IMPACT OF VAGINAL MICROBIOTA ON GENITAL TISSUE AND

IMPACT OF VAGINAL MICROBIOTA ON GENITAL TISSUE AND PLASMA CONCENTRATIONS OF DAPIVIRINE Sharon L Hillier, Leslie A Meyn, Katherine Bunge, Michele Austin, Bernard J Moncla, Charlene S. Dezzutti, Brid Devlin, Mark Marzinke, Craig Hendrix and Lisa Rohan for the FAME Team University of Pittsburgh and Magee-Womens Research Institute, Pittsburgh, PA; the International Partnership for Microbicides, Silver Springs, MD and Johns Hopkins University, Baltimore, MD International AIDS Conference Paris, France July 25, 2017
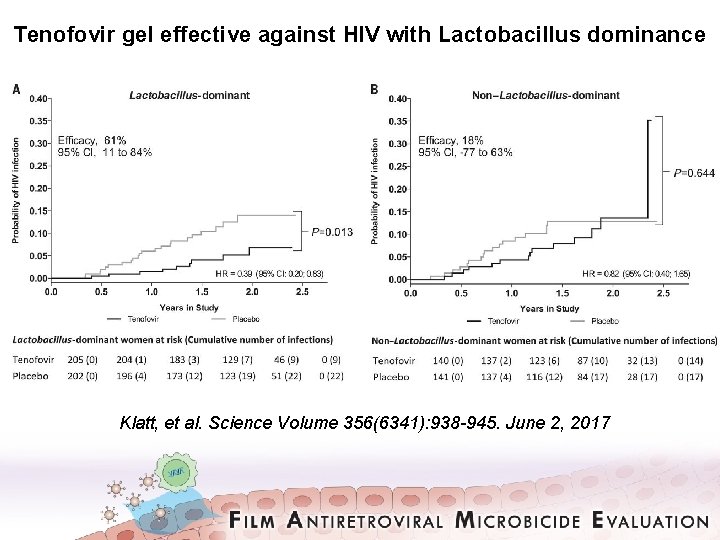
Tenofovir gel effective against HIV with Lactobacillus dominance Klatt, et al. Science Volume 356(6341): 938 -945. June 2, 2017
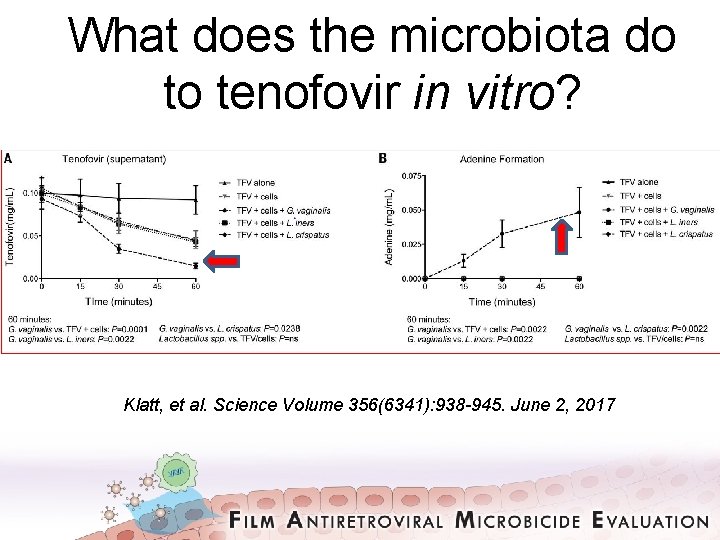
What does the microbiota do to tenofovir in vitro? Klatt, et al. Science Volume 356(6341): 938 -945. June 2, 2017

Limitations of Published Data • Women having greater numbers of partners and more frequent sexual activity at greater risk of BV and HIV; association between BV and apparent decreased efficacy in CAPRISA -04 trial could be attributed to unmeasured differences in behavior • Degradation of tenofovir in presence of G vaginalis was demonstrated within hours in vitro, but this may not replicate in vivo conditions. Klatt, et al. Science Volume 356(6341): 938 -945. June 2, 2017
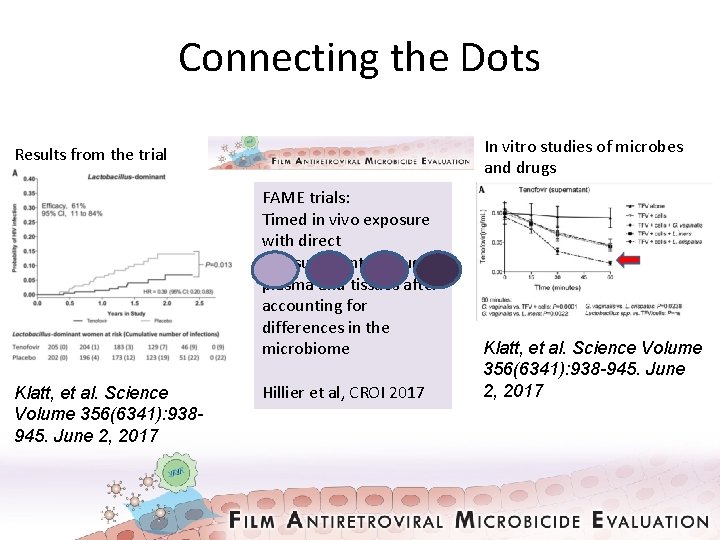
Connecting the Dots In vitro studies of microbes and drugs Results from the trial FAME trials: Timed in vivo exposure with direct measurement of drug in plasma and tissues after accounting for differences in the microbiome Klatt, et al. Science Volume 356(6341): 938945. June 2, 2017 Hillier et al, CROI 2017 Klatt, et al. Science Volume 356(6341): 938 -945. June 2, 2017

Objective and Hypothesis • Objective: To evaluate whether vaginal microbiota associated with BV impact dapivirine concentrations in genital tract tissues and plasma following vaginal application. • Hypothesis: Vaginal microbiota associated with BV will not decrease genital tract tissue and plasma levels of dapivirine

Study Population • 66 healthy, nonpregnant women from Pittsburgh, PA, USA using effective contraception • Asymptomatic, HIV-, negative for chlamydia, gonorrhea, trichomoniasis • Characteristics: 76% unmarried 67% White, 27% Black
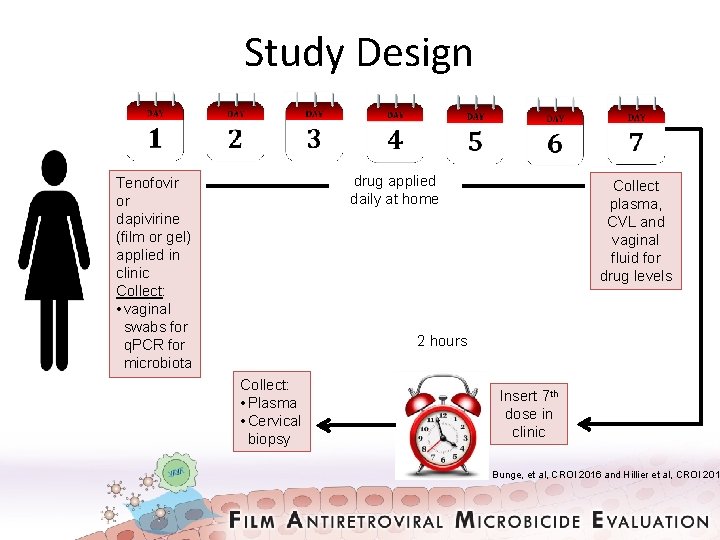
Study Design drug applied daily at home Tenofovir or dapivirine (film or gel) applied in clinic Collect: • vaginal swabs for q. PCR for microbiota Collect plasma, CVL and vaginal fluid for drug levels 2 hours Collect: • Plasma • Cervical biopsy Insert 7 th dose in clinic Bunge, et al, CROI 2016 and Hillier et al, CROI 201

Methods • q. PCR performed from vaginal swab samples collected at baseline – Gardnerella vaginalis – Lactobacillus crispatus, L jensenii, L gasseri, • Bacterial vaginosis detected using Nugent criteria from a Gram stained vaginal smear collected at baseline • Statistics: – Relationship between vaginal microbiota and TFV or dapivirine concentrations was assessed using linear regression models – A quadratic term was included in the tenofovir models with G. vaginalis to improve model fit. – Reported P-values are from the global F-test.
Tenofovir and Dapivirine: Tissue and Plasma LB dominant Vaginal administration of tenofovir or dapivirine BV Genital epithelium Plasma
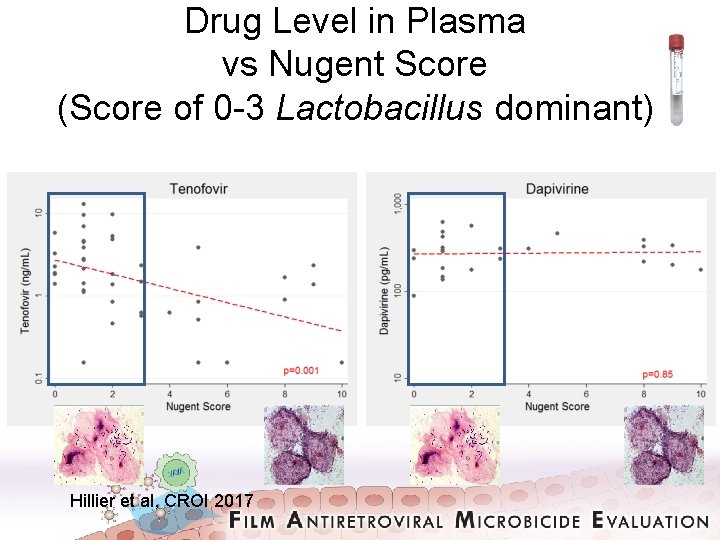
Drug Level in Plasma vs Nugent Score (Score of 0 -3 Lactobacillus dominant) Hillier et al, CROI 2017
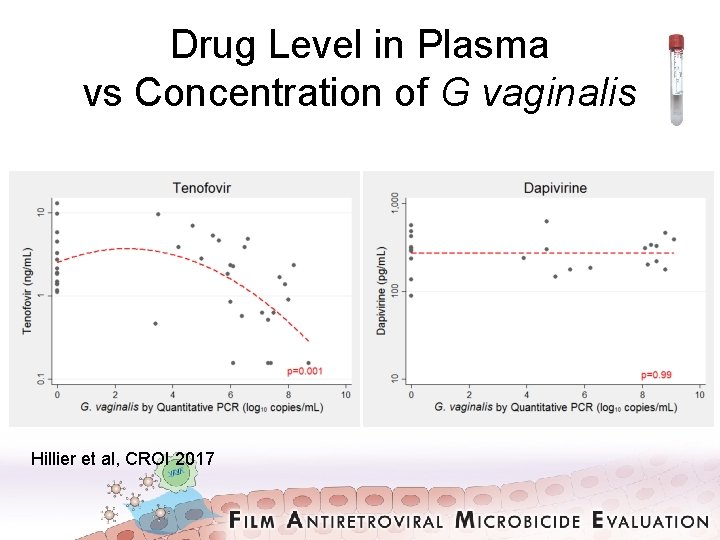
Drug Level in Plasma vs Concentration of G vaginalis Hillier et al, CROI 2017
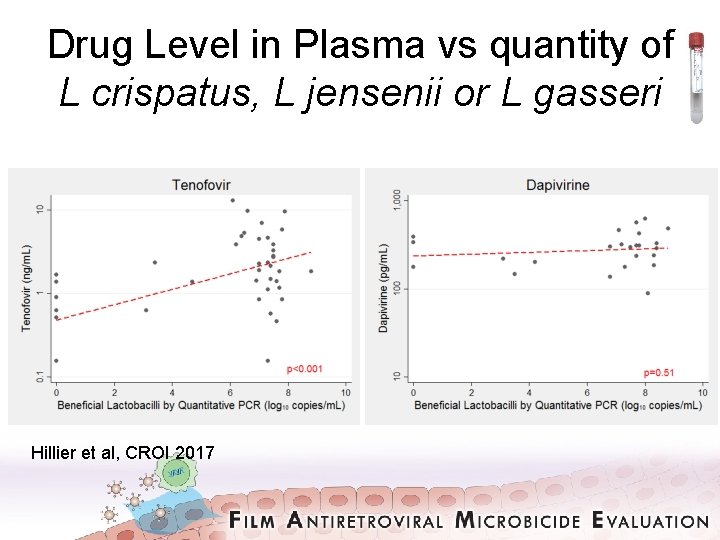
Drug Level in Plasma vs quantity of L crispatus, L jensenii or L gasseri Hillier et al, CROI 2017
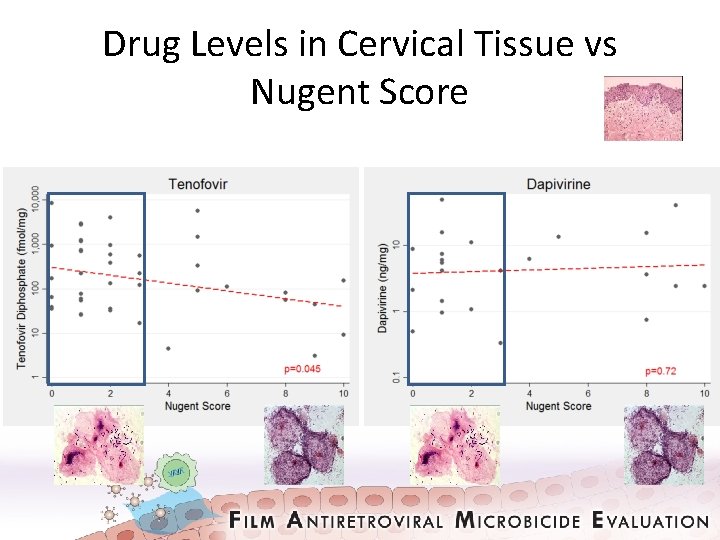
Drug Levels in Cervical Tissue vs Nugent Score
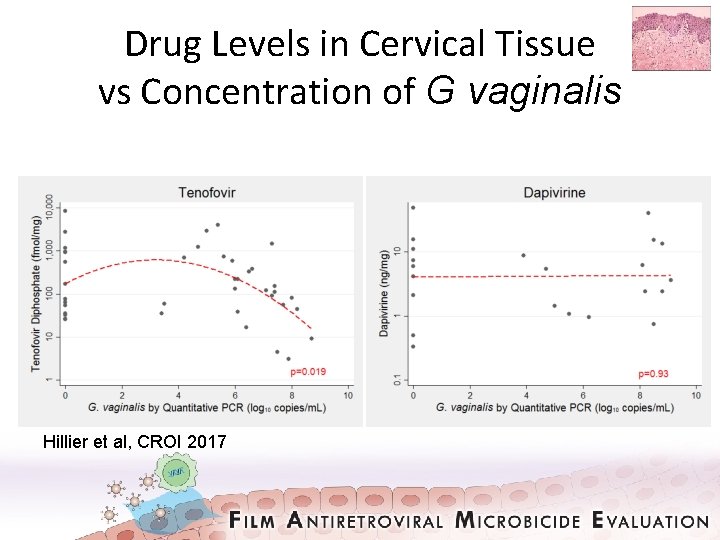
Drug Levels in Cervical Tissue vs Concentration of G vaginalis Hillier et al, CROI 2017
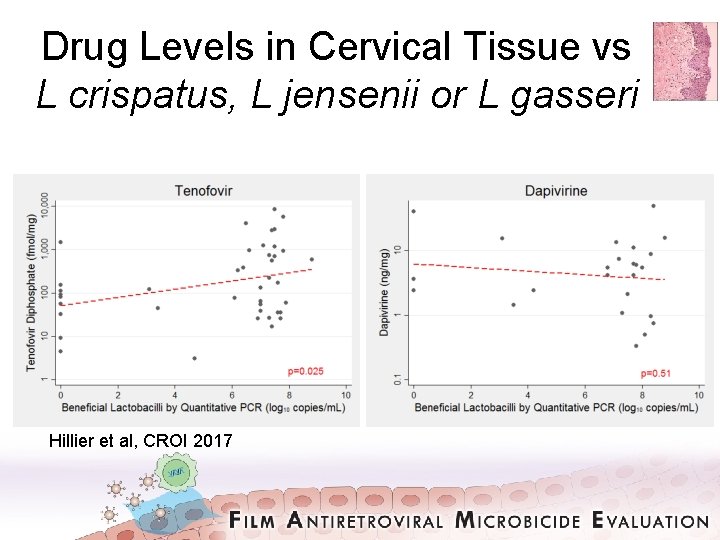
Drug Levels in Cervical Tissue vs L crispatus, L jensenii or L gasseri Hillier et al, CROI 2017

Conclusions • In contrast to tenofovir, genital and plasma concentrations of dapivirine were not decreased by bacteria associated with BV. • These data suggest that the levels of dapivirine following vaginal application should not be impacted by the microbiota associated with bacterial vaginosis. • This is supportive of the ASPIRE study results – 40% of women had bacterial vaginosis at baseline. – No difference in the efficacy of the dapivirine ring in women with bacterial vaginosis and in those without bacterial vaginosis at baseline (39% vs. 21%, P=0. 4 for interaction). (NEJM 2017 Mar 9; 376: 995 -6 • This emphasizes the need for HIV-1 prevention products that work in women with vaginal dysbiosis.

Acknowledgements • Study participants • National Institute of Allergy and Infectious Diseases, National Institutes of Health, Grant AI 082639 and UL 1 TR 000005. • CONRAD was the regulatory sponsor of the tenofovir trial and donated tenofovir drug substance and some study product. • IPM was the regulatory sponsor for the dapivirine trial and donated drug substance and study product.
- Slides: 18