Implantation of BalloonExpandable THVs in Native Mitral Valves

Implantation of Balloon-Expandable THVs in Native Mitral Valves with Severe MAC Mayra Guerrero, MD, FACC, FSCAI Director of Cardiac Structural Interventions North. Shore University Health. System CRT 2018 Washington, DC March 4 th, 2018 Evanston Hospital

Disclosure Statement of Financial Interest Within the past 12 months, I or my spouse/partner have had a financial interest/arrangement or affiliation with the organization(s) listed below. Affiliation/Financial Relationship • Research Grant Support, Proctor • Consultant, Speaker’s Bureau Company • Edwards Lifesciences • Abbott, Boston Scientific Off label use of products and investigational devices will be discussed in this presentation Evanston Hospital
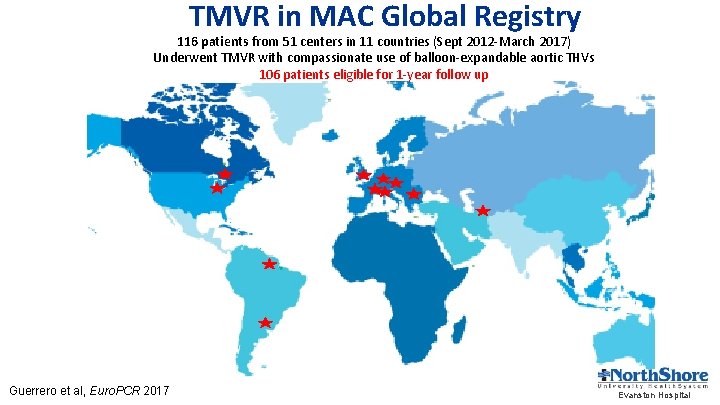
TMVR in MAC Global Registry 116 patients from 51 centers in 11 countries (Sept 2012 -March 2017) Underwent TMVR with compassionate use of balloon-expandable aortic THVs 106 patients eligible for 1 -year follow up Guerrero et al, Euro. PCR 2017 Evanston Hospital
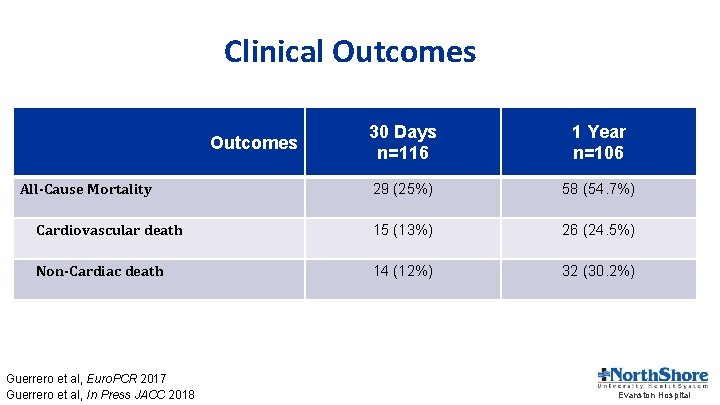
Clinical Outcomes 30 Days n=116 1 Year n=106 29 (25%) 58 (54. 7%) Cardiovascular death 15 (13%) 26 (24. 5%) Non-Cardiac death 14 (12%) 32 (30. 2%) Outcomes All-Cause Mortality Guerrero et al, Euro. PCR 2017 Guerrero et al, In Press JACC 2018 Evanston Hospital
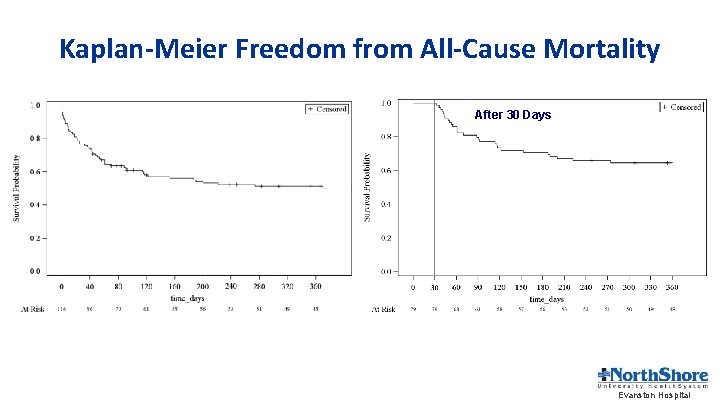
Kaplan-Meier Freedom from All-Cause Mortality After 30 Days Evanston Hospital
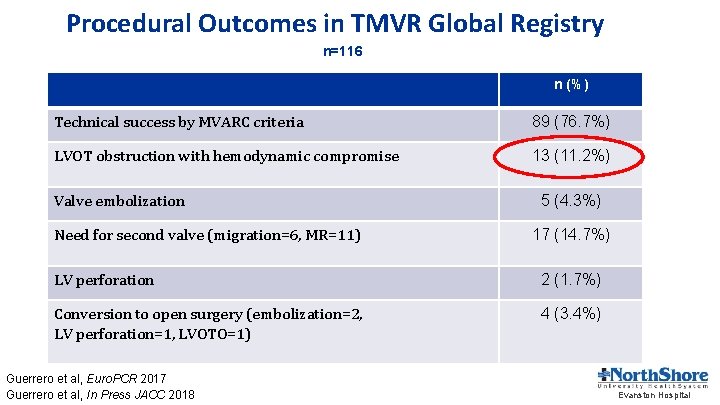
Procedural Outcomes in TMVR Global Registry n=116 n (%) Technical success by MVARC criteria 89 (76. 7%) LVOT obstruction with hemodynamic compromise 13 (11. 2%) Valve embolization Need for second valve (migration=6, MR=11) 5 (4. 3%) 17 (14. 7%) LV perforation 2 (1. 7%) Conversion to open surgery (embolization=2, LV perforation=1, LVOTO=1) 4 (3. 4%) Guerrero et al, Euro. PCR 2017 Guerrero et al, In Press JACC 2018 Evanston Hospital
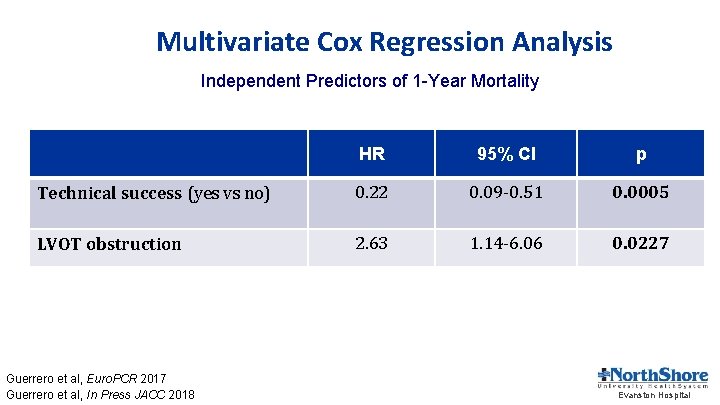
Multivariate Cox Regression Analysis Independent Predictors of 1 -Year Mortality HR 95% CI p Technical success (yes vs no) 0. 22 0. 09 -0. 51 0. 0005 LVOT obstruction 2. 63 1. 14 -6. 06 0. 0227 Guerrero et al, Euro. PCR 2017 Guerrero et al, In Press JACC 2018 Evanston Hospital
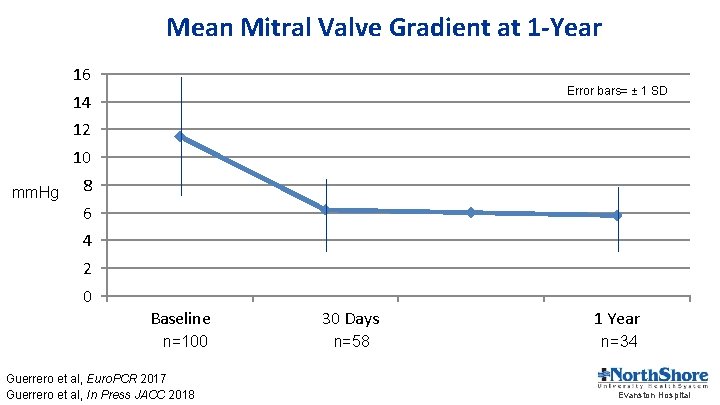
Mean Mitral Valve Gradient at 1 -Year 16 Error bars= ± 1 SD 14 12 10 mm. Hg 8 6 4 2 0 Baseline n=100 Guerrero et al, Euro. PCR 2017 Guerrero et al, In Press JACC 2018 30 Days 1 Year n=58 n=34 Evanston Hospital
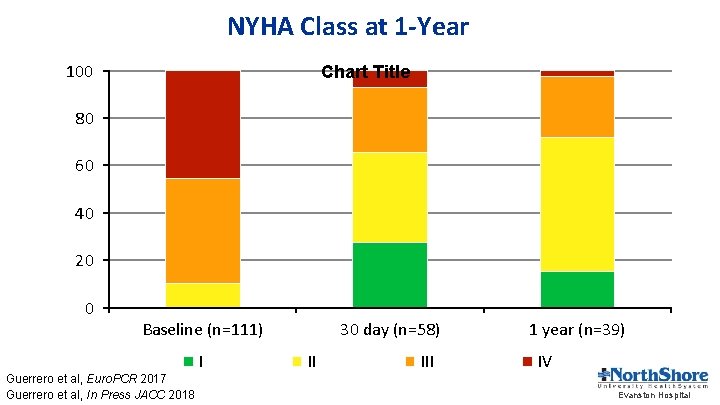
NYHA Class at 1 -Year 100 Chart Title 80 60 40 20 0 Baseline (n=111) I Guerrero et al, Euro. PCR 2017 Guerrero et al, In Press JACC 2018 30 day (n=58) II III 1 year (n=39) IV Evanston Hospital

2017 ESC/EACTS Guidelines for the management of valvular heart disease Baumgartner et al, Eur Heart J 2017; 00: 1 -53. Evanston Hospital
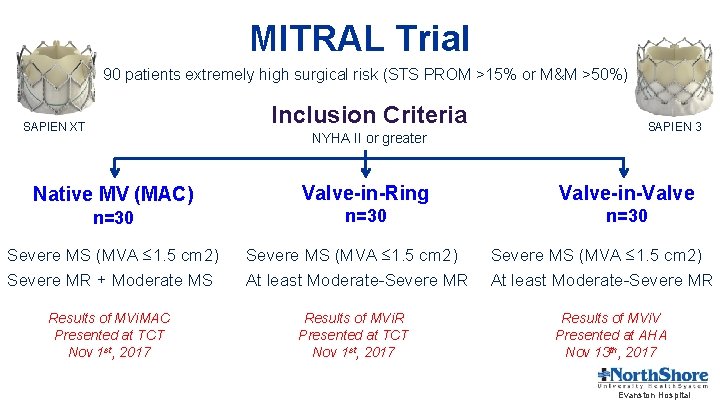
MITRAL Trial 90 patients extremely high surgical risk (STS PROM >15% or M&M >50%) Inclusion Criteria SAPIEN XT NYHA II or greater Native MV (MAC) n=30 SAPIEN 3 Valve-in-Ring Valve-in-Valve n=30 Severe MS (MVA ≤ 1. 5 cm 2) Severe MR + Moderate MS Severe MS (MVA ≤ 1. 5 cm 2) At least Moderate-Severe MR Results of MVi. MAC Presented at TCT Nov 1 st, 2017 Results of MVi. R Presented at TCT Nov 1 st, 2017 Severe MS (MVA ≤ 1. 5 cm 2) At least Moderate-Severe MR Results of MVi. V Presented at AHA Nov 13 th, 2017 Evanston Hospital
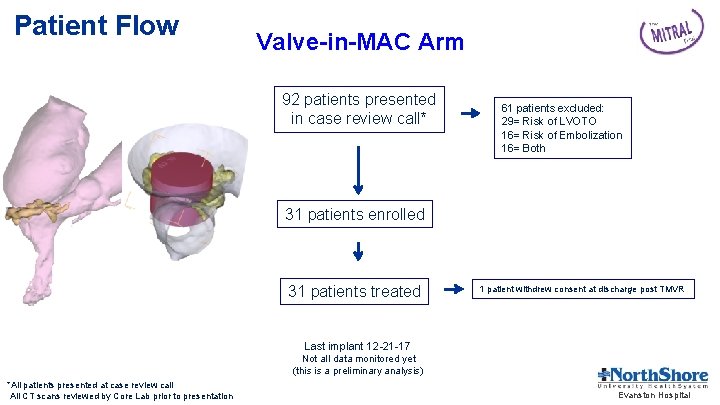
Patient Flow Valve-in-MAC Arm 92 patients presented in case review call* 61 patients excluded: 29= Risk of LVOTO 16= Risk of Embolization 16= Both 31 patients enrolled 31 patients treated 1 patient withdrew consent at discharge post TMVR Last implant 12 -21 -17 Not all data monitored yet (this is a preliminary analysis) *All patients presented at case review call All CT scans reviewed by Core Lab prior to presentation Evanston Hospital
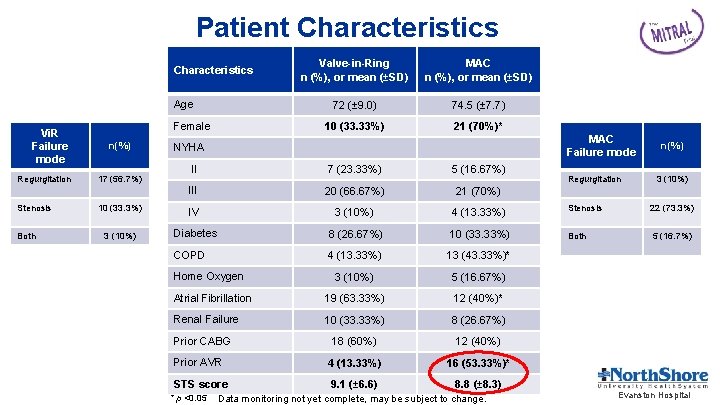
Patient Characteristics Age Vi. R Failure mode Regurgitation Stenosis Both Female n(%) Valve-in-Ring n (%), or mean (±SD) MAC n (%), or mean (±SD) 72 (± 9. 0) 74. 5 (± 7. 7) 10 (33. 33%) 21 (70%)* MAC Failure mode NYHA n(%) II 7 (23. 33%) 5 (16. 67%) III 20 (66. 67%) 21 (70%) 10 (33. 3%) IV 3 (10%) 4 (13. 33%) Stenosis 22 (73. 3%) 3 (10%) Diabetes 8 (26. 67%) 10 (33. 33%) Both 5 (16. 7%) COPD 4 (13. 33%) 13 (43. 33%)* 3 (10%) 5 (16. 67%) Atrial Fibrillation 19 (63. 33%) 12 (40%)* Renal Failure 10 (33. 33%) 8 (26. 67%) 18 (60%) 12 (40%) Prior AVR 4 (13. 33%) 16 (53. 33%)* STS score 9. 1 (± 6. 6) 8. 8 (± 8. 3) 17 (56. 7%) Home Oxygen Prior CABG * p <0. 05 Data monitoring not yet complete, may be subject to change. Regurgitation 3 (10%) Evanston Hospital
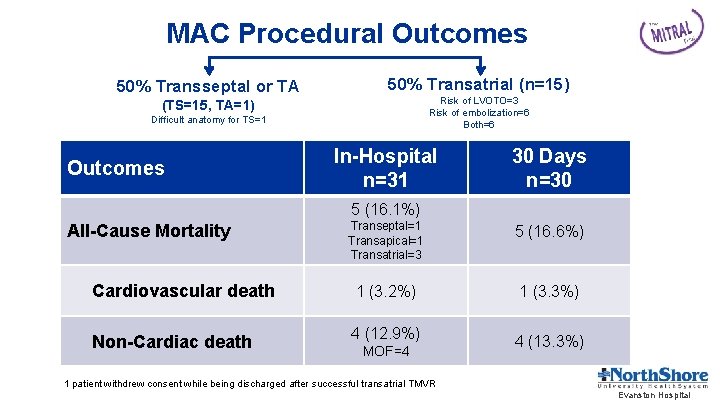
MAC Procedural Outcomes 50% Transseptal or TA 50% Transatrial (n=15) Risk of LVOTO=3 Risk of embolization=6 Both=6 (TS=15, TA=1) Difficult anatomy for TS=1 Outcomes In-Hospital n=31 30 Days n=30 5 (16. 1%) Transeptal=1 Transapical=1 Transatrial=3 5 (16. 6%) Cardiovascular death 1 (3. 2%) 1 (3. 3%) Non-Cardiac death 4 (12. 9%) All-Cause Mortality MOF=4 4 (13. 3%) 1 patient withdrew consent while being discharged after successful transatrial TMVR Evanston Hospital

Role of Alcohol Septal Ablation 16 non-transatrial TMVRprocedures Transseptal=15, Transapical=1 1 st TMVR in the trial (TS) was complicated with LVOTO Treated with bail-out alcohol ablation (Dr. O’Neill) 2 st TMVR in the trial (TA) was complicated with LVOTO Treated with bail-out alcohol ablation at Evanston Hospital LVOT gradient recurred the following day Bail out Proof of concept Generated concept of Preemptive ablation weeks prior to TMVR 14 additional transseptal TMVR procedures (7 pretreated with alcohol septal ablation weeks prior to TMVR) 100% discharged from the hospital 100% alive at 30 days Evanston Hospital
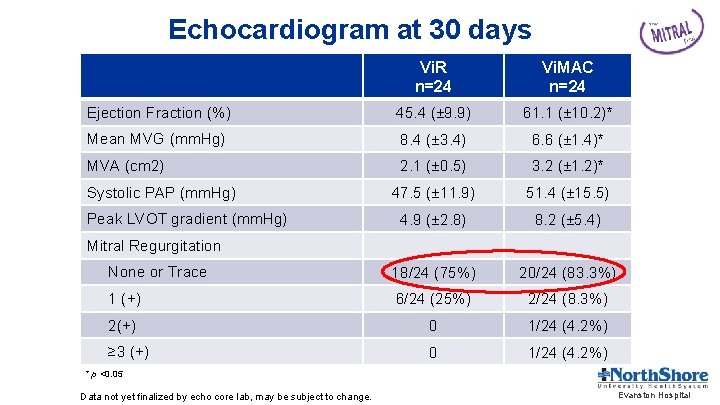
Echocardiogram at 30 days Vi. R n=24 Vi. MAC n=24 Ejection Fraction (%) 45. 4 (± 9. 9) 61. 1 (± 10. 2)* Mean MVG (mm. Hg) 8. 4 (± 3. 4) 6. 6 (± 1. 4)* MVA (cm 2) 2. 1 (± 0. 5) 3. 2 (± 1. 2)* 47. 5 (± 11. 9) 51. 4 (± 15. 5) 4. 9 (± 2. 8) 8. 2 (± 5. 4) None or Trace 18/24 (75%) 20/24 (83. 3%) 1 (+) 6/24 (25%) 2/24 (8. 3%) 2(+) 0 1/24 (4. 2%) ≥ 3 (+) 0 1/24 (4. 2%) Systolic PAP (mm. Hg) Peak LVOT gradient (mm. Hg) Mitral Regurgitation * p <0. 05 Data not yet finalized by echo core lab, may be subject to change. Evanston Hospital
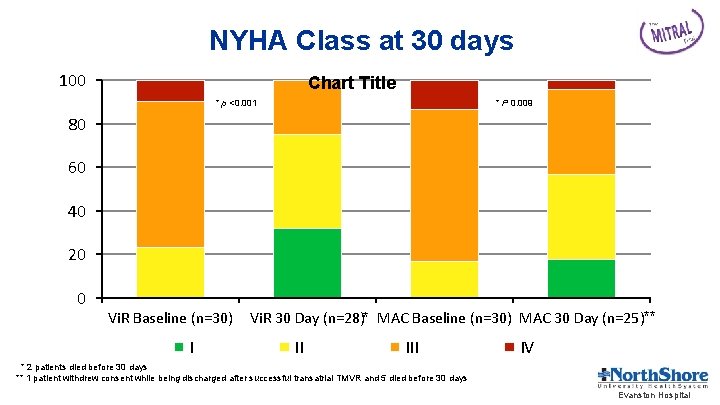
NYHA Class at 30 days 100 Chart Title * p <0. 001 * P 0. 009 80 60 40 20 0 Vi. R Baseline (n=30) I Vi. R 30 Day (n=28)* MAC Baseline (n=30) MAC 30 Day (n=25)** II IV * 2 patients died before 30 days ** 1 patient withdrew consent while being discharged after successful transatrial TMVR and 5 died before 30 days Evanston Hospital
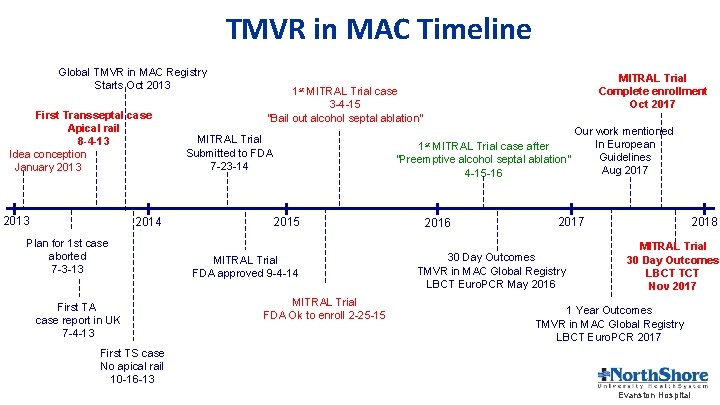
TMVR in MAC Timeline Global TMVR in MAC Registry Starts Oct 2013 First Transseptal case Apical rail 8 -4 -13 Idea conception January 2013 2014 Plan for 1 st case aborted 7 -3 -13 First TA case report in UK 7 -4 -13 MITRAL Trial Complete enrollment Oct 2017 1 st MITRAL Trial case 3 -4 -15 “Bail out alcohol septal ablation” MITRAL Trial Submitted to FDA 7 -23 -14 2015 MITRAL Trial FDA approved 9 -4 -14 MITRAL Trial FDA Ok to enroll 2 -25 -15 Our work mentioned In European MITRAL Trial case after Guidelines “Preemptive alcohol septal ablation” Aug 2017 4 -15 -16 1 st 2016 2018 2017 30 Day Outcomes TMVR in MAC Global Registry LBCT Euro. PCR May 2016 MITRAL Trial 30 Day Outcomes LBCT TCT Nov 2017 1 Year Outcomes TMVR in MAC Global Registry LBCT Euro. PCR 2017 First TS case No apical rail 10 -16 -13 Evanston Hospital
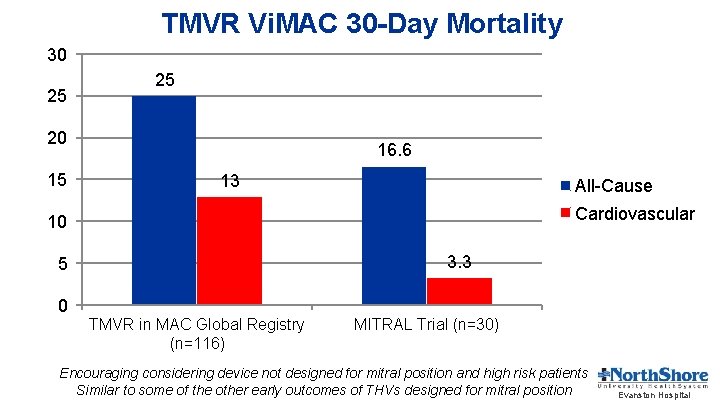
TMVR Vi. MAC 30 -Day Mortality 30 25 25 20 15 16. 6 13 All-Cause Cardiovascular 10 3. 3 5 0 TMVR in MAC Global Registry (n=116) MITRAL Trial (n=30) Encouraging considering device not designed for mitral position and high risk patients Similar to some of the other early outcomes of THVs designed for mitral position Evanston Hospital

SITRAL Trial Surgical Implantation of TRAnscatheter va. Lve in Native MAC Baylor Research Institute PI Robert Smith, MD 30 patients Start April 2016 Transatrial TMVR with SAPIEN 3 www. clinicaltrials. gov Evanston Hospital
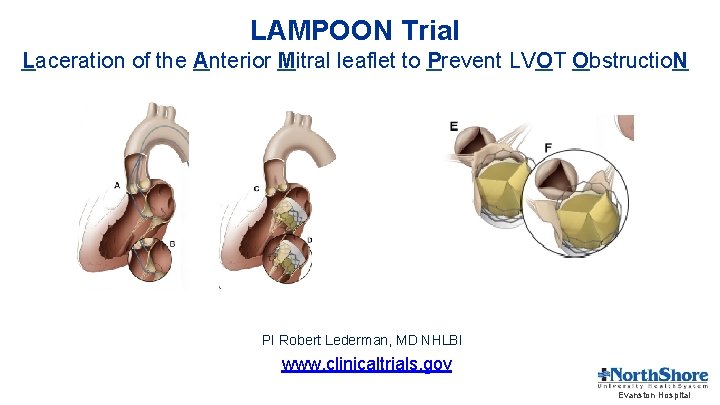
LAMPOON Trial Laceration of the Anterior Mitral leaflet to Prevent LVOT Obstructio. N PI Robert Lederman, MD NHLBI www. clinicaltrials. gov Evanston Hospital

Next Steps Short-term goals MITRAL trial expansion Further improve patient selection and overall outcomes Standardize techniques Evanston Hospital

Next Steps Short-term goals MITRAL trial expansion Further improve patient selection and overall outcomes Standardize techniques Long-term goals FDA approval for TMVR in MAC Change in guidelines Evanston Hospital
New TMVR in MAC Timeline Change in Guidelines FDA Approval 201? TVT data Post-Approval 20? ? Evanston Hospital

Conclusions TMVR in MAC is feasible but challenging and associated with complications Outcomes have improved with better patient selection and techniques Most patients have high risk of LVOTO and require risk reduction strategies Pre-emptive alcohol septal ablation facilitates successful TS TMVR in selected patients Cardiac CT analysis is key to improve patient selection and outcomes Transatrial approach allows resection of anterior leaflet to decrease LVOTO risk and sutures to decrease embolization risk, but is more invasive and associated with M&M Vi. MAC may become a reasonable alternative for high surgical risk patients with favorable anatomy, but techniques require further refinement Evanston Hospital
- Slides: 25