In the name of GOD 1 Zeinab Mokhtari

In the name of GOD 1

Zeinab Mokhtari 2 06 -Jan-2010
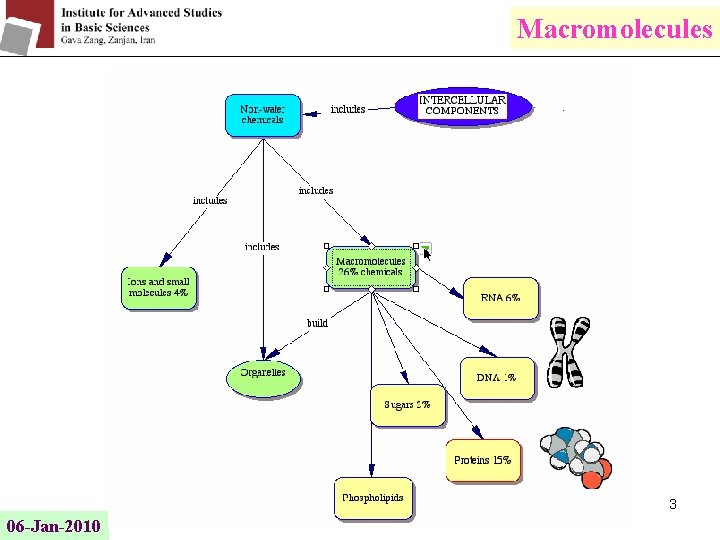
Macromolecules 3 06 -Jan-2010
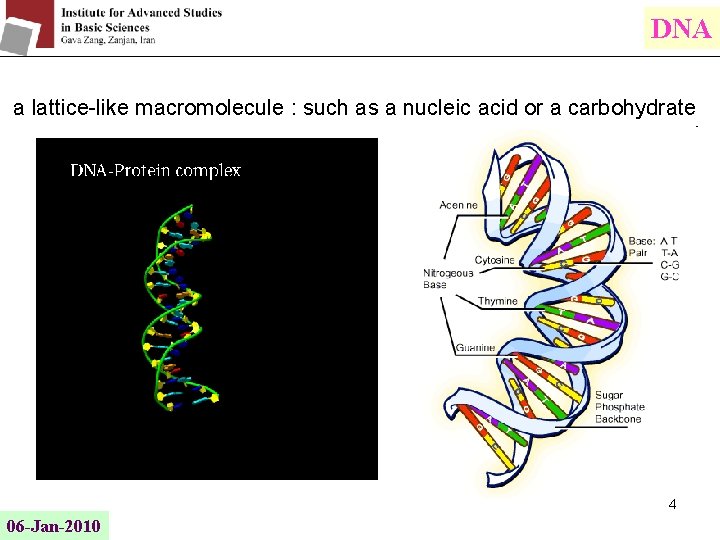
DNA a lattice-like macromolecule : such as a nucleic acid or a carbohydrate 4 06 -Jan-2010

Macromolecules N : potential binding sites per macromolecule l : the length of the ligand Thermodynamic parameters (k , H, …) governers of the behavior of the system The number of ligand molecules bound per macromolecule 5 06 -Jan-2010
![Scatchard plot ν versus [L] : hyperbolic representation, requires nonlinear regression analysis Scatchard plot Scatchard plot ν versus [L] : hyperbolic representation, requires nonlinear regression analysis Scatchard plot](http://slidetodoc.com/presentation_image_h2/452e67def7c65150b723c591351c0252/image-6.jpg)
Scatchard plot ν versus [L] : hyperbolic representation, requires nonlinear regression analysis Scatchard plot is a transformed representation. v /[L] as a function of ν N equivalent and independent binding sites and a ligand with size l = 1 a linear plot 06 -Jan-2010 Several classes of binding sites, or interacting binding sites, or a ligand with size l > 1 a curved plot 6

Mc. Ghee and von Hippel Valid for infinite homogeneous lattices with: any ligand size, l any ligand dissociation constant, k any level of cooperativity interaction between ligands bound contiguously, ω Extensions of this theory for : finite lattices heterogeneous systems ( the binding sites are not homogeneously distributed throughout the macromolecule) different classes of binding sites present in the macromolecule different ligand binding modes a mixture of different ligands 7 06 -Jan-2010

Case I : non-cooperative (independent) ligand binding Case II : cooperative ligand binding 8 06 -Jan-2010

ITC : Isothermal titration calorimetry An excellent tool for studying biomolecular reactions The heat associated with a binding process is measured. Injections of a ligand solution from a computer-controlled syringe into a macromolecule solution placed in a thermostatized cell. 9 06 -Jan-2010

The syringe rotates in place during the ITC experiment. The end of the syringe has been adapted to provide continuous mixing in the ITC cell. The plunger is computercontrolled and injects precise volumes of ligand. The total concentration of each reactant in the cell after injection i : 10 06 -Jan-2010

Case I Lineal lattice-like macromolecule with noncooperative ligand binding: macromolecule with N equivalent and independent binding sites and ligand size l ≥ 1 11 06 -Jan-2010

Case I linear in ν only if l = 1 As binding proceeds to saturation, it is more difficult to find l free consecutive binding sites when l is large. Then, the larger the ligand size l, the larger such effect. an (l + 1)-order polynomial equation A nonlinear regression : the optimal values of N, l, k, and H from the experimental data. 06 -Jan-2010 12
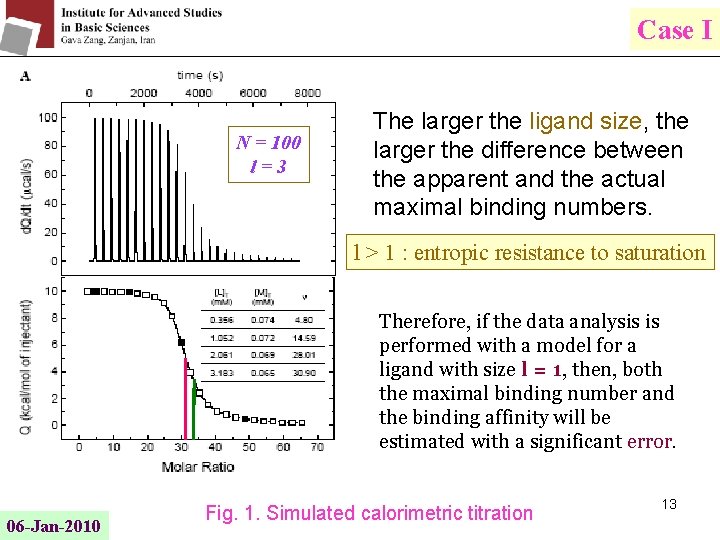
Case I N = 100 l=3 The larger the ligand size, the larger the difference between the apparent and the actual maximal binding numbers. l > 1 : entropic resistance to saturation Therefore, if the data analysis is performed with a model for a ligand with size l = 1, then, both the maximal binding number and the binding affinity will be estimated with a significant error. 06 -Jan-2010 Fig. 1. Simulated calorimetric titration 13
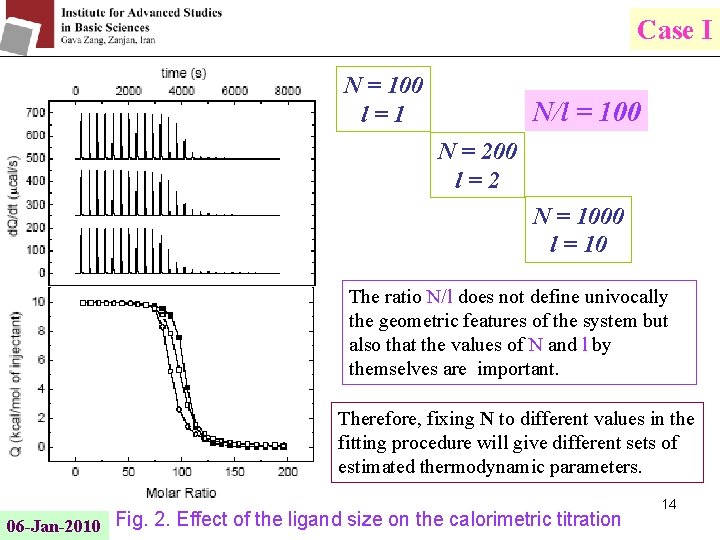
Case I N = 100 l=1 N/l = 100 N = 200 l=2 N = 1000 l = 10 The ratio N/l does not define univocally the geometric features of the system but also that the values of N and l by themselves are important. Therefore, fixing N to different values in the fitting procedure will give different sets of estimated thermodynamic parameters. 06 -Jan-2010 Fig. 2. Effect of the ligand size on the calorimetric titration 14

Case II Lineal lattice-like macromolecule with cooperative ligand binding: macromolecule with N equivalent and dependent binding sites and ligand size l ≥ 1 15 06 -Jan-2010

Case II an (l + 3)-order polynomial equation 06 -Jan-2010 16

Case II 1≤ω<∞ Positive cooperativity ω=1 No cooperation 0≤ω<1 Negative cooperativity It is obvious that at a given value of the ligand size l, there will be a value of the interaction parameter ω that almost compensates the negative entropic effect from l, resulting in a nearly linear plot. Difficult discriminating between different situations ITC allows discriminating between different cases. Interaction between bound ligands : an additional contribution to the overall enthalpy of binding (The interaction or cooperativity enthalpy h) 06 -Jan-2010 17

Case II h : the enthalpy associated with the interaction of nearest neighbor bound ligands νisol : the partial number of ligand molecules bound isolated νsc : the partial number of ligand molecules bound with only one nearest neighbor (singly contiguous) νdc : the partial number of ligand molecules bound with two 18 nearest neighbors (doubly contiguous) 06 -Jan-2010

Case II 19 06 -Jan-2010
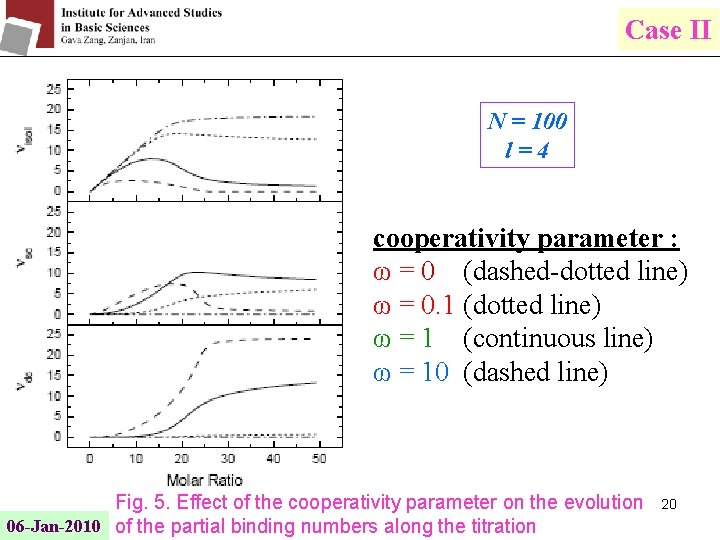
Case II N = 100 l=4 cooperativity parameter : ω = 0 (dashed-dotted line) ω = 0. 1 (dotted line) ω = 1 (continuous line) ω = 10 (dashed line) Fig. 5. Effect of the cooperativity parameter on the evolution 06 -Jan-2010 of the partial binding numbers along the titration 20
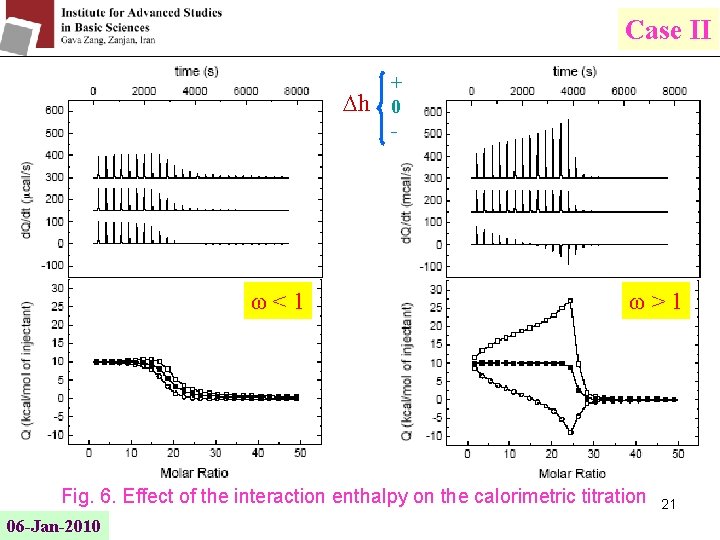
Case II h ω<1 + 0 - ω>1 Fig. 6. Effect of the interaction enthalpy on the calorimetric titration 06 -Jan-2010 21
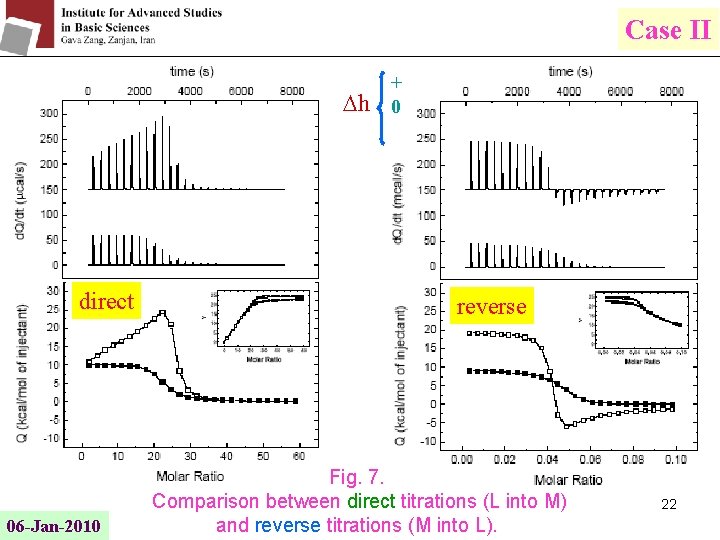
Case II h direct 06 -Jan-2010 + 0 reverse Fig. 7. Comparison between direct titrations (L into M) and reverse titrations (M into L). 22

Tha 23

y a d a s i r e t h g u a. l t ted u o as h t i n i w l w p y a a h d C A ie l r a h C 24
- Slides: 24