Implantable Devices for Pulmonary Hypertension William T Abraham

Implantable Devices for Pulmonary Hypertension William T. Abraham, MD, FACP, FACC, FAHA, FESC, FRCP Professor of Medicine, Physiology, and Cell Biology College of Medicine Distinguished Professor The Ohio State University Columbus, Ohio

William T Abraham, MD Employee, Stockholder: V-Wave Medical

Pulmonary Arterial Hypertension (PAH) • • • Prevalence: 30, 000 U. S. patients Incidence: 3, 500 – 5, 250 new U. S. cases /year Average at diagnosis: 36 years Female predominance: 2: 1 Median survival: 5 years Pathology: intimal proliferation/inflammation and plexiform lesion development. End-stage with thrombosis and vessel obliteration. • Treatment: No Cure; only palliation; minor impact on survival • • • Pharmacological Treatment Expensive: ~$100 K-150 K USD annually Balloon atrial septostomy (BAS) – guideline indicated treatment but low adoption due to limited clinical data, and high incidence of septostomy closure Lung transplantation 3% 5% 6% 10% Schistosomiasis HIV Heritable 25% Drugs / toxins Portal Hypertension CHD 47% CTD Idiopathic WHO Class % of population Class 1 10 -20% Class 2 40% Class 3 40% Class 4 5 -10%

PAH Patient Survival by Functional Class REVEAL Registry

Atrial Septostomy for PAH Rationale and Guidelines • • • Patients with Eisenmenger’s syndrome and patients with IPAH with a PFO have a survival advantage Recommended technique is graded intraprocedural balloon dilation atrial septostomy (BAS), based on multiple case series totaling ~300 patients with long-term follow-up BAS decompresses the RA/RV, increases LV preload and CO, improves systemic O 2 transport despite arterial desaturation, and increases 6 MWD No RCTs evaluating survival but patients survive average of 65 months One-third of BAS patients undergo retreatment after 6 months Other techniques, e. g. fenestrated ASD closure device, diabolo stent, etc. , are considered to be experimental due to very limited reporting and short follow-up and questionable long-term patency
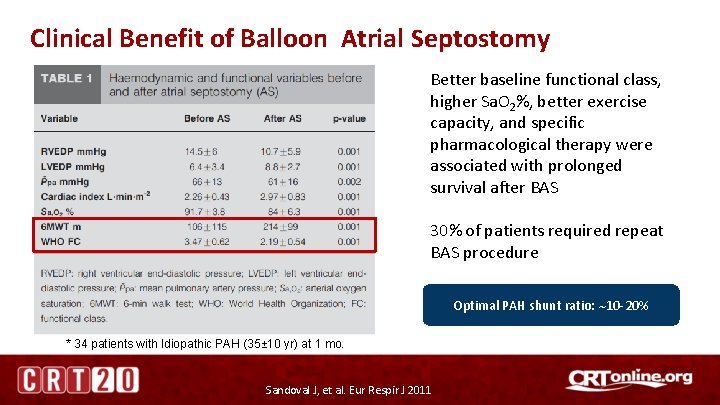
Clinical Benefit of Balloon Atrial Septostomy Better baseline functional class, higher Sa. O 2%, better exercise capacity, and specific pharmacological therapy were associated with prolonged survival after BAS 30% of patients required repeat BAS procedure Optimal PAH shunt ratio: 10 -20% * 34 patients with Idiopathic PAH (35± 10 yr) at 1 mo. Sandoval J, et al. Eur Respir J 2011

Devices for Atrial Septostomy

Inter-Atrial Shunt Devices

Ongoing Interatrial Shunt Device Trials in PAH • PROPHET Trial (clinicaltrials. gov identifier NCT 03022851) – OUS pilot open-label study – 30 PAH patients (includes pediatric patients) – Feasibility and safety study • RELIEVE-PAH Trial (clinicaltrials. gov identifier NCT 03838445) – North American FDA IDE Early Feasibility Study – 10 -15 patients – Feasibility and safety study
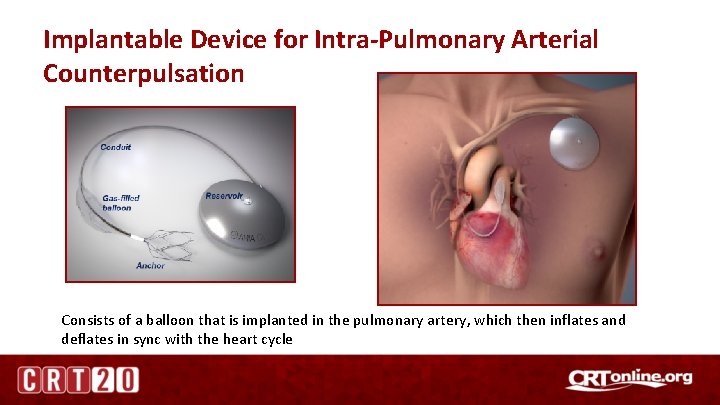
Implantable Device for Intra-Pulmonary Arterial Counterpulsation Consists of a balloon that is implanted in the pulmonary artery, which then inflates and deflates in sync with the heart cycle

First-in-Human Study Design • Medical University of Vienna (Irene Lang, MD) • Prospective, single-center, nonrandomized, single arm, acute • n = 10 subjects • Key inclusion criteria – WHO group 1 PH (PAH) – NYHA Functional Class III – Subjects on standard of care drug therapy • Endpoints – Safety – Efficacy
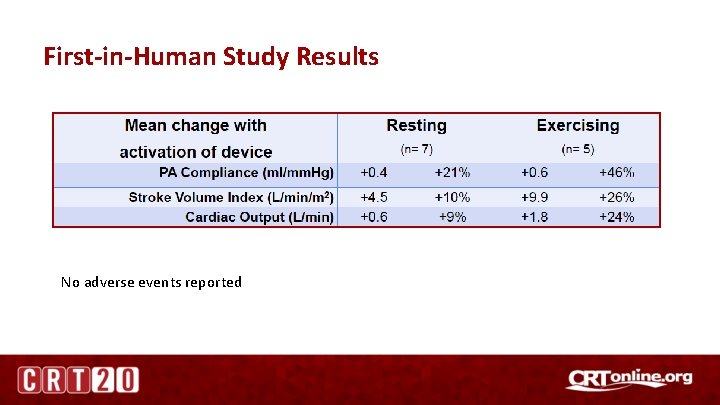
First-in-Human Study Results No adverse events reported
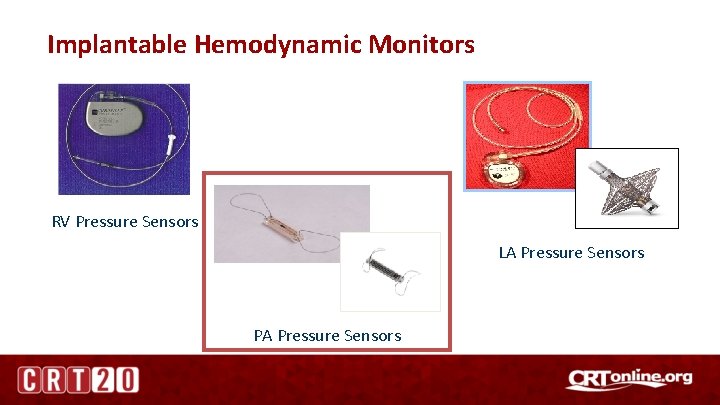
Implantable Hemodynamic Monitors RV Pressure Sensors LA Pressure Sensors PA Pressure Sensors
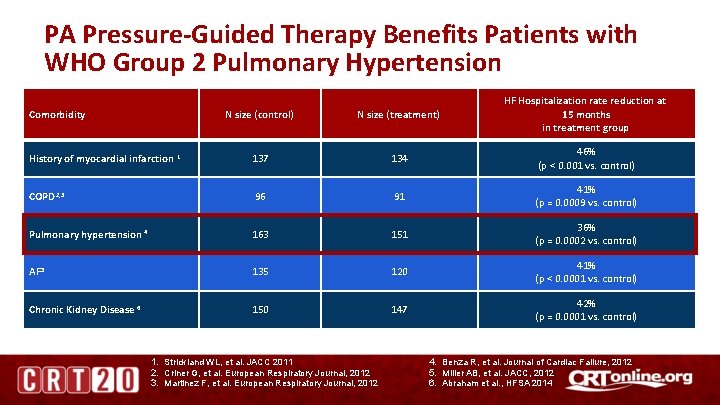
PA Pressure-Guided Therapy Benefits Patients with WHO Group 2 Pulmonary Hypertension N size (control) N size (treatment) HF Hospitalization rate reduction at 15 months in treatment group History of myocardial infarction 1 137 134 46% (p < 0. 001 vs. control) COPD 2, 3 96 91 41% (p = 0. 0009 vs. control) Pulmonary hypertension 4 163 151 36% (p = 0. 0002 vs. control) AF 5 135 120 41% (p < 0. 0001 vs. control) Chronic Kidney Disease 6 150 147 42% (p = 0. 0001 vs. control) Comorbidity 1. Strickland WL, et al. JACC 2011 2. Criner G, et al. European Respiratory Journal, 2012 3. Martinez F, et al. European Respiratory Journal, 2012 4. Benza R, et al. Journal of Cardiac Failure, 2012 5. Miller AB, et al. JACC, 2012 6. Abraham et al. , HFSA 2014
- Slides: 14