In Bio Ltd In Bio Ltd ANTIBODIES FOR

In. Bio Ltd. __________________ In. Bio Ltd. ANTIBODIES FOR THERAPEUTIC AND DIAGNOSTIC APPLICATIONS Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ Facts on In. Bio • Established in Tallinn, 1999 • General business idea: to develop new effective and selective skin cancer therapeutics and diagnostics. • The company was initiated by a group of scientists of National Institute of Chemical Physics and Biophysics and Karolinska Institute, Sweden Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826
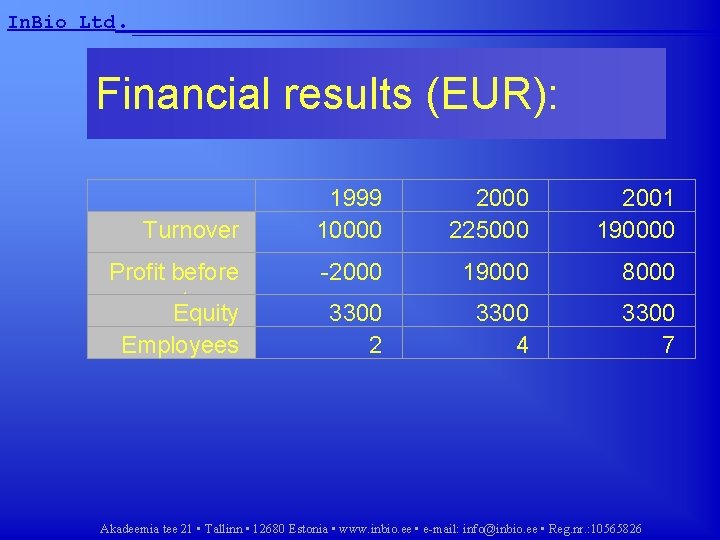
In. Bio Ltd. __________________ Financial results (EUR): Turnover 1999 10000 225000 2001 190000 Profit before taxes Equity Employees -2000 19000 8000 3300 2 3300 4 3300 7 Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ Background for product development • Cancer is the second most common cause of death in the developed world • The limits of usefulness are being approached for conventional therapies and further advances will require principally new approaches • The foreseen products of our company are prototypes of new anti-cancer drugs that are characterised by: – – -high degree of selectivity -efficient delivery Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ What is antibody? • Antibodies are made by a class of white blood cells • They are naturally present in the blood • Biological function is to perform defence functions • Each antigen causes the formation of a specific antibody • Recombinant DNA technology allows the engineer antibodies • It is possible to produce complete antibodies in cell culture systems. Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ Reasons to use antibodies in drug development • Can be rationally designed and easily made • Can be engineered to add fragmenmts that enhance their therapeutic potential • Have no intrinsic toxicity as based on the natural molecules • Monoclonal antibodies (m. Abs)will not fail at the drug safety testing (phase I clinical trial stages) • Technology is now advanced enough for therapeutic applications of m. Abs Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ What is new in our technology? • Our technology combines two different platforms: • Monoclonal antibodies against cancer-specific antigens – Modification of antibody sequence to make these m. Abs membrane permeable • Such approach helps to utilise intracellular cancerspecific targets that can not be usually reached • Focus on skin cancer, first targets are GLI proteins Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ What are GLI proteins? • GLI proteins are gene switches • GLI proteins are involved in development and lead cells to divide • Abnormal activation of GLI proteins lead to cancer • GLI proteins represent an attractive target for anticancer drug development Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826
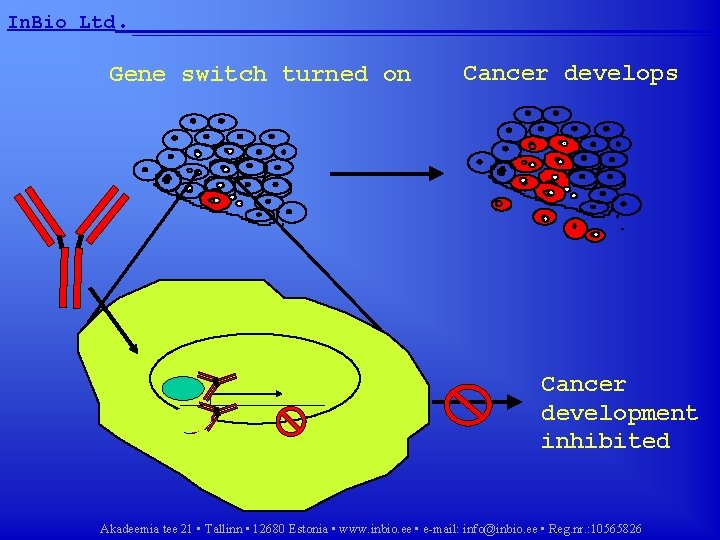
In. Bio Ltd. __________________ Gene switch turned on Cancer develops Cancer development inhibited Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826
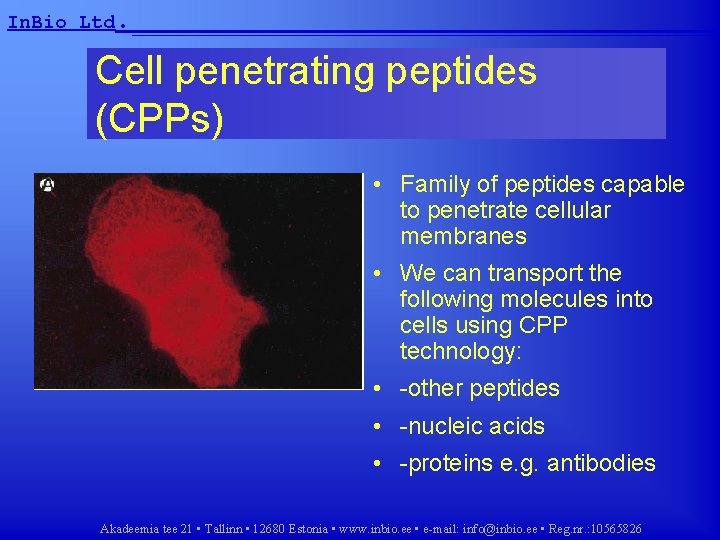
In. Bio Ltd. __________________ Cell penetrating peptides (CPPs) • Family of peptides capable to penetrate cellular membranes • We can transport the following molecules into cells using CPP technology: • -other peptides • -nucleic acids • -proteins e. g. antibodies Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ World antibody market: • Currently more than 200 monoclonal antibodies are in development • Targeted disorders include cancer, heart disease, infectious diseases and autoimmune diseases • Market is still in its inception stage, • Market size estimated at nearly $2. 8 billion in 1999, Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ World antibody market: • Market forecast growth to almost $9. 8 billion in 2004. • Expected therapeutic antibody average annual growth rate is projected at 21, 8% • Diagnostic/therapeutic antibody AAGR will grow at fast nearly 50% rate • Main Target Markets for In. Bio: • Scandinavian Market • U. S. market Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ Timing and positioning • Antibody based drugs are in the position of significant commercial growth • In. Bio enters the market at the moment when all developers seek for new approaches • In. Bio has new technology to make antibodies more effective • In. Bio has involved outstanding specialists from all fields that are required to accomplish our business plan Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826

In. Bio Ltd. __________________ Benefits for the investor: • Will invest in profitable business at evolving market • Will invest in area that is significantly improving the well-being of the mankind • Will get possession rights of a unique knowhow and new technologies that will realise in new type of therapeutic and diagnostic tools Akadeemia tee 21 • Tallinn • 12680 Estonia • www. inbio. ee • e-mail: info@inbio. ee • Reg. nr. : 10565826
- Slides: 14