Importance of Ancillary Supplies for Subcutaneous Immunoglobulin Infusion

Importance of Ancillary Supplies for Subcutaneous Immunoglobulin Infusion: Management of the Local Infusion Site Diane Ochoa, 1* Christine Curtis, 2 Carla Duff, 3 Patty Riley, 4 Elyse Murphy, 4 Annette Zampelli 4 1 Dallas Allergy and Immunology, Dallas, TX; 2 Cook Children’s Medical Center−Infectious Disease Clinic, Fort Worth, TX; 3 University of South Florida, Tampa, FL; 4 CSL Behring, LLC, King of Prussia, PA The International Nursing Group for Immunodeficiencies October 3 -6, 2012, Florence, Italy *Current affiliation is Maxim Healthcare, Dallas, TX

Disclosures and Acknowledgments • DO, CC, and CD are nurse consultants for CSL Behring. EM, PR, and AZ are employees of CSL Behring. • This presentation was supported by CSL Behring, LLC. • Medical writing and editorial support was provided by Daniel Mc. Callus, Ph. D, of Complete Publication Solutions, LLC, and was funded by CSL Behring, LLC.

Ancillary Supplies Use During SCIG Administration • Subcutaneous immunoglobulin (SCIG) – An effective treatment for patients with primary immunodeficiency disease (PIDD) • Ancillary supplies for SCIG therapy – May contribute to the development of issues at the local infusion site – Adjustment may reduce the incidence and severity of infusion related issues SCIG administration to the abdomen

Ancillary Supplies Used During SCIG Administration Ancillary supplies for SCIG therapy – Disposable • Needle sets • Tubing • Antiseptic preparation • Post-infusion dressing • Tape – Non-disposable • Roller/cassette pump • Syringe driver pump
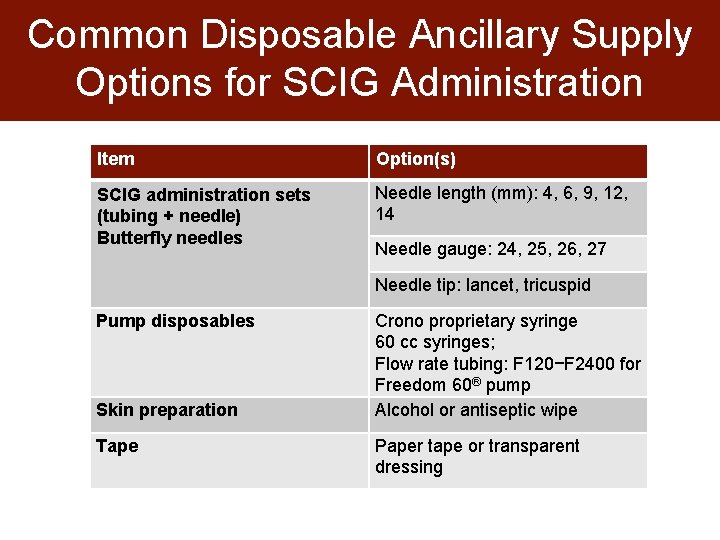
Common Disposable Ancillary Supply Options for SCIG Administration Item Option(s) SCIG administration sets (tubing + needle) Butterfly needles Needle length (mm): 4, 6, 9, 12, 14 Needle gauge: 24, 25, 26, 27 Needle tip: lancet, tricuspid Pump disposables Skin preparation Tape Crono proprietary syringe 60 cc syringes; Flow rate tubing: F 120−F 2400 for Freedom 60® pump Alcohol or antiseptic wipe Paper tape or transparent dressing
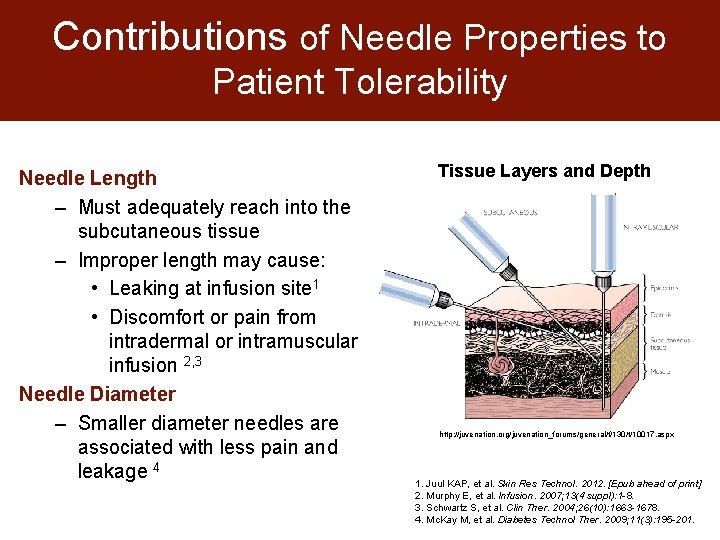
Contributions of Needle Properties to Patient Tolerability Needle Length – Must adequately reach into the subcutaneous tissue – Improper length may cause: • Leaking at infusion site 1 • Discomfort or pain from intradermal or intramuscular infusion 2, 3 Needle Diameter – Smaller diameter needles are associated with less pain and leakage 4 Tissue Layers and Depth http: //juvenation. org/juvenation_forums/general/f/130/t/10017. aspx 1. Juul KAP, et al. Skin Res Technol. 2012. [Epub ahead of print] 2. Murphy E, et al. Infusion. 2007; 13(4 suppl): 1 -8. 3. Schwartz S, et al. Clin Ther. 2004; 26(10): 1663 -1678. 4. Mc. Kay M, et al. Diabetes Technol Ther. 2009; 11(3): 195 -201.

Impact of Needle Properties on Patient Tolerability Puncture in Simulated Tricuspid Skin 1 Needle Type – Type of skin puncture may affect the development of infusion related issues – Lancet needles result in more coring, bleeding, and tissue necrosis than tricuspid needles 1 Lancet 1. Selafon A and Baker PM. Presented at National Home Infusion Association Annual Conference, Phoenix, AZ, April 23 -26, 2012.

Impact of Flawed Needles on Patient Tolerability Needles “Out of the Box” Damaged During Manufacturing 3 Needles may be made inconsistently 1 – Tip damage may occur during manufacturing or handling Flawed/damaged/blunted needles – May cause pain – Result in inefficient or improper delivery of product 200 X Needle tip damage (~ 10 microns) – Associated with patients’ perception of pain 2 Patients or caregivers – Should thoroughly inspect needles 1. Parker RK and White PF. Anesth Analg. 1997; 85(5): 1101 -1104. – Use only if undamaged 2. Kinast P. Med Device Technol. 1992; 3(6): 46 -49. 3. Selafon A and Baker PM. Presented at National Home Infusion Association Annual Conference, Phoenix, AZ, April 23 -26, 2012.

Contributions of Other Ancillary Supplies to Patient Tolerability • Tubing – Size is a determinant of infusion rate, which may influence tolerability • Antiseptic preparation and post-infusion dressing – May affect skin sensitivity • Tape – May lead to local irritation at the site of application

Case Studies Demonstrating the Effects of Ancillary Supplies on Local Tolerability Case Study 1 Patient Description • 10 -year-old • 18 -kg female Diagnosis Common variable immuno -deficiency disease Initial SCIG and Technical/ Ancillary Clinical Supplies Complaints Lyophilized IVIG • Redness, reconstituted to swelling, and 16% given SC leaking at infusion sites • 3 g • 20 m. L • Resolved by • 2 sites (inner the evening of thigh) infusions • 6 -mm needle • Weekly Treatment Adjustment(s) Changed from a 6 -mm to a 9 -mm needle Outcome • Decreased swelling and leaking from sites • Patient has tolerated SCIG infusions well • Switched to 20% SCIG after product available

Case Studies Demonstrating the Effects of Ancillary Supplies on Local Tolerability Case Study 2 Patient Description • 5 -year-old • 18 -kg Diagnosis Hypogammaglobulinemia Initial SCIG and Technical/ Ancillary Clinical Supplies Complaints 20% SCIG • Tegaderm tape not sticking to • 3 g skin • 15 m. L • 2 sites (thigh) • SCIG needle • 26 -gauge, 12 - displaced during mm needle infusion • Weekly • Use of thigh sites limited mobility of child Treatment Adjustment(s) • Changed from Tegaderm tape to silk tape attached in “x” pattern over needle • Used tincture of benzoin on edges of silk tape to secure • Switched to abdomen sites Outcome Improvement and resolution of all issues reported

Case Studies Demonstrating the Effects of Ancillary Supplies on Local Tolerability Case Study 3 Patient Description • 9 -year-old • 27 -kg male • Naive to SCIG treatment Diagnosis • X-linked agammaglobulinemia Initial SCIG and Technical/ Ancillary Clinical Supplies Complaints 20% SCIG • Swelling • 5 g • 24 m. L • 3 sites (abdomen) • 6 -mm needle • F 180 -rate tubing (4 m. L/hr/site) • Weekly • Redness • Severe discomfort • Leakage at site Treatment Adjustment(s) • Reduced infusion rate by changing to F 120 -rate tubing (2. 32 m. L/hr/site) • Reduced site volume by adding a fourth site; leaking at site still occurred • Secondary adjustment of a 6 -mm to a 9 -mm needle Outcome Improvement and resolution of issues reported

Case Studies Demonstrating the Effects of Ancillary Supplies on Local Tolerability Case Study 4 Patient Description • 25 -year-old • 67 -kg female • Avid runner Diagnosis Common variable immuno -deficiency disease Initial SCIG and Technical/ Ancillary Clinical Treatment Supplies Complaints Adjustment(s) 10% SCIG • Severe burning • Changed from a 6 -mm to a 9 • 10 g • Edema mm needle; no • 100 m. L improvement • 4 sites • Pain lasting 2 -3 • 27 -gauge, 6 days • Changed to mm needle 16% SCIG • Biweekly • Interfered with product once running available Outcome • Improvement and resolution of issues reported • Able to run same day as infusion • Switched to 20% SCIG after product available; well tolerated

Treatment Algorithm for Patients With Technical or Clinical Complaints During or Following Initial SCIG Regimen • Should be followed for patients who experience tolerability problems beyond the mild, transient effects that may occur after SCIG

Conclusions • Careful attention to technical or clinical complaints at the local infusion site warrants reassessment of infusion regimen including supplies. • Case studies demonstrate that adjustment of, or changes to, ancillary supplies may decrease the occurrence and/or severity of infusion related issues. • Ancillary supplies should be adjusted before changing the SCIG product. • Alterations in the choice of ancillary supplies can: o Improve the patient experience with SCIG administration o Positively impact patient quality of life and medication adherence
- Slides: 15