Implications of Batch Processing for Manufacture of Human

Implications of Batch Processing for Manufacture of Human Cells, Tissues and Cellular and Tissue-Based Products Experience of a Commercial Manufacturer of Pituitary-derived Growth Hormone A. Eshkol, Ph. D. , Vice President for Scientific Affaires, Serono International SA, Geneva A. Eshkol, TSEAC, June 26 -27, 2002

Background Human Growth Hormone had been extracted from Pituitary Glands from the 1950’s. Until the 70’s acetone dried glands were used and basically 2 methods – Raben and Wilhelmi - were employed. The products contained aggregates and dimers and were not a purified h. GH product. From the 70’s onwards extraction and purification was done on frozen glands. The methods were milder (no strong acid or alkali) but included chromatographic exclusion of dimers, aggregates and other impurities. A. Eshkol, TSEAC, June 26 -27, 2002

Safety Depends on: • Raw Material - Biological Source • Process - Validation - Decontamination - Cross Contamination - Batch Size • Testing - QC A. Eshkol, TSEAC, June 26 -27, 2002

Source – h. GH from Human Hypophyses • Pituitary glands collected Post Mortem • Majority of Donors Older People • Donors with Nervous System Disease or Mental Illness Excluded • Mostly no Brain Histology available • Conclusion : No certainty of CJD-free Donors A. Eshkol, TSEAC, June 26 -27, 2002

h. GH and CJD • In March 1985 the NIH learned that a 22 year old man died of CJD. He received h. GH from 1966 -1976. • In April 1985 two additional deaths due to CJD were revealed. • Such a risk was considered by some investigators, and a validation study determining the clearance potential of the purification process was initiated in 1979 in the UK and published in August 1985 – Taylor et al. , The Lancet, Aug. 3, 1985, 260 -262. This study demonstrated that the specific procedure employed the Lumley-Jones method (Lumley-Jones et al. J Endocrinol 82: 77 -86) – had a high capacity for the Scrapie agent clearance. A. Eshkol, TSEAC, June 26 -27, 2002

Process for h. GH Preparation Multiple steps including: • Aqueous phase protein extraction • Selective Isoelectric Precipitation – p. H 4. 9 • Solubilization and concentration • Chromatography on Sephacryl SF 200 • Collection of monomeric h. GH • Millipore ultrafiltration – 100, 000 dalton cut-off * • 6 M Urea Treatment * * Introduced in 1985 A. Eshkol, TSEAC, June 26 -27, 2002
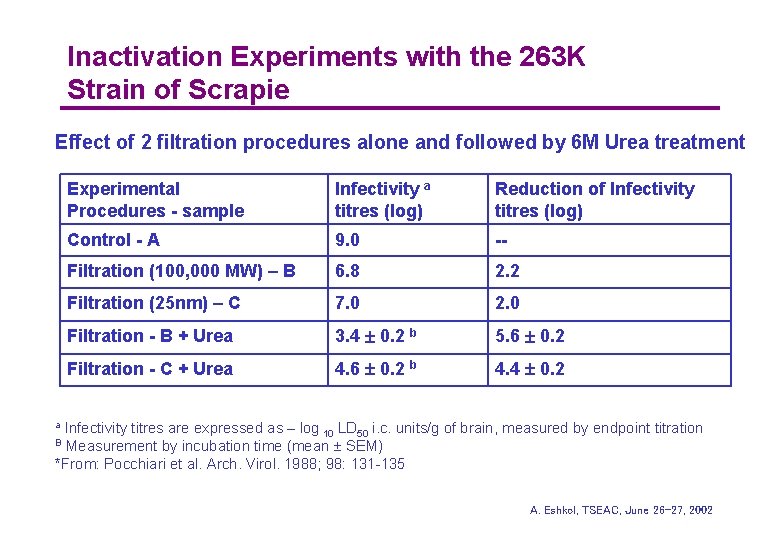
Inactivation Experiments with the 263 K Strain of Scrapie Effect of 2 filtration procedures alone and followed by 6 M Urea treatment Experimental Procedures - sample Infectivity a titres (log) Reduction of Infectivity titres (log) Control - A 9. 0 -- Filtration (100, 000 MW) – B 6. 8 2. 2 Filtration (25 nm) – C 7. 0 2. 0 Filtration - B + Urea 3. 4 0. 2 b 5. 6 0. 2 Filtration - C + Urea 4. 6 0. 2 b 4. 4 0. 2 Infectivity titres are expressed as – log 10 LD 50 i. c. units/g of brain, measured by endpoint titration Measurement by incubation time (mean SEM) *From: Pocchiari et al. Arch. Virol. 1988; 98: 131 -135 a B A. Eshkol, TSEAC, June 26 -27, 2002

Background and Decontamination Checking 1. Trial Run / Background contamination h. GH purification procedure without scrapie challenge was run till after the Sephacryl chromatography step. The monomeric h. GH was concentrated and the whole product inoculated i. c. into 66 hamsters. None developed scrapie. A. Eshkol, TSEAC, June 26 -27, 2002
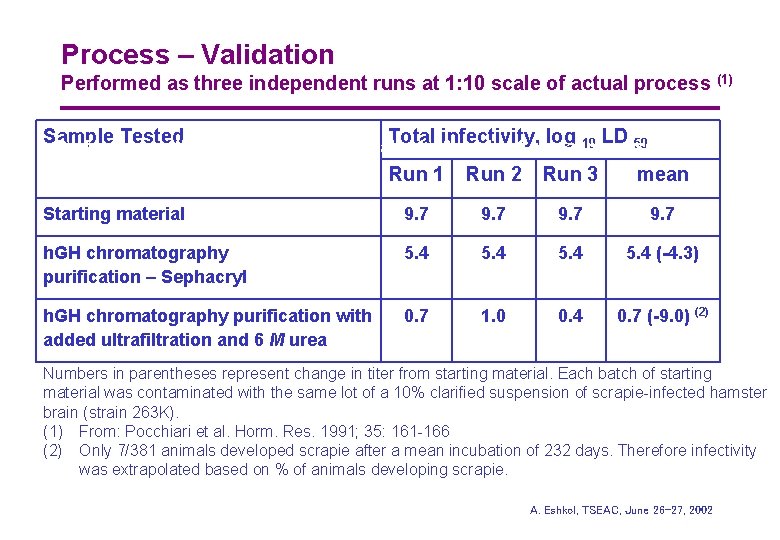
Process – Validation Performed as three independent runs at 1: 10 scale of actual process Sample Tested Total infectivity, log (1) LD 50 (1) Performed as three independent runs at a 1: 10 scale of actual 10 process Run 1 Run 2 Run 3 mean Starting material 9. 7 h. GH chromatography purification – Sephacryl 5. 4 (-4. 3) h. GH chromatography purification with added ultrafiltration and 6 M urea 0. 7 1. 0 0. 4 0. 7 (-9. 0) (2) Numbers in parentheses represent change in titer from starting material. Each batch of starting material was contaminated with the same lot of a 10% clarified suspension of scrapie-infected hamster brain (strain 263 K). (1) From: Pocchiari et al. Horm. Res. 1991; 35: 161 -166 (2) Only 7/381 animals developed scrapie after a mean incubation of 232 days. Therefore infectivity was extrapolated based on % of animals developing scrapie. (2) Only 7/381 animals developed scrapie after a mean incubation of A. 232 days. Therefore Eshkol, TSEAC, June 26 -27, infectivity 2002 was extrapolated based on % of animals developing scrapie.

Background and Decontamination Checking 2. Control Run / Decontamination At the end of the validation/clearance experiment, laboratory equipment (glassware, columns, ultrafiltration equipment) was decontaminated either by steam autoclaving at 134°C (2 bars) for 1 h or, if not feasible, by overnight soaking in a 1 N Na. OH solution containing 1% sodium dodecyl sulfate. Then the h. GH purification was performed using the decontaminated equipment. Aliquots of 0. 05 ml of monomeric h. GH (post Sephacryl) were inoculated into 12 hamsters. Following ultrafiltration and Urea treatment, the entire product was inoculated into 135 hamsters. None showed clinical signs of disease nor histological lesions in the brain. A. Eshkol, TSEAC, June 26 -27, 2002
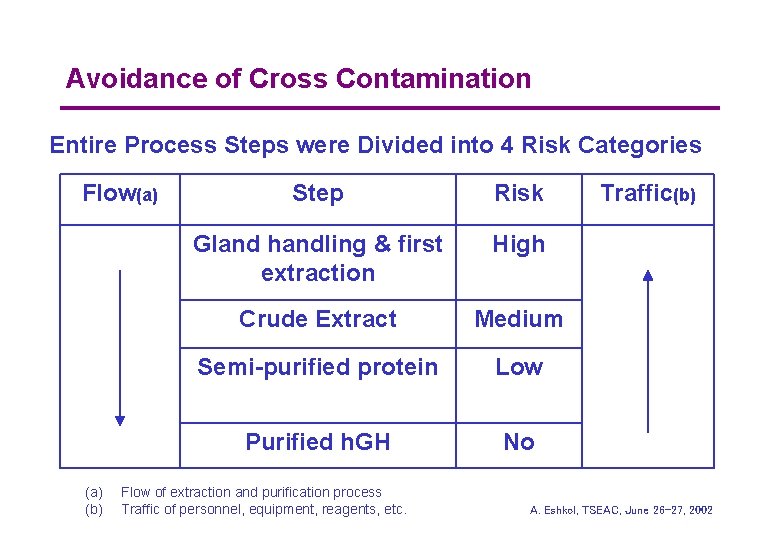
Avoidance of Cross Contamination Entire Process Steps were Divided into 4 Risk Categories Flow(a) (b) Step Risk Gland handling & first extraction High Crude Extract Medium Semi-purified protein Low Purified h. GH No Flow of extraction and purification process Traffic of personnel, equipment, reagents, etc. Traffic(b) A. Eshkol, TSEAC, June 26 -27, 2002

Batch Contamination / Batch Size Assumption : CJD bearing donors - 1/10, 000 (1) Batch size Contaminated batches Max. No. of vials (2) per batch 5, 000 1/2 25, 000 2, 000 1/5 10, 000 500 -600 (3) 1/10 – 1/20 2, 500 (1) Brown, Gajdusek, Gibbs & Asher ; NEJM, 1985; 313: 728 -731 (2) Of 4 IU of h. GH (3) Serono Procedure A. Eshkol, TSEAC, June 26 -27, 2002

Calculated Level (Worst Case) of Contamination of h. GH following Production Procedures With and Without Added Ultrafiltration and Urea Treatment Steps h. GH purification with filtration/urea Infectivity (log. LD 50) in pituitary batch 4. 0 Infectivity (log. LD 50) per h. GH batch -0. 3 -5. 0 Infectivity (log. LD 50) per h. GH vial -4. 7 -9. 4 Probability that a single vial is contaminated 1 in 50, 000 1 in 2. 5 billion Probability that 1 patient receives a contaminated vial during course of treatment 1 in 64 1 in 3. 2 million (1) From: Pocchiari et al. , 1991 A. Eshkol, TSEAC, June 26 -27, 2002
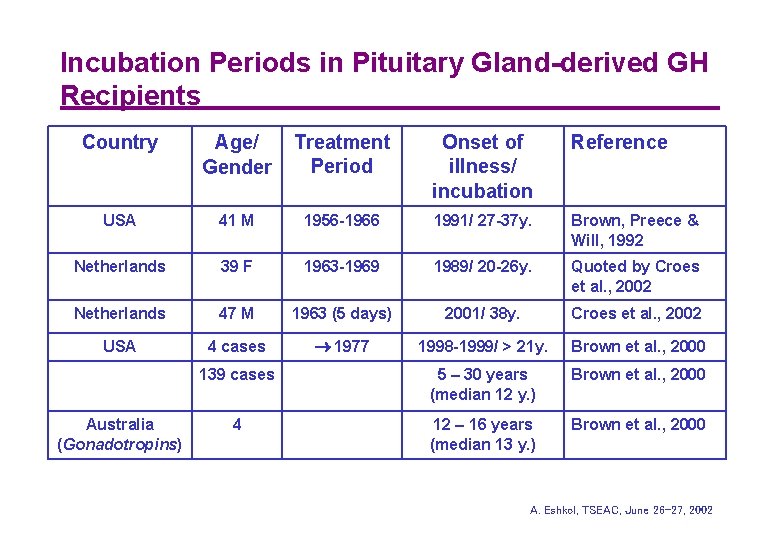
Incubation Periods in Pituitary Gland-derived GH Recipients Country Age/ Gender Treatment Period Onset of illness/ incubation Reference USA 41 M 1956 -1966 1991/ 27 -37 y. Brown, Preece & Will, 1992 Netherlands 39 F 1963 -1969 1989/ 20 -26 y. Quoted by Croes et al. , 2002 Netherlands 47 M 1963 (5 days) 2001/ 38 y. Croes et al. , 2002 USA 4 cases 1977 1998 -1999/ > 21 y. Brown et al. , 2000 139 cases 5 – 30 years (median 12 y. ) Brown et al. , 2000 4 12 – 16 years (median 13 y. ) Brown et al. , 2000 Australia (Gonadotropins) A. Eshkol, TSEAC, June 26 -27, 2002

Conclusions: 1. A protein like h. GH (single chain, non-glycosilated) can be purified with a large safety margin provided that: • In the procedure, steps are included which can significantly reduce and inactivate CJD/TSE agents. • The process is carried out under strict conditions and includes effective decontamination procedures. • Care is taken to avoid cross-contamination. A. Eshkol, TSEAC, June 26 -27, 2002

2. Can total elimination of potential infectivity be guaranteed ? • Serono has had no CJD cases reported, but in view of some very long incubation periods reported, we remain vigilant. If there was residual infectivity in some batches, it must have been extremely low. • Total absence (? ) of risk would also need assurance of the safety of the raw material, which is particularly important in the case of older age group donors. • Currently available test methods do not have the sensitivity which would make this feasible. A. Eshkol, TSEAC, June 26 -27, 2002

References • • P. Brown, M. Preece, J. -P. Brandel, T. Sato, L. Mc. Shane, I. Zerr, A. Fletcher, M. Pocchiari, N. R. Cashman, J. H. Aignaux, L. Cervenáková, J. Fradkin, L. B. Schonberger, S. J. Collins. Neurology 2002; 55: 1075 -1081. P. Brown, M. A. Preece, R. G. Will. Lancet 1992; 340: 24 -27. P. Brown, D. C. Gajdusek, C. J. Gibbs Jr, D. M. Asher. NEJM 1985; 313: 728731. E. A. Croes, G. Roks, G. H. Jansen, P. C. G. Nijssen, C. M. van Duijn. J Neurol Neurosurg Psychiatry 2002; 72: 792 -793. R. Lumley-Jones, G. Benker, P. R. Salacinski, T. J. Lloyd, P. J. Lowry. J Endocrinol 1979; 82: 77 -86. M. Pocchiari, S. Peano, A. Conz, A. Eshkol, F. Maillard, P. Brown, C. J. Gibbs Jr, Y. Geng Xl, E. Tenham-Fisher, G. Macchi. Horm Res 1991; 35: 161 -166. M. Pocchiari, G. Macchi, S. Peano, A. Conz. Arch Virol 1988: 131 -135. D. M. Taylor, H. Fraser, P. R. Salacinski, A. G. Dickinson, P. A. Robertson, P. J. Lowry. Lancet 1985: 260 -262. A. Eshkol, TSEAC, June 26 -27, 2002
- Slides: 17