Improving Physicochemical Properties of Biopharmaceutical Drug Candidates David

Improving Physicochemical Properties of Biopharmaceutical Drug Candidates David Litzinger, Ph. D Director, Pharmaceutical Sciences Amylin Pharmaceuticals, Inc. PEGS Conference May 20, 2010 Boston, MA
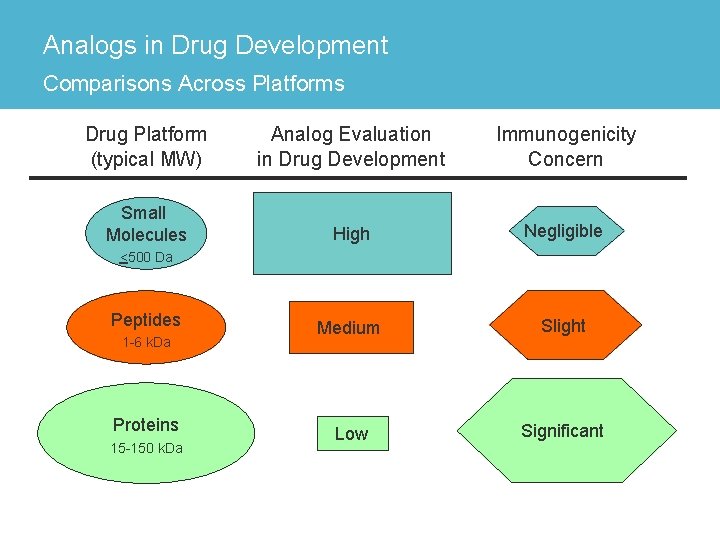
Analogs in Drug Development Comparisons Across Platforms Drug Platform (typical MW) Analog Evaluation in Drug Development Immunogenicity Concern Small Molecules High Negligible Medium Slight Low Significant <500 Da Peptides 1 -6 k. Da Proteins 15 -150 k. Da
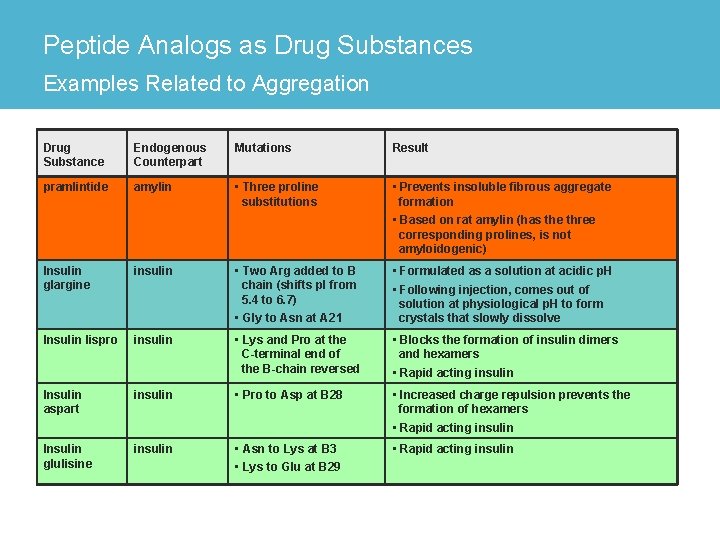
Peptide Analogs as Drug Substances Examples Related to Aggregation Drug Substance Endogenous Counterpart Mutations Result pramlintide amylin • Three proline substitutions • Prevents insoluble fibrous aggregate formation • Based on rat amylin (has the three corresponding prolines, is not amyloidogenic) Insulin glargine insulin • Two Arg added to B chain (shifts p. I from 5. 4 to 6. 7) • Gly to Asn at A 21 • Formulated as a solution at acidic p. H • Following injection, comes out of solution at physiological p. H to form crystals that slowly dissolve Insulin lispro insulin • Lys and Pro at the C-terminal end of the B-chain reversed • Blocks the formation of insulin dimers and hexamers • Rapid acting insulin Insulin aspart insulin • Pro to Asp at B 28 • Increased charge repulsion prevents the formation of hexamers • Rapid acting insulin Insulin glulisine insulin • Asn to Lys at B 3 • Lys to Glu at B 29 • Rapid acting insulin

Peptide and Protein Optimization Example Options for Improving Physical Stability Approaches to Improving Physical Stability* Mutational Chemical Modification – Mutations based on superior properties in alternate species √ – Decrease hydrophobicity √ √ – Increase hydrophilicity √ √ – Increase net charge √ √ – Changing the p. I √ √ – Polymer conjugation * More that one approach can be combined √

Glucose-Dependent Insulinotropic Polypeptide Example of Analog Evaluation in Drug Development > Glucose-dependent Insulinotropic Polypeptide (GIP) – 42 -amino acid hormone synthesized and secreted from intestinal K-cells – Integral role in regulating insulin secretion and response – Amylin Pharmaceuticals currently investigating GIP as a possible mono- or combination therapy for Type 2 Diabetes Mellitus > Development Challenges – Native GIP rapidly inactivated by dipeptidyl peptidase-IV (DPP-IV) and has a very short half-life – Development of GIP analogs challenging due to poor solubility > Second Generation Effort (G 2) – G 1 effort addressed DPP-IV metabolism, optimized activity – G 2 GIP analogs identified and evaluated for improved solubility • In Silico modeling used for primary sequences analysis • p. H-solubility profile, physical and chemical stability were screened • CD used to monitor secondary structure
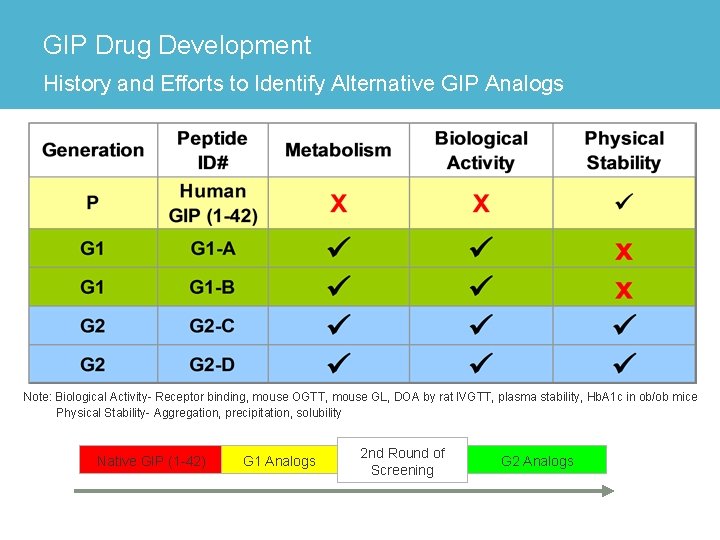
GIP Drug Development History and Efforts to Identify Alternative GIP Analogs Note: Biological Activity- Receptor binding, mouse OGTT, mouse GL, DOA by rat IVGTT, plasma stability, Hb. A 1 c in ob/ob mice Physical Stability- Aggregation, precipitation, solubility Native GIP (1 -42) G 1 Analogs 2 nd Round of Screening G 2 Analogs
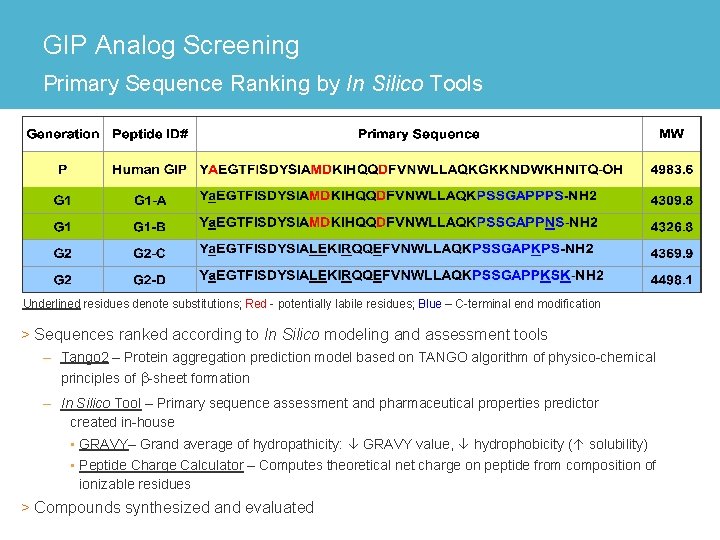
GIP Analog Screening Primary Sequence Ranking by In Silico Tools Underlined residues denote substitutions; Red - potentially labile residues; Blue – C-terminal end modification > Sequences ranked according to In Silico modeling and assessment tools – Tango 2 – Protein aggregation prediction model based on TANGO algorithm of physico-chemical principles of b-sheet formation – In Silico Tool – Primary sequence assessment and pharmaceutical properties predictor created in-house • GRAVY– Grand average of hydropathicity: GRAVY value, hydrophobicity ( solubility) • Peptide Charge Calculator – Computes theoretical net charge on peptide from composition of ionizable residues > Compounds synthesized and evaluated
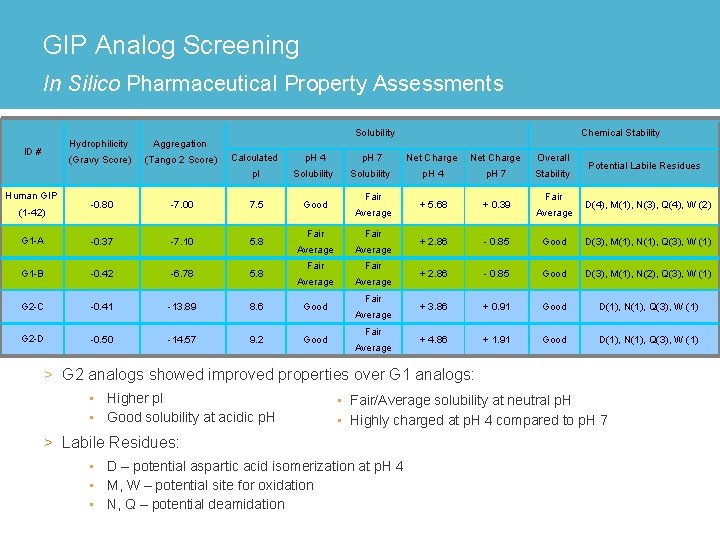
GIP Analog Screening In Silico Pharmaceutical Property Assessments ID # Human GIP Hydrophilicity Aggregation (Gravy Score) (Tango 2 Score) Solubility Calculated p. H 4 p. I Solubility Good -0. 80 -7. 00 7. 5 G 1 -A -0. 37 -7. 10 5. 8 G 1 -B -0. 42 -6. 78 5. 8 G 2 -C -0. 41 -13. 89 8. 6 Good G 2 -D -0. 50 -14. 57 9. 2 Good (1 -42) p. H 7 Solubility Fair Average Fair Average Fair Average Chemical Stability Net Charge Overall Stability Potential Labile Residues p. H 4 p. H 7 + 5. 68 + 0. 39 + 2. 86 - 0. 85 Good D(3), M(1), N(1), Q(3), W (1) + 2. 86 - 0. 85 Good D(3), M(1), N(2), Q(3), W (1) + 3. 86 + 0. 91 Good D(1), N(1), Q(3), W (1) + 4. 86 + 1. 91 Good D(1), N(1), Q(3), W (1) Fair Average D(4), M(1), N(3), Q(4), W (2) > G 2 analogs showed improved properties over G 1 analogs: • Higher p. I • Good solubility at acidic p. H • Fair/Average solubility at neutral p. H • Highly charged at p. H 4 compared to p. H 7 > Labile Residues: • D – potential aspartic acid isomerization at p. H 4 • M, W – potential site for oxidation • N, Q – potential deamidation
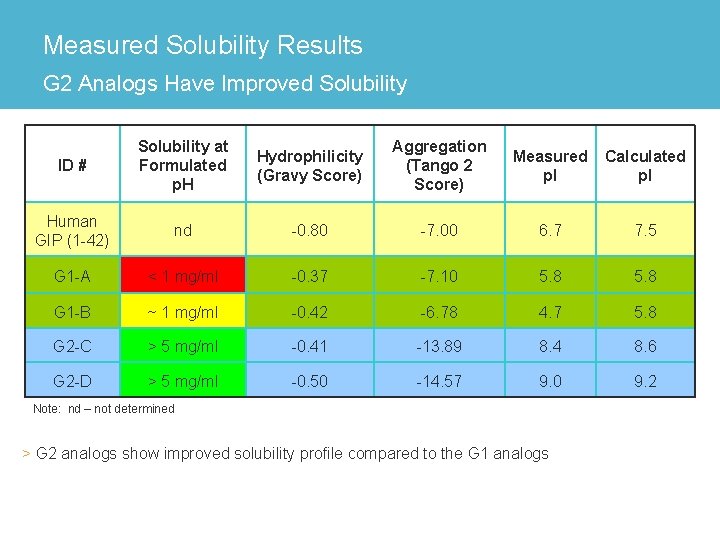
Measured Solubility Results G 2 Analogs Have Improved Solubility ID # Solubility at Formulated p. H Hydrophilicity (Gravy Score) Aggregation (Tango 2 Score) Measured p. I Calculated p. I Human GIP (1 -42) nd -0. 80 -7. 00 6. 7 7. 5 G 1 -A < 1 mg/ml -0. 37 -7. 10 5. 8 G 1 -B ~ 1 mg/ml -0. 42 -6. 78 4. 7 5. 8 G 2 -C > 5 mg/ml -0. 41 -13. 89 8. 4 8. 6 G 2 -D > 5 mg/ml -0. 50 -14. 57 9. 0 9. 2 Note: nd – not determined > G 2 analogs show improved solubility profile compared to the G 1 analogs
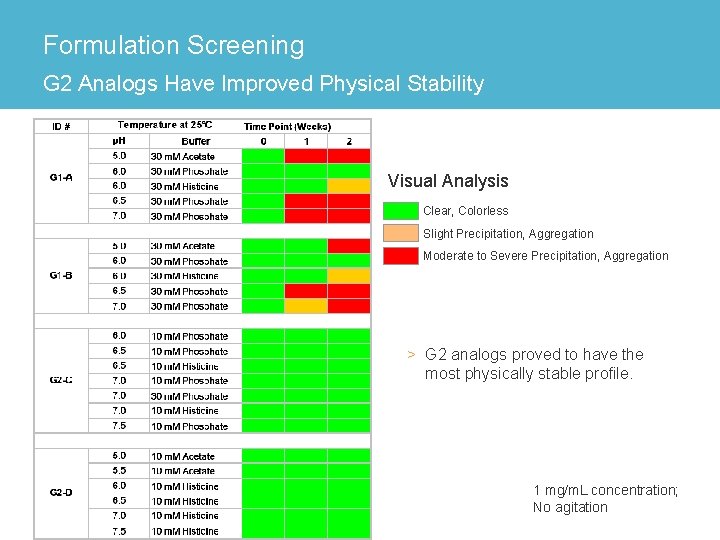
Formulation Screening G 2 Analogs Have Improved Physical Stability Visual Analysis Clear, Colorless Slight Precipitation, Aggregation Moderate to Severe Precipitation, Aggregation > G 2 analogs proved to have the most physically stable profile. 1 mg/m. L concentration; No agitation
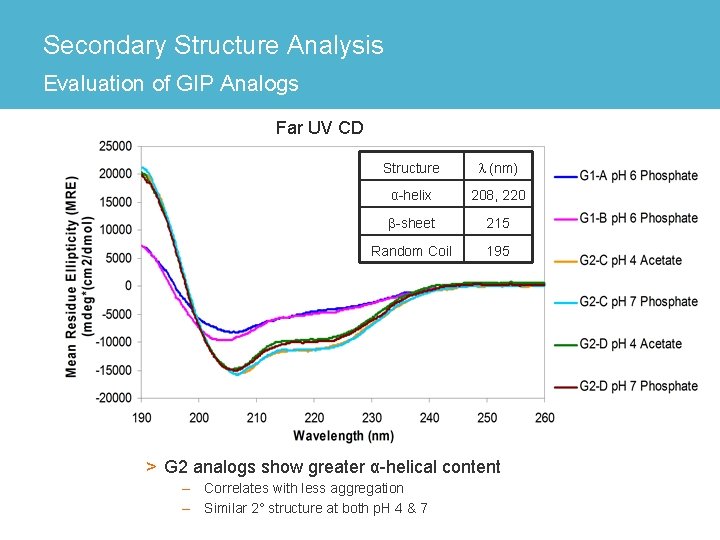
Secondary Structure Analysis Evaluation of GIP Analogs Far UV CD Structure l (nm) α-helix 208, 220 β-sheet 215 Random Coil 195 > G 2 analogs show greater α-helical content – Correlates with less aggregation – Similar 2° structure at both p. H 4 & 7

GIP Analog Optimization Conclusions > G 1 analogs demonstrated improved biological efficacy and longer duration of action compared to native GIP, but had poor physical stability > G 2 analogs showed both improved efficacy and physical stability – Experimental results correlated well with their higher net charge and more negative GRAVY scores predicted in silico. – At 1 mg/m. L concentrations were physically and chemically stable under the tested conditions with little to no visible aggregation. – Secondary structure is predominantly α-helical in liquid state (p. H 4. 0 and p. H 7. 0)
Metreleptin Compound Properties and Obesity Treatment Approaches • 16. 2 kd 147 amino acids, (native leptin 146 AA) • Isoelectric point 6. 1 • Single disulfide bond • No free cysteines • Limited solubility at neutral p. H, 2 -3 mg/m. L, higher at lower p. H • Four helix bundle tertiary structure > Amgen pursued leptin monotherapy as a treatment for obesity – High dose, up to 0. 3 mg/kg (~30 mg per injection) – Heymsfield et al. (1999) JAMA > Amylin is evaluating leptin in combination with pramlintide for treatment of obesity – Lower dose – Roth et al. (2008) PNAS
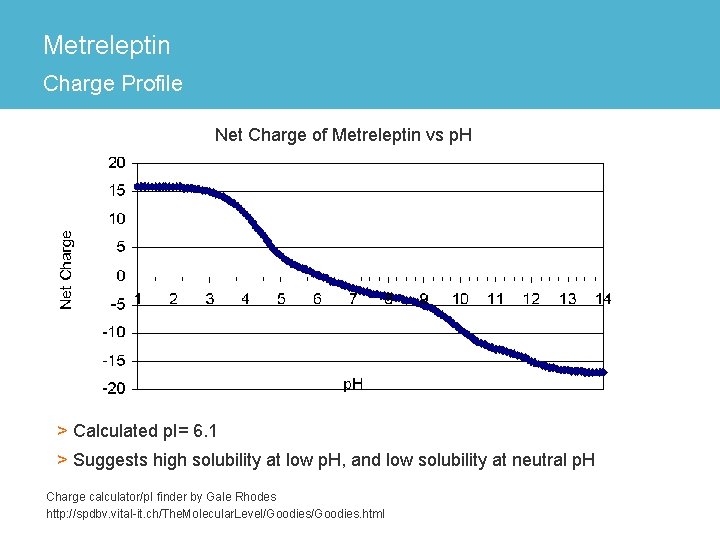
Metreleptin Charge Profile Net Charge of Metreleptin vs p. H > Calculated p. I= 6. 1 > Suggests high solubility at low p. H, and low solubility at neutral p. H Charge calculator/p. I finder by Gale Rhodes http: //spdbv. vital-it. ch/The. Molecular. Level/Goodies. html
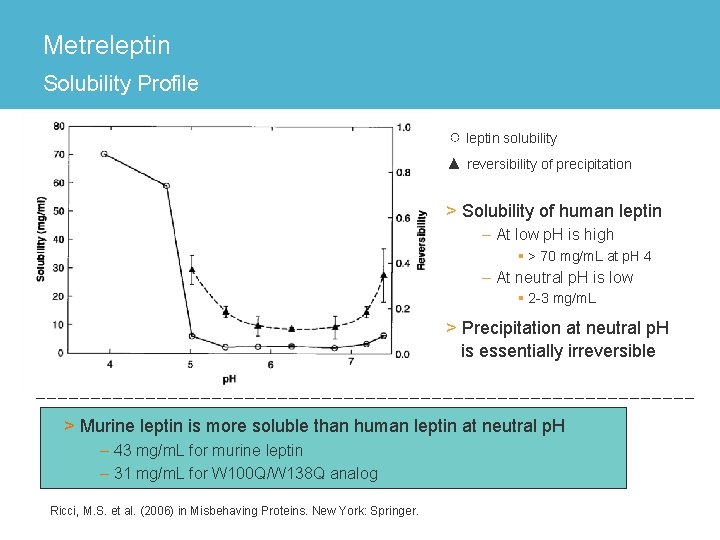
Metreleptin Solubility Profile ○ leptin solubility ▲ reversibility of precipitation > Solubility of human leptin – At low p. H is high § > 70 mg/m. L at p. H 4 – At neutral p. H is low § 2 -3 mg/m. L > Precipitation at neutral p. H is essentially irreversible > Murine leptin is more soluble than human leptin at neutral p. H – 43 mg/m. L for murine leptin – 31 mg/m. L for W 100 Q/W 138 Q analog Ricci, M. S. et al. (2006) in Misbehaving Proteins. New York: Springer.

Human and Murine Leptin Amino Acid Sequence Comparison > Comparison of human and murine leptin sequences 0 10 20 30 40 50 60 70 80 90 100 110 LAFSKSCHLP LAFSKSCSLP WASGLETLDS QTSGLQKPES 130 140 HUMAN: MVPIQKVQDD MURINE: MVPIQKVQDD HUMAN: DFIPGLHPIL MURINE: DFIPGLHPIL HUMAN: LENLRDLLHV MURINE: LENLRDLLHL 120 HUMAN: STEVVALSRL MURINE: STEVVALSRL TKTLIKTIVT TLSKMDQTLA SLSKMDQTLA QGSLQDMLWQ QGSLQD I LQQ RINDISHTQS VYQQILTSMP VYQQVLTSLP LDLSPGC LDVSPEC Residues that differ between the human and murine sequences are in red. Note that the first methionine residue associated with E. coli production is not counted. – Differ at 22 sites – Sequence differences of particular significance in solubility/aggregation properties VSSKQKVTGL VSAKQRVTGL SRNVIQISND SQNVLQIAND LGGVLEASGY LDGVLEASLY

Metreleptin Surface Modeling Electrostatic Surface Red = Basic (+) Blue = Acidic(-) Hydrophobicity Surface Brown = Lipophilic Blue = Hydrophilic, charged Trp 138 > Surface modeling shows region around Trp 138 has potential role in aggregation – Low electrostatic potential – High lipophilicity Benchware 3 DExplorer (Tripos)
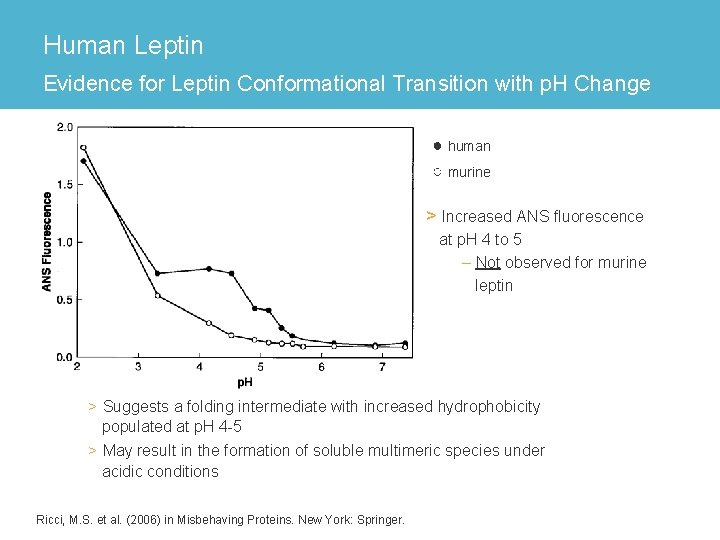
Human Leptin Evidence for Leptin Conformational Transition with p. H Change ● human ○ murine > Increased ANS fluorescence at p. H 4 to 5 – Not observed for murine leptin > Suggests a folding intermediate with increased hydrophobicity populated at p. H 4 -5 > May result in the formation of soluble multimeric species under acidic conditions Ricci, M. S. et al. (2006) in Misbehaving Proteins. New York: Springer.
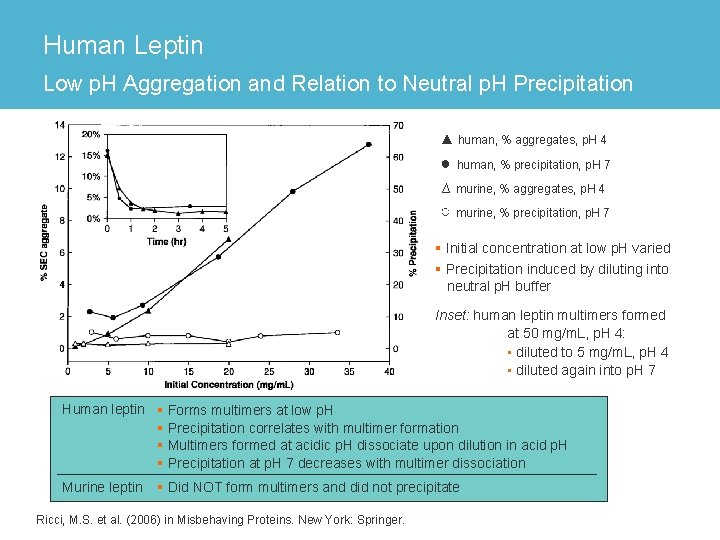
Human Leptin Low p. H Aggregation and Relation to Neutral p. H Precipitation ▲ human, % aggregates, p. H 4 ● human, % precipitation, p. H 7 ∆ murine, % aggregates, p. H 4 ○ murine, % precipitation, p. H 7 § Initial concentration at low p. H varied § Precipitation induced by diluting into neutral p. H buffer Inset: human leptin multimers formed at 50 mg/m. L, p. H 4: • diluted to 5 mg/m. L, p. H 4 • diluted again into p. H 7 Human leptin § Forms multimers at low p. H § Precipitation correlates with multimer formation § Multimers formed at acidic p. H dissociate upon dilution in acid p. H § Precipitation at p. H 7 decreases with multimer dissociation Murine leptin § Did NOT form multimers and did not precipitate Ricci, M. S. et al. (2006) in Misbehaving Proteins. New York: Springer.

Human Leptin Proposed Aggregation Mechanism Increased hydrophobicity at acidic p. H not observed** N Multimers not observed* Murine Leptin I U Iassoc Precipitation not observed* precipitation * Under conditions in which human leptin precipitated, and formed multimers. N: native state I: folding intermediate ** As observed for human leptin in ANS studies. U: unfolded conformer Iassoc: folding intermediate self associated into a soluble multimer Ricci, M. S. et al. (2006) in Misbehaving Proteins. New York: Springer.
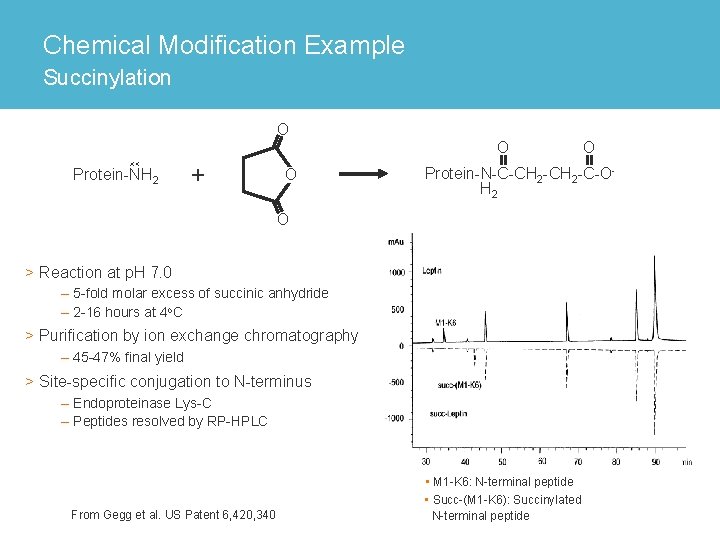
Chemical Modification Example Succinylation O O Protein-NH 2 + O O Protein-N-C-CH 2 -C-OH 2 O > Reaction at p. H 7. 0 – 5 -fold molar excess of succinic anhydride – 2 -16 hours at 4 o. C > Purification by ion exchange chromatography – 45 -47% final yield > Site-specific conjugation to N-terminus – Endoproteinase Lys-C – Peptides resolved by RP-HPLC From Gegg et al. US Patent 6, 420, 340 • M 1 -K 6: N-terminal peptide • Succ-(M 1 -K 6): Succinylated N-terminal peptide
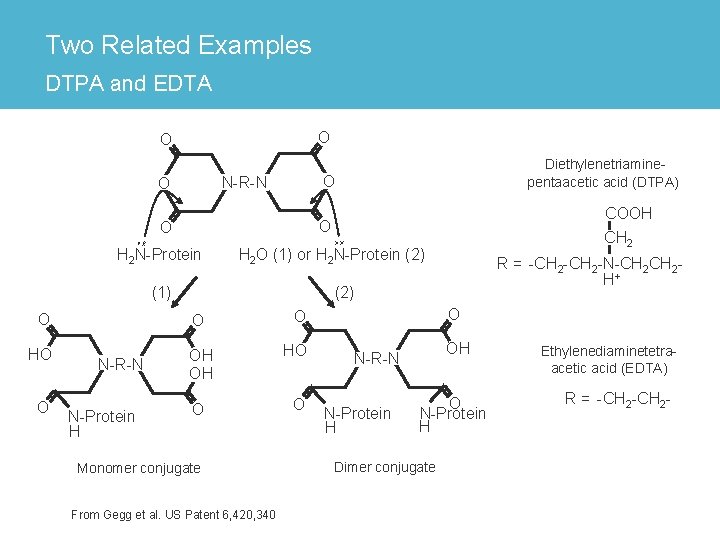
Two Related Examples DTPA and EDTA O O O N-R-N O H 2 N-Protein H 2 O (1) or H 2 N-Protein (2) (1) O O HO OH OH HO O O N-Protein H R = -CH 2 -N-CH 2 H+ (2) O O COOH CH 2 O O N-R-N Diethylenetriaminepentaacetic acid (DTPA) Monomer conjugate From Gegg et al. US Patent 6, 420, 340 O OH N-R-N N-Protein H O N-Protein H Dimer conjugate Ethylenediaminetetraacetic acid (EDTA) R = -CH 2 -
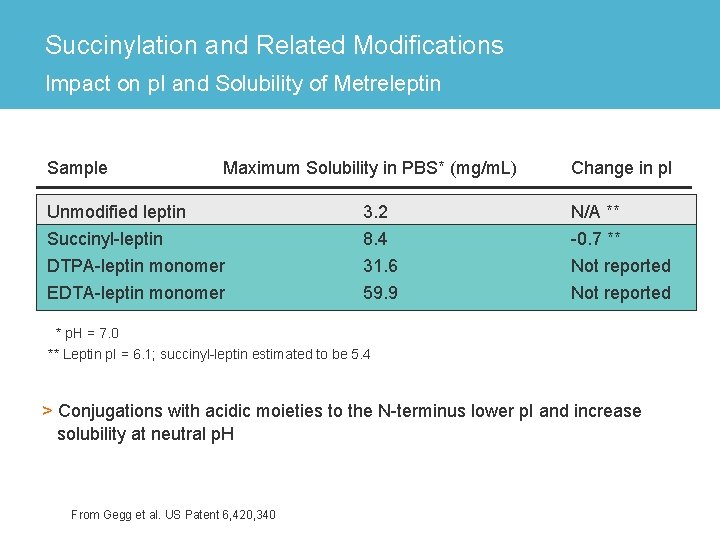
Succinylation and Related Modifications Impact on p. I and Solubility of Metreleptin Sample Maximum Solubility in PBS* (mg/m. L) Change in p. I Unmodified leptin 3. 2 N/A ** Succinyl-leptin DTPA-leptin monomer 8. 4 31. 6 -0. 7 ** Not reported EDTA-leptin monomer 59. 9 Not reported * p. H = 7. 0 ** Leptin p. I = 6. 1; succinyl-leptin estimated to be 5. 4 > Conjugations with acidic moieties to the N-terminus lower p. I and increase solubility at neutral p. H From Gegg et al. US Patent 6, 420, 340
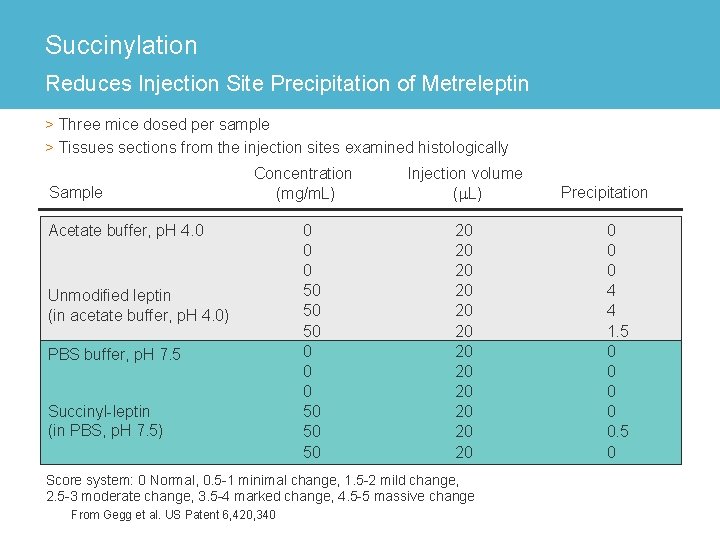
Succinylation Reduces Injection Site Precipitation of Metreleptin > Three mice dosed per sample > Tissues sections from the injection sites examined histologically Sample Concentration (mg/m. L) Acetate buffer, p. H 4. 0 Unmodified leptin (in acetate buffer, p. H 4. 0) PBS buffer, p. H 7. 5 Succinyl-leptin (in PBS, p. H 7. 5) 0 0 0 50 50 50 Injection volume (m. L) 20 20 20 Score system: 0 Normal, 0. 5 -1 minimal change, 1. 5 -2 mild change, 2. 5 -3 moderate change, 3. 5 -4 marked change, 4. 5 -5 massive change From Gegg et al. US Patent 6, 420, 340 Precipitation 0 0 0 4 4 1. 5 0 0 0. 5 0
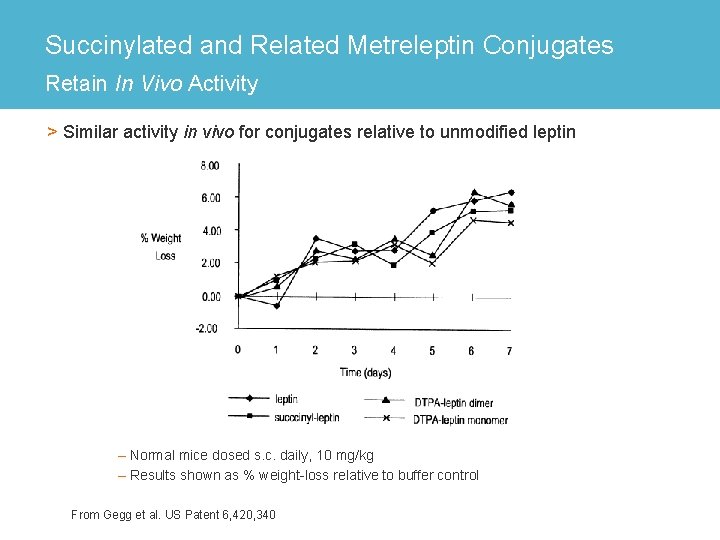
Succinylated and Related Metreleptin Conjugates Retain In Vivo Activity > Similar activity in vivo for conjugates relative to unmodified leptin – Normal mice dosed s. c. daily, 10 mg/kg – Results shown as % weight-loss relative to buffer control From Gegg et al. US Patent 6, 420, 340

Polymer Conjugation Example PEGylation > What is PEGylation? – Covalent attachment of poly(ethylene glycol) (PEG) – Example PEGylation reagent: Methoxy cap CH 3 O-(CH 2 -O)n-CH 2 -X Reactive group > Why PEGylation? – Slow clearance/maintain circulating concentrations/reduce dose frequency – Increase solubility – Reduce aggregation – Reduce proteolysis – Reduce immunogenicity – In several approved products
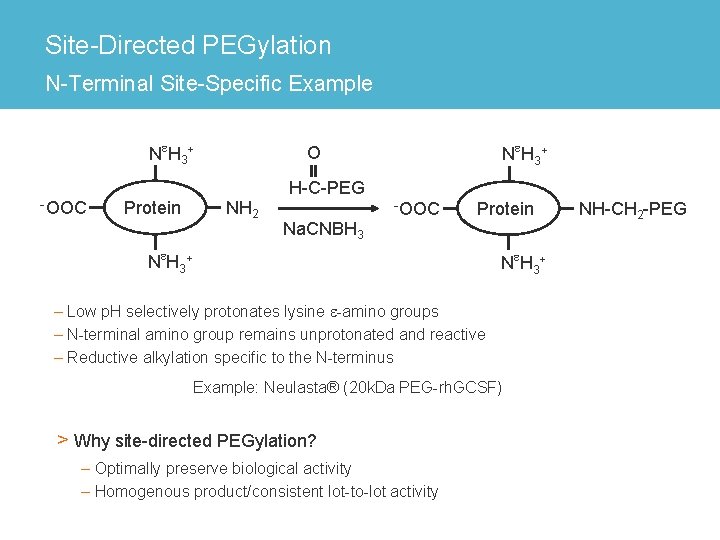
Site-Directed PEGylation N-Terminal Site-Specific Example Ne. H 3+ -OOC Protein Ne. H 3+ O NH 2 H-C-PEG Na. CNBH 3 -OOC Protein Ne. H 3+ – Low p. H selectively protonates lysine e-amino groups – N-terminal amino group remains unprotonated and reactive – Reductive alkylation specific to the N-terminus Example: Neulasta® (20 k. Da PEG-rh. GCSF) > Why site-directed PEGylation? – Optimally preserve biological activity – Homogenous product/consistent lot-to-lot activity NH-CH 2 -PEG
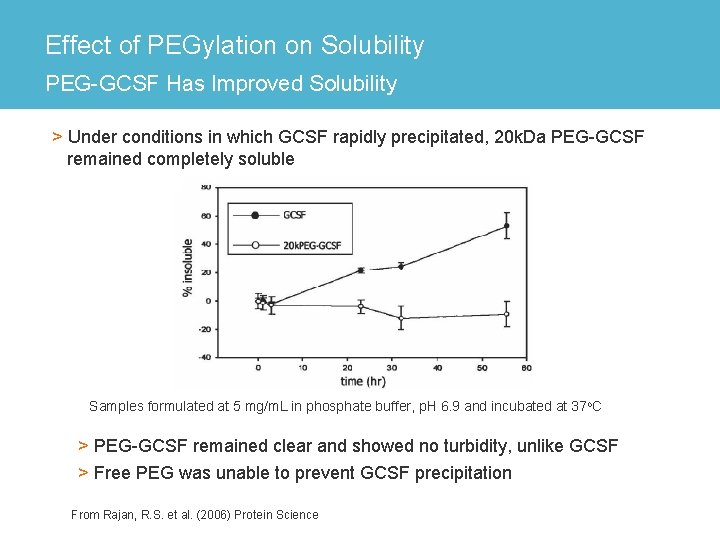
Effect of PEGylation on Solubility PEG-GCSF Has Improved Solubility > Under conditions in which GCSF rapidly precipitated, 20 k. Da PEG-GCSF remained completely soluble Samples formulated at 5 mg/m. L in phosphate buffer, p. H 6. 9 and incubated at 37 o. C > PEG-GCSF remained clear and showed no turbidity, unlike GCSF > Free PEG was unable to prevent GCSF precipitation From Rajan, R. S. et al. (2006) Protein Science
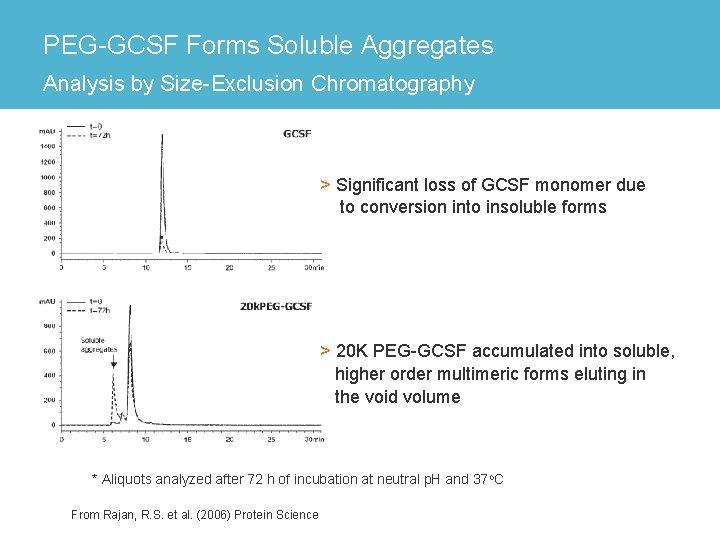
PEG-GCSF Forms Soluble Aggregates Analysis by Size-Exclusion Chromatography > Significant loss of GCSF monomer due to conversion into insoluble forms > 20 K PEG-GCSF accumulated into soluble, higher order multimeric forms eluting in the void volume * Aliquots analyzed after 72 h of incubation at neutral p. H and 37 o. C From Rajan, R. S. et al. (2006) Protein Science

PEGylation and Aggregation Mechanism Findings > PEGylation does not alter the linkages or heterogeneity of the aggregates – Resolubilized GCSF and PEG-GCSF soluble aggregates comparison • Both included a mixture of monomer, dimers, trimers, and higher order multimers • Multimers in both cases were covalent, disulfide-linked • Similar extent of covalent formation > PEGylation does not alter the helix-to-sheet transition that accompanies aggregation – GCSF and PEG-GCSF showed similar starting FTIR spectral profiles as well as temperature-induced conversion to b-sheet – The GCSF precipitate and the PEG-GCSF soluble aggregate showed similar extent of b-sheet content by FTIR analysis > PEGylation confers improved solvation by water molecules – In phase partition studies, GCSF aggregates partitioned to octanol while PEG-GCSF aggregates remained in the aqueous phase From Rajan, R. S. et al. (2006) Protein Science

Aggregation and Drug Development Improving the Drug Compound > Identify potential issues early – Dose level, dose concentration – Solubility at physiological p. H – Manufacturing, shipping and handling > Consider strategy to reduce aggregation – Remove aggregates during manufacture – Formulate to prevent aggregate formation – Modify the compound to reduce/remove aggregation potential > Generally, testing compounds early is preferred – Logistical benefit, test compounds while in vitro and in vivo screens are in process (rather than restarting assays) – Opportunity to solve before Candidate Nomination
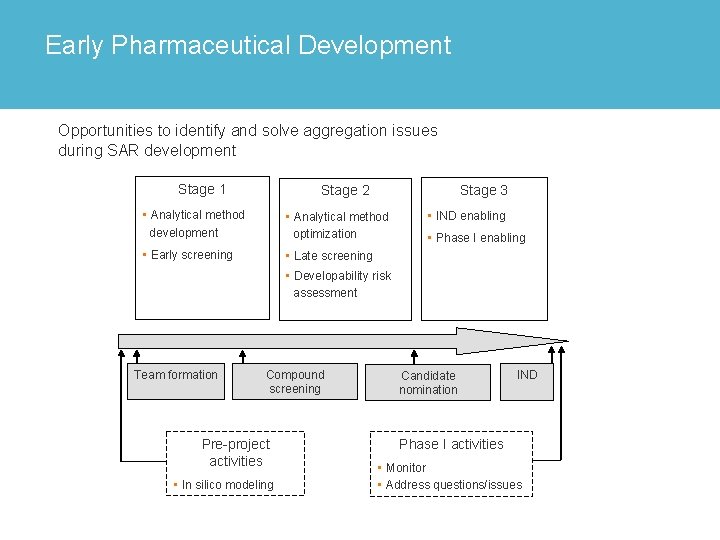
Early Pharmaceutical Development Opportunities to identify and solve aggregation issues during SAR development Stage 1 Stage 3 Stage 2 • Analytical method development • Analytical method optimization • Early screening • Late screening • IND enabling • Phase I enabling • Developability risk assessment Team formation Compound screening Pre-project activities • In silico modeling Candidate nomination IND Phase I activities • Monitor • Address questions/issues

Acknowledgments and References Acknowledgments Pharmaceutical Sciences Steven Ren Derrick Katayama Ellen Padrique Johnny Gonzales Jenny Jin Biology Diane Hargrove Eric Kendall Augustine Cho Krystyna Tatarkiewicz Slave Gedulin Biology, cont’d Pam Smith Christine Villescaz Tina Whisenant Lynn Jodka Kim De. Conzo Julie Hoyt Jenne Pierce Amy Carroll Aung Lwin Informatics Eugene Coats Robert Feinstein Paul Nelson Research Chemistry Odile Levy Ramina Nazarbaghi Lawrence D’Souza John Ahn Bioanalytical Chemistry Swati Gupta Kristine De Dios Liying Jiang References M. S. Ricci et al. (2006) Mutational Approach to Improve Physical Stability of Protein Therapeutics Susceptible to Aggregation. In Misbehaving Proteins (Murphy RM and Tsai AM, ed) pp 331 -350. New York: Springer. Gegg, C. and Kinstler, O. (2002) Chemical modification of proteins to improve biocompatibility and bioactivity. US Patent 6, 420, 340 Rajan, R. S. et al. (2006) Modulation of protein aggregation by polyethylene glycol Conjugation: GCSF as a case study. Protein Science 15: 1063 -1075.
- Slides: 33