Implantable Devices Katrina Hoffman FNP ACHPN Samaritan Evergreen

Implantable Devices Katrina Hoffman FNP, ACHPN Samaritan Evergreen Hospice Fall 2018

Objectives • Identify legal and ethical considerations in removing implanted devices at the end of life. • Identify methods used for both planned and urgent implanted device deactivation. • Define different methods to reduce prosthetic parts after death. • Recognize the need for more information on LVAD device discontinuation at end of life.

Outline • Definition of Implantable Devices • Pacemakers and ICD’s • Basics of devices • How to deactivate ICD and pacemaker in the home • Implantable device and Prosthetic recovery after death • What types of implantable or prosthetic devices can we reuse and how? • LVAD (Left Ventricular Assist Device) • Basics of LVAD’s • How to go about deactivating the device at time of death

Wikepedia Definition: Implantable Devices An implant is a medical device manufactured to replace a Missing biological structure Support a damaged biological structure Enhance an existing biological structure Medical implants are man-made devices, in contrast to a transplant, which is a transplanted biomedical tissue.

Pacemakers and ICD (implantable cardioverter defibrillator) devices Pacemaker and ICD’s 1. Basics of devices 2. How to deactivate ICD and more rarely Pacemaker 3. What happens after pacemaker is sent to mortuary. Prosthetic device recovery LVAD 1. Basics of devices 2. How to deactivate at end of life
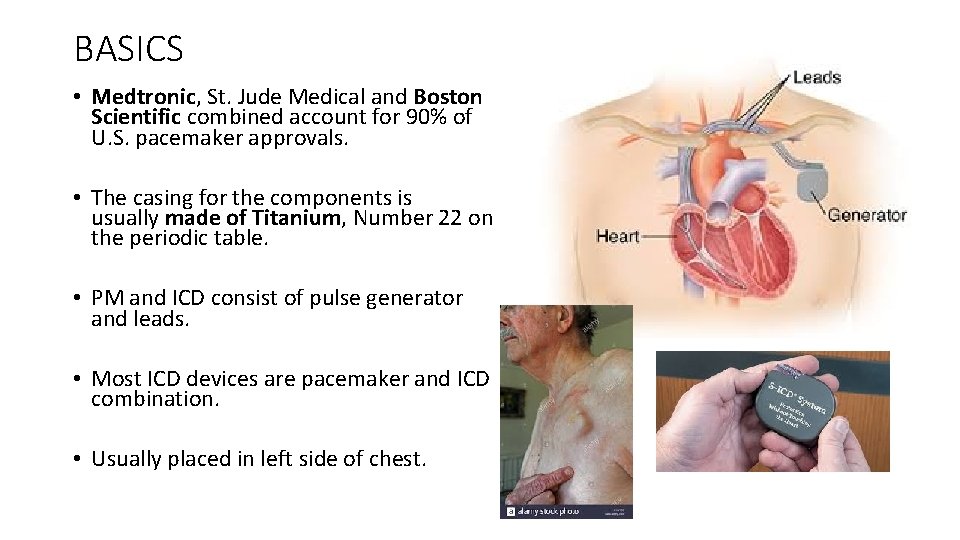
BASICS • Medtronic, St. Jude Medical and Boston Scientific combined account for 90% of U. S. pacemaker approvals. • The casing for the components is usually made of Titanium, Number 22 on the periodic table. • PM and ICD consist of pulse generator and leads. • Most ICD devices are pacemaker and ICD combination. • Usually placed in left side of chest.
ICD An ICD is larger in size than a regular pacemaker and many ICD models include all the features of a regular pacemaker. How ICDs Work • Continually detect the R-R interval. • If an ICD detects too many short R-R intervals, it decides between antitachycardia pacing (less battery use, more tolerable for the patient) or shock. • Charging time is 6 -15 seconds, after which many ICDs will reconfirm an arrhythmia prior to actually shocking. • 10% of shocks are due to a non-VF non-VT rhythm, most commonly due to SVT.

End of Life Deactivation • Results of a survey of 414 American hospices published in 2010 in Annals of Internal Medicine showed that a little less than half (42%) of hospice patients had the shock function of their ICDs turned off, and a little more than half (58%) of the hospices reported that at least one of their patients had been shocked by his or her ICD in the past year. • Only 10% of the hospices had a policy addressing ICD deactivation. • Policy Model for ICD deactivation on this website: www. health. harvard. edu/healthextra

Pacemaker deactivation While there may be more agreement about the deactivation of implantable cardioverter-defibrillators than of pacemakers amongst practicing clinicians, the Heart Rhythm Society issued a consensus statement in collaboration with many professional groups (including the American College of Cardiology, the American Academy of Hospice and Palliative Medicine, and the American Heart Association) which effectively erased any ethical distinction between types of implanted devices being deactivated and endorsed a patient’s right to have one’s pacemaker deactivated.

Deactivation of ICD • A new battery usually lasts between 4 -7 years. Most patients who have received shocks from their ICDs describe them as startling, jolting and unsettling, but not painful • Some people say it feels like being hit with a baseball bat or being kicked by a horse. Most people feel it more in their back than their chest.

Informed Consent Pearls • Turning off the ICD will not cause death. • Deactivating the defibrillator (shocking) function of the ICD does not deactivate the pacemaker function of the ICD. • Deactivating the ICD will not be painful and dying will not be more painful if the device is turned off. • If the patient’s circumstances change following the deactivation, the patient can request re-activation. • Discussions relating to ICD deactivation should be initiated early, rather than at the terminal stage. • There may be a logistical delay between the request for deactivation and this process being carried out. • Delivery of shocks near the end of life may be ineffective, painful to the patient and distressing to patients, their care givers and relatives.

How to deactivate ICD • Contact cardiology clinic for deactivation or pacemaker rep. IF URGENT • All pacemakers respond to a magnet by switching to an asynchronous pacing mode at a programmed atrioventricular (AV) delay and a fixed magnet rate depending on the manufacturer, device model, and the status of the battery. • The programmed mode DDD switches to DOO, VI switches to VOO and AAI switches to AOO

Medical Magnets can be found at cardiology clinics, ER, OR, or critical care settings. Bar shaped or more likely donut shaped Can be purchased online on Amazon, AAA Wholesale, American Science & Surplus, Medtronic, St. Jude, or e. Bay. Usually 10 -20 dollars Strength of magnet is 90 Gauss-The gauss, abbreviated as G or Gs, is the cgs unit of measurement of magnetic flux density (or “magnetic induction”)

Auditory or Vibratory Alerts • Implantable cardioverter defibrillators have either an auditory or a vibratory alert feature; only Medtronic and Boston Scientific ICDs provide an auditory confirmation of the suspension of anti-tachycardia therapy on magnet application. • If a tone is not heard, a stethoscope may be used to identify auditory response, particularly in obese patients and in those with sub-muscular implantations. • In Medtronic or Boston Scientific devices, if no tone is heard even after repositioning the magnet: • a depleted battery; • all the alerts are programmed to remain ‘OFF', as in Medtronic devices; • the device is programmed to ignore the magnet, as in Boston Scientific devices, • the magnetic field does not reach the device due to sheer depth, as in the case of those with deeper (abdominal or submuscular) implants or in very obese patients.

Boston Scientific- Tachy Mode • Magnet changes the tachy mode on certain models • On-Off: Magnet held on pacer 30 seconds • Beep tone for 30 seconds then long tone Permanent Off • OK to take off magnet • Beeping longer than 30 seconds Tachy therapy off when magnet stays in place. • Long beep means already programmed to OFF

Biotronik • Devices from one manufacturer (Biotronik) will be inhibited by a magnet for only 8 hours. • Biotronik device (or if the manufacturer is unknown) the magnet must be removed for a few seconds and then reapplied every 7 hours. • When a magnet is used for emergency deactivation, arrangements should be made as soon as is practicable for definitive deactivation, using a programmer.

Pacemaker and ICD after death • Pacemakers and ICDs are often taken out of the body after death but can be buried if family chooses • These devices are always removed before cremation because the batteries can explode when heated. • The same goes for spinal cord stimulators that treat pain and some types of internal pumps for administering drugs, since they contain electronics too.

Pacemakers Recycled • Until recently, nearly all pacemakers removed for cremation were simply thrown away as medical waste. • US and UK have rules against reusing medical waste.

Recycle • At $4, 000 for a pacemaker and $20, 000 for an ICD, a second-hand implant is the only way that millions of people will be able to afford this life-saving equipment. • Charities like Heartbeat International, World Medical Relief Inc, and Pace 4 Life recycle used pacemakers for use worldwide. www. heartbeatsaveslives. org • The journal Annals of Internal Medicine recently published the results of a US program called: • “Project My Heart Your Heart” • 75 patients who received second-hand ICDs showed no evidence of infection or malfunction. • The group are now applying for FDA approval to send recycled heart devices overseas.

Cremation and Metals in the body • Cremation is the process that converts human remains to ashes and mineral fragments through combustion and oxidation. • The furnace used to disintegrate the corpse should reach temperatures up to 1, 800 degrees Fahrenheit. • The melting points of certain metals commonly used by dentist and doctors are: Gold: 1, 947 degrees Fahrenheit Titanium: 3, 034 degrees Fahrenheit Steel: 2, 500 degrees Fahrenheit Amalgam (mercury, silver, tin, copper, etc. ): Varied

Metal Recycling • The gold used in tooth fillings is often a “dental gold” alloy that will not survive the high temperatures of the furnace. It must be removed from the decedent’s teeth before the cremation process begins. A dentist must be called out to remove the gold filling. Sometimes the fee charged by the dentist is higher than the value of the dental gold extracted. • Any other metal that survives the cremation furnace is usually recycled. • The metals that withstand the heat of the cremation are removed • After that, the methods of disposal differ by crematory but may include donation or sale to a medical recycling company. • Usually free of charge • Metal is sold and portion of proceeds returned to crematorium or donated to local charity. • Materials collected by them are not sold into markets that will reuse them for orthopedic purposes without being recycled and re-melted first. • If a metal recycling company is not used, the metal may be buried or discarded. • Find out more at Ortho Metals or Implant Recycling websites

Standing with Hope Project • Prosthetic limbs to Ghana. • The Metro Davidson County Detention Facility
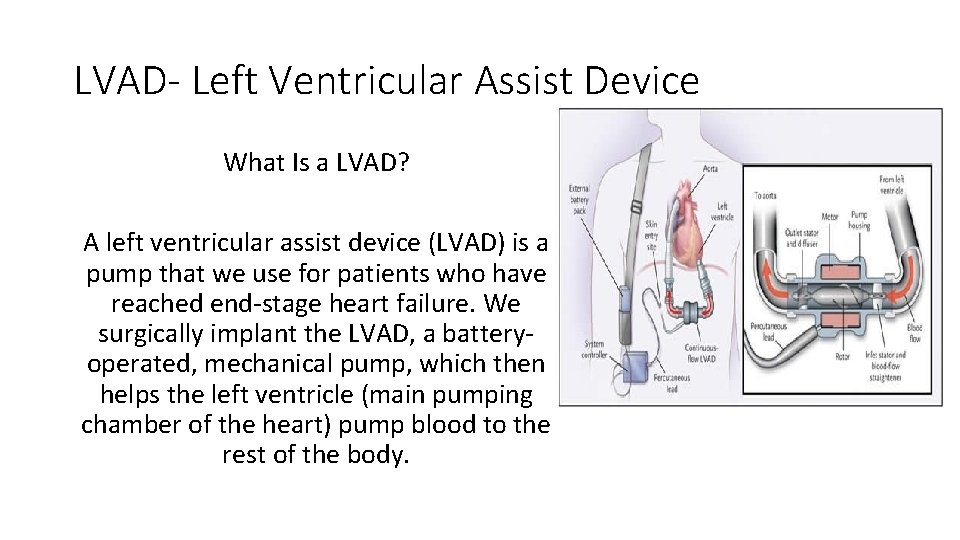
LVAD- Left Ventricular Assist Device What Is a LVAD? A left ventricular assist device (LVAD) is a pump that we use for patients who have reached end-stage heart failure. We surgically implant the LVAD, a batteryoperated, mechanical pump, which then helps the left ventricle (main pumping chamber of the heart) pump blood to the rest of the body.
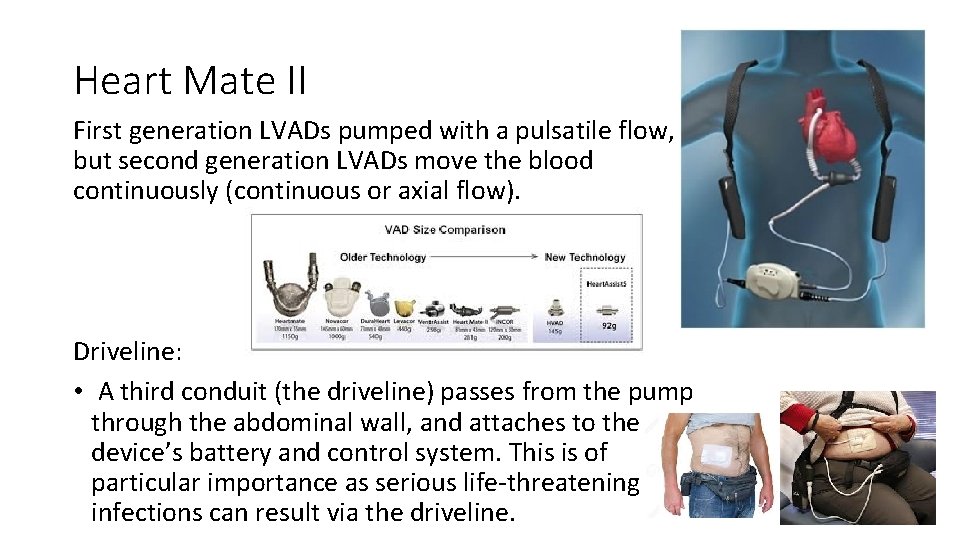
Heart Mate II First generation LVADs pumped with a pulsatile flow, but second generation LVADs move the blood continuously (continuous or axial flow). Driveline: • A third conduit (the driveline) passes from the pump through the abdominal wall, and attaches to the device’s battery and control system. This is of particular importance as serious life-threatening infections can result via the driveline.

MCS Mechanical circulatory support • • LVAD RVAD BIVAD TAH- Total artificial heart • Fast Fact #296

Who are eligible for LVAD End stage heart failure (stage D) patients refractory to medical management: • Used as a bridge to heart transplant (BT) • Restoration of a failing heart • Destination therapy (DT)(an alternative to transplant) who are not eligible / unwilling to undergo a heart transplant • Those that were BT but no longer are candidate then become DT status

Why LVAD? 1. Bridge-to-transplant therapy: This is a lifesaving therapy for patients awaiting a heart transplant. Patients use the LVAD until a heart becomes available. In some cases, the LVAD is able to restore the failing heart, eliminating the need for a transplant. 2. Destination therapy: Some patients are not candidates for heart transplants. In this case, patients can receive long-term treatment using an LVAD, which can prolong and improve patients' lives.
Statistics • Currently, approximately 5, 000 to 6, 000 patients are treated annually with LVADs. Currently, the number of LVAD implantations exceeds the number of annual heart transplantations worldwide • 2 year survival is 70% • The mean age of the Medicare beneficiaries was 61 years old, while 78 percent were male and 80 percent were white • The model projected that LVADs cost $726, 200 over a six-year period. The readmissions costs after LVAD implantation were$268, 700, the cost of outpatient care was $219, 500, the cost of device implantation was $175, 400 and the cost of heart transplantation was $62, 600.

What Are the Benefits and Risks of LVAD Implantation? Benefits: A better quality of life (less fatigue, more strength, and better breathing) and longer survival. 79% improve NYHA from IV to 1 and health related QOL is improved 79% are alive at 2 yrs. Some VAD patients have been on support for up to seven years. Complications related to LVAD therapy: • Bleeding • Gut 22%, Brain • • Clot Stroke-25% Infection (driveline) 10 -21% some say 49% Sepsis 36% Rehospitalization 94% Right heart dysfunction Hemolysis or Thrombosis of pump 10% High degree of ongoing medical care and device care

Conditions leading to VAD deactivation • Thrombotic / Hemorrhagic stroke effecting quality of life • Overwhelming Sepsis • Multi organ system failure • Poor quality of life despite LVAD treatment e. g. chronic infections, recurrent bleeding, revolving door hospitalizations • Development of serious secondary comorbidities e. g. cancer, advance dementia, renal failure needing HD

Ethical Considerations LVAD deactivation “at the request of a patient or surrogate is not considered physician-assisted death but rather allowing natural death to occur. ” Some legal scholars consider LVAD deactivation “a form of physicianaid-in-dying” or “an act of euthanasia” based on the surgical reconfiguration of the circulation. 1. A substantial number of clinicians also consider LVAD deactivation as a form of euthanasia

General moral consensus Ventricular assist devices carry the same moral weight of other lifesustaining or extending technology. Therefore, a decision capable patient and/or their designated surrogate decision maker have the moral right to choose to discontinue such technology’s use.

Ethical Principles When a decision is made to deactivate the LVAD or if the patient (or surrogate decision maker) requests to have a device turned off, principles of autonomy and self-determination dictate that it should be turned off. Failure to do so is legally termed “assault” Makdisi, Tony, and George Makdisi. “Ethical Challenges and Terminal Deactivation of Left Ventricular Assist Device. ” Annals of Translational Medicine 5. 16 (2017): 331. PMC. Web. 10 Sept. 2018.

Important Steps prior to Deactivation • Confirm and document DNR and DNI status • Confirm the treatment plan goals • Address any patient/family or professional ethical concerns • Make direct recommendations about what treatments should be stopped, instead of asking families treatment-by-treatment what they want • Reassured that patients can be kept comfortable without life sustaining treatments. • Allow ample opportunities of visitation for families and loved ones

(Cont’d) Important Steps prior to Deactivation of VADs • Describe what can be expected after deactivation • Inform life expectancy (minutes to days) to patient and family in advance • Discontinue all non-symptom-directed monitoring • Establish adequate symptom control prior to deactivation • Administer comfort medications prior to deactivation to ensure adequate circulation of the drugs • Discontinue other life-sustaining treatments (e. g. artificial nutrition/hydration, antibiotics, dialysis, ventilator)

Medication Protocol prior to Deactivation of VADs • IV bolus dose of an opioid (i. e. morphine 2 -10 mg IV) for rapid onset of dyspnea/labored respirations • IV bolus dose of a benzodiazepine (lorazepam 1 -2 mg IV) if anxiety is anticipated. • Consider an IV continuous infusion of sedating medication • Titrate medications to control labored respirations and achieve the desired state of sedation prior to deactivation. • Have additional medication drawn up and ready to administer at the bedside if needed. • If distress is noted, utilize additional bolus doses of opioids and benzodiazepines (e. g. morphine 5 -10 mg IV push q 10 min, and/or midazolam, 2 -4 mg IV push q 10 min, until distress is relieved). • The symptom relief is more important than specific dose. A goal should be to keep the respiratory rate < 20 and eliminate grimacing, agitation, and

How to Deactivate LVADs • Unscrew small black nickel-sized battery in “System Driver” (also called controller) to disable back up alarms. • Press alarm silence button on System Driver (controller). • Remove power from controller by removing both cables coming from the main power base unit (simultaneous removal of both cables will limit alarms). • Detach controller from patient (cord going from LVAD driveline to percutaneous cable. • Move quick as alarms are not disabled for long- Low Flow alarm and low battery.

References Kramer, D, Mitchell, S. , and Brock, D. “Deactivation of Pacemakers and Implantable Cardioverter. Defibrillators. ” Prog Carciovasc Dis (2012): 290 -299. Makdisi, Tony, and George Makdisi. “Ethical Challenges and Terminal Deactivation of Left Ventricular Assist Device. ” Annals of Translational Medicine 5. 16 (2017): 331. PMC. Web. 10 Sept. 2018. Mc. Ilvennan, CK, et al. "Deactivation of Left Ventricular Assist Devices: Differing Perspectives of Cardiology and Hospice/Palliative Medicine Clinicians. " Journal Cardiology Failure (2016): 708 -712. Mohamed, Rady, J Verheijde and MB BChir. "End-Of-Life Care and Deactivation of left ventricular assist devices. " Critical Care Medicine 42. 7 (2014). Palliative Care Network of Wisconsin. Mechanical Circulatory support in advanced heart failure #205. Fast Facts and Concepts. 2017. Palliative Care Network of Wisconsin. " Cardiac Pacemakers at the end of Life #111. " 2017. Fast Facts and Concepts. Panke, J, George Ruiz and Tonya Elliott. "Discontinuation of a left ventricular assist device in the home hospice setting. " Journal of Pain and Symptom Management 52. 2 (2016): 313 -317. Shreibati, B, et al. "Cost-effectiveness of left ventricular assist devices in Ambulatory Patients with advanced heart failure. " JACC Heart Failure (2017): 110 -119. Smith, Elisa M. , and Jennifer Franzwa. “Chronic Outpatient Management of Patients with a Left Ventricular Assist Device. ” Journal of Thoracic Disease 7. 12 (2015): 2112– 2124. PMC. Web. 10 Sept. 2018.
- Slides: 38