Implementation of the proposed Analytical Procedure Lifecycle USP


- Slides: 11

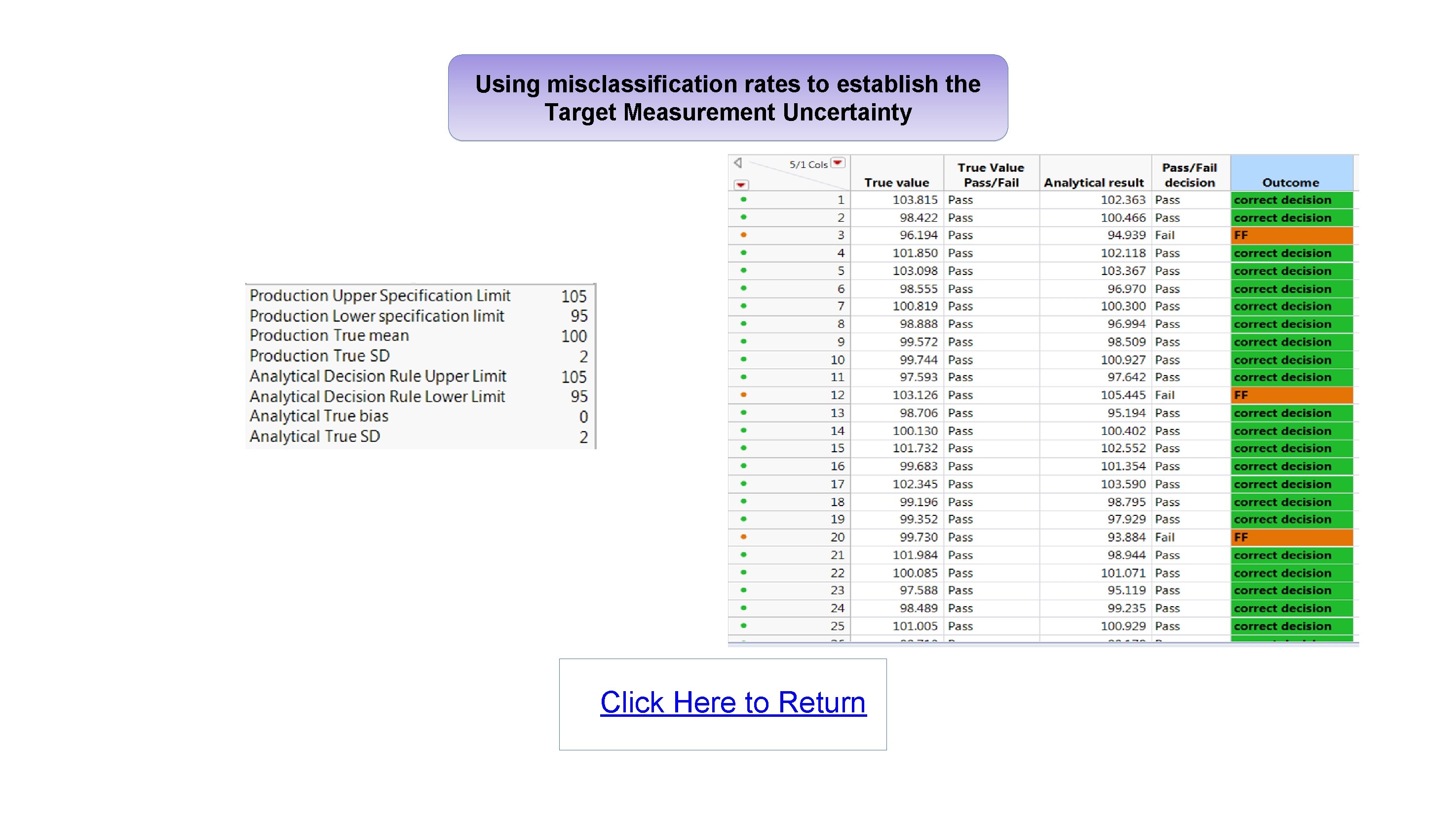
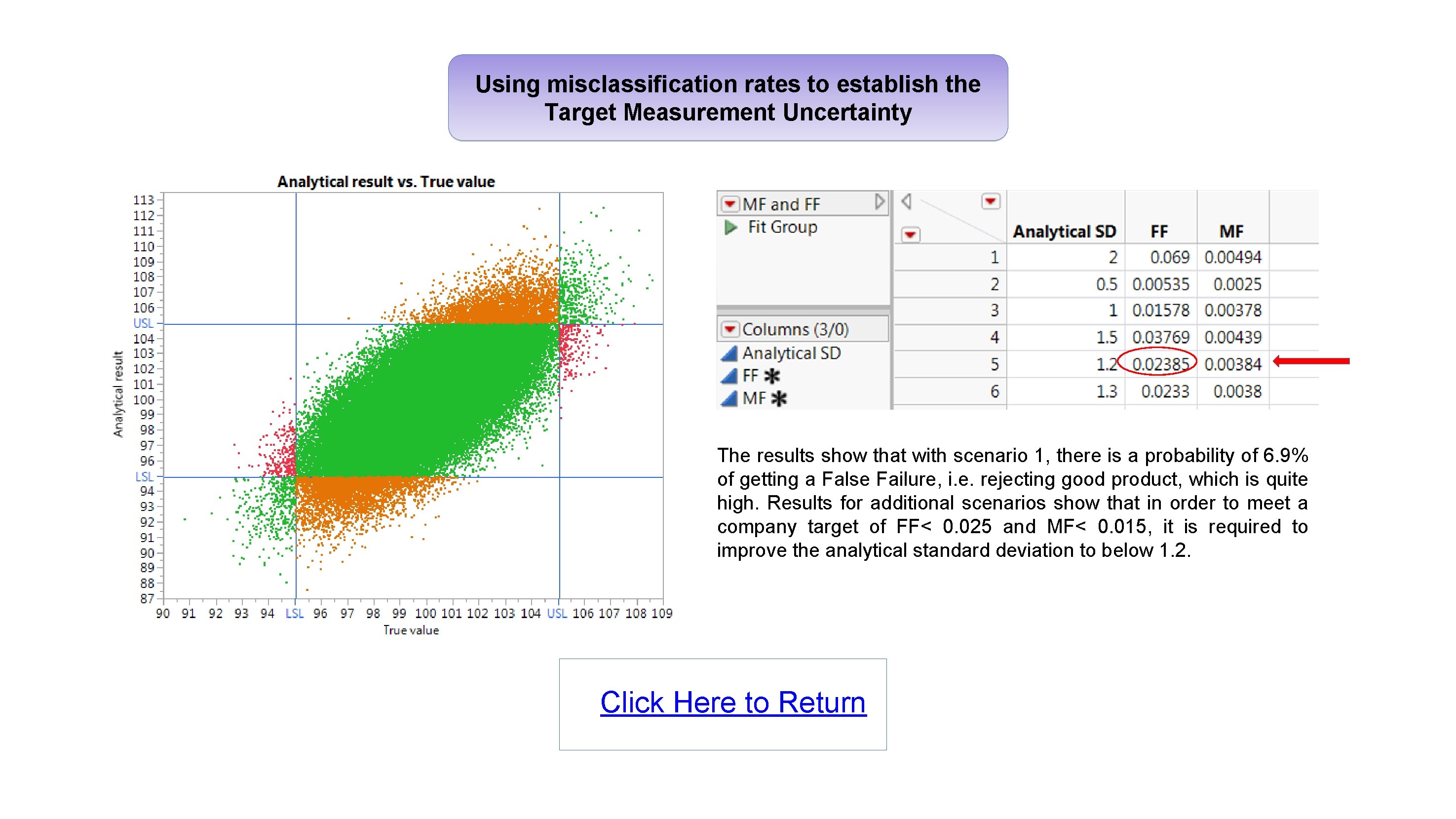
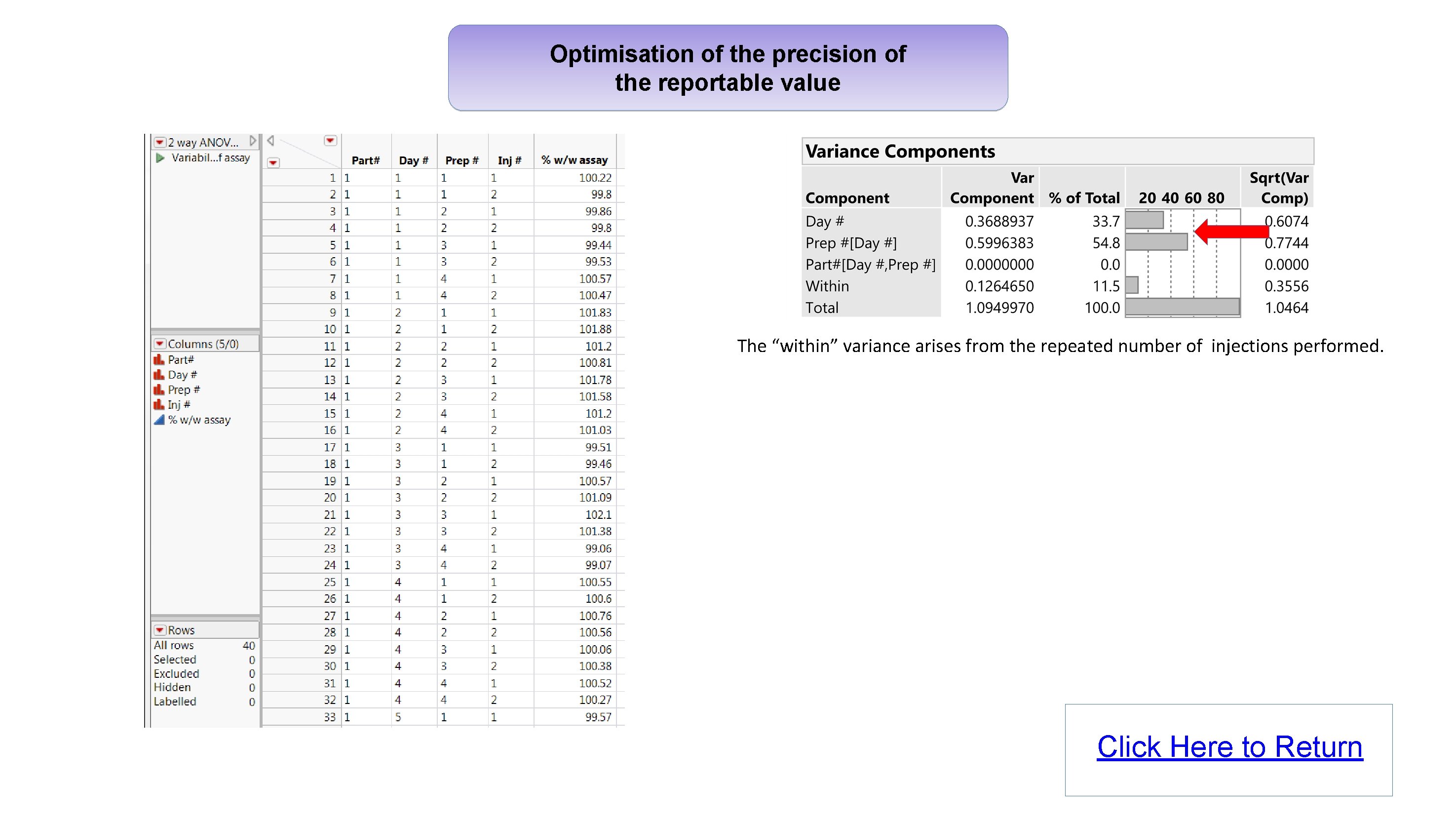
Implementation of the proposed Analytical Procedure Lifecycle USP <1220> general chapter: Automate the calculations and visualise the results using JMP Christel Kronig - Dr Reddy’s Laboratories Abstract Analytical method development and validation in the pharmaceutical industry is currently evolving with more emphasis now being placed on understanding the measurement error in an effort to ensure methods of analysis are fit for purpose. The USP Validation and Verification Expert Panel recently published a new draft general chapter 1 “The Analytical Procedure Lifecycle”. Several stimuli articles were also published 2, 3, 4, providing details of calculations proposed to support the development and qualification of analytical methods, including: • Using misclassification rates to establish the Target Measurement Uncertainty. • Optimisation of the precision of the reportable value and calibration format. • Verifying that the analytical procedure meets the Analytical Target Profile. These calculations represent a significant step change compared with what has traditionally been performed, and to ensure adoption of these new concepts, it will be important for analytical scientists to have access to easy-to-use tools which can process the results and visualise the outcome. Using misclassification rates to establish the Target Measurement Uncertainty Let’s look at the example 4 of a drug product with a specification for assay of 95. 0 -105. 0%, and a production mean of 100% with a standard deviation of 2. The analytical method used to measure the assay for this drug product has no bias, but has a standard deviation of 2. The product is declared within specification if the result is in the range 95. 0 -105. 0%. SCENARIO 1 To understand the impact of the measurement error from the analytical method, a simulation is carried out. 100, 000 data points with true value and analytical result values are generated to fit the above scenario. The resulting probabilities of getting a correct / incorrect decision can be automatically calculated for different scenarios using a script. RESULTS OF SIMULATED DATA FOR SCENARIO 1 This poster looks at some of the calculations and graphs detailed in these stimuli articles and show they can easily be performed, automated and visualised in JMP with the use of scripts. Glossary • FF : False Failure ( Product is good, but result is Out Of Specification ) • MF : Missed Faults ( Product is bad, but result is In Specification ) • ATP : Analytical Target Profile • TMU : Target Measurement Uncertainty (Total error budget for the method) • Bias : Difference between Measured Value and True value. • Intermediate Precision : Measure of variability which includes several sources of error (e. g. day, instrument, analyst) • NMT : Not More Than Optimisation of the precision of the reportable value Here we look at the results of an example study 5 to evaluate the precision of an analytical method used to measure the assay of a drug product: • k = 5 series (Day #) • m = 4 sample preps per series (Prep #) • n = 2 injections per sample prep (Inj #) What is the biggest source of error for this method? STANDARD JMP ANALYSIS: VARIANCE COMPONENTS (NESTED) The data shows that the largest error contribution arises from the number of series and sample preps. We need to optimize the precision of the reportable value and find out how many series, sample preps and injections should be done routinely to minimize the error 3, 5. Calculations and graphs were automated with a script. INTERMEDIATE PRECISION AS A FUNCTION OF k, m AND n MISCLASSIFICATION RATES FOR MULTIPLE SCENARIOS The results show that with scenario 1, there is a probability of 6. 9% of getting a False Failure, i. e. rejecting good product, which is quite high. Results for additional scenarios show that in order to meet a company target of FF< 0. 025 and MF< 0. 015, it is required to improve the analytical standard deviation to below 1. 2. ( Click Figures to Enlarge ) VIDEO Results show that for this method: : • Having two series will provide the biggest reduction in measurement error. Performing dual preparations would also help further reduce the error. • Performing multiple injections will not help to reduce the error, so a single injection only is required for routine testing. Next slide

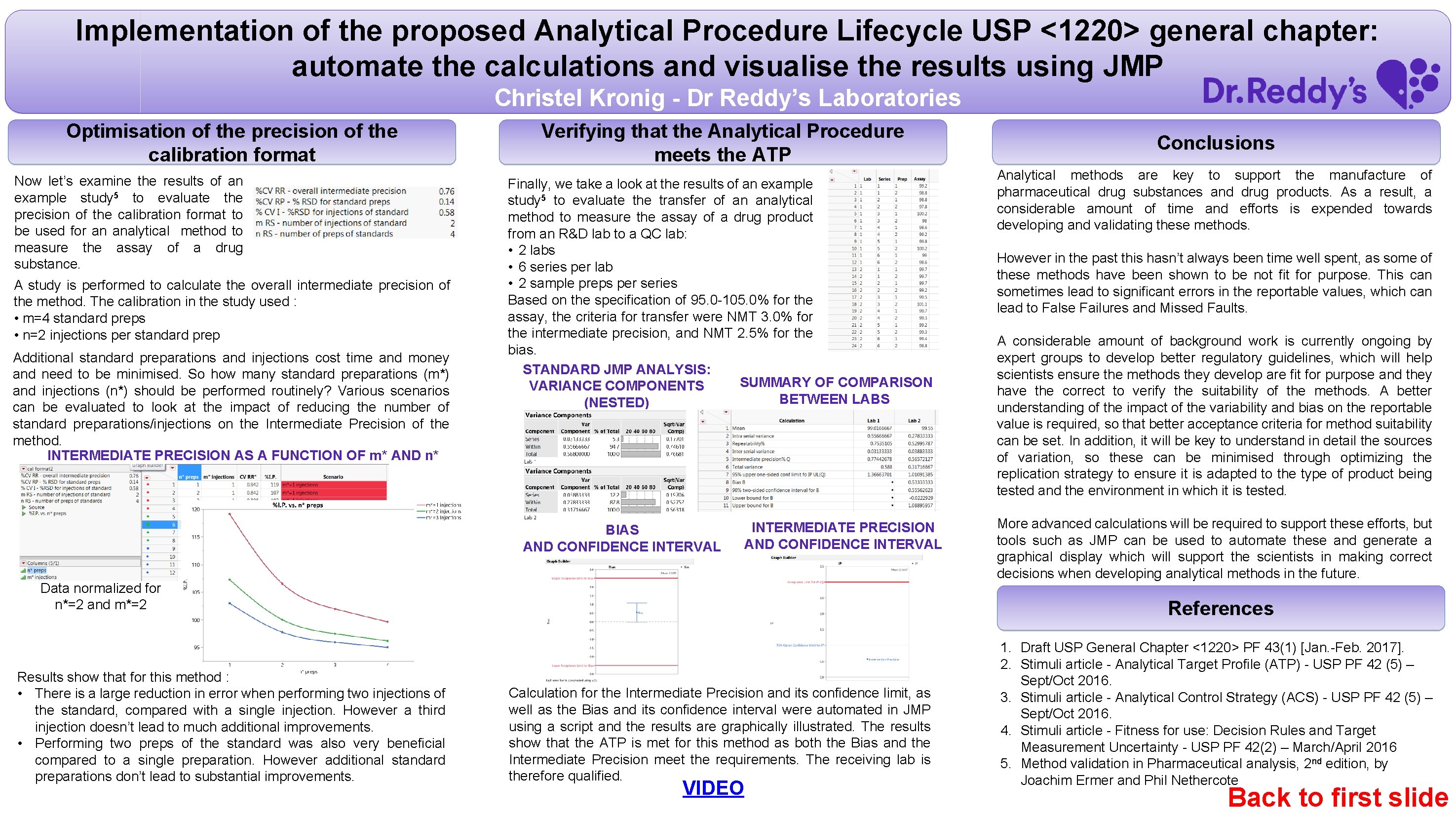
Implementation of the proposed Analytical Procedure Lifecycle USP <1220> general chapter: automate the calculations and visualise the results using JMP Christel Kronig - Dr Reddy’s Laboratories Optimisation of the precision of the calibration format Now let’s examine the results of an example study 5 to evaluate the precision of the calibration format to be used for an analytical method to measure the assay of a drug substance. A study is performed to calculate the overall intermediate precision of the method. The calibration in the study used : • m=4 standard preps • n=2 injections per standard prep Additional standard preparations and injections cost time and money and need to be minimised. So how many standard preparations (m*) and injections (n*) should be performed routinely? Various scenarios can be evaluated to look at the impact of reducing the number of standard preparations/injections on the Intermediate Precision of the method. INTERMEDIATE PRECISION AS A FUNCTION OF m* AND n* Verifying that the Analytical Procedure meets the ATP Finally, we take a look at the results of an example study 5 to evaluate the transfer of an analytical method to measure the assay of a drug product from an R&D lab to a QC lab: • 2 labs • 6 series per lab • 2 sample preps per series Based on the specification of 95. 0 -105. 0% for the assay, the criteria for transfer were NMT 3. 0% for the intermediate precision, and NMT 2. 5% for the bias. STANDARD JMP ANALYSIS: SUMMARY OF COMPARISON VARIANCE COMPONENTS BETWEEN LABS (NESTED) BIAS AND CONFIDENCE INTERVAL INTERMEDIATE PRECISION AND CONFIDENCE INTERVAL Data normalized for n*=2 and m*=2 Results show that for this method : • There is a large reduction in error when performing two injections of the standard, compared with a single injection. However a third injection doesn’t lead to much additional improvements. • Performing two preps of the standard was also very beneficial compared to a single preparation. However additional standard preparations don’t lead to substantial improvements. Conclusions Analytical methods are key to support the manufacture of pharmaceutical drug substances and drug products. As a result, a considerable amount of time and efforts is expended towards developing and validating these methods. However in the past this hasn’t always been time well spent, as some of these methods have been shown to be not fit for purpose. This can sometimes lead to significant errors in the reportable values, which can lead to False Failures and Missed Faults. A considerable amount of background work is currently ongoing by expert groups to develop better regulatory guidelines, which will help scientists ensure the methods they develop are fit for purpose and they have the correct to verify the suitability of the methods. A better understanding of the impact of the variability and bias on the reportable value is required, so that better acceptance criteria for method suitability can be set. In addition, it will be key to understand in detail the sources of variation, so these can be minimised through optimizing the replication strategy to ensure it is adapted to the type of product being tested and the environment in which it is tested. More advanced calculations will be required to support these efforts, but tools such as JMP can be used to automate these and generate a graphical display which will support the scientists in making correct decisions when developing analytical methods in the future. References Calculation for the Intermediate Precision and its confidence limit, as well as the Bias and its confidence interval were automated in JMP using a script and the results are graphically illustrated. The results show that the ATP is met for this method as both the Bias and the Intermediate Precision meet the requirements. The receiving lab is therefore qualified. VIDEO 1. Draft USP General Chapter <1220> PF 43(1) [Jan. -Feb. 2017]. 2. Stimuli article - Analytical Target Profile (ATP) - USP PF 42 (5) – Sept/Oct 2016. 3. Stimuli article - Analytical Control Strategy (ACS) - USP PF 42 (5) – Sept/Oct 2016. 4. Stimuli article - Fitness for use: Decision Rules and Target Measurement Uncertainty - USP PF 42(2) – March/April 2016 5. Method validation in Pharmaceutical analysis, 2 nd edition, by Joachim Ermer and Phil Nethercote Back to first slide

Using misclassification rates to establish the Target Measurement Uncertainty Click Here to Return

Using misclassification rates to establish the Target Measurement Uncertainty The results show that with scenario 1, there is a probability of 6. 9% of getting a False Failure, i. e. rejecting good product, which is quite high. Results for additional scenarios show that in order to meet a company target of FF< 0. 025 and MF< 0. 015, it is required to improve the analytical standard deviation to below 1. 2. Click Here to Return

Optimisation of the precision of the reportable value The “within” variance arises from the repeated number of injections performed. Click Here to Return

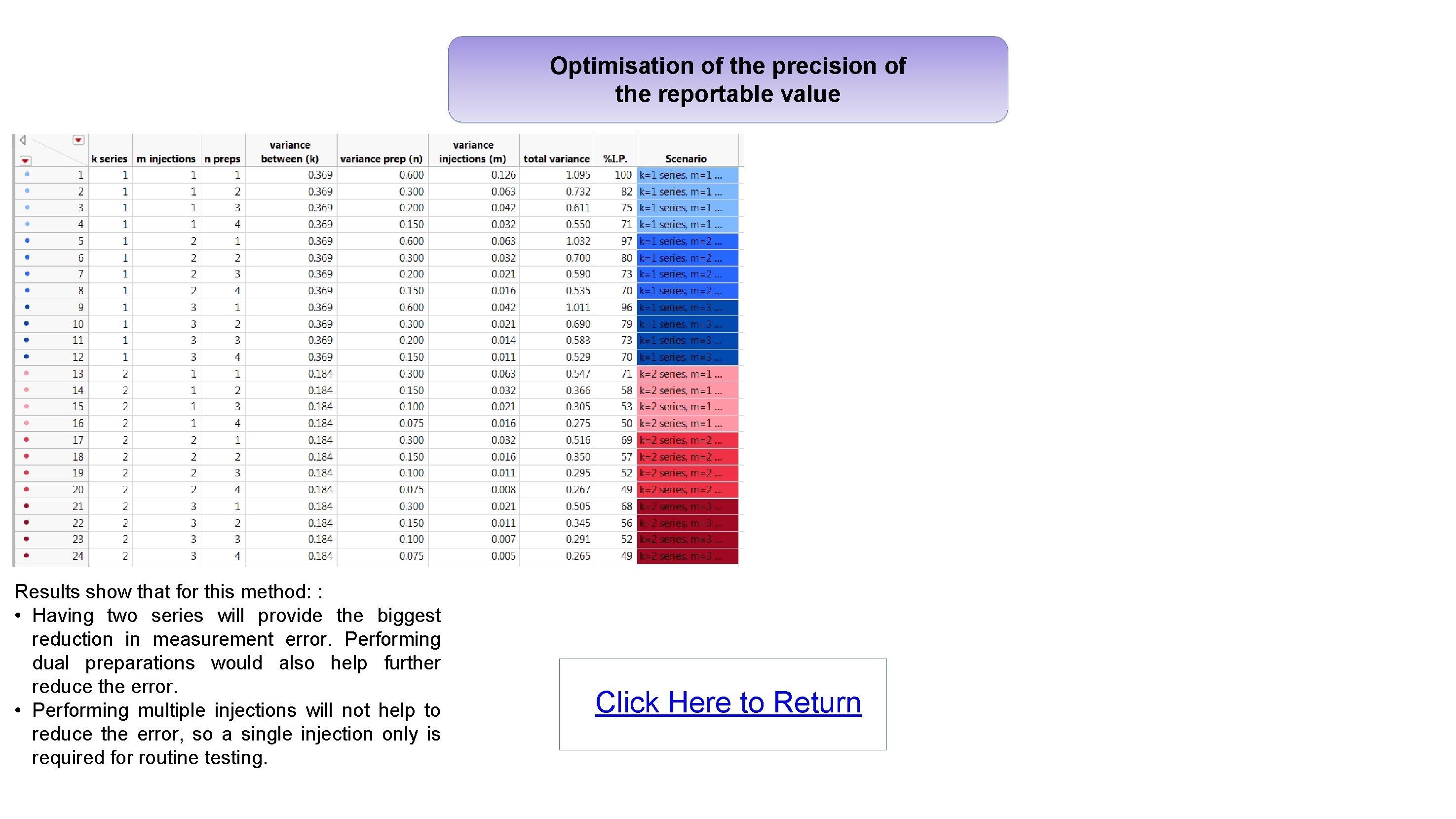
Optimisation of the precision of the reportable value Results show that for this method: : • Having two series will provide the biggest reduction in measurement error. Performing dual preparations would also help further reduce the error. • Performing multiple injections will not help to reduce the error, so a single injection only is required for routine testing. Click Here to Return

Optimisation of the precision of the reportable value Click Here to Return

Optimisation of the precision of the calibration format Results show that for this method : • There is a large reduction in error when performing two injections of the standard, compared with a single injection. However a third injection doesn’t lead to much additional improvements. • Performing two preps of the standard was also very beneficial compared to a single preparation. However additional standard preparations don’t lead to substantial improvements. Data normalized for n*=2 and m*=2 Click Here to Return

Verifying that the Analytical Procedure meets the ATP The “within” variance arises from the repeated number of preps performed. Click Here to Return

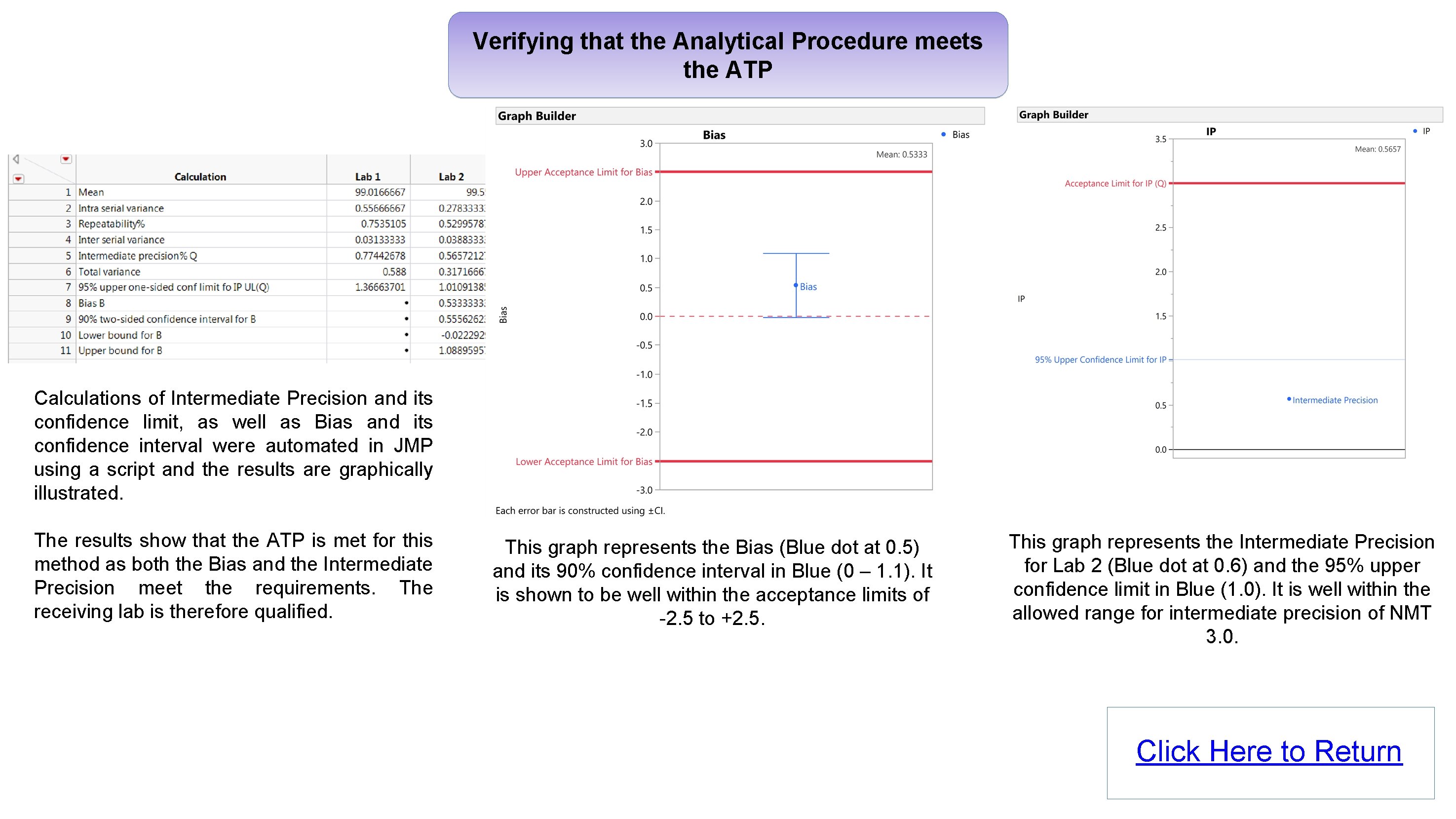
Verifying that the Analytical Procedure meets the ATP Calculations of Intermediate Precision and its confidence limit, as well as Bias and its confidence interval were automated in JMP using a script and the results are graphically illustrated. The results show that the ATP is met for this method as both the Bias and the Intermediate Precision meet the requirements. The receiving lab is therefore qualified. This graph represents the Bias (Blue dot at 0. 5) and its 90% confidence interval in Blue (0 – 1. 1). It is shown to be well within the acceptance limits of -2. 5 to +2. 5. This graph represents the Intermediate Precision for Lab 2 (Blue dot at 0. 6) and the 95% upper confidence limit in Blue (1. 0). It is well within the allowed range for intermediate precision of NMT 3. 0. Click Here to Return

Verifying that the Analytical Procedure meets the ATP Click Here to Return