Impurities in New Drug Substances ICH Topic Q

ﺍﻟﺸﻮﺍﺋﺐ ﻓﻲ ﺍﻷﺪﻭﻳﺔ ﺍﻟﺠﺪﻳﺪﺓ Impurities in New Drug Substances ICH Topic Q 3 A (R) – Revised Guidelines in Oct-2006.

CONTENTS 1. Preamble 2. Classification of Impurities 3. Rationale for the Reporting and Control of Impurities 3. 1 Organic Impurities 3. 2 Inorganic Impurities 3. 3 Solvents 4. Analytical Procedures 5. Reporting Impurity Content of Batches 6. Specification Limits for Impurities

1. Preamble Impurities in new drug substances are addressed from two perspectives: Chemistry Aspects includes – – classification and identification of impurities, report generation, setting specifications, and a brief discussion of analytical procedures; and Safety Aspects includes specific guidance for qualifying impurities that were not present in batches of new drug substance used in safety and clinical studies and/or impurity levels substantially higher than in those batches. Threshold limits are defined at or below which, qualification is not needed.

2. CLASSIFICATION OF IMPURITIES Impurities may be classified into the following categories: – Organic Impurities (Process- and Drug-Related) – Inorganic Impurities – Residual Solvents

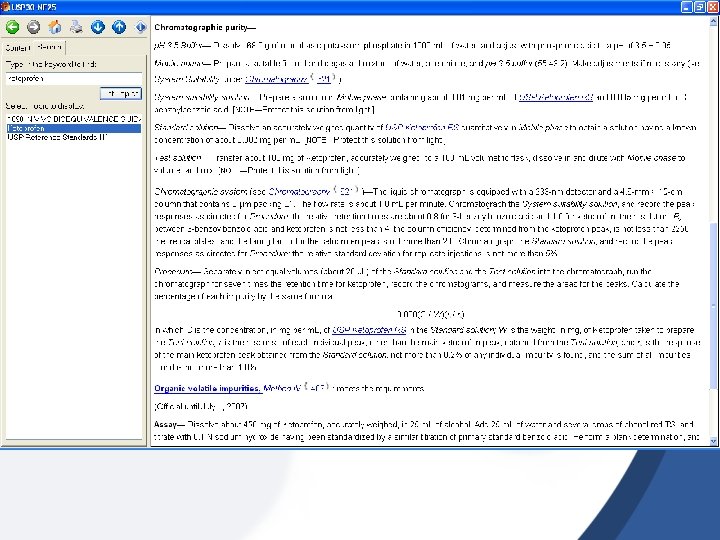
Source of Impurities impurities may arise during the manufacturing process and/or storage of the new drug substance [a] Organic Impurities; They may be identified or unidentified, volatile or non-volatile, and include · Starting Materials · By-Products · Intermediates · Degradation Products · Reagents, Ligands and Catalysts Solvents are organic or inorganic liquids used during the manufacturing process
![[B] Inorganic impurities; may derive from the manufacturing process. They are normally known and [B] Inorganic impurities; may derive from the manufacturing process. They are normally known and](http://slidetodoc.com/presentation_image_h/30c817421bc21a64b858c078cf19b4f5/image-6.jpg)
[B] Inorganic impurities; may derive from the manufacturing process. They are normally known and identified and include: · Reagents, Ligands and Catalysts · Heavy metals or other residual metals · Inorganic Salts

3. RATIONALE FOR THE REPORTING AND CONTROL OF IMPURITIES 3. 1 Organic Impurities The applicant should summarize those actual and potential impurities most likely to arise during the synthesis, purification, and storage of the new drug substance. The applicant should summarize the laboratory studies conducted to detect impurities in the new drug substance. This summary should include test results of batches manufactured during the development process and batches from the proposed commercial process, as well as results of intentional degradation studies used to identify potential impurities arising during storage.

3. 2 Inorganic Impurities Inorganic impurities are normally detected and quantitated using pharmacopoeial or other appropriate procedures. Carry-over of catalysts to the new drug substance should be evaluated during development. The need for inclusion or exclusion of inorganic impurities in the new drug substance specifications should be discussed. Limits should be based on pharmacopoeial standards or known safety data.

4. ANALYTICAL PROCEDURES Organic impurity levels can be measured by a variety of techniques, including; those which compare an analytical response for an impurity to that of an appropriate reference standard or to the response of the new drug substance itself. - Use Diode-Array Detector - Use LC/MS or GC-MS
![Assay of impurities, how? • [1] Known impurities; use RS substance sold by USP Assay of impurities, how? • [1] Known impurities; use RS substance sold by USP](http://slidetodoc.com/presentation_image_h/30c817421bc21a64b858c078cf19b4f5/image-10.jpg)
Assay of impurities, how? • [1] Known impurities; use RS substance sold by USP or Eu. Pharm. Or use working standard • [2] Unknown impurities; measured as if it is the principle drug (use correction factor or not)

5. REPORTING IMPURITY CONTENT OF BATCHES • Batches of Clinical study, safety, stability studies. • Report what? ; – – – Identified impurity(ies) Un-identified impur. Total impurities Analytical method used (with validation) Tabulated data (impurities amount) Representative chromatogram(s) to show impurity peak(s) and the anal. method performance (impu-separation)

For each batch of the new drug substance, the report should include: · Batch Identity and Size · Date of Manufacture · Site of Manufacture · Manufacturing Process · Impurity Content, Individual and Total · Use of Batches · Reference to Analytical Procedure Used
![6. SPECIFICATIONS FOR IMPURITIES Obtain Specs of Known and unknown impur. From; [1] USP 6. SPECIFICATIONS FOR IMPURITIES Obtain Specs of Known and unknown impur. From; [1] USP](http://slidetodoc.com/presentation_image_h/30c817421bc21a64b858c078cf19b4f5/image-13.jpg)
6. SPECIFICATIONS FOR IMPURITIES Obtain Specs of Known and unknown impur. From; [1] USP or BP [2] Drug supplier/manufacturer Limit of impurities from USP/BP or supplier, +Chromatogram +and method of separation. (Potential impurities).

The new drug substance specifications should include, where applicable, limits for: Organic Impurities 1. Each Specified Identified Impurity 2. Each Specified Unidentified Impurity at a level greater than (>) the qualification/identification threshold · Any Unspecified Impurity, with a limit of not more than (_) the qualification/identification threshold 1. Total Impurities Residual Solvents Inorganic Impurities
![Limit of impurities As per ICH Guidelines; [1] Get the cited limit of impurities Limit of impurities As per ICH Guidelines; [1] Get the cited limit of impurities](http://slidetodoc.com/presentation_image_h/30c817421bc21a64b858c078cf19b4f5/image-15.jpg)
Limit of impurities As per ICH Guidelines; [1] Get the cited limit of impurities (known, unknown, total) from USP or BP or Eur. Pharm [2] From Supplier or manufacturer [3] If not available in official monographs apply the below rule; Maximum Daily Dose Qualification Threshold and Identification Threshold Reporting Threshold * 2 g / day 0. 1% or 1 mg per day intake (whichever is lower) 0. 05% >2 g / day 0. 05% 0. 03%


- Slides: 18