Improving access to generic biosimilar medicines in Bulgaria

Improving access to generic & biosimilar medicines in Bulgaria and in the EU Sófia, 14 th November 2013 Adrian van den Hoven Director General EGA – European Generic medicines Associations 1

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 2

The European Generic Medicines and Biosimilar Medicines Association (EGA) is at the heart of Europe’s medicine industry and is essential to EU Public Health. 1 Patients 2 Quality 3 Value 4 Sustainability 5 Partnership

The EU generic medicines industry is highly committed to Europe’s health Generic medicines bring savings of 54% 35€ Bn p. a. in the EU 28 1000 More than generic companies directly employing 150, 000 European citizens Generic medicines account for almost volume One of the most competitive sectors in Europe 21% value 7% of turnover to R&D exporting to more than 100 countries outside the EU 4
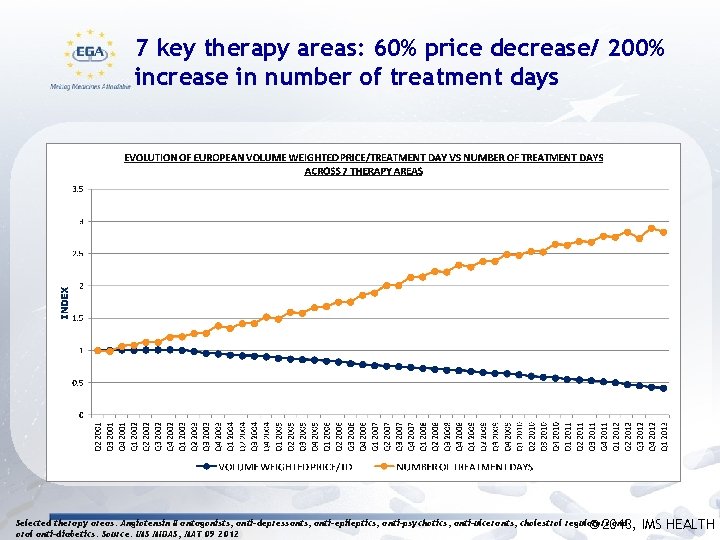
7 key therapy areas: 60% price decrease/ 200% increase in number of treatment days Selected therapy areas: Angiotensin II antagonists, anti-depressants, anti-epileptics, anti-psychotics, anti-ulcerants, cholestrol regulators and © 2013, oral anti-diabetics. Source: IMS MIDAS, MAT 09 2012 IMS HEALTH
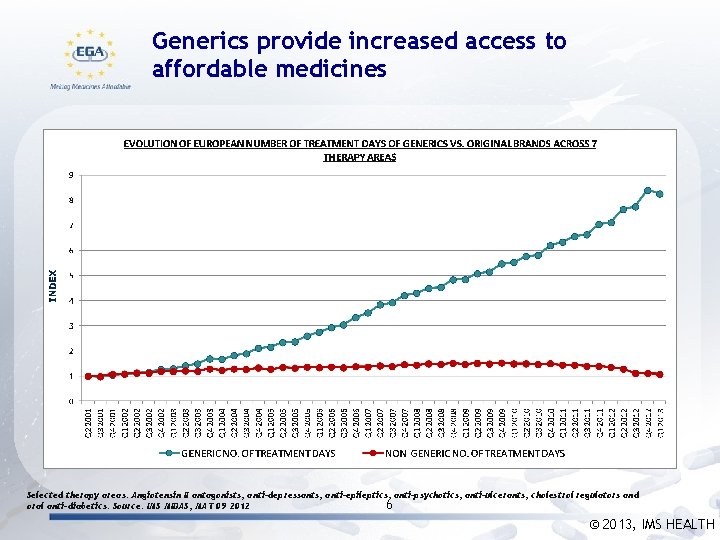
Generics provide increased access to affordable medicines Selected therapy areas: Angiotensin II antagonists, anti-depressants, anti-epileptics, anti-psychotics, anti-ulcerants, cholestrol regulators and oral anti-diabetics. Source: IMS MIDAS, MAT 09 2012 6 © 2013, IMS HEALTH

Diabetes patients treated in Europe Projection for EU 28, 2013 Prevalence: 8. 5%, adult population aged above 15 years Total number of treated patients: 36 million Patients treated by generic medicine: 20 million Generic share: 54% ATC classes: A 10 drugs used in diabetes Source: International Diabetes Federation (IDF), Diabetes Atlas; European Community Health Indicators (ECHI) estimate based on IMS MIDAS Internal 7

Hypertension patients treated in Europe Projection for EU 28, 2013 Prevalence: total hypertension 40% of population aged above 15 years; 19% - treated hypertension, Total number of treated patients: 82 million Patients treated by generic medicine: 48 million Generic share: 59% ATC classes: C 2 antihypertensives Source: Euro Barometer; BMC Public Health - Internal estimate based on IMS MIDAS 8

Generic Medicines generate savings “(…)policies promoting the use of generic medicines are often at the forefront in the attempt to increase the cost-effectiveness of medicinal products”. EC-FIN “Policies to promote greater use of generic medicines are likely to lower pharmaceutical expenditure and enhance efficiency” –WHO Report Source: European Commission and Mrazek & Frank, 2004 in WHO - Health policy responses to the financial crisis in Europe report). 9

Lipitor generics help NHS save £ 1 m per day (in the UK), Pharma Times, 15 May The NHS saved more than £ 350 million in the first 12 months after the patent expiration of Pfizer’s Lipitor last year. (…), now costs just £ 1. 89 – the price tag for Lipitor comes in at £ 13 for the same pack size and dose, according to the British National Formulary. 10

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 11
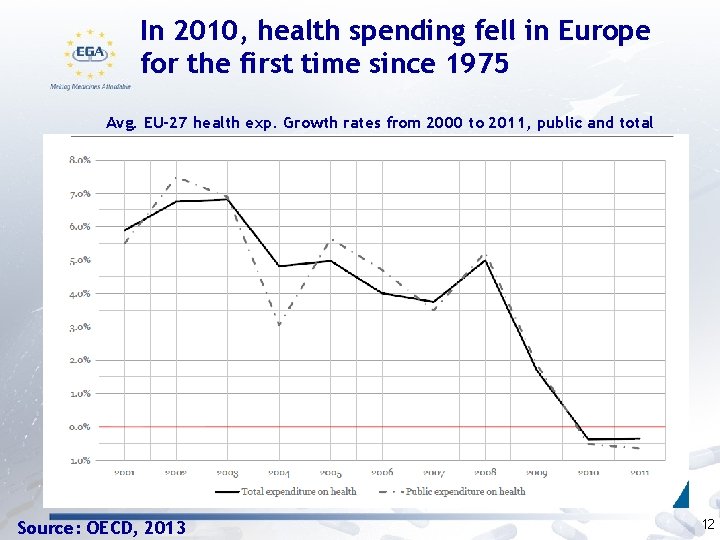
In 2010, health spending fell in Europe for the first time since 1975 Avg. EU-27 health exp. Growth rates from 2000 to 2011, public and total Source: OECD, 2013 12
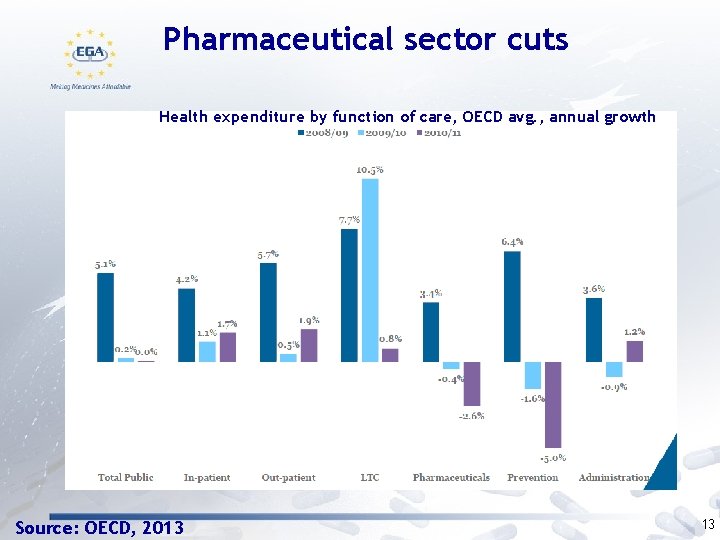
Pharmaceutical sector cuts Health expenditure by function of care, OECD avg. , annual growth Source: OECD, 2013 13
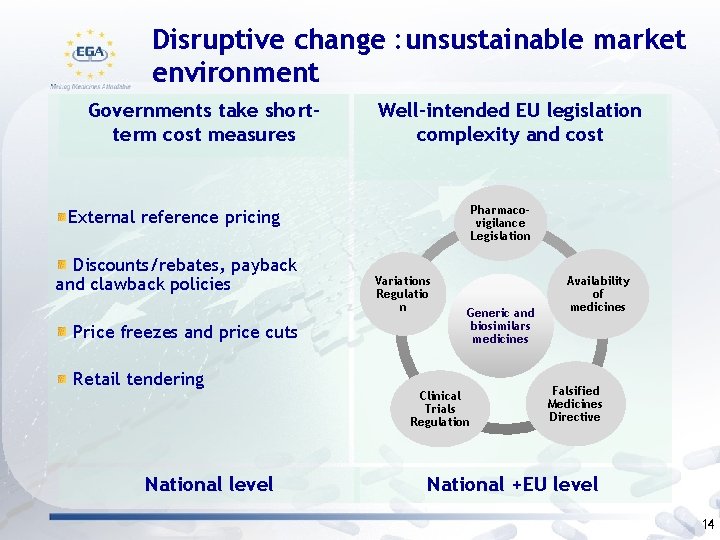
Disruptive change : unsustainable market environment Governments take shortterm cost measures Well-intended EU legislation complexity and cost Pharmacovigilance Legislation External reference pricing Discounts/rebates, payback and clawback policies Price freezes and price cuts Retail tendering National level Variations Regulatio n Generic and biosimilars medicines Clinical Trials Regulation Availability of medicines Falsified Medicines Directive National +EU level 14

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 15
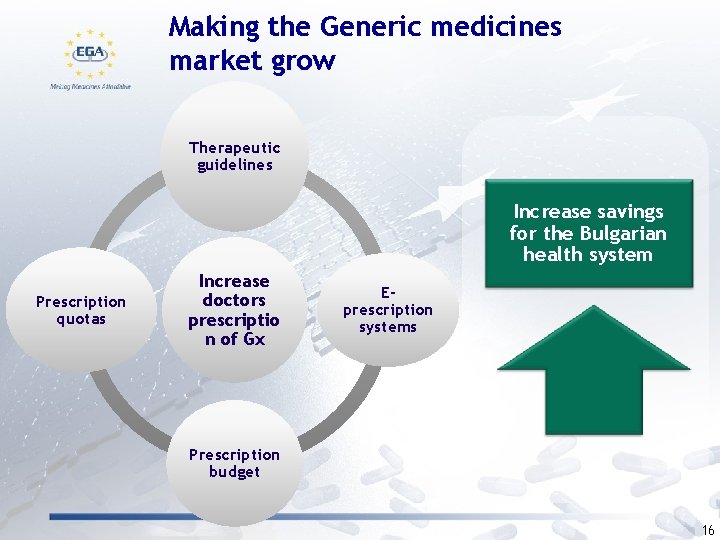
Making the Generic medicines market grow Therapeutic guidelines Increase savings for the Bulgarian health system Prescription quotas Increase doctors prescriptio n of Gx Eprescription systems Prescription budget 16

Making the Generic medicines market grow Incentives for pharmacies to dispense generic medicines Establish remuneration linked to the level of achievement of a substitution goal Remuneration scheme that incentivises the dispensing of the lower price medicines Increase savings for the Bulgarian Health system 17

External Reference Pricing Not for generics Key risks when introducing ERP: 1. 2. 3. 4. 5. Generic medicines prices below profitability level Artificial downward price spiral (procurement and tendering) Price linkage – ERP is applied twice Inability to launch generics in some markets Divergences in updating price lists WHO : ERP not appropriate for generics 18

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 19
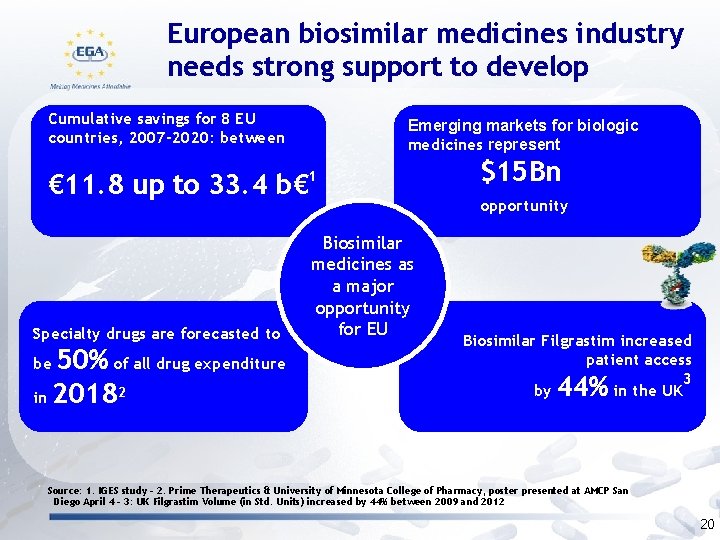
European biosimilar medicines industry needs strong support to develop Cumulative savings for 8 EU countries, 2007 -2020: between Emerging markets for biologic medicines represent € 11. 8 up to 33. 4 b€ 1 Specialty drugs are forecasted to 50% of all drug expenditure in 20182 be Biosimilar medicines as a major opportunity for EU $15 Bn opportunity Biosimilar Filgrastim increased patient access 3 by in the UK 44% Source: 1. IGES study – 2. Prime Therapeutics & University of Minnesota College of Pharmacy, poster presented at AMCP San Diego April 4 - 3: UK Filgrastim Volume (in Std. Units) increased by 44% between 2009 and 2012 20
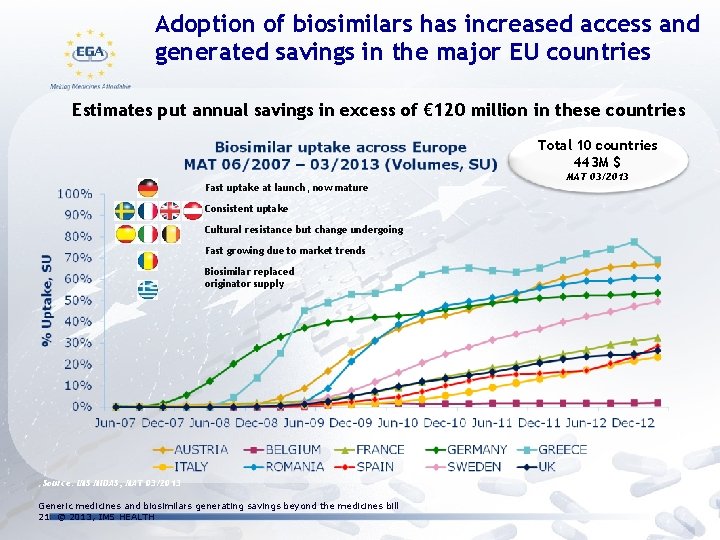
Adoption of biosimilars has increased access and generated savings in the major EU countries Estimates put annual savings in excess of € 120 million in these countries Total 10 countries 443 M $ Fast uptake at launch, now mature Consistent uptake Cultural resistance but change undergoing Fast growing due to market trends Biosimilar replaced originator supply Source: IMS MIDAS, MAT 03/2013 Generic medicines and biosimilars generating savings beyond the medicines bill 21 © 2013, IMS HEALTH MAT 03/2013
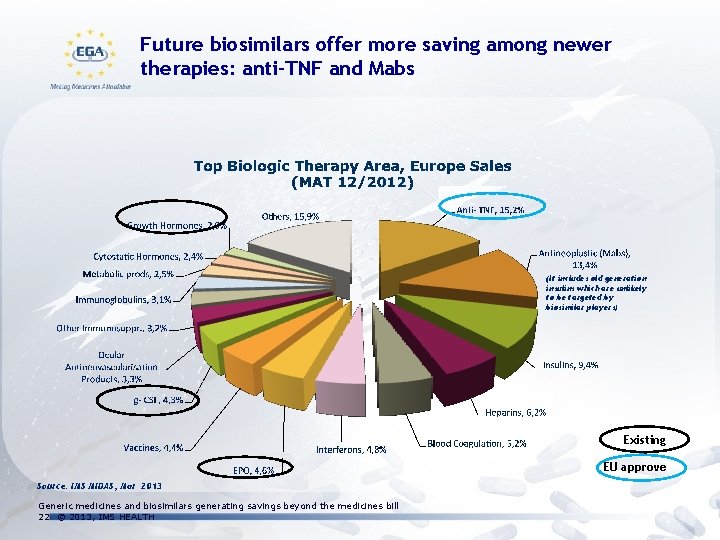
Future biosimilars offer more saving among newer therapies: anti-TNF and Mabs (It includes old generation insulins which are unlikely to be targeted by biosimilar players) Existing EU approved Source: IMS MIDAS, Mar 2013 Generic medicines and biosimilars generating savings beyond the medicines bill 22 © 2013, IMS HEALTH

Biosimilars and the way ahead Information for doctors : Clear, unbiased information on the scientific concept of biosimilars, approval process, safety and efficacy Member States: Guidelines to incentivize physicians to prescribe biosimilars; reimbursement rules for biosimilars uptake Reimbursement: remove hurdles or delays to pricing and reimbursement approval to increase savings and patient access Advanced Manufacturing Rule: allow European based industry to compete early in emerging markets; Bulgaria 10 % of EU manufacturing

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 24
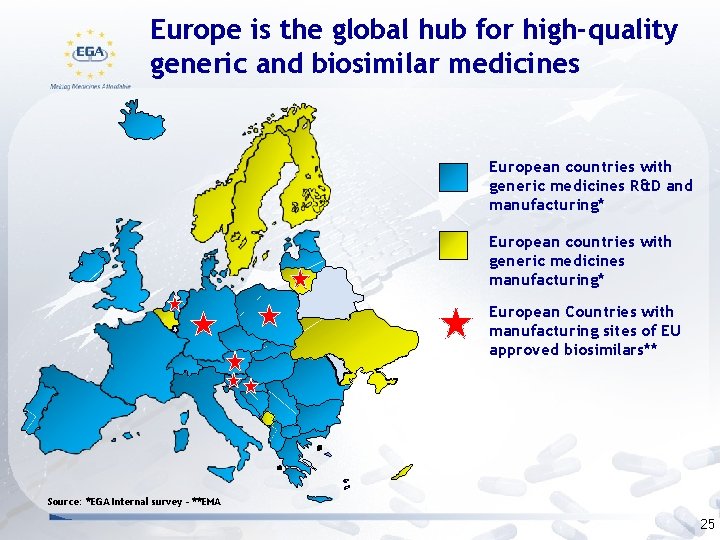
Europe is the global hub for high-quality generic and biosimilar medicines European countries with generic medicines R&D and manufacturing* European countries with generic medicines manufacturing* European Countries with manufacturing sites of EU approved biosimilars** Source: *EGA Internal survey - **EMA 25
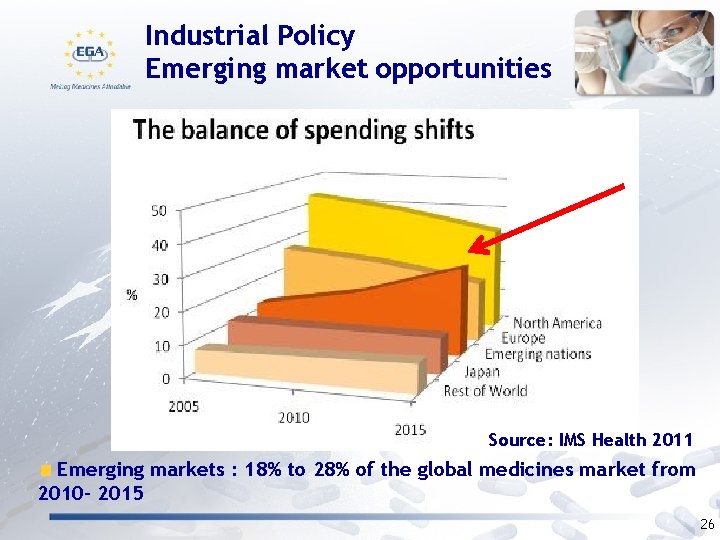
Industrial Policy Emerging market opportunities Source: IMS Health 2011 Emerging markets : 18% to 28% of the global medicines market from 2010 - 2015 26
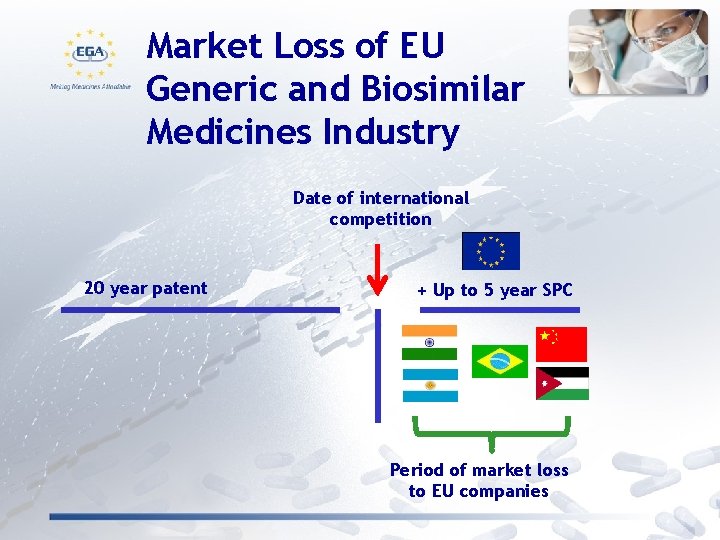
Market Loss of EU Generic and Biosimilar Medicines Industry Date of international competition 20 year patent + Up to 5 year SPC Period of market loss to EU companies

Executive summary Patient access to high-quality medicines The healthcare environment Supporting the Generic medicines market Savings through the use of Biosimilar medicines Boosting Manufacturing Conclusion 28
Stronger collaboration is needed to foster industry and to compete at global level Generate savings Generic and biosmilars Medicines Industry EU and Member States authorities Increase patient access Jobs and Growth 29

Conclusion Medicines provide huge value to patients and healthcare systems Stability of pricing for generic and biosimilar medicines is needed Well thought out regulations that achieve patient safety and reduce complexity are key It is time to support the generic and biosimilar medicines industries 30

благодаря Adrian@egagenerics. com 31
- Slides: 31