IMPORTANT RULE All poisoned patients should be managed


IMPORTANT RULE All poisoned patients should be managed as if they have a potentially life-threatening intoxication, although they appear normal

Initial assessment & stabilization of VFs 2. Definitive care of poisoning cases: 1. a) Measures to identify the toxic agent. I. Preventio. History and circumstantial b) c) d) e) evidence II. Symptoms and signs (including toxidromes) III. Investigations IV. Postmortem picture Decrease further absorption. Antidote therapy (if available). Enhance elimination (excretion) of the toxic substance. Treatment of systemic complications resulting from poisoning 3. A secondary survey for: INFECTION, TRAUMA, METABOLIC DISTURBANCE

STABILIZATION OF THE PATIENT � The initial approach to the poisoned patient should be essentially similar in every case, irrespective of the toxin ingested. � Similar to the initial approach to the trauma patient � Applied as initial approach to all patients in ED irrespective of the mechanism of injury. � This approach can be termed as routine poison management

INITIAL ASSESSMENT & STABILIZATION �The Basic Life Support including ABCDE should be applied before all other considerations. � Many toxic substances can lead to potentially life-threatening changes that should be identified and treated quickly. �Many poisoned patients require only supportive therapy alone.
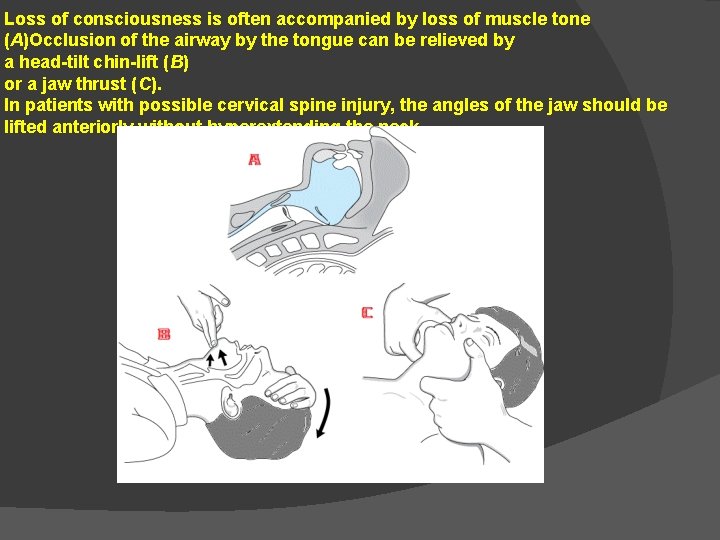
Loss of consciousness is often accompanied by loss of muscle tone (A)Occlusion of the airway by the tongue can be relieved by a head-tilt chin-lift (B) or a jaw thrust (C). In patients with possible cervical spine injury, the angles of the jaw should be lifted anteriorly without hyperextending the neck.

Disability (Neurological): A decreased level of consciousness is the most common serious complication of drug overdose or poisoning. �Coma � C. N. S depressant: barbiturate, benzodiazepine and opiate � Hypoglycemic agent and alcohol. � Hypoxic agent (CO). � Metabolic agent (Salicylate, methanol). � Other as organophosphate. For patients with altered consciousness or respiratory depression of unknown cause give "coma cocktail“: � � Dextrose (Hypoglycemia) Thiamine (Alcohol-related Amnestic Syndromes) Naloxone (Opiates) O 2 (Hypoxia)

Coma Cocktail According to the age of the patient: loss of consciousness of unknown cause or persistence of symptoms, try coma cocktail: 1. 100% oxygen 2. Glucose (Important) 3. Naloxone 4. Thiamine � Coma cocktail is administered for a diagnostic and therapeutic reasons.

DEFINITIVE CARE WITH POISONING

Try to identify the poison � Accurate and complete history � History from sources other than patient (family, friends, pharmacist, & pill bottles at the scene). � Attempt to establish the time and amount of the ingestion

CAREFUL PHYSICAL EXAM � Based on knowledge of drug pharmacology � The vital signs should be monitored accurately. � Level of consciousness pupillary size and reaction to light. � Skin examination and breath odors � Recognizing toxidromes. � Exam should include evaluation for head trauma, focal neurological findings, needle track marks.

INVESTIGATION S 1. TOXICOLOGY SCREENING 2. ESSENTIAL CLINICAL LABORATORY TESTS

Toxicology Screening � Quantitative serum levels of Acetaminophen, Salicylates, Digoxin, Iron, Lithium, Methanol, Theophylline, Phenobarb, Ethylene glycol may influence therapy. �Toxico screening provides direct evidence of ingestion, but must not affect initial management (first 6 -8 hrs) and should not await results. �The management must depend mainly on the clinical status and available lab studies

TOXICOLOGY SCREENING Comprehensive blood and urine screening is of little practical value in the initial care of the poisoned patient. On the other hand, specific toxicologic analyses and quantitative levels of certain drugs may be extremely helpful. Urine and gastric specimens are the best samples for broad qualitative screening. Blood samples should be saved for possible quantitative testing, but blood is not a good specimen for screening for many common drugs, including psychotropic agents,

LIMITATIONS OF TOXICOLOGY 1. SCREENS Comprehensive toxicology screens may look specifically for only 40– 50 drugs out of more than 10, 000 possible drugs or toxins (or 6 million chemicals). ○ However, these 40– 50 drugs account for more than 80% of overdoses. 2. The time factor for reading toxicology screening is very long. 3. If the initial urine screen is done too soon after ingestion. The drugs found on the screen may not be responsible for the symptoms seen, especially if the drugs are not quantitated.

1. 2. 3. 4. 5. 6. 7. Investigations CBC LFT RFT Electrolytes, CPK Blood Sugar Arterial Blood Gas � CO, Met. Hgb, Cyanide toxicity � Respiratory or metabolic acidosis Example (Salicylates) Urinalysis � Ferric chloride test for salicylates � Ketones ( salicylates, ketoacidosis ) � Calcium oxalate crystals (ethylene glycol and oxalic acid )

Osmolal Gap OG = (measured serum osmolality) – (calculated osmlality) � Calculated osmolality: [2(Na+) + (Glucose/18) + (BUN/2. 8)]. � Normal Serum osmolality is 285 -300 m. Osm/kg. � Normal osmolal gap is 8 -12 m. Osm/kg. � Elevation due to presence of unmeasured, lowmolecular weight molecules that are osmotically active: � Methanol � Ethylene glycol � Diuretics, such as glycerol, manitol, sorbitol � Isopropanol � Ethanol

Anion Gap � AG is an indirect measure of phosphates, sulfates, & organic acids. Na + unmeasured cations = (Cl + HCO 3) + unmeasured anions Na – (Cl + HCO 3) = unmeasured anions – unmeasured cations Anion gap = (Na ) – (Cl + HCO 3) � Normal anion gap is 12 -16 m. Eq/L.

a. An elevated anion-gap acidosis is usually caused by an accumulation of lactic acid but may also be caused by other unmeasured acid anions such as formate (eg, methanol poisoning) or oxalate (eg, ethylene glycol poisoning). b. In any patient with an elevated anion gap, also check the osmolar gap; a combination of elevated anion and osmolar gaps suggests poisoning by methanol or ethylene glycol. Note: Combined osmolar- and anion gap elevation may also be seen with severe alcoholic ketoacidosis and diabetic ketoacidosis. c. A narrow anion gap may occur with an overdose by lithium, bromide, or nitrate, all of which can increase the serum chloride level measured by some laboratory instruments.

�Causes of high anion gap metabolic acidosis: excessive acid production or with addition of exogenous acids: �Carbon monoxide, Cyanide. �Alcoholic ketoacidosis �Toluene �Methanol �Uremia �Diabetic ketoacidosis �Paraldehyde, Phenformin �Iron, Isoniazid �Lactic acidosis �Ethylene glycol �Salicylates

Causes of low anion gap with acidosis Acetazolamide � Amiloride � Ammonium chloride � Amphotericin B � Bromide � Iodide � Lithium � Polymyxin B � Spironolactone � Toluene �

PREVENTION OF FURTHER ABSORPTION A- Dermal Exposure Remove all clothing. � Washing skin gently with soap and water for at least 30 minutes. � Forceful washing may damage skin and promotes further absorption. � Protection of medical staff B- Eye Exposure � Washing conjunctiva with running water or normal saline for 20 minutes. Solid corrosives should be removed by forceps. C- GIT Exposure � Gastrointestinal Decontamination Induction of emesis Gastric lavage Activated Charcoal Activated charcoal Cathartics Whole bowel irrigation

INDUCTION OF EMESIS Syrup Ipecac � The only safe method for induction of vomiting. Poison Treatment in the Home. Pediatrics 2003; 112; 1182 � From the root of Cephalus Ipecachuana: emetine & cephaline � Early phase: within 30 minutes by direct GIT stimulation. � Late phase: after 30 minutes through action on CTZ. � Dose: Adults: 30 ml; 15 ml for children more than 2 Yrs, 5 -10 ml for children 6 months to 2 yrs. Not more than 2 doses � If emesis does not occur within 30 minutes, move around the patient, if no emesis a second dose can be given. � Syrup of ipecac should no longer be used routinely as a poison treatment intervention in the home.

INDUCTION OF EMESIS Contraindications 1. Convulsions. 2. Corrosives. 3. Hydrocarbons. 4. Sharp objects (e. g. needles). 5. Coma 6. Decreased gag reflex. 7. Severe CVS disease or respiratory distress or emphysema. 8. Recent surgical intervention. 9. Hemorrhagic tendencies (varices, active peptic ulcer, thrombocytopenia). 10. Previous significant vomiting (spontaneously). 11. Less than 6 m of age (not well developed gag reflex).

GASTRIC LAVAGE used in hospitals when emesis was failed or there was contraindication for it. � Gastric lavage is effective in the first 4 -6 hrs after ingestion. � Technique: An assistant with suction machine should be available. 2. Dentures, mucous, vomitus should be removed from patient's mouth. 3. Proper tube size to be selected according to the patient age. 1.

Complications: 1. 2. 3. 4. 5. 6. 7. Bradycardia, especially, in cases of organophosphate or digitalis toxicity. Laryngospasm and cyanosis. Vomiting & Aspiration pneumonia. Stress reaction that may cause hypertension, and or tachycardia. Hyponatremia, if tape water is used in children. Mechanical gut injury. Faulty introduction of the tube in the trachea Contraindications: � Absolute contraindications: 1. 2. 3. Corrosives Froth producing substances as shampoo or liquid soap. Oesophageal varices or peptic ulcer 1. 2. 3. coma. Convulsions. Petroleum distillates. � Relative contraindications

ACTIVATED CHARCOAL � Adsorption of a wide variety of drugs and chemicals. � It is not digested; it stays inside the GI tract and eliminates the toxin when the person has a bowel movement. � Adult dose is 1 gm/kg.

Substances not adsosorbed by AC 1. 2. 3. 4. 5. 6. 7. 8. 9. Corrosives Alcohols Cyanide Oils Glycols Metals (Iron, Lithium, Lead, Mercury, . . ) Petroleum distillates Sodium chloride Sodium hypochlorite bleach

ACTIVATED CHARCOAL Contraindications 1) Coma 2) Intestinal obstruction. 3) Corrosives 4) If an oral antidote is given 5) Hydrocarbons

CATHARTICS (laxatives) These are substances that enhance the passage of material through GIT and decrease the time of contact between the poison and the absorptive surfaces of the stomach and intestine. a) Osmotic cathartics: increase osmotic pressure in the lumen, as Mg sulfate. b) Irritant cathartics: act by increasing motility, such as caster oil. Contraindications: � � GIT hemorrhage. Recent bowel surgery. Intestinal obstruction. Renal failure for magnesium salts.

WHOLE BOWEL The. IRRIGATION goal of WBI is to clean GIT from nonabsorbed ingested toxins. Polyethylene glycol electrolyte solutions are used, should be administered by gravity. Indications � � � Ingestion of a toxin that is known to be poorly adsorbed by A charcoal. Ingestion of massive amounts of drugs / impractical AC Ingestion of sustained-release or enteric-coated preparations (e. g. aspirin) Removal of ingested packets of illegal drugs (body packers). Ingestion of large amount of drugs that may form concretions or bezoars Treatment of suspected drug concretion � � Continual rise in measurable toxin concentrations despite charcoal administration). Drugs that may form concretions: Salicylates, Barbiturates, Carbamazepine, Enteric coated or sustained release tablets, Meprobamate.

ANTIDOTES

DEFINITION � according to � International Programme on Chemical Safety � United Nations Environmental Programme � WHO A THERAPEUTIC SUBSTANCE USED TO COUNTERACT THE TOXIC ACTION(S) OF A SPECIFIED XENOBIOTIC

ANTIDOTES Classification According to mode of action 1 - Physical 2 - Chemical 3 - physiological According to mechanism of action 1 - Interacts with the poison to form a nontoxic complex that can be excreted e. g. chelators 2 - Accelerate the detoxification of the poison: N-acetylcysteine, thiosulfate. 3 - Decrease the rate of conversion of the poison into its toxic metabolites: Ethanol, Fomepizole. 4 - Compete the poison for certain receptors: Naloxone. 5 - Block the receptors through which the toxic effects of the poison are mediated: atropine 6 - Bypass the effect of the poison: O 2 treatment in CO and cyanide toxicity. 7 - Antibodies to the poison: digiband, antivenoms.

1 - Physical Antidotes Agents used to interfere with poisons through physical properties, not change their nature a) Adsorbing: the main example is activated charcoal. b) Coating: a mixture of egg & milk makes a coat over the mucosa. It can be effective in corrosives, but not in cases of organophosphate. It decreases efficacy of AC. c) Dissolving: 10% alcohol or glycerine for carbolic acid.

2 - Chemical Antidotes a) Oxidizing: Amyl Nitrite is used in cyanide toxicity. b) Reducing: � Methylene Blue: it is used with Met. Hb producers: nitrates, nitrites, phenacetin, chlorates, sulfonamides. � Vitamin C: used for drugs causing Met-Hb. c) Precipitating: � Starch: it makes blue precipitate with iodine. 50 gm in glass water. � Egg albumin, gelatin, skimmed milk: in mercury but should be rapidly washed as the precipitate dissolves in excess albumin.

3 - Physiological (Pharmacological) Antidotes a) Antagonism 1 - Competitive Antagonists Naloxone: Naltrexone: Opiate dependence; longer action with affinity for mu receptors. Flumazenil: antagonist for benzodiazepines Atropine: organophosphate, carbamate, and other parasympathomimetic (pilocarpine, muscarine). It is used as antidote to correct bradycardia caused by morphine, digitalis, aconitine, β-blockers, and calcium channel blockers. .

2) Non-Competitive Antagonism Anticonvulsants: for treatment of convulsants. Calcium gluconate: used for: a) Calcium channel blockers especially verapamil. b) Black widow spider bite. c) Lead colic d) Oxalic acid. Pralidoxime (2 -PAM): Ch. E activator by breaking the alkylphosphate-Ch. E bond. It is used in organophosphate toxicity to form an oxime-phosphate complex and leaves the Ch. E enzyme activated. Contraindicated in carbamate toxicity because of short duration of action. Diacetyl Monoxime (DAM): same action as PAM with more BBB penetration and reactivation of Ch. E in CNS. Physostigmine: counteract the anticholinergic effects such as in severe atropine or TCA toxicities.

b) Chelating Agents: Unite metallic poisons to form soluble, nonionizable, less toxic, and easily excreted chelates. 1 - Dimercaprol (BAL) � prototype chelator (bisulfide molecule) � removes intracellular and extracellular lead � lipid-soluble drug and must be administered IM only � It has the typical sulfide odor and patients often complain of the taste � It is the first chelator used in encephalopathic individuals. Rapidly crosses the BBB. Adverse effects fever, pain at the injection site, nausea, vomiting, headache, and sterile abscess formation. It is recommended to alkalinize urine as the chelate may dissociate in acidic urine. Indications: Lead, Arsenic, inorganic Mercury, Copper, Gold. Contraindications: a) Liver failure as metallic chelate is excreted mainly through the bile. b) G 6 PD deficiency. c) Transtoxicity or concurrent iron supply. d) Organic Mercury; risk of neurotoxicity.

2 - Calcium disodium edentate (Ca Na 2 EDTA) Unites with the metal which takes place of its calcium. � It is water soluble and may be used IM or IV � It allows extracellular lead to be renally eliminated � In cases of lead encephalopathy, BAL should be given first to avoid redistribution of lead mobilized by Ca. Na 2 EDTA to CNS. � � 3 - D-Penicillamine: Hydrolysis product of penicillin approved for the treatment of Wilson disease and cystinosis. Effective orally and has few adverse effects. � a) It can chelate lead even in low blood levels. � b) It is effective in children with blood levels 20 -40 µg/100 ml.

4 - Succimer (Di-Mercapto Succinic Acid; DMSA) The best for lead chelation in children with a lead level higher than 45 µg/d. L. An effective oral chelator that approaches the effect of both Ca. Na 2 EDTA and BAL. 5 - Dimerval (DMPS) Has become antidote for most heavy metal intoxications. In the oral form and in a water-based parenteral form. 6 - Cobalt edetate (Kelocyanor) Cobalt compounds directly chelate cyanide, thus reducing its toxicity, bypassing the formation of a toxic (methemoglobin) intermediate. Kelocyanor is thought to work faster than any of the Met. Hb-formers.

N-Acetyl Cysteine (NAC): Antidote in acetaminiophen toxicity: 1 -Precursor for glutathione. NAC is converted to cysteine, which can replenish glutathione stores. 2 - NAC also directly detoxifies acetaminophen toxic metabolite to nontoxic metabolites. 3 -NAC can provide a substrate for sulfation a) Oral NAC: It is effective in preventing hepatotoxicity regardless of the initial acetaminophen level if it is started within 8 hrs of ingestion. b) Intravenous NAC: IV administration of NAC is recommended for selected patients, including those with GIT bleeding or obstruction, potential fetal toxicity, or an inability to tolerate oral NAC.

d) Antibodies (Immunology-based Antidotes) 1 - Digoxin Specific Antibody Fragment (FAB fragment Digiband): Life-threatening arrhythmias Initial potassium level >5 m. Eq/l. Digoxin SL >10 ng/m. L at 6 -8 h postingestion Digoxin SL >15 ng/m. L in an acute ingestion Ingestion >10 mg in healthy adults or > 4 mg in childre 2 - Polyvalent Snake Antivenom: 3 - Scorpion Antivenom: Indications include: a) All children and old patients presenting with any of systemic manifestations. b) Patients with CV disease. 4 - Antibotulism Serum:

4) Other Antidotes a) Vitamins Vitamin K: a) Agents causing hypoprothrombonemia: organic arsenic, salicylates, coumarin. b) Vitamin A toxicity: inhibits flora, antagonism of hepatic action of vit K. Vitamin B 12 (Hydroxycobalamin): contains cobalt ion, able to bind to cyanide with greater affinity than cytochrome oxidase to form nontoxic cyanocobalamin that is excreted in urine. Glucagon: is used in β-blocker poisoning to stimulate the βadrenergic nerves on a receptor different from that occupied by the β-blocker.

Cyanide Antidote Kit Step I Amyl nitrite & Sodium nitrite: induce methemoglobinemia so that cyanide, with high affinity for ferric iron (Fe 3+), may attach to it to form cyanomethemoglobin, rather than to the iron of the cytochrome, thus restoring or allowing cellular respiration � Step II Sodium Thiosulfate: detoxification of the formed cyanomethemoglobin compound so that it can be excreted. The presence of thiosulfate in the blood allows rhodanese to detoxify Cyano-Met. Hb by catalyzing the formation of thiocyanate, which is nontoxic and rapidly excreted in the urine. �

ENHANCEMEN T OF EXCRETION OF ABSORBED POISONS

Forced Diuresis It is a simple method for some poisons. It is effect is increase with manipulation of urine p. H. It is efficient only in poisons with the following properties: � Substances excreted mainly by kidneys. � Substances with low volume distribution. � Substances with low protein binding. Types: 1 - Fluid Diuresis 2 - Osmotic Diuresis: mannitol 10%, which is excreted by renal tubules leading to increases in its osmotic pressure

Manipulation of Urine p. H This method acts depending on the extent of ionization (p. Ka) a) Forced alkaline diuresis b) Acid diuresis It is uncommonly used method for certain substances such as amphetamine. It is a dangerous method because of the risk of myoglobin precipitation in renal tubules.

Dialysis by allowing toxic substances to pass through semipermeable membrane depending on the concentration gradient. It is beneficial when renal function is impaired. Dialyzable substances for good results must have: A) Low volume distribution. B) Low molecular weight. C) Low protein binding. Examples for dialyzable substances: alcohols, barbiturates, salicylates. Examples for nondialyzable substances: opiates, atropine, antidpressants.

a) Peritoneal dialysis Acts by considering peritoneum as semipermeable membrane. Complications: intra-abdominal bleeding, perforation of abdominal organs, peritonitis, dehydration or overhydration. Contraindications: pregnancy, abdominal hernia, or respiratory distress.

b) Hemodialysis The semipermeable membrane is a cellulose bag (artificial kidney). Indicated when the condition is deteriorating despite proper treatment, or in toxicities with potentially lethal blood levels. Complications: Hypotension. Bleeding tendency (due to heparin). Electrolyte imbalance. Cross infections. Muscle cramps. Air embolism.

Hemoperfusion Using equipment and vascular access similar to that for hemodialysis, the blood is pumped directly through a column containing an adsorbent material (either charcoal or Amberlite resin). Systemic anticoagulation is required, often in higher doses than for hemodialysis, and thrombocytopenia is a common complication. 1. Because the drug or toxin is in direct contact with the adsorbent material, drug size, water solubility, and protein binding are less important limiting factors. 2. For most drugs, hemoperfusion can achieve greater clearance rates than hemodialysis. For example, the hemodialysis clearance for phenobarbital is 60– 80 m. L/min, whereas the hemoperfusion clearance is 200– 300 m. L/min.

Contraindications: Patients with coagulopathy. Patients with uncorrected hypotension. Complications: Thrombocytopenia. Hypocalcemia. Hypoglycemia. Hypotension. Adsorption of therapeutic drugs.
- Slides: 54