/02, 339 gel explants Physician Observations n Laboratory Observations n Mechanical Testing n Sharp-edge Analyses n 9
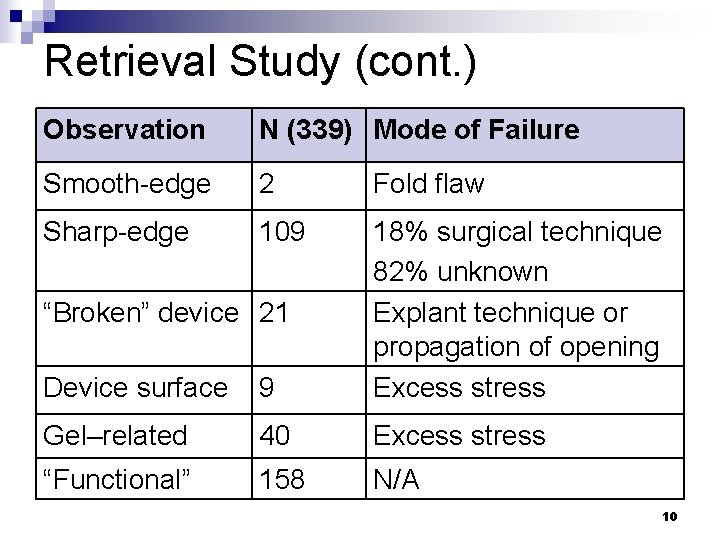
Retrieval Study (cont. ) Observation N (339) Mode of Failure Smooth-edge 2 Fold flaw Sharp-edge 109 Device surface 9 18% surgical technique 82% unknown Explant technique or propagation of opening Excess stress Gel–related 40 Excess stress “Functional” 158 N/A “Broken” device 21 10

Shelf Life n Device and package testing n 2. 5 -year shelf life date on package label (2 years real + ½ year accelerated) 11

Conclusions – Mechanical & Other Gel Cohesion Testing n Gel Bleed Testing n Fatigue Testing n Retrieval Study n Shelf Life n 12

Chemistry Overview Sam Arepalli, Ph. D.

Device Materials n Shell, middle (barrier) layer: Diphenyldimethylsiloxane copolymer, 15 mole% diphenyl n Shell, inner/outer (base) layers: Diphenyldimethylsiloxane copolymer, 5 mole% diphenyl n Patch, outer layer: Peroxide cure silicone elastomer n Patch, inner (barrier) layer: Dimethyl, methyltrifluoropropylsiloxane n Silicone Gel: Two-part platinum cure gel n Silicone adhesive: Oxime cure RTV silicone 14

Extent of Crosslinking n Shell: 3. 4 crosslinked units/molecule (Sol Fraction Method) n Gel: 3. 5 -7. 5 mm (Penetrometer) 15

Volatiles n Shell: 1, 1, 1 trichloroethane (279 µg) Isopropyl alcohol (251 µg) 16

Extractables n Gravimetric analysis n Gel permeable chromatography n FTIR analysis n Qualitative and quantitative analysis 17

GC-MS Analysis n Cyclicoligosiloxanes up to D 10 not detectable. n Higher cyclic and linear oligosiloxanes concentrations comparable to those of saline-filled breast implants. 18

Metal Analysis n Shell: Sn (0. 05 ppm); Pt (3. 3 ppm) n Patch: Sn (6. 6 ppm); Pt (2. 6 ppm) n Gel: Sn (0. 06 ppm); Pt (4 ppm) 19

Silica Analysis n Amorphous silica (X-ray diffraction) n No free silica present (Electrospectroscopy) 20

Conclusions - Chemistry Shell and gel tested separately n Degree of crosslinking n Volatiles n Metals n Extractables ¨Gravimetric analysis ¨GPC ¨FTIR ¨GC-MS n 21

Toxicology Overview David Berkowitz, Ph. D. , V. M. D.

Six Testing Categories Pharmacokinetics n Biocompatibility n Subchronic Toxicity n Reproductive and Teratogenicity n Immunotoxicology n Genotoxicity and Carcinogenesis Testing n 23

Pharmacokinetics 30 days after implantation, only 0. 06% of radiolabeled Gel left the implant site. n Lower molecular weight siloxanes (e. g. , D 4 and D 5) diffuse out of the implants at a slow rate. n 24

Biocompatibility Testing Cytotoxicity n Irritation and Sensitization n Acute Systemic Toxicity n Implantation Testing (Subchronic Toxicity) n Hemolysis n Pyrogenicity n 25
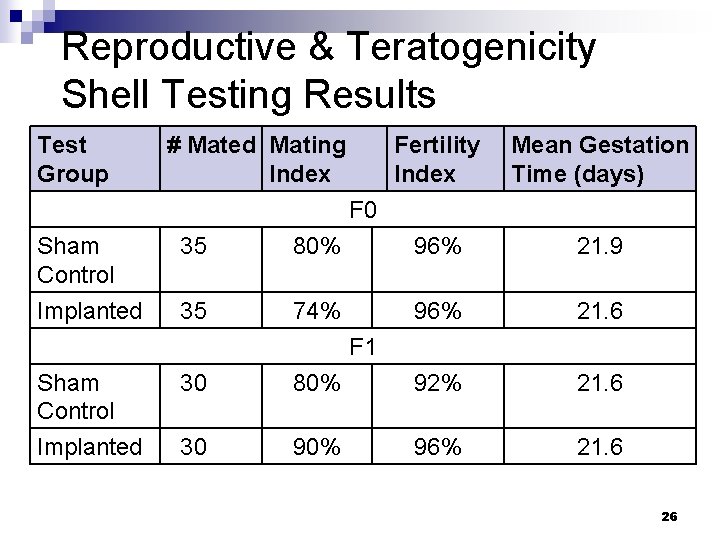
Reproductive & Teratogenicity Shell Testing Results Test Group # Mated Mating Index Fertility Index Mean Gestation Time (days) F 0 Sham Control 35 80% 96% 21. 9 Implanted 35 74% 96% 21. 6 F 1 Sham Control Implanted 30 80% 92% 21. 6 30 90% 96% 21. 6 26
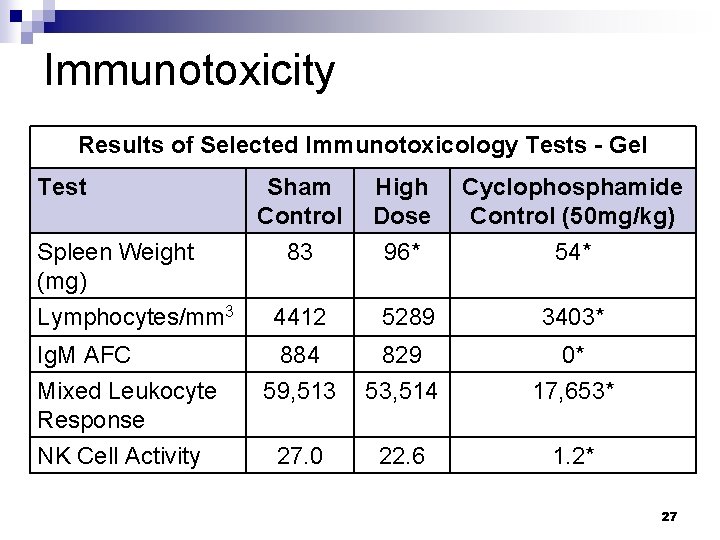
Immunotoxicity Results of Selected Immunotoxicology Tests - Gel Test Spleen Weight (mg) Lymphocytes/mm 3 Ig. M AFC Mixed Leukocyte Response NK Cell Activity Sham Control High Dose Cyclophosphamide Control (50 mg/kg) 83 96* 54* 4412 5289 3403* 884 59, 513 829 53, 514 0* 17, 653* 27. 0 22. 6 1. 2* 27

Genotoxicity n Bacterial Mutagenesis n Mammalian Cell Forward Mutation Assay n Chromosomal Aberration Assay n Mammalian Cell Transformation Assay 28

Carcinogenicity n 2 -year studies including gross and microscopic pathology. n Gel – Longer time to tumor and longer survival time than polyethylene control. n Shell – Shorter survival time than sham and control. Differences attributable to foreign body carcinogenesis. 29

Clinical Data Overview Sahar M. Dawisha, M. D. Medical Officer

Summary of Studies 1. 2. 3. n n n Core Study—Started 1999. Adjunct Study—Started 1998. 1990 Study—Started 1990. All open label, prospective, multicenter. Yearly F/U in Core Study & 1990 Study. All collected local complications. 31

Core Study Majority of Safety and Effectiveness data. n Augmentation, Reconstruction, Revision. n Yearly F/U to 10 years after implantation. n Only study with prospective MRI screening for asymptomatic rupture in 34% of 940 total patients. n Only study with QOL and CTD signs/symptoms collected. n 32

Adjunct Study Intended to make the implants available for reconstruction and revision indications. n Collected local complications at 1, 3, and 5 years after implantation surgery. n Unlimited sample size. n Enrollment is ongoing. n 33

1990 Study Majority of patients: augmentation indication. n Yearly F/U to 5 years. n Data from 4 of 11 styles presented. n 34

Core Study Results 35
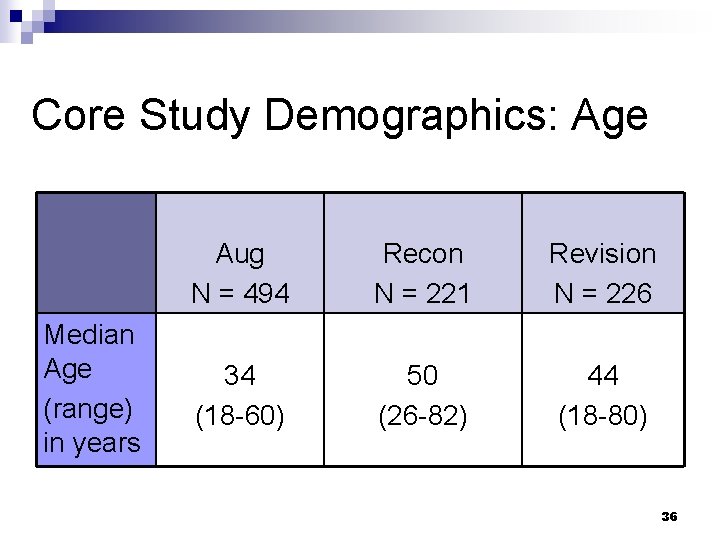
Core Study Demographics: Age Median Age (range) in years Aug N = 494 Recon N = 221 Revision N = 226 34 (18 -60) 50 (26 -82) 44 (18 -80) 36

Core Augmentation Cohort 37

Patient Disposition—Core Augmentation 494 Patients (987 devices) enrolled. n 90% of 489 expected patient F/U at 2 years. n 81% of 398 expected patient F/U at 3 years. n ¨ 1 Death ¨ 13 Implant Removals ¨ 76 Lost to Follow-up 38
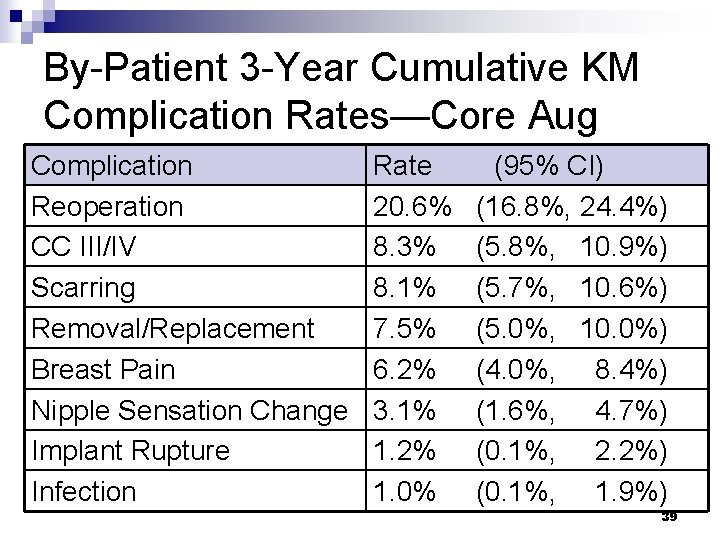
By-Patient 3 -Year Cumulative KM Complication Rates—Core Aug Complication Reoperation CC III/IV Scarring Removal/Replacement Breast Pain Nipple Sensation Change Implant Rupture Infection Rate 20. 6% 8. 3% 8. 1% 7. 5% 6. 2% 3. 1% 1. 2% 1. 0% (95% CI) (16. 8%, 24. 4%) (5. 8%, 10. 9%) (5. 7%, 10. 6%) (5. 0%, 10. 0%) (4. 0%, 8. 4%) (1. 6%, 4. 7%) (0. 1%, 2. 2%) (0. 1%, 1. 9%) 39

Reoperation—Core Augmentation 248 Additional procedures in 112 reoperations through 3 years in 94 of the 494 patients (19. 1%). n Capsule related: 79 of 248 procedures (31. 9%). n Removal with replacement: 51 of 248 procedures (20. 6%). n 40
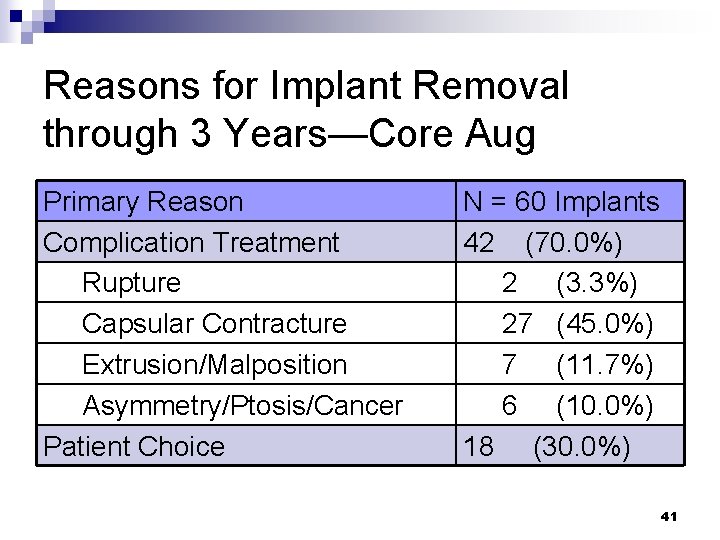
Reasons for Implant Removal through 3 Years—Core Aug Primary Reason Complication Treatment Rupture Capsular Contracture Extrusion/Malposition Asymmetry/Ptosis/Cancer Patient Choice N = 60 Implants 42 (70. 0%) 2 (3. 3%) 27 (45. 0%) 7 (11. 7%) 6 (10. 0%) 18 (30. 0%) 41

Asymptomatic Implant Rupture Screening—Core Augmentation n n n 166 Patients (331 implants) enrolled. At 1 year: 139 patients (87%) of expected underwent MRI screening. At 3 years: 83 patients (64% of expected) underwent screening. Total of 145 patients (289 implants) who had at least one MRI screening. 3 Implants reported ruptured. Silent rupture rate: 1. 2% (0. 0%, 2. 6%) through 3 years, by-implant. 42

Implant Ruptures—Core Augmentation n No MRI Screening/Symptomatic Ruptures ¨ 2 implants (out of 698) ruptured. ¨ 2? Additional implants reported as intact. ¨ Unknown asymptomatic rupture rate. *Overall by-implant rupture rate: 0. 6% (0. 1%, 1. 1%) through 3 years: • 3 Asymptomatic/silent + 2 Symptomatic. • Excludes potential silent ruptures in No MRI. • Excludes 2 additional symptomatic ruptures. 43

Other Safety Information—Core Augmentation No increase in reports of reproductive or lactation problems. n 32 post-implant breast disease reports: 1 malignant, 29 benign, 2 unconfirmed. n 12 post-implant abnormal mammogram reports: 1 no disease; 11 benign. n 1 New CTD: RA. n 44
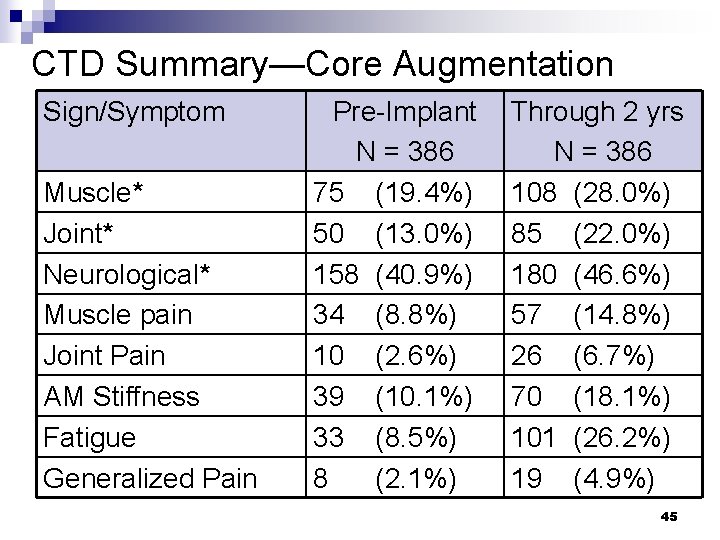
CTD Summary—Core Augmentation Sign/Symptom Muscle* Joint* Neurological* Muscle pain Joint Pain AM Stiffness Fatigue Generalized Pain Pre-Implant N = 386 75 (19. 4%) 50 (13. 0%) 158 (40. 9%) 34 (8. 8%) 10 (2. 6%) 39 (10. 1%) 33 (8. 5%) 8 (2. 1%) Through 2 yrs N = 386 108 (28. 0%) 85 (22. 0%) 180 (46. 6%) 57 (14. 8%) 26 (6. 7%) 70 (18. 1%) 101 (26. 2%) 19 (4. 9%) 45

Effectiveness—Core Augmentation Most patients completing 2 years of followup reported being satisfied, but declines in mean satisfaction over time. n Mean General QOL measures worsened over time. n Some Specific QOL measures improved (TSCS, Body Esteem--Total, Sexual Attractiveness, and Weight); while, others declined over time (Rosenberg Self Esteem, Body Esteem-Physical). n 46

Core Reconstruction Cohort 47

Patient Disposition—Core Reconstruction 221 Patients (361 devices) enrolled. n 95% of 205 expected patient F/U at 2 years. n 91% of 116 expected patient F/U at 3 years. n ¨ 7 Deaths ¨ 16 Implant Removals ¨ 11 Lost to Follow-up 48
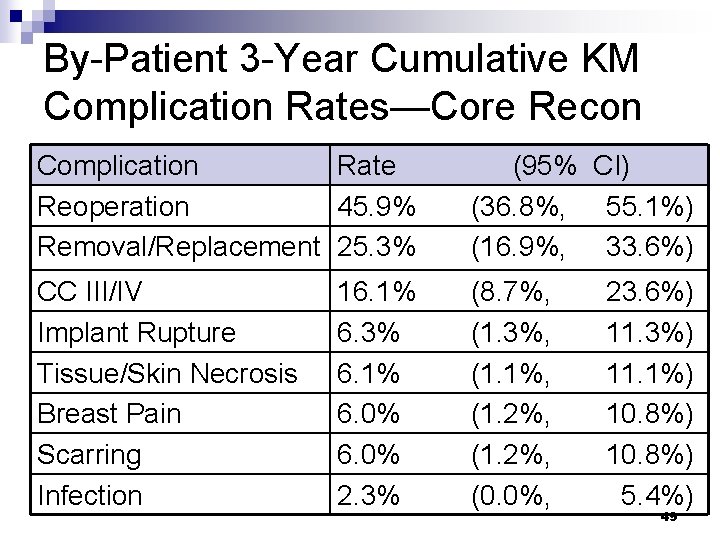
By-Patient 3 -Year Cumulative KM Complication Rates—Core Recon Complication Rate Reoperation 45. 9% Removal/Replacement 25. 3% (95% CI) (36. 8%, 55. 1%) (16. 9%, 33. 6%) CC III/IV Implant Rupture Tissue/Skin Necrosis Breast Pain Scarring Infection (8. 7%, (1. 3%, (1. 1%, (1. 2%, (0. 0%, 16. 1% 6. 3% 6. 1% 6. 0% 2. 3% 23. 6%) 11. 3%) 11. 1%) 10. 8%) 5. 4%) 49

Reoperation—Core Reconstruction 242 Additional procedures in 127 reoperations through 3 years in 92 of the 221 patients (41. 6%). n Capsule related: 54 of 242 procedures (22. 3%). n Removal with replacement: 51 of 242 (21. 1%). n Scar revision/wound repair: 47 of 242 (19. 4%). n 50
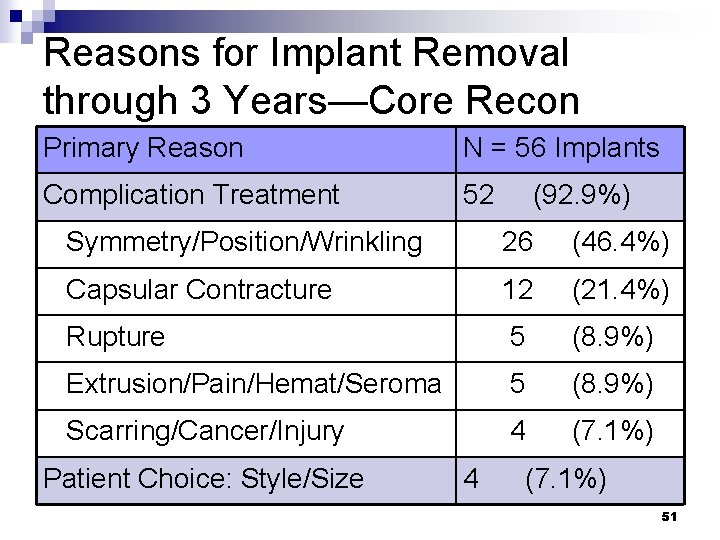
Reasons for Implant Removal through 3 Years—Core Recon Primary Reason N = 56 Implants Complication Treatment 52 (92. 9%) Symmetry/Position/Wrinkling 26 (46. 4%) Capsular Contracture 12 (21. 4%) Rupture 5 (8. 9%) Extrusion/Pain/Hemat/Seroma 5 (8. 9%) Scarring/Cancer/Injury 4 (7. 1%) Patient Choice: Style/Size 4 (7. 1%) 51

Asymptomatic Implant Rupture Screening—Core Reconstruction 108 Patients (184 implants) enrolled. n Total of 101 patients (170 implants) at least one MRI screening (93. 5% of expected). n 8 implants ruptured. n Silent rupture rate: 4. 7% (1. 5%, 7. 9%) byimplant through 3 years. n Only 2 patients (2 implants) with 2 nd screening at 3 years. n 52

Implant Ruptures—Core Recon n No MRI Screening/Symptomatic Ruptures ¨ 5 implants (out of 191) ruptured. ¨ Unknown asymptomatic rupture rate. *Overall by-implant rupture rate: 4. 2% (2. 0%, 6. 5%) through 3 years: • 8 Asymptomatic/silent + 5 Symptomatic. • Excludes potential silent ruptures in No MRI Group (53% of Core Reconstruction implants). 53

Other Safety Information—Core Reconstruction No increase in reports of reproductive or lactation problems. n 5 New reports of breast malignancy: recurrence or metastasis. n 1 New report of CTD: Scleroderma. n 54
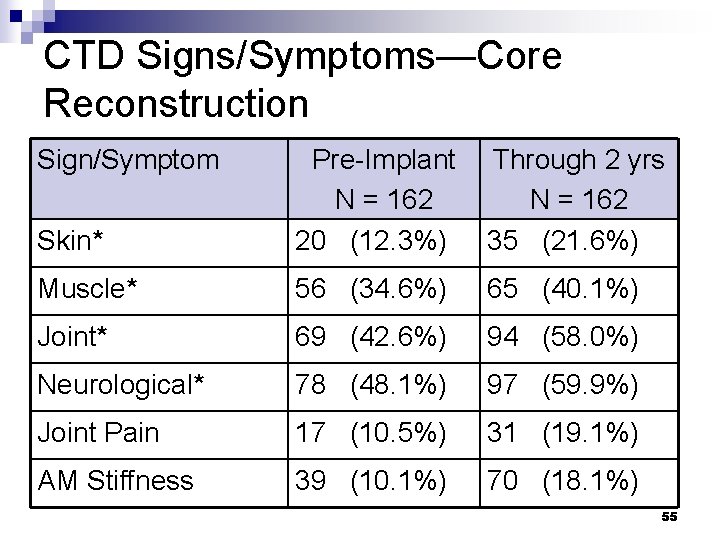
CTD Signs/Symptoms—Core Reconstruction Sign/Symptom Skin* Pre-Implant N = 162 20 (12. 3%) Through 2 yrs N = 162 35 (21. 6%) Muscle* 56 (34. 6%) 65 (40. 1%) Joint* 69 (42. 6%) 94 (58. 0%) Neurological* 78 (48. 1%) 97 (59. 9%) Joint Pain 17 (10. 5%) 31 (19. 1%) AM Stiffness 39 (10. 1%) 70 (18. 1%) 55

Effectiveness—Core Reconstruction n Most patients completing 2 years of follow-up reported being satisfied, but declines in mean satisfaction over time. Mean General QOL measures improved over time. Some specific QOL measures improved (Semantic Differential, Body Esteem-Sexual Attractiveness); while others worsened (TSCS, Rosenberg Self Esteem, Body Esteem-Total) 56

Core Revision Cohort 57

Patient Disposition through 3 years —Core Revision 225 Patients (432 devices) enrolled. n 87% of 216 expected patient F/U at 2 years. n 83% of 192 expected patient F/U at 3 years. n ¨ 4 Deaths ¨ 10 Implant Removals ¨ 32 Lost to Follow-up 58
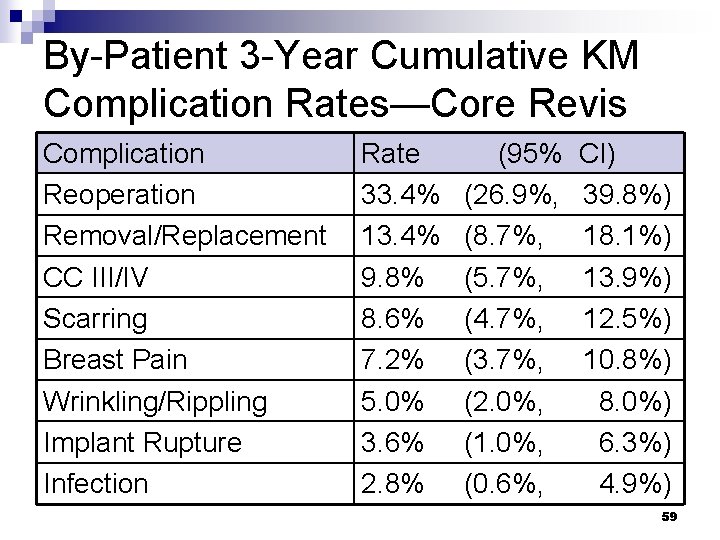
By-Patient 3 -Year Cumulative KM Complication Rates—Core Revis Complication Reoperation Removal/Replacement CC III/IV Scarring Breast Pain Wrinkling/Rippling Implant Rupture Infection Rate 33. 4% 13. 4% 9. 8% 8. 6% 7. 2% 5. 0% 3. 6% 2. 8% (95% (26. 9%, (8. 7%, (5. 7%, (4. 7%, (3. 7%, (2. 0%, (1. 0%, (0. 6%, CI) 39. 8%) 18. 1%) 13. 9%) 12. 5%) 10. 8%) 8. 0%) 6. 3%) 4. 9%) 59

Reoperation—Core Revision 190 Additional procedures in 100 reoperations through 3 years in 70 of the 225 patients (31. 1%). n Capsule related: 23 of 190 procedures (27. 9%). n Removal with replacement: 41 of 190 procedures (21. 6%). n 60
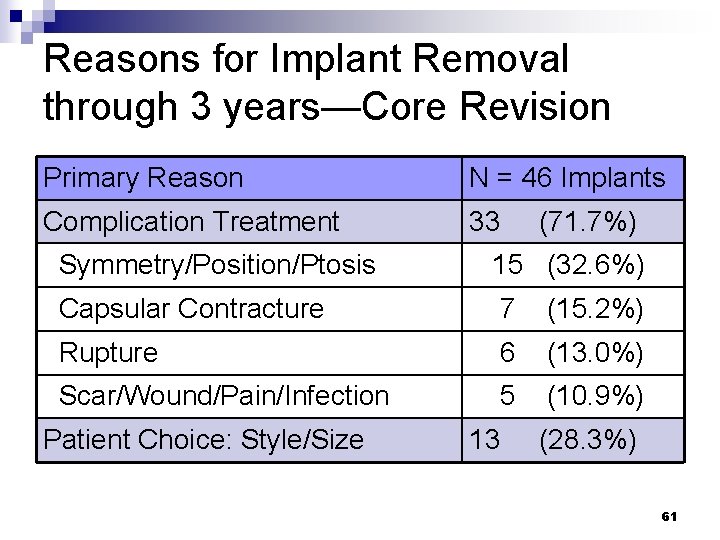
Reasons for Implant Removal through 3 years—Core Revision Primary Reason N = 46 Implants Complication Treatment 33 (71. 7%) Symmetry/Position/Ptosis 15 (32. 6%) Capsular Contracture 7 (15. 2%) Rupture 6 (13. 0%) Scar/Wound/Pain/Infection 5 (10. 9%) Patient Choice: Style/Size 13 (28. 3%) 61

Asymptomatic Implant Rupture Screening—Core Revision 77 Patients (148 implants) enrolled. n Total of 72 patients (138 implants) at least 1 MRI screening (93. 5% of expected). n 4 implants ruptured. n Silent rupture rate: 2. 9% (0. 1%, 5. 7%) byimplant through 3 years. n Only 1 patient (2 implants) with 2 nd MRI screening at 3 years. n 62

Implant Ruptures—Core Revision n No MRI Screening/Symptomatic Ruptures ¨ 4 implants (out of 294) ruptured. ¨ Unknown asymptomatic rupture rate. *Overall by-implant rupture rate: 2. 2% (0. 7%, 3. 7%) through 3 years: • 4 Asymptomatic/silent + 4 Symptomatic. • Excludes potential silent ruptures from No MRI Group (68% of Core Revision Implants). 63

Other Safety Information—Core Revision No increase in reports of reproductive or lactation problems. n 13 New reports of breast disease: all benign. n 1 New report of CTD: Fibromyalgia. n 64
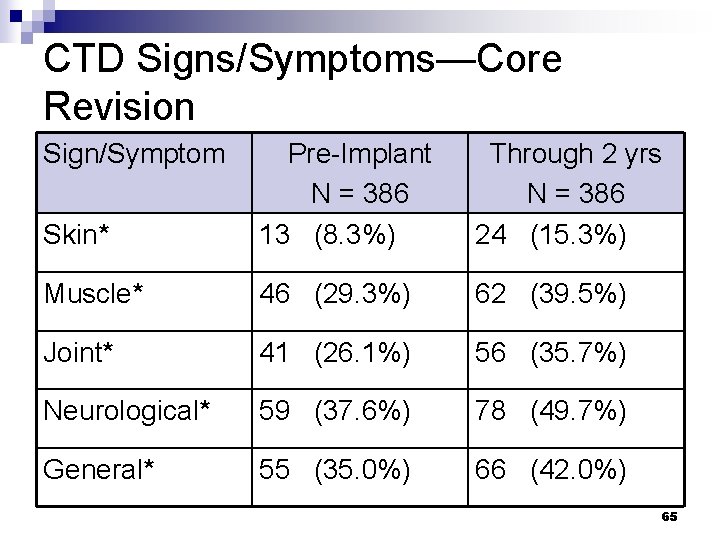
CTD Signs/Symptoms—Core Revision Sign/Symptom Skin* Pre-Implant N = 386 13 (8. 3%) Through 2 yrs N = 386 24 (15. 3%) Muscle* 46 (29. 3%) 62 (39. 5%) Joint* 41 (26. 1%) 56 (35. 7%) Neurological* 59 (37. 6%) 78 (49. 7%) General* 55 (35. 0%) 66 (42. 0%) 65

Effectiveness—Core Revision Most patients completing 2 years of followup reported being satisfied, but declines in mean satisfaction over time. n Mean General QOL measures worsened over time. n Some specific measures improved (Body Esteem—Sexual Attractiveness); while all others worsened. n 66

Comparison to Mc. Ghan Saline Breast Implant Data Cannot compare rupture rates. n Historical control group. n Confidence intervals not overlapping for reoperation, removal, capsular contracture. n 67

Adjunct Study n n n Reconstruction. Revision. ~50% F/U at 1 year. ~20% F/U at 3 years. Complication rates comparable to Core Study. 1990 Study n n n Augmentation. 70% F/U at 5 years. Complication rates at 3 years comparable to Core Study. 68

Summary Reoperation most frequent complication. n Capsular contracture reoperation most common procedure. n Most implants removed to treat a complication. n CTD signs/symptoms increase over time. n Patient satisfaction high but decreases over time; General QOL measures improved for reconstruction; Body Esteem-Sexual Attractiveness only specific measure consistently improved. n 69

Summary—Implant Rupture Implant rupture rate is under ascertained. n Most implant ruptures are asymptomatic: n ¨ Asymptomatic: 15 of 26 total implant ruptures. ¨ Asymptomatic rupture rate (MRI) based on 34% of implants and 1 year data. ¨ Overall rupture rate excludes asymptomatic ruptures in 66% of implants. ¨ Almost all asymptomatic ruptures were intracapsular. 70

Thank You!

Statistical Overview Telba Irony, Ph. D. Mathematical Statistician Division of Biostatistics

Statistical Analyses: Core Study • Prospective • Multi-Center • 10–year study • Reported follow-up time points: • 4 weeks, 6 months, 1, 2, 3 years • All patients traversed the 2 -year window • A large fraction of patients traversed the 3 -year window 73

Core Study Augmentation § 494 patients and 83% traversed the 3 -year visit. § 398 patients were expected at 3 years. § Actual # of patients at 3 years: 322 (19% lost to follow-up) Reconstruction § 221 patients and 58% traversed the 3 -year visit. § 116 patients were expected at 3 years. § Actual # of patients at 3 years: 105 (9% lost to follow-up ) Revision § 225 patients and 91% traversed the 3 -year visit. § 192 patients were expected at 3 years. § Actual # of patients at 3 years: 160 (17% lost to follow-up) 74

Descriptive Nature of Studies n There were no claims, targets, or control groups in this study. n Descriptive statistics: No hypothesis tests. n Sample size: Reflected in the width (i. e. precision) of the confidence interval. n Decision makers should assess the adequacy of the precision of the results when weighing the risks and benefits of the implants. 75

Safety Endpoints - Rates § § Adverse Events Implant Rupture Reoperations Implant Replacement/Removal Employed Statistical Techniques 1. Kaplan - Meier analyses 2. “Prevalence” 3. “Incidence” 76

Kaplan-Meier Analyses Kaplan-Meier analyses were conducted on the time to first occurrence of each adverse event. Result: Estimated probability that a patient will experience the adverse event from the time of implant up to the considered time point. Advantage: Patients who were lost to follow -up provide information up to the time they left the study. Independence Assumption for Censoring 77

Kaplan-Meier Analyses (cont. ) n n n Days were used as units of time for the computation of the rates (alleviates interval censoring bias). Assumption: reporting is accurate to the date Correlation among adverse events: not taken into account Competing Risks Problem: To solve it, all patients that experienced a complication were returned to the “pool” of patients who could experience another complication. Exception: Implant Removal 78
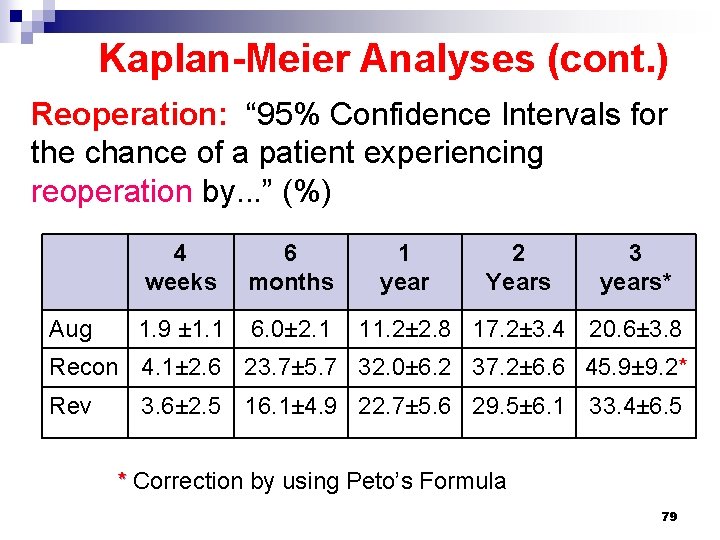
Kaplan-Meier Analyses (cont. ) Reoperation: “ 95% Confidence Intervals for the chance of a patient experiencing reoperation by. . . ” (%) Aug 4 weeks 6 months 1. 9 ± 1. 1 6. 0± 2. 1 1 year 2 Years 3 years* 11. 2± 2. 8 17. 2± 3. 4 20. 6± 3. 8 Recon 4. 1± 2. 6 23. 7± 5. 7 32. 0± 6. 2 37. 2± 6. 6 45. 9± 9. 2* Rev 3. 6± 2. 5 16. 1± 4. 9 22. 7± 5. 6 29. 5± 6. 1 33. 4± 6. 5 * Correction by using Peto’s Formula 79

Other Statistical Techniques Prevalence: percentage of patients seen at a given follow-up visit, who are experiencing a specific adverse event. (given they returned to the follow-up) Incidence: percentage of patients seen at a given follow-up visit who are experiencing the adverse event not experienced at earlier visits (given they returned to the follow-up). Disadvantage: Both measures are very sensitive to biases generated by losses to follow up. 80

Additional Safety Information Connective Tissue Disease Signs and Symptoms • The frequencies of patients reporting 8 categories of signs and symptoms of CTD before implantation was compared to the frequencies 2 years after implantation. • For all cohorts, all 8 frequencies increased after implantation. The only exception was the urinary symptom: the frequency remained the same in the Reconstruction group. 81

Additional Safety Information Connective Tissue Disease Signs and Symptoms • To assess the statistical significance of the increases in the frequencies, the sponsor used a Bonferroni correction that was too conservative for this case. It did not take into account possible correlations among signs and symptoms. • Consequence: It was difficult to detect statistical significance. 82

Additional Safety Information Connective Tissue Disease Signs and Symptoms • Despite the conservative statistical analysis, the increase in the frequency of some signs and symptoms was statistically significant. • However, the clinical interpretation is problematic: no control group 83

Medical Device Surveillance & Literature Overview S. Lori Brown, Ph. D. , M. P. H. Office of Surveillance and Biometrics

Medical Device Reporting n What is Medical Device Reporting (MDR)? n MDR is the mechanism for the Food and Drug Administration to receive significant medical device adverse events from manufacturers, importers, and user facilities 85

CDRH Surveillance Databases n Manufacturer And User Facility Device Experience (MAUDE) database - MDR and Med. Watch reports are entered into database - 1992 -present n Alternative Summary Reporting (ASR) – 1995 -present n Device Experience Network (DEN) – 1984 -1996 86

Surveillance is NOT the equivalent of a clinical study: n Rates cannot be calculated because of under-reporting of adverse events n Number of individuals at risk (denominator) is unknown. It is not appropriate to use the number of devices sold as the denominator! 87

Surveillance does not always establish causality: n Accuracy and completeness not verified n Cannot always establish a causal link between a death or injury and the listed device(s) 88

Surveillance Reports Are n Important for providing a signal of a potential problem with a regulated medical product 89
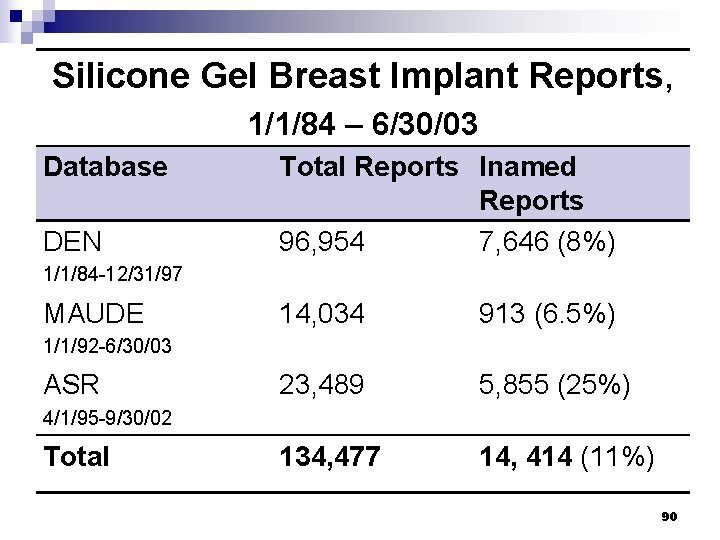
Silicone Gel Breast Implant Reports, 1/1/84 – 6/30/03 Database DEN Total Reports Inamed Reports 96, 954 7, 646 (8%) 1/1/84 -12/31/97 MAUDE 14, 034 913 (6. 5%) 23, 489 5, 855 (25%) 134, 477 14, 414 (11%) 1/1/92 -6/30/03 ASR 4/1/95 -9/30/02 Total 90

Device Problems Inamed (MAUDE Database) Explanted n Rupture n Migration n 35% 32% 3% 91

Patient Problems Inamed (MAUDE Database) n n n Pain Headache Surgical procedure Capsular contracture CTD Fatigue 15% 13% 9% 7% 6% 5% 92

MAUDE Analysis: Breast Implant Rupture During Mammography n Between 1992 and 2002, FDA received 33 adverse event reports describing breast implant rupture during mammography n An additional 8 reports described mammography as possible cause of subsequently detected ruptures 93

MAUDE Analysis: Reproductive/ 2 nd Generation Issues n 130 reports from MAUDE that described injury or illness in mothers or their children attributed to breast implants n 89 of these reports asserted that children were ill due to mother’s implants but provided no details of illness n 23 reports described illness in children n 5 reports attributed birth defects to mother’s breast implants n 9 reports described difficulty nursing 94

Literature Review Reproductive/second generation issues n Connective tissue disease n Fibromyalgia n Cancer n Mammography n Neurologic disease n Breast implants and mortality n Resurgery and local complications n Rupture and gel migration n 95

Literature on Reproductive/ nd 2 Generation Issues n Illness in children of mothers with implants (Levine, Teuber, Signorello, Kjøller) n Birth defects (Signorello, Kjøller) n Breast feeding by mothers with implants ¨ Silicon(e) in breast milk (Semple et al, 1998) ¨ Ability to breast feed (Neifert, Hurst, Hughes, Strom) n Limited information on these issues 96

Connective Tissue Disease n Meta-analyses of relation between silicone breast implants and risk of connective-tissue disease (Janowski et al, 2000) n Institute of Medicine review of safety of silicone breast implants concluded that “[these studies] do not support an association between connective tissue disease, combined or individually, for these diseases in women with silicone breast implants…” (1999) 97
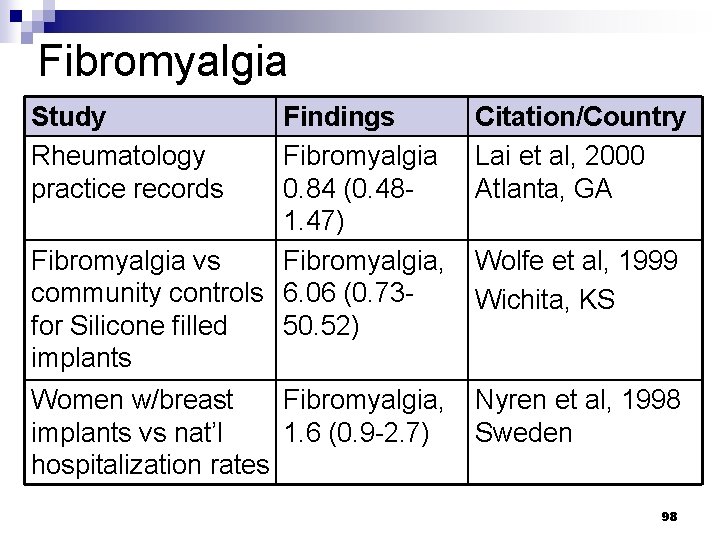
Fibromyalgia Study Rheumatology practice records Findings Fibromyalgia 0. 84 (0. 481. 47) Fibromyalgia vs Fibromyalgia, community controls 6. 06 (0. 73 for Silicone filled 50. 52) implants Citation/Country Lai et al, 2000 Atlanta, GA Women w/breast Fibromyalgia, implants vs nat’l 1. 6 (0. 9 -2. 7) hospitalization rates Nyren et al, 1998 Sweden Wolfe et al, 1999 Wichita, KS 98
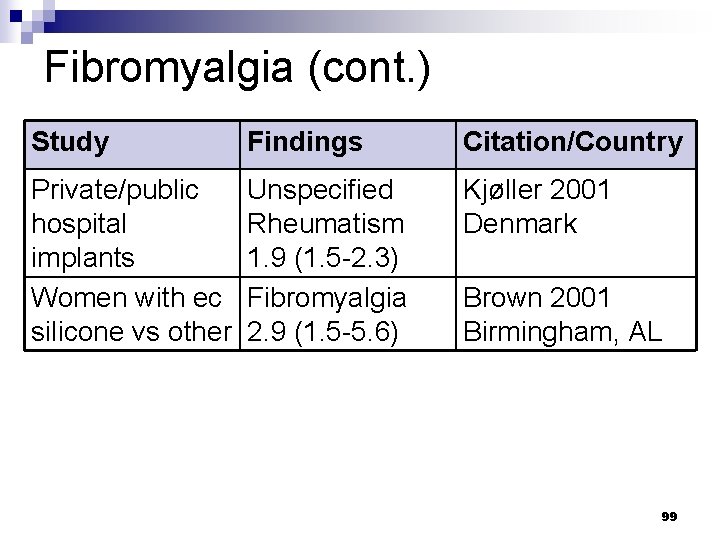
Fibromyalgia (cont. ) Study Findings Citation/Country Private/public hospital implants Women with ec silicone vs other Unspecified Rheumatism 1. 9 (1. 5 -2. 3) Fibromyalgia 2. 9 (1. 5 -5. 6) Kjøller 2001 Denmark Brown 2001 Birmingham, AL 99
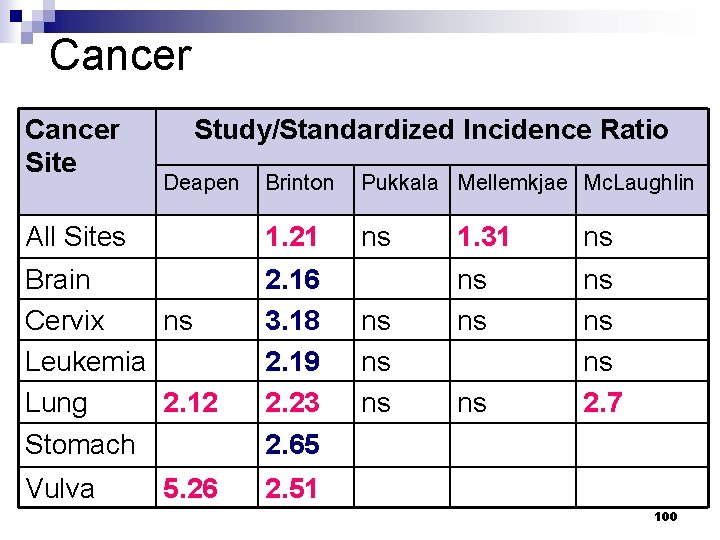
Cancer Site Study/Standardized Incidence Ratio Brinton Pukkala Mellemkjae Mc. Laughlin All Sites 1. 21 ns Brain Cervix ns Leukemia Lung 2. 12 2. 16 3. 18 2. 19 2. 23 Stomach 2. 65 Vulva Deapen 5. 26 ns ns ns 1. 31 ns ns ns 2. 7 ns 2. 51 100

Mammography Implant rupture during mammography n Implants obscure 22 -83% of breast tissue (Hayes et al, 1988) n Modified techniques needed (Ecklund et al, 1988) n Breast cancer detection delayed but no difference in mortality (Brinton et al, 2000) n Tumor size, lymph node involvement, histopathology similar (Cahan et al, 1995) n 101

Neurologic Disease n Swedish population based cohort found no increase in MS, ALS, Meniere’s syndrome, but significant increase in neurological disease in general (1. 7, 1. 1 -2. 6) (Nyren et al, 1998) n Danish study found no increase in specific neurologic diagnoses; neurologic disease in general slightly increased but not statistically significant (1. 7, 0. 9 -2. 9) (Winther et al, 1998) n Similar findings in breast reduction comparison groups in both studies n Both studies based on hospitalization 102
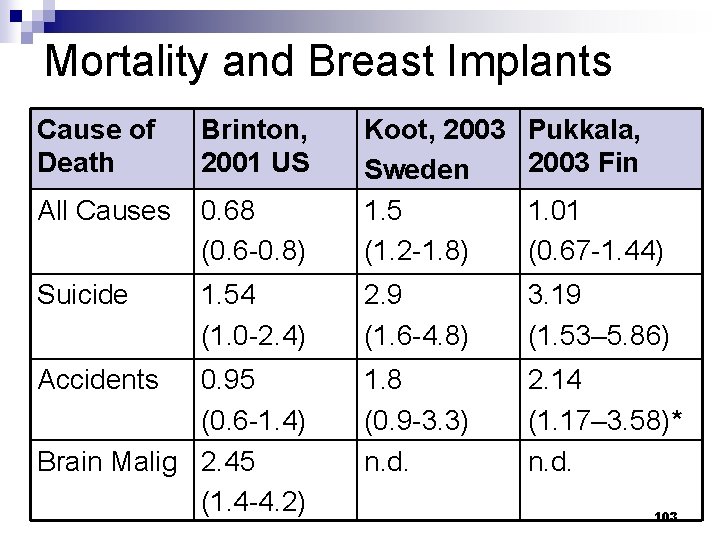
Mortality and Breast Implants Cause of Death Brinton, 2001 US Pukkala, 2003 Fin 0. 68 (0. 6 -0. 8) Koot, 2003 Sweden 1. 5 (1. 2 -1. 8) All Causes Suicide 1. 54 (1. 0 -2. 4) 2. 9 (1. 6 -4. 8) 3. 19 (1. 53– 5. 86) 0. 95 (0. 6 -1. 4) Brain Malig 2. 45 (1. 4 -4. 2) 1. 8 (0. 9 -3. 3) n. d. 2. 14 (1. 17– 3. 58)* n. d. Accidents 1. 01 (0. 67 -1. 44) 103

Resurgery and Local Complications Rates for additional surgery in numerous studies reported to be about 33% of women - common reason for resurgery is capsular contracture in several studies Gabriel, 1997 n Gutowski, 1997 n Brown, 2001 n 28% of 749 fu 8 yr 21% of 504 fu 6 yr 33% of 907 mt 11. 5 yr 104
Local Complications Capsular contracture n Breast pain n Infection n Hematoma n Implant extrusion n Changes in nipple sensation n Rashes n Chest wall skeletal changes n Calcification n Rupture n Gel migration n Etc. n 105

Breast Implant Rupture and Gel Migration n Breast implant rupture by MR in 344 B’ham women found 55% of implants ruptured affecting 68% of women, 22% ruptures extracapsular, 17 yr median (Brown et al, 2000) n Breast implant rupture by MR in 271 Danish women found 26% of implants ruptured affecting 36% of women, 22% ruptures extracapsular, 10 yr median (Holmich et al, 2001) 106

Breast Implant Rupture and Gel Migration (cont. ) n Extracapsular spread of silicone gel reported in 1123% of ruptured implants across several series n Frequency or severity of distant migration not known n Migration may result in gel/oil in lymph nodes, intraductal extension of gel, granuloma formation, transcutaneous leakage of gel, ulceration, tissue destruction, scarring n Silicone in tissues confirmed by imaging, microscopic examination of granulomatous response 107

Proposed Postapproval Study & Labeling Overview Samie Allen

Proposed Postapproval Study n Core Study Protocol ¨ Yearly follow-up with physician through 10 years ¨ MRI assessments at 1, 3, 5, 7, and 9 years n 2 -Phase Postapproval Study ¨ Phase I – continued evaluations as per IDE protocol through 5 -year timepoint ¨ Phase II – patient mail-in surveys for 6 -10 -year timepoints (no MRI assessments) 109

Proposed Labeling n Directions For Use (package insert) n Patient Brochure ¨Focus Group Study 110

Conclusion of FDA’s Presentation

Panel Questions

Panel Question 1 Prospective MRI screening for asymptomatic rupture was conducted in a subset of Core Study participants (approximately 34%). Complete MRI screening data are available for the 1 -year post-operative timepoint for each indication and partial 3 year data are available for the augmentation indication at the time of database closure. Continued MRI screening of this Core Study subset is planned for at years 3, 5, 7, and 9 after implantation. Of the 15 implant ruptures that Inamed reports as confirmed at the time of database closure, the majority--9 implants (60%)--were initially detected by MRI screening and were asymptomatic: Core Augmentation, 0 of 3 ruptures; Core Reconstruction, 6 of 8 ruptures; and Core Revision, 3 of 5 ruptures. Additionally, published literature on silicone gel implant rupture, although not specific to Inamed’s implants, indicates that rupture rate increases significantly with implant age and that depending on implant type, manufacturer, and age, between 26% (median implant age 12 years) and 55% (median implant age 16. 4 years) of implants assessed by MRI had MRI evidence of rupture. Please discuss the adequacy of the information to determine the safety of this product with respect to asymptomatic rupture. 113

Panel Question 2 Potential long-term and general health effect issues for these implants include the risk of cancer(s), connective tissue disorders (typical and atypical), gel migration, interference of implant on ability of mammography to detect tumors in implanted breasts, interference with breast feeding, reproductive/teratogenic effects, and the later effects on offspring from women with implants. To address these issues, Inamed utilized historical published literature, which is not specific to Inamed’s implants, as well as animal studies on their product. Please discuss the adequacy of the literature and preclinical testing to determine the safety of this product with respect to long-term and general health 114 effects.

Panel Question 3 Considering the safety data reported for the augmentation group: n local complications reported in Core Study, Adjunct Study, and AR 90 Study n asymptomatic/silent rupture information based on approximately 30% of the patients in the Core Study with only the first of 5 prospective serial screenings with complete data n published historical literature and animal data to address long term and general health effects. Given these data, and that the augmentation patient generally has breast implant surgery at a younger age which includes childbearing years compared to the other indications, is there reasonable assurance that the device is safe for augmentation patients? 115

Panel Question 4 Considering the safety data reported for the reconstruction and revision groups: n local complications reported in Core Study, Adjunct Study, and AR 90 Study n asymptomatic/silent rupture information based on approximately 30% of the patients in the Core Study with only the first of 5 prospective serial screenings with complete data n published historical literature and animal data to address long term and general health effects. Given these data, and that reconstruction and revision patients generally undergo breast implantation at an older age than augmentation patients, is there reasonable assurance that the device is safe for reconstruction and revision 116 patients?

Panel Question 5 To evaluate device effectiveness, Inamed collected data on patient satisfaction and health status/quality of life (e. g. , SF-36, MOS-20, Body Esteem Scale, etc. ). Based on these data, has Inamed adequately demonstrated reasonable assurance of effectiveness of the implants for each of the augmentation, reconstruction, and revision indications? 117

Panel Question 6 Given the information in question 1 and if you recommend approval of the PMA, please address the following with respect to labeling for the device: n Provide your recommendations for the frequency and method of screening for asymptomatic rupture, given that prospective screening for asymptomatic rupture in not currently routinely performed. n Provide your recommendations for the necessity of explantation of asymptomatic implant ruptures. 118

Panel Question 7 Inamed provided a brief description of their postapproval study plan. The Core Study protocol, as well as informed consent, currently requires yearly follow-up with a physician. Inamed is now proposing a change to the study requirements as follows. More specifically, Inamed is proposing a 2 -phase postapproval study. Phase I involves continued physician evaluation as per the IDE protocol through a patient’s 5 -year follow-up timepoint. Phase II involves mail-in surveys completed by the patient from their 6 to 10 -year follow-up timepoints. In the proposed Phase II protocol, for example, MRI screening for asymptomatic rupture would not be captured. Given this proposal and if you recommend approval of the PMA: n Please comment on the method of data collection (mailed survey) from the 6 -10 -year timepoints, given that the Core Study protocol as well as informed consent currently calls for prospective yearly follow-up. n In addition, please describe any other specific endpoints that should be captured as part of their postapproval study. For example, in the proposed protocol, silent rupture would not be captured. 119
- Slides: 119