IMPLANTABLE ORTHOPEDIC SENSORS Group 4 Derek Sheesley Sunil
IMPLANTABLE ORTHOPEDIC SENSORS Group 4 Derek Sheesley Sunil Shah Michael Iskhakov Marina Louis Anthony Jones

Design Goal � � � Issues arise within prostheses without the doctors or wearers knowledge Estimations ok but more exact measurements needed Sensor that will [6]: � Give real time information � Alert the wearer when a problem arises
![Constraints � Size � 1 cm 3 � Wireless sensor [6] 2 -5 millimeters Constraints � Size � 1 cm 3 � Wireless sensor [6] 2 -5 millimeters](http://slidetodoc.com/presentation_image/60e5561dce82f5bc916963ec901b2f2f/image-3.jpg)
Constraints � Size � 1 cm 3 � Wireless sensor [6] 2 -5 millimeters across 500 microns thick � Biocompatibility (must be…) � compatible with implant and body micro-disc electrode arrays, poly hema hydrogels[3] � capable of withstanding forces in body

Constraints � � � Risk/Benefit Factor � Biosensor must be made so that the risk of failure, as well as risk of further damage, is reduced Minimally Invasive � Surgery should be kept minimal � Use of ultra-thin flexible biosensors (<100 micrometers thick) to reduce size of incisions and risk of injury during surgery [5] Affordable � Must be relatively cheap and be able to be mass-produced
![Criteria � Strong, biocompatible material � Stainless steel, titanium[2] � Non-biocompatible materials coated with Criteria � Strong, biocompatible material � Stainless steel, titanium[2] � Non-biocompatible materials coated with](http://slidetodoc.com/presentation_image/60e5561dce82f5bc916963ec901b2f2f/image-5.jpg)
Criteria � Strong, biocompatible material � Stainless steel, titanium[2] � Non-biocompatible materials coated with PEG PDMS [3] � Collection � Good accuracy and precision � Reduce noise with gyroscopic system [5] � Telemetric data collection [6] or

Criteria � Monitor state of implant and surrounding area � Temperature, p. H, force, pressure, etc. � Early warning system � Power Supplies � Kinetic and Thermoelectric energy harvesters [1] � Single inductor-capacitor component [4] � Wireless (no electrical components)

WEAKNESSES FDA rationale: High risk to benefit ratio • Highly Invasive • Number of parameters measured • Signal Collection a)Noise [5] b)Patient Confidentiality •

STRENGTHS
Accurate Diagnostics
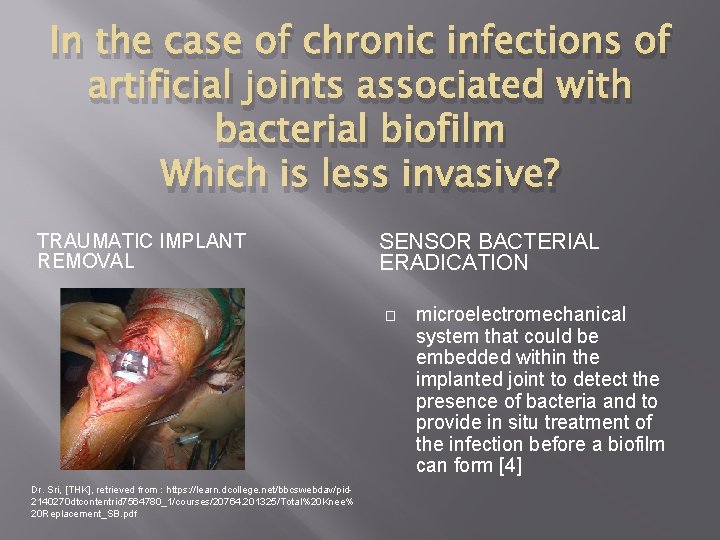
In the case of chronic infections of artificial joints associated with bacterial biofilm Which is less invasive? TRAUMATIC IMPLANT REMOVAL SENSOR BACTERIAL ERADICATION � Dr. Sri, [THK], retrieved from : https: //learn. dcollege. net/bbcswebdav/pid 2140270 dtcontentrid 7564780_1/courses/20764. 201325/Total%20 Knee% 20 Replacement_SB. pdf microelectromechanical system that could be embedded within the implanted joint to detect the presence of bacteria and to provide in situ treatment of the infection before a biofilm can form [4]

Signal Collection Is Patient confidentiality at risk when a form of wireless communication is used to read a sensor?

Conclusion � Incorporate a sensor into orthopedic prosthetics that will monitor their condition as they are being used by patients. � Example Prosthetics such as: Knee, Hip, Spine. � Sensors to record and transmit readings in concentration changes of specific substances present in surrounding blood and tissue. [8] � Chemicals, Biological substances, Ions, etc. � Sensor can’t be more invasive than its partner prosthetic; must be compatible both with orthopedic implant and patient. � Side effects of transmitting sensor can’t compromise the benefits of implant

Conclusion � Transmission must be coherent, accurate and precise. � Possible depending on coating of sensor, such as a functionalized polymer coasting[8] � Ultimately, sensor will aid in gathering data to design a more efficient prosthetic.
![SOURCES [1] Andrea Cadei, et al, . 2013. Kinetic and thermal energy harvesters for SOURCES [1] Andrea Cadei, et al, . 2013. Kinetic and thermal energy harvesters for](http://slidetodoc.com/presentation_image/60e5561dce82f5bc916963ec901b2f2f/image-14.jpg)
SOURCES [1] Andrea Cadei, et al, . 2013. Kinetic and thermal energy harvesters for implantable medical devices and biomedical autonomous sensors. Measurement Science and Technology, vol 25. Retrieved from: http: //iopscience. iop. org/0957 -0233/25/1/012003/pdf/09570233_25_1_012003. pdf [2] Ehrlich G. et al, . 2006. Engineering Approaches for the Detection and Control of Orthopaedic Biofilm Infections. National Institute of Health (437): 59– 66. Retrieved from: http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 151327 [3] Gusphyl Justin, et al, . 2008. Biometric hydrogels for biosensor implant biocompatibility: electrochemical characterization using micro-disc electrode arrays(MDEAs). Biomed Microdevices, vol 11: 103 -115. [4] Rensselaer Polytechnic Institute, 2012. Implantable, Wireless Sensors Share Secres of Healing Tissues. Retrieved from: http: //search. proquest. com/docview/922474572 [5] Sirivisot S. et al, . 2006. Developing Biosensors for Monitoring Orthopedic Tissue Growth. Material Research Society, vol. 950. Retrieved from: http: //journals. cambridge. org/action/display. Abstract? from. Page=online&aid=8014 891 [6] Su-Jin K. et al, . 2012. Evaluation of the biompatibility of a coating material for an implantable bladder volume sensor. The Kaohsiung Journal of Medical Sciences, vol. 28, Issue 3: 123 -129. Retrieved from: http: //www. sciencedirect. com/science/article/pii/S 1607551 X 11002397
![SOURCES [7] Umbrecht, et at, . 2010. Wireless implantable passive strain sensor: design, fabrication SOURCES [7] Umbrecht, et at, . 2010. Wireless implantable passive strain sensor: design, fabrication](http://slidetodoc.com/presentation_image/60e5561dce82f5bc916963ec901b2f2f/image-15.jpg)
SOURCES [7] Umbrecht, et at, . 2010. Wireless implantable passive strain sensor: design, fabrication and characterization. Journal of Micromechanics and Microengineering vol. 20, 14. Retrieved from: http: //iopscience. iop. org/0960 -1317/20/8/085005/pdf/0960 -1317_20_8_085005. pdf [8] Guenther M. , Gerlach G. , et al. 2008. Hydrogel-based Sensor for a Rheochemical Characterization of Solutions. Sensors and Actuators B: Chemical. Transducers '07/Eurosensors XXI. Volume 132, Issue 2, 16 June 2008, Pages 471– 476. Available: http: //www. sciencedirect. com/science/article/pii/S 0925400507009094

Questions?
- Slides: 16