Implementation Issues for Chemoprevention of Breast Cancer Jack

Implementation Issues for Chemoprevention of Breast Cancer Jack Cuzick, Ph. D. Wolfson Institute of Preventive Medicine St Bartholomew’s Medical School London, United Kingdom
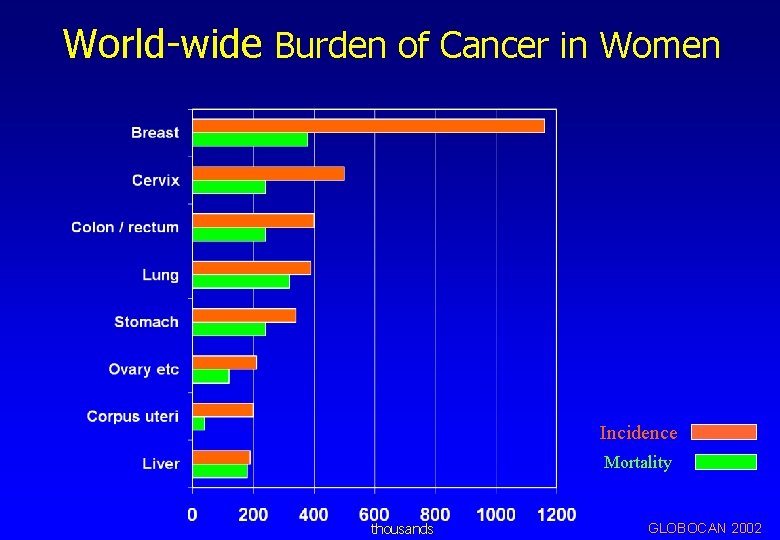
World-wide Burden of Cancer in Women Incidence Mortality thousands GLOBOCAN 2002
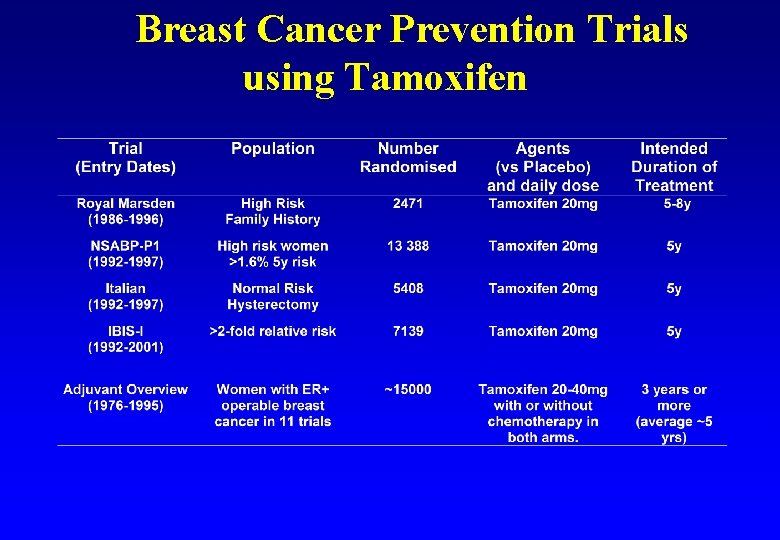
Breast Cancer Prevention Trials using Tamoxifen
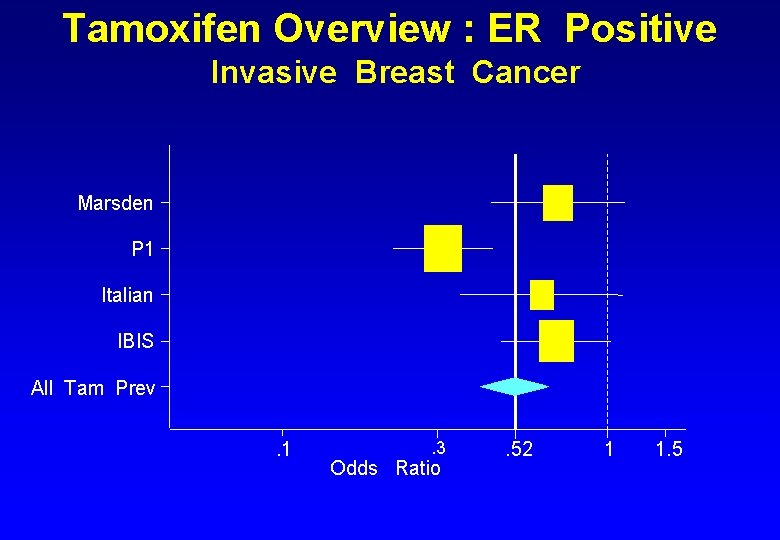
Tamoxifen Overview : ER Positive Invasive Breast Cancer Marsden P 1 Italian IBIS All Tam Prev. 1 . 3 Odds Ratio . 52 1 1. 5

Outcome in 1000 women at high risk of breast cancer followed for 5 years No Treatment Tamoxifen for 5 years Breast Cancer 30 19 VTE 6 12 Endometrial Cancer 2 5

Prevention Trials using Raloxifene
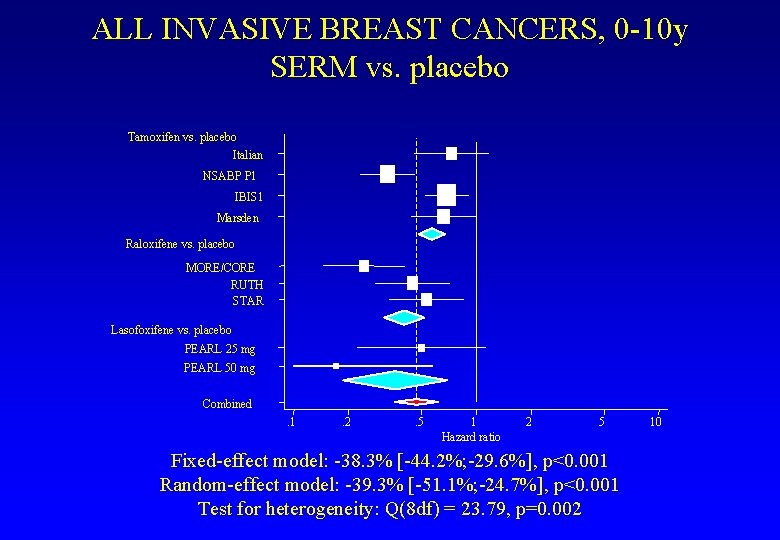
ALL INVASIVE BREAST CANCERS, 0 -10 y SERM vs. placebo Tamoxifen vs. placebo Italian NSABP P 1 IBIS 1 Marsden Raloxifene vs. placebo MORE/CORE RUTH STAR Lasofoxifene vs. placebo PEARL 25 mg PEARL 50 mg Combined. 1 . 2 . 5 1 Hazard ratio 2 5 Fixed-effect model: -38. 3% [-44. 2%; -29. 6%], p<0. 001 Random-effect model: -39. 3% [-51. 1%; -24. 7%], p<0. 001 Test for heterogeneity: Q(8 df) = 23. 79, p=0. 002 10
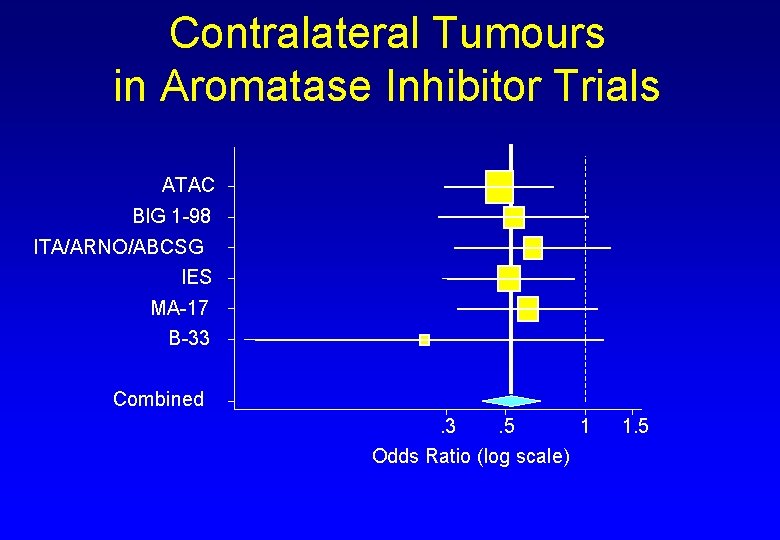
Contralateral Tumours in Aromatase Inhibitor Trials ATAC BIG 1 -98 ITA/ARNO/ABCSG IES MA-17 B-33 Combined. 3. 5 1 Odds Ratio (log scale) 1. 5
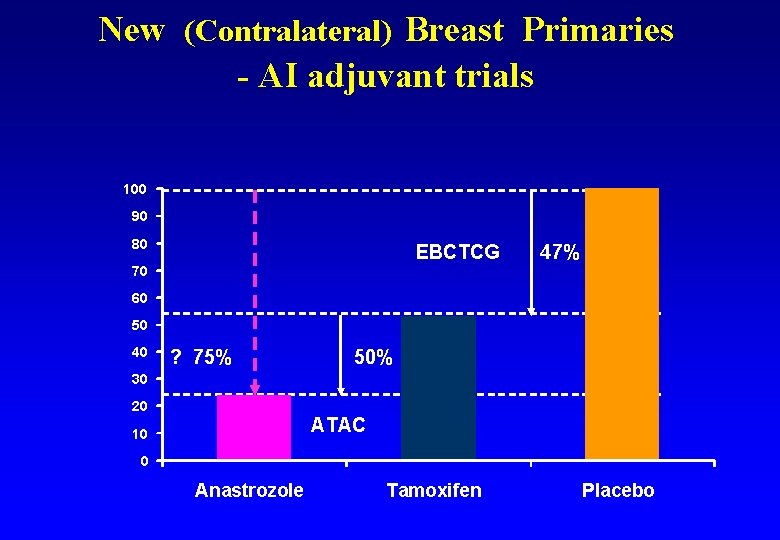
New (Contralateral) Breast Primaries - AI adjuvant trials 100 90 80 EBCTCG 47% 70 60 50 40 ? 75% 50% 30 20 ATAC 10 0 Anastrozole Tamoxifen Placebo
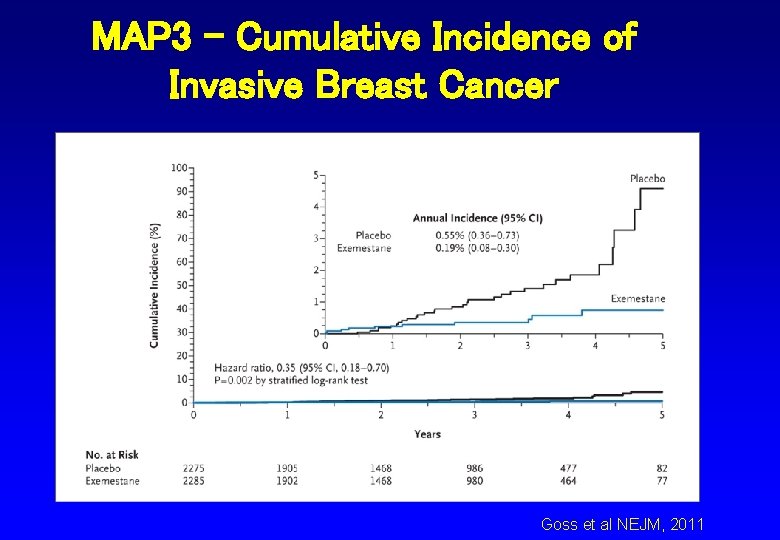
MAP 3 - Cumulative Incidence of Invasive Breast Cancer Goss et al NEJM, 2011

IBIS II- PREVENTION STRATUM • High Risk Post-menopausal women, aged 40 -70. • Placebo controlled 2 -arm trial for high risk • 5 Year Treatment RANDOMISATION PLACEBO ANASTROZOLE 1 mg n = 4, 000 High Risk

Implementation Issues • No agents licensed for prevention in Europe • Tamoxifen and Raloxifene approved in the US • Only manufacturer can apply for license • All drugs off patent protection
- Slides: 12