Impact of Using Fixed Dose Combinations FDCs versus


Impact of Using Fixed Dose Combinations (FDCs) versus Single Dose Combination Among New Pulmonary Smear Positive Patients in Bangladesh Dr. Md. Khurshid Alam Hyder, Jr. Consultant National TB Control Programme, Directorate General of Health Services Ministry of Health & Family Welfare, Dhaka, Bangladesh
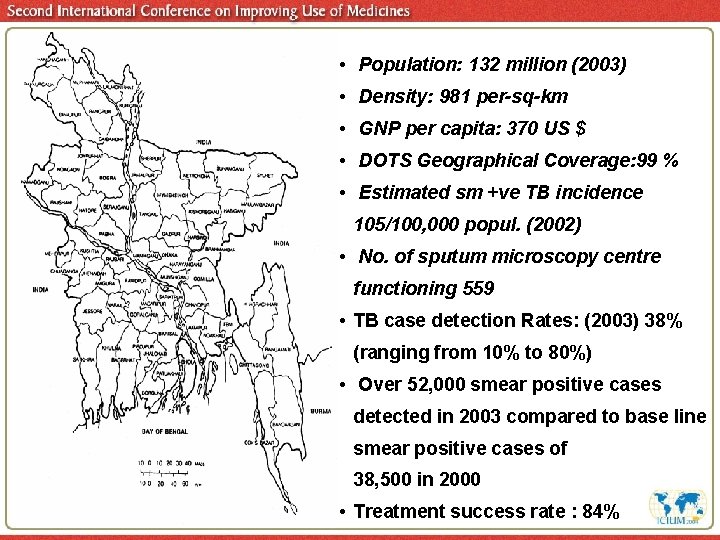
• Population: 132 million (2003) • Density: 981 per-sq-km • GNP per capita: 370 US $ • DOTS Geographical Coverage: 99 % • Estimated sm +ve TB incidence 105/100, 000 popul. (2002) • No. of sputum microscopy centre functioning 559 • TB case detection Rates: (2003) 38% (ranging from 10% to 80%) • Over 52, 000 smear positive cases detected in 2003 compared to base line smear positive cases of 38, 500 in 2000 • Treatment success rate : 84%

Year implementation of DOTS started: 1993 Total population : 132 million (2003) Estimated population 2000 2001 2002 2003 DOTS coverage (Geographical) 95% 98% 99% Estimated all new cases 290, 000 302, 000 307, 000 310, 000 Total cases notified 73, 977 81, 833 Estimated new smear positive cases 130, 000 136, 000 138, 000 140, 000 New Smear positive cases notified 38, 484 (29. 9%) 40, 771 (31. 4%) 46, 771 (33. 8%) 52, 500 (38%) New Smear positive cases treated successfully under DOTS 83% 84% 84% (upto 3 rd quarter 2002) 74, 252 86, 000
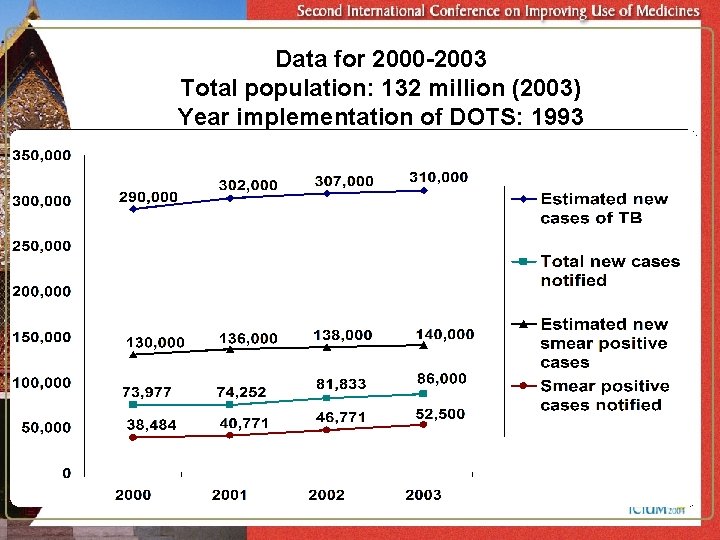
Data for 2000 -2003 Total population: 132 million (2003) Year implementation of DOTS: 1993
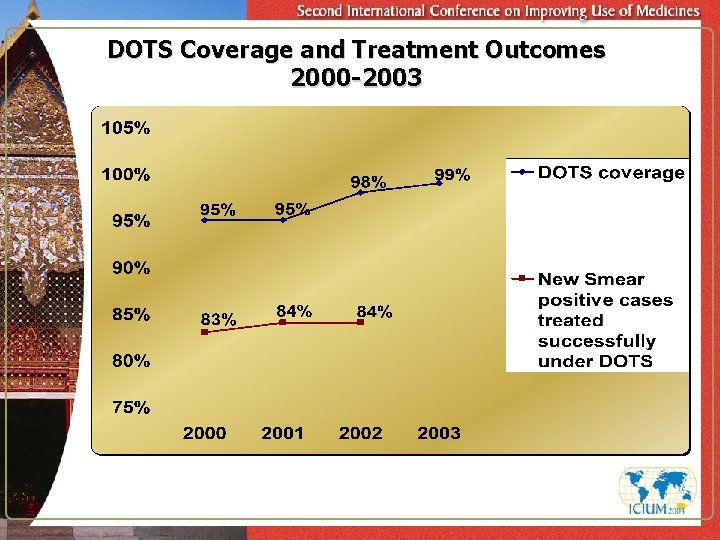
DOTS Coverage and Treatment Outcomes 2000 -2003

Background on the involvement of NGOs § The NGOs under the banner of Leprosy Coordination Committee were working for leprosy control since 1972. The Damien Foundation has been treating TB cases since 1991 § BRAC has been working for TB control in a limited area since 1984 § Bangladesh introduced DOTS in 1993, however the formal collaboration with NGOs was 1 st initiated in 1994 through two separate MOUs § MOUs have been signed between NSDP, UPHCP and Public-Private Pilot Project for Urban TB Programme

Principles of Collaboration § Mutual respect and trust § Commitment to follow NTP guidelines § Pulling of resources to maximize the use of expertise of partners to make the programme cost effective § Periodic review of programme performance

NTPs initiatives to involve other stake holder § National TB Control Programme has recently revised the national guidelines § Introduced Fixed Dosed Combination from 1 st quarter 2004 § Availability of FDC’s from Global Drug Facility ensured

Global Drug Facility - Services § Grants of first line drugs, to support DOTS expansion § A direct procurement mechanism for countries and NGOs, to buy drugs for use in DOTS programmes § A web-based tool for placing orders and tracking shipments § A white list of ‘pre-qualified’ manufacturers of quality TB drugs

Global Drug Facility - Bangladesh § § § § Applied - March 02 Approved for 3 years 1 st year (55, 700 patients; $700, 000) Received 1 st year assignment of 4 FDCs and 2 FDCs, separate Isoniazid, Ethambutol in 2003 Technical assistance - GDF country visit (May 02), GDF monitoring visit (Sept 03) 2 nd year request (97, 000 patients; $1. 2 million) Request for 2 nd year is for 4 FDCs and 2 FDCs which is under process Bought additional drugs through GDF Direct Procurement mechanism in 2003

Background Information of the Study § NTP Bangladesh recently introduced FDCs in treatment regimen to rationalize the use of fewer tablets compared to a larger number of tablets in a single drug combination to increase compliance § The objectives of the study are to assess the impact of FDCs on efficacy, sputum conversion and side effects among new pulmonary smear positive patients compared to those on single drug combinations under DOT § Prospective study with comparison series § Two Public Urban Chest Disease Clinics, and Centers of NSDP at Dhaka were selected

Methodology Two phases of intervention § BCC among service providers at the study centers during last two quarters of 2003 § Development of questionnaire for collection of information § Implementation among two groups of new pulmonary smear positive patients – FDCs and Single Drug Combination started from first quarter of 2004
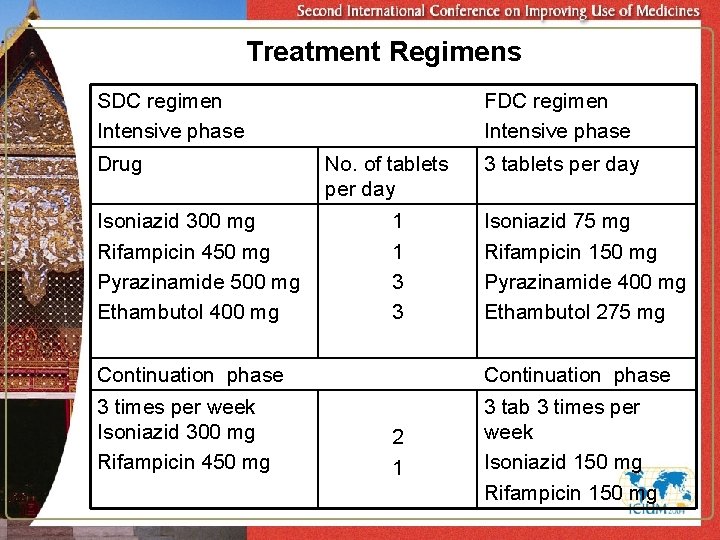
Treatment Regimens SDC regimen Intensive phase Drug Isoniazid 300 mg Rifampicin 450 mg Pyrazinamide 500 mg Ethambutol 400 mg FDC regimen Intensive phase No. of tablets per day 1 1 3 3 3 tablets per day Isoniazid 75 mg Rifampicin 150 mg Pyrazinamide 400 mg Ethambutol 275 mg Continuation phase 3 times per week Isoniazid 300 mg Rifampicin 450 mg 3 tab 3 times per week Isoniazid 150 mg Rifampicin 150 mg 2 1

Preliminary Results §Total patients enrolled for the study (Jan-Feb, 2004): 32 §SDCs: 14 §FDCs: 18 Sex of 32 new smear positive patients by regimen Regimen Males Females Total SDCs 9 (64%) 5 (36%) 14 (44%) FDCs 12 (67%) 6 (33%) 18 (56%) Total 21 (65. 5%) 11 (34. 5%) 32

Preliminary Results (Cont. . ) Frequency of all complaints during the first month of intensive phase of treatment by regimen Regimen Gastro. Skin Joint Pain intestinal reaction/ Itching SDCs 2 (14 %) - 3 (21 %) FDCs 2 (11 %) - 1 (6 %)

Inference § Too early to comment as implementation started in 1 st qr. 2004, however, preliminary results shows that more patients on SDCs have joint pains as compared to patients on FDCs

Expected Outcome from the Study § Efficacy of FDCs in the treatment regimen of NTP in Bangladesh § Sputum conversion rates comparison among new pulmonary smear positive patients using SDCs and FDCs § Knowledge of Adverse/Side effect using SDCs and FDCs § Provide information to the policy makers and program management on use of FDCs in NTP of Bangladesh

Thank You
- Slides: 19