IMPLICATIONS OF UKPDS GSK Advisory Board 24 May

IMPLICATIONS OF UKPDS GSK Advisory Board 24 May 2003 Dr. J. R. Conway
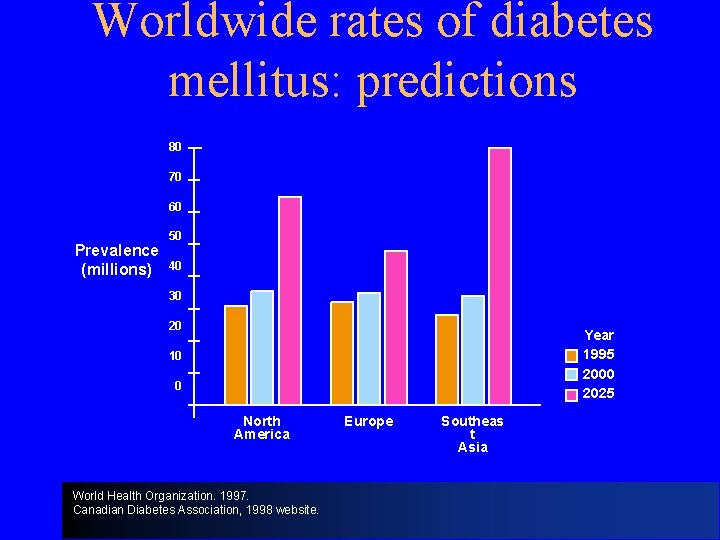
Worldwide rates of diabetes mellitus: predictions 80 70 60 Prevalence (millions) 50 40 30 20 Year 1995 2000 2025 10 0 North America World Health Organization. 1997. Canadian Diabetes Association, 1998 website. Europe Southeas t Asia

2. 2 Million Canadians Have Diabetes Mellitus Frequency of diagnosed and undiagnosed diabetes and IGT, by age (U. S. data - Harris) Harris. Diabetes Care 1993; 16: 642 -52.

Cardiovascular Disease Risk is Increased 2 to 4 Times Framingham study: diabetes and CAD mortality at 20 -year follow-up Haffner Am J Cardiol 1999; 84: 11 J-4 J.

UK Prospective Diabetes Study multi-centre randomised controlled trial of different therapies of Type 2 diabetes

UK Prospective Diabetes Study Does an intensive glucose control policy reduce the risk of complications of diabetes?

Blood Glucose Control Study : Aims to determine whether • improved glucose control of Type 2 diabetes will prevent clinical complications • therapy with – sulphonylurea - first or second generation – insulin – metformin – has any specific advantage or disadvantage

Patient Characteristics 5102 newly diagnosed Type 2 diabetic patients age 25 - 65 years mean gender male : female ethnic group Caucasian Asian 10% Afro-caribbean Body Mass Index mean fasting plasma glucose (fpg) 11. 5 mmol/L Hb. A 1 c median hypertensive 39% 53 y 59 : 41% 82% 8% 28 kg/m 2 median 9. 1 %

Randomisation of Treatment Policies Main Randomisation n=4209 (82%) 342 allocated to metformin 3867 Conventional Policy 30% (n=1138) Intensive Policy 70% (n=2729) Sulphonylurea n=1573 Insulin n=1156

Actual Therapy

Any Diabetes Related Endpoint 1401 of 3867 patients (36%) First occurrence of any one of: • diabetes related death • non fatal myocardial infarction, heart failure or angina • non fatal stroke • amputation • renal failure • retinal photocoagulation or vitreous haemorrhage • cataract extraction or blind in one eye

Microvascular Endpoints (cumulative) renal failure or death, vitreous haemorrhage or photocoagulation 346 of 3867 patients (9%)

Hb. A 1 c cross-sectional, median values
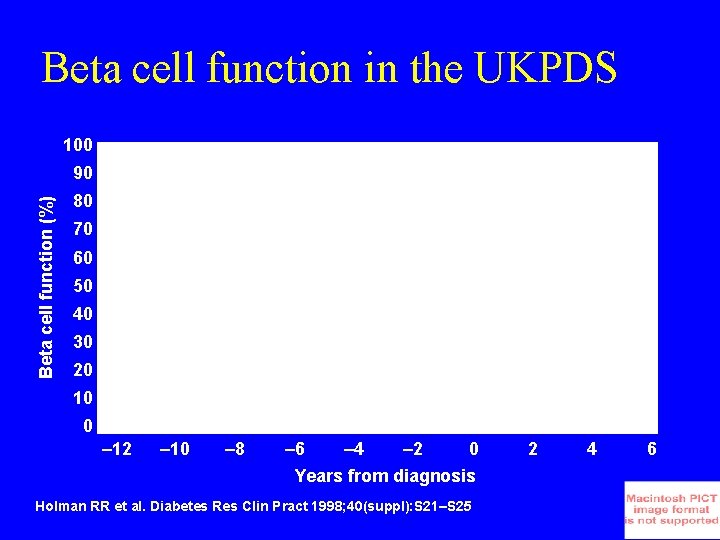
Beta cell function in the UKPDS 100 Beta cell function (%) 90 80 70 60 50 40 30 20 10 0 – 12 – 10 – 8 – 6 – 4 – 2 0 Years from diagnosis Holman RR et al. Diabetes Res Clin Pract 1998; 40(suppl): S 21–S 25 2 4 6

WHAT’S THE PROBLEM • It used to be easy diet+Dia. Beta/glyburide metformin do as you’re told • We must reach glucose targets -CDA guidelines; UKPDS; Kumamoto see you later • It doesn’t work poor control vascular complications

A BIG ISSUE • glyburide works-then fails • metformin works-then fails • insulin, using standard regimens, works-then fails

UKPDS TREATMENT FAILURE • On SU treatment: 5%/year • Hb. A 1 c increased 0. 3%/year

UKPDS monotherapy failure A 1 c<0. 07 at 9 years % UKPDS JAMA 1999 281 2005

COMBINATION THERAPY • • • Achieves better blood glucose levels Less side-effects than high dose monotherapy Delays use of insulin Patients more prepared for aggressive therapies ? Protects beta-cell function M. Riddle Am J Med 2000; 108(6 A) 15 S-22 S

Insulin resistance: an underlying problem Time Insulin resistance Insulin production Glucose level Non. Pre. Type 2 diabetes Opara JU, Levine JH, South Med J. 1997; 90: 1162 -1168.

Pathophysiology Type 2 Diabetes: Underlying Defects Beta-cell function Insulin resistance Type 2 diabetes Other defects: lipolysis release of NEFA hepatic glucose production Adapted from Matthaei et al. Endocrine Reviews 2000; 21: 585 -618. Adapted from Frayn. Br J Nutr 2000; 83(suppl 1): S 71 -S 77.
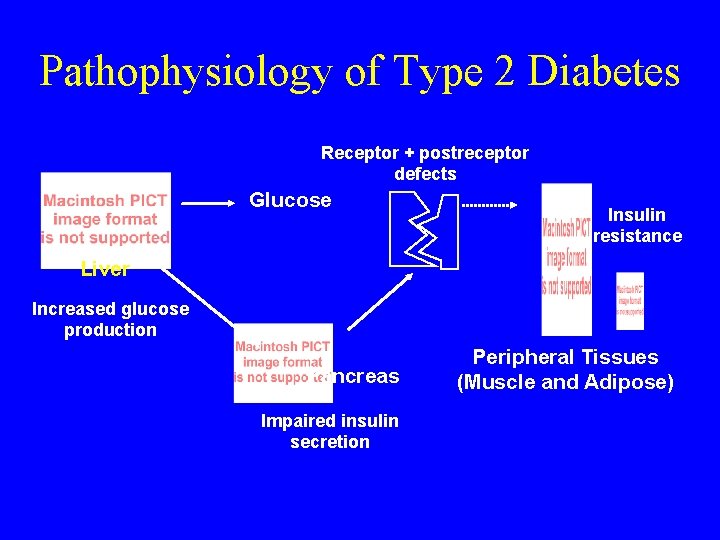
Pathophysiology of Type 2 Diabetes Receptor + postreceptor defects Glucose Insulin resistance Liver Increased glucose production Pancreas Impaired insulin secretion Adapted from Saltiel et al. Diabetes 1996; 45: 1661 -1669. Peripheral Tissues (Muscle and Adipose)

Metabolic syndrome Insulin resistance Obesity Diabetes Hypertension Dyslipidaemia Atherosclerosis risk

THE ARGUMENT • Insulin insufficiency • Insulin resistance
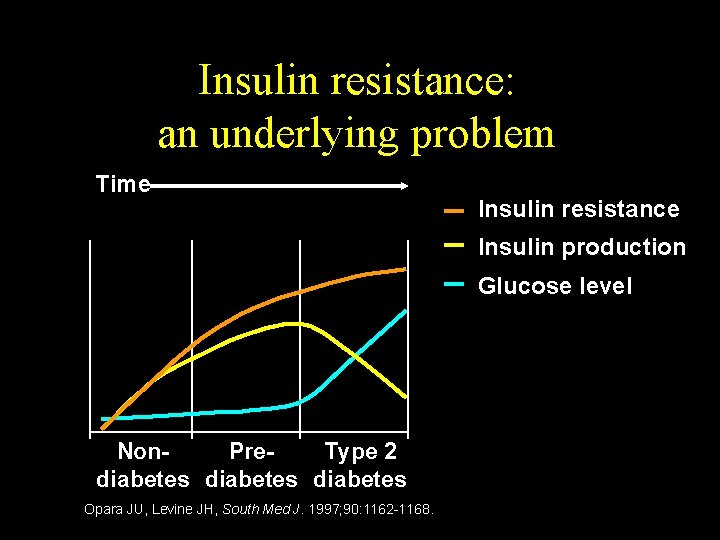
Insulin resistance: an underlying problem Time Insulin resistance Insulin production Glucose level Non. Pre. Type 2 diabetes Opara JU, Levine JH, South Med J. 1997; 90: 1162 -1168.
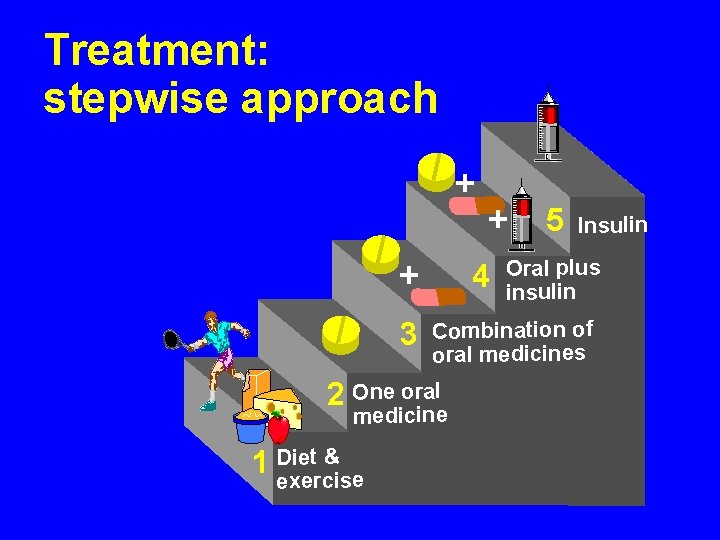
Treatment: stepwise approach + + 3 4 5 Insulin Oral plus insulin Combination of oral medicines 2 One oral medicine t& 1 Die exercise +
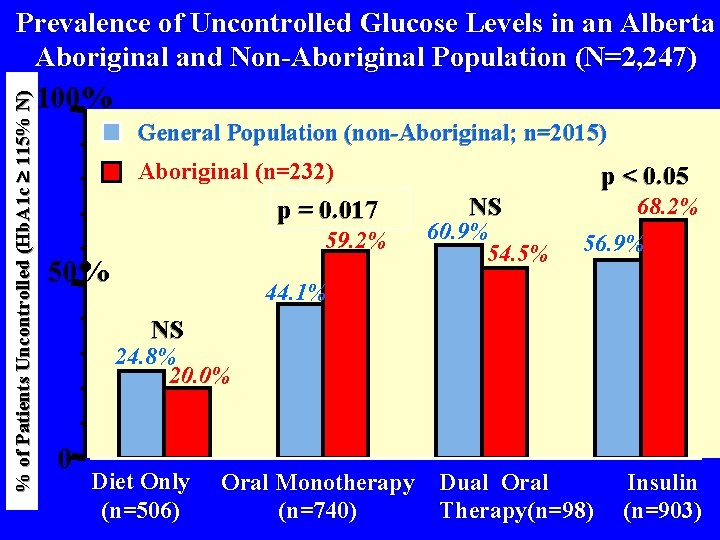
% of Patients Uncontrolled (Hb. A 1 c 115% N) Prevalence of Uncontrolled Glucose Levels in an Alberta Aboriginal and Non-Aboriginal Population (N=2, 247) 100% General Population (non-Aboriginal; n=2015) Aboriginal (n=232) p = 0. 017 59. 2% 50% p < 0. 05 NS 60. 9% 54. 5% 68. 2% 56. 9% 44. 1% NS 24. 8% 20. 0% 0 Diet Only (n=506) Oral Monotherapy Dual Oral (n=740) Therapy(n=98) Insulin (n=903)

Table 3: UNCONTROLLED DIABETES DURATION on THERAPY ** years * * p<0. 001 ** p=0. 009 age MONO DUAL INSULIN

Thiazolidinediones: • Rosiglitazone -Avandia • Pioglitazone -Actos
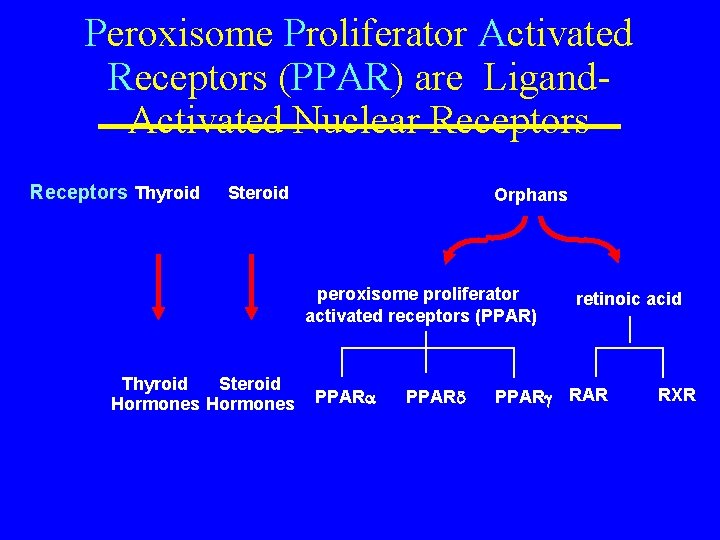
Peroxisome Proliferator Activated Receptors (PPAR) are Ligand. Activated Nuclear Receptors Thyroid Steroid Orphans peroxisome proliferator activated receptors (PPAR) Thyroid Steroid Hormones PPAR retinoic acid PPAR RXR
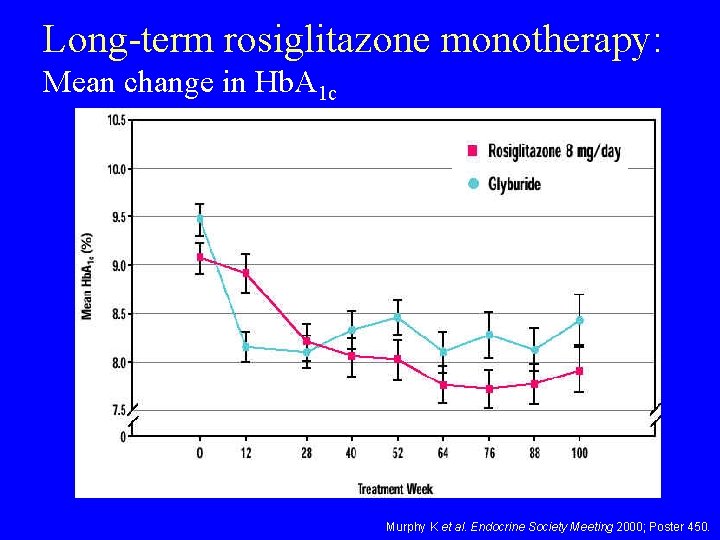
Long-term rosiglitazone monotherapy: Mean change in Hb. A 1 c Murphy K et al. Endocrine Society Meeting 2000; Poster 450.

Fasting Plasma Glucose Conway, R; Rosiglitazone in Family Practice, CDA Oct 2002

Hb. A 1 c over 40 months Conway, R; Rosiglitazone in Family Practice, CDA, Oct 2002

Glycemic parameters by body mass index (BMI): Rosiglitazone added to metformin BMI > 30 kg/m : Extension study (18 months) Effect of BMI: Double-blind studies (26 weeks) 2 9. 5 Patients completing 18 months o metformin + RSG therapy (N = 12 9. 0 0. 5 Hb. A 1 c (%) Mean Change from Baseline in Hb. A 1 C (%) 1 0 -0. 5 8. 0 7. 5 -1 -1. 5 7. 0 BMI < 25 BMI 25– 30 BMI > 30 0. 0 0 MET + placebo MET + RSG 4 mg/day MET + RSG 8 mg/day 3 6 9 12 Months 15 18

Long-Term Durability of Rosiglitazone as Monotherapy or in Combination Therapy in Patients with Type 2 Diabetes Gould E, Cobitz, A. Presented at 84 th Annual Meeting of the Endocrine Society, San Francisco, CA, June 19 -22, 2002 #P 1 -60
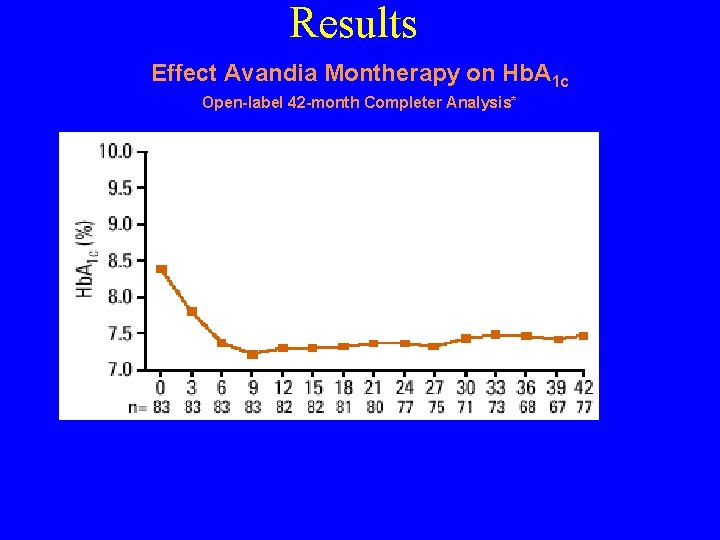
Results Effect Avandia Montherapy on Hb. A 1 c Open-label 42 -month Completer Analysis* *Patients who received Avandia 8 mg qd and 4 mg bid for at least 42 months during 2 double-blind, 26 -week, placebo-controlled trials and their open label extensions. Completer analysis limited by potential bias towards responders to treatment, and small numbers of patients at various time points. 1. Gould E, et al. Presented at 84 th Annual Meeting of the Endocrine Society, San Francisco, CA, June 19 -22, 2002 #P 1 -60
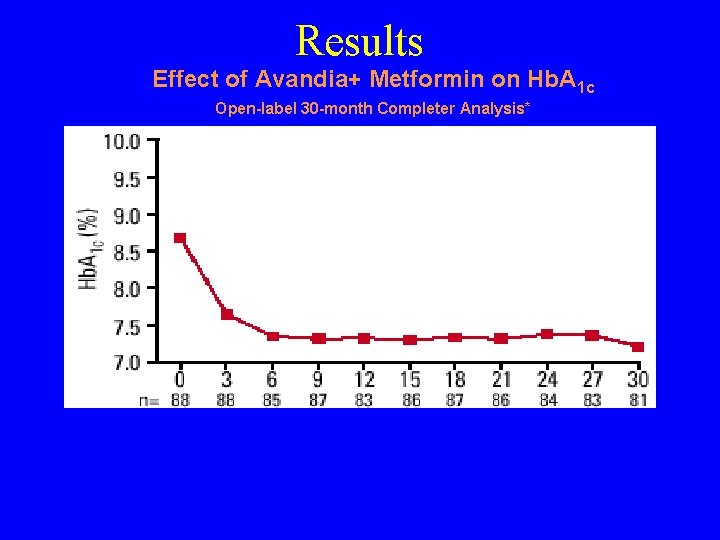
Results Effect of Avandia+ Metformin on Hb. A 1 c Open-label 30 -month Completer Analysis* *Patients who received Avandia 4 mg bid plus 2. 5 g/day of metformin for at least 30 months during 1 double-blin 26 -week, placebo-controlled trial and its open label extension. Completer analysis limited by potential bias towards responders to treatment, and small numbers of patients at various time points. 1. Gould E, et al. Presented at 84 th Annual Meeting of the Endocrine Society, San Francisco, CA, June 19 -22, 2002 #P 1 -60
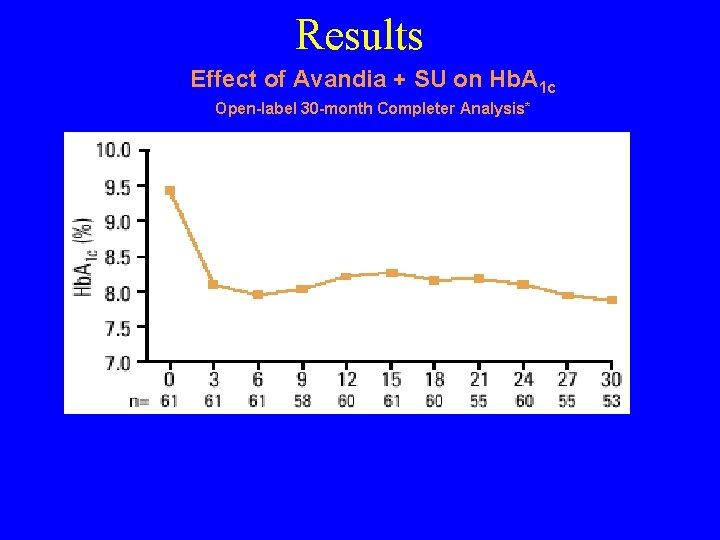
Results Effect of Avandia + SU on Hb. A 1 c Open-label 30 -month Completer Analysis* *Patients who received Avandia 2 mg bid plus glyburide for at least 30 months during 1 double-blind, 26 -week, placebo-controlled trial and its open label extension. Completer analysis limited by potential bias towards responders to treatment, small numbers of patients at of various time points. 1. Gould E, etand al. Presented at 84 th Annual Meeting the Endocrine Society, San Francisco, CA, June 19 -22, 2002 #P 1 -60

WHAT HAS CHANGED • We must treat the Metabolic Syndrome (insulin resistance) -glucose levels -blood pressure -lipids

ORAL AGENTS Dose Response
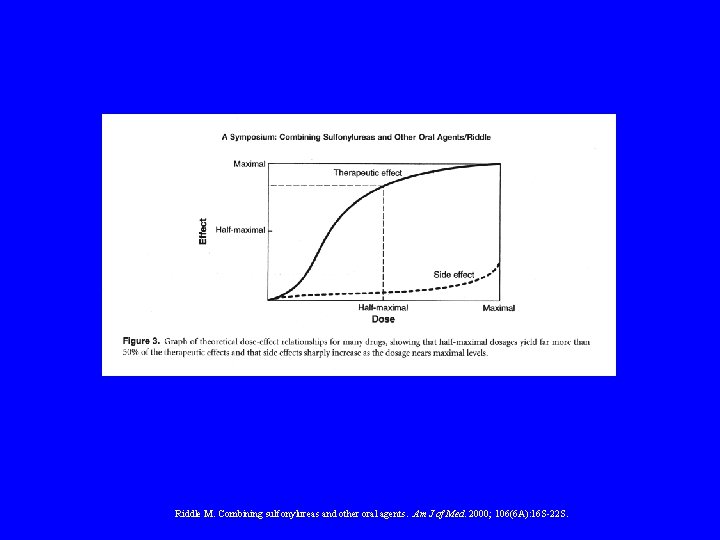
Riddle M. Combining sulfonylureas and other oral agents. Am J of Med. 2000; 106(6 A): 16 S-22 S.
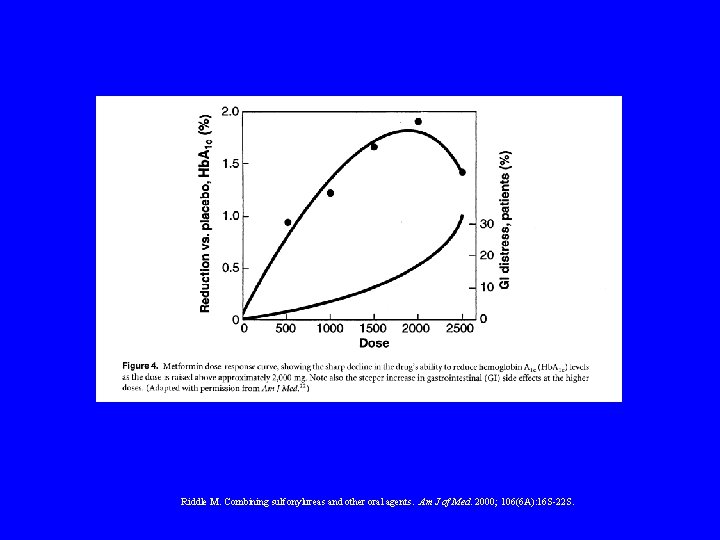
Riddle M. Combining sulfonylureas and other oral agents. Am J of Med. 2000; 106(6 A): 16 S-22 S.
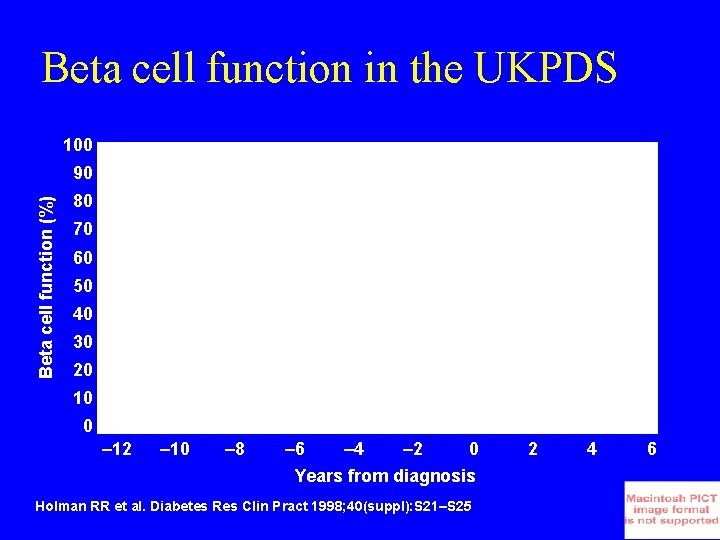
Beta cell function in the UKPDS 100 Beta cell function (%) 90 80 70 60 50 40 30 20 10 0 – 12 – 10 – 8 – 6 – 4 – 2 0 Years from diagnosis Holman RR et al. Diabetes Res Clin Pract 1998; 40(suppl): S 21–S 25 2 4 6

NON-EVIDENCE-BASED THOUGHTS • Use two agents early-on in treatment • Consider a ‘glitazone’ + metformin, or fast-acting insulin secretor

A Peek at the Future • Fast-acting insulin secretors: gliclazide MR/repaglinide/nateglinide • Metformin • TZD rosiglitazone/pioglitazone • Statin • ACE/ARB • Insulin
- Slides: 45