IMMUNOTHERAPY Vanesa Gregorc MD IRCCS San Raffaele Milano










![RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ, RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ,](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-24.jpg)
![RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ, RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ,](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-26.jpg)
![START TRIAL – STUDY DESIGN stage IIIA/B post CT/RT (DC) [TITLE] START TRIAL – STUDY DESIGN stage IIIA/B post CT/RT (DC) [TITLE]](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-31.jpg)


- Slides: 37

IMMUNOTHERAPY Vanesa Gregorc MD IRCCS San Raffaele, Milano

LUNG CANCER AS AN MEDIATED DISEASE AND IMMUNOTHERAPY n IMMUNOLOGICTARGET FOR Although many immunotherapies are being investigated in NSCLC, currently none have been FDA approved, as vaccines and other immune-modulators have yet to demonstrate considerable improvements in. OS in phase III clinical trials

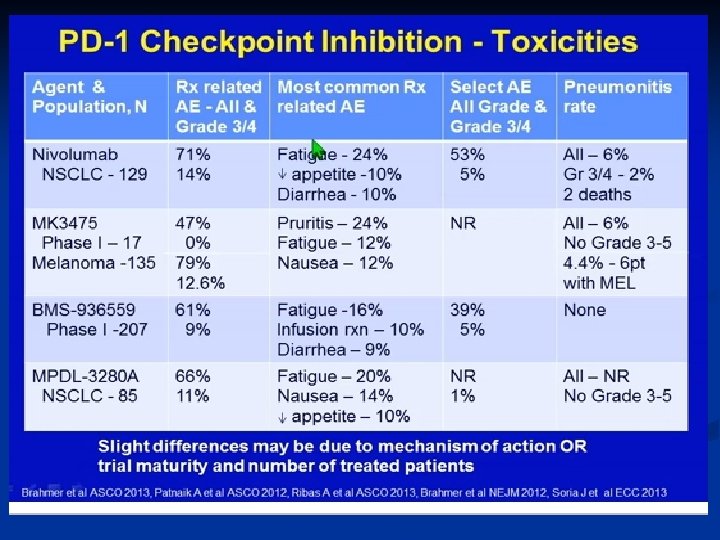
NOVEL ASPECTS Immune-related response criteria n Endpoints n The immune-related toxicities n All differ considerably from conventional cytotoxic agents and targeted therapies

RESPONSE EVALUATION FOR IMMUNOTHERAPY • Permissive not restrictive • RECIST/m. WHO modified by immunological criteria • First radiological examination the most critical (pseudoprogressions) • ir. Responders with new lesions also but decrease in baseline lesions • PD should be confirmed after 4 w Wolchok JD, et al Clin Cancer Res 2009; 15: 7412 -7420.

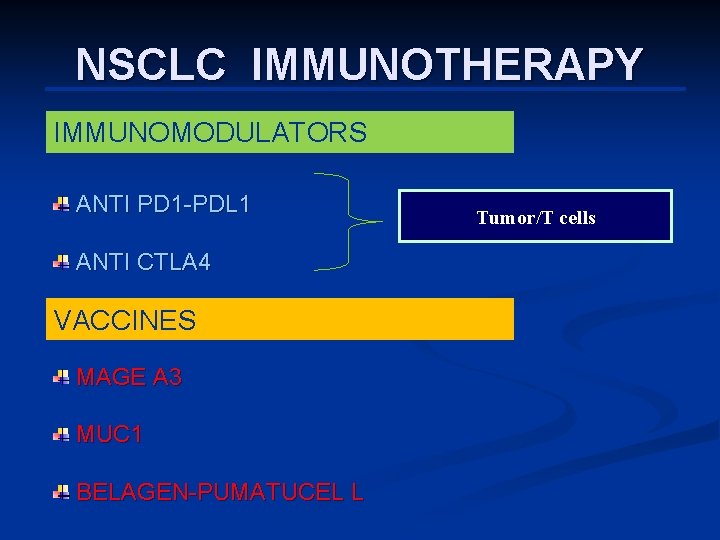
NSCLC IMMUNOTHERAPY IMMUNOMODULATORS ANTI PD 1 -PDL 1 ANTI CTLA 4 VACCINES MAGE A 3 MUC 1 BELAGEN-PUMATUCEL L Tumor/T cells

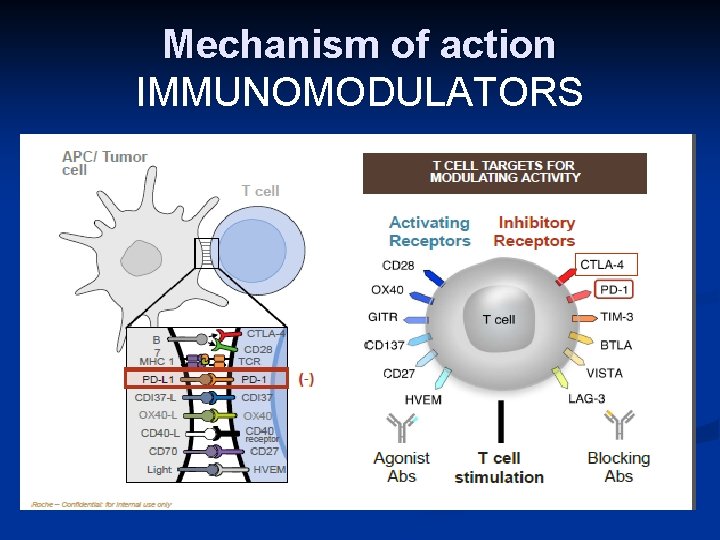
Mechanism of action IMMUNOMODULATORS

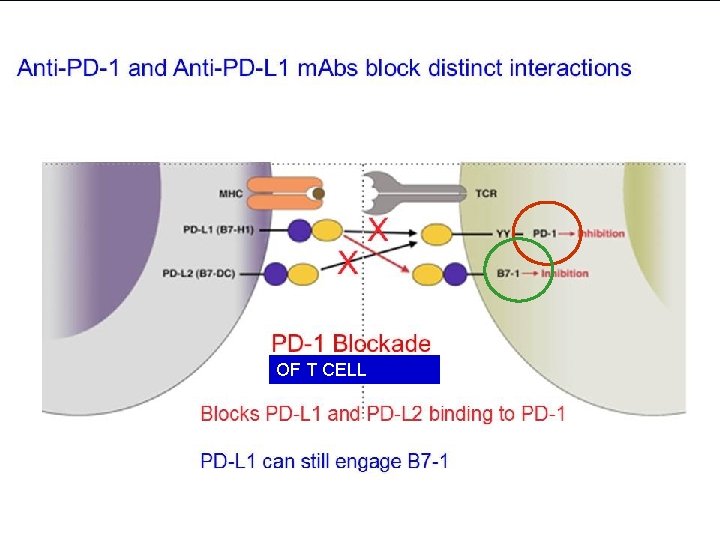
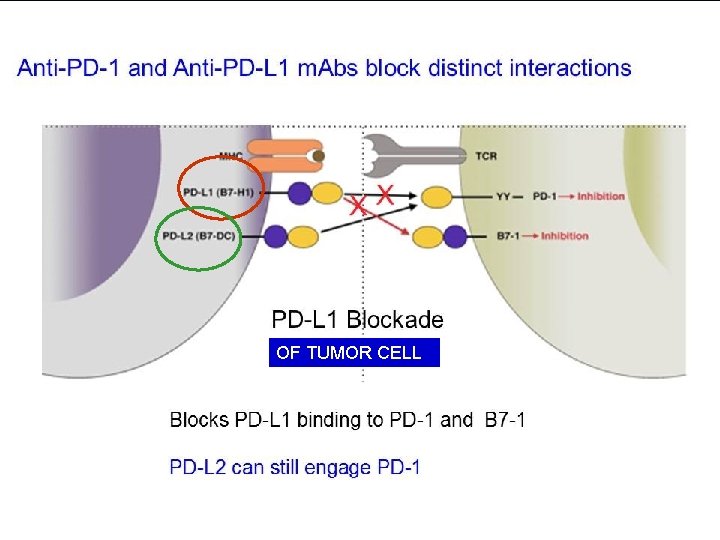
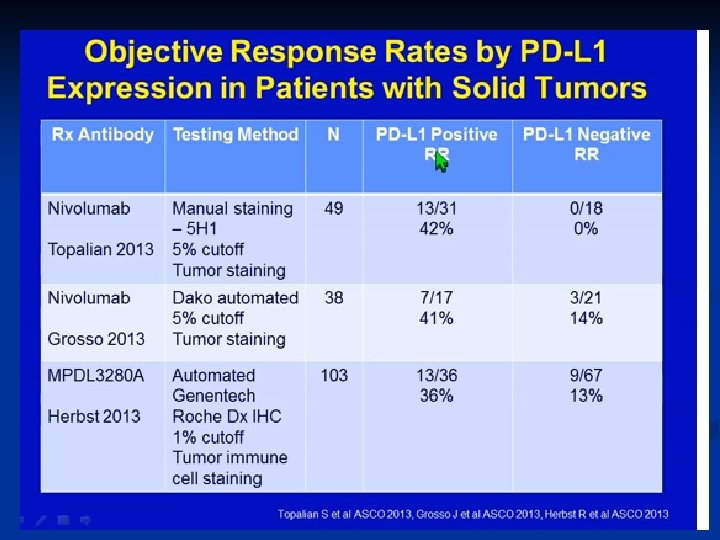
PROGRAMMED DEATH-1 IMMUNE CHECKPONT (PD-1) n n PD-1 (cell receptor) is overexpressed on NSCLC-infiltrating T cells and these are functionally exhausted cells Ligands: PDL-1 and PDL-2 (tumor cell /APC) Higher tumoral PDL-1 expression correlates with decreased OS RATIONALE Blocking the PD-1 or PDL-1 pathway would restore/promote the function of chronically exhausted tumor-specific T cells and decrease tumor-induced immune Zhang Y et al. Cell Mol Immunol. 2010; 7: 389 - 395.

OF T CELL

OF TUMOR CELL

CLINICAL DEVELOPMENT OF INHIBITORS OF PD-1 IMMUNE CHECKPOINT

Inhibitors of PD-1 – Phase I trial CT-O 11 (0. 26. 0 mg/kg N=17 0. 2 -6. 0 mg/kg IV

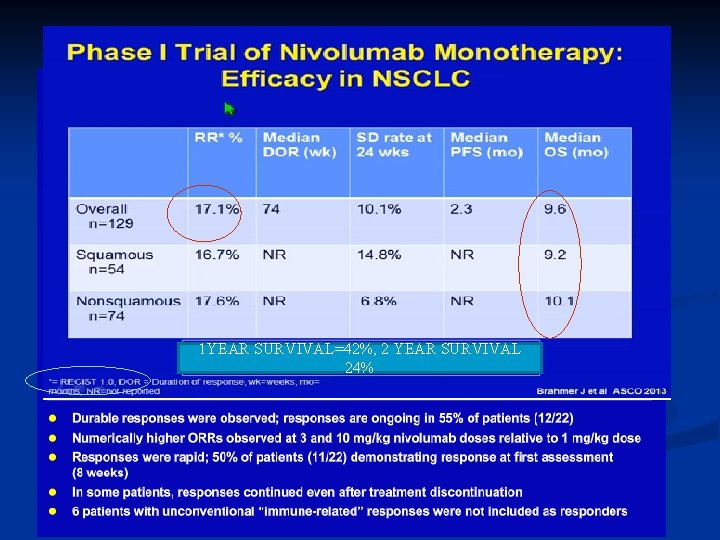
1 YEAR SURVIVAL=42%, 2 YEAR SURVIVAL 24%

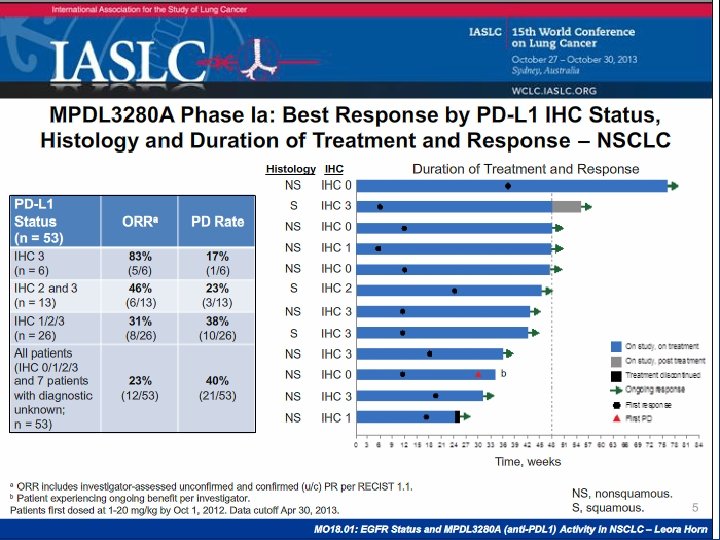
Lambrolizumab in NSCLC Garon E. et al Abstract MO 18. 02 IALS 2013

INHIBITORS OF PDL 1 - phase I trial

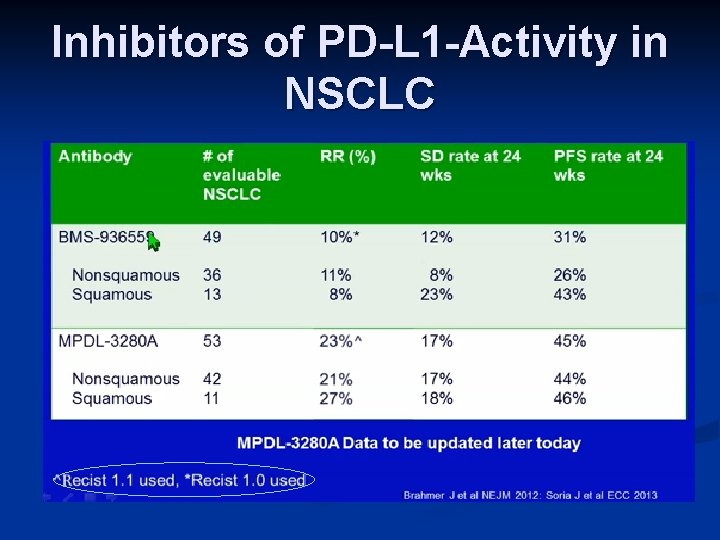
Inhibitors of PD-L 1 -Activity in NSCLC

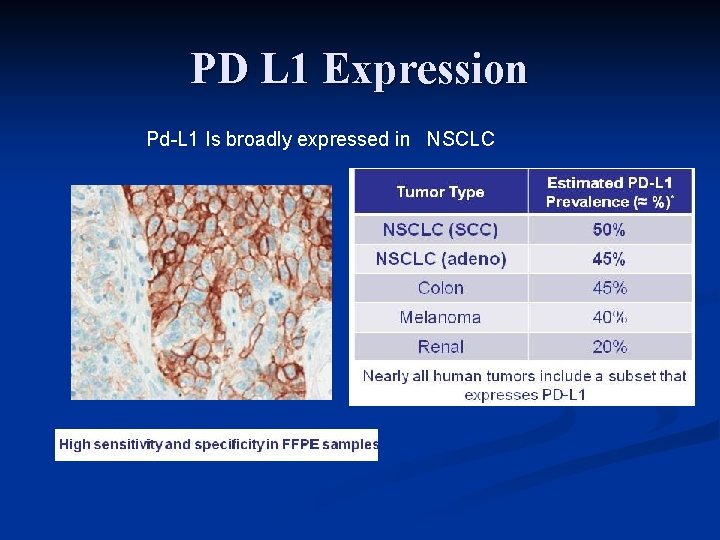
PD L 1 Expression Pd-L 1 Is broadly expressed in NSCLC



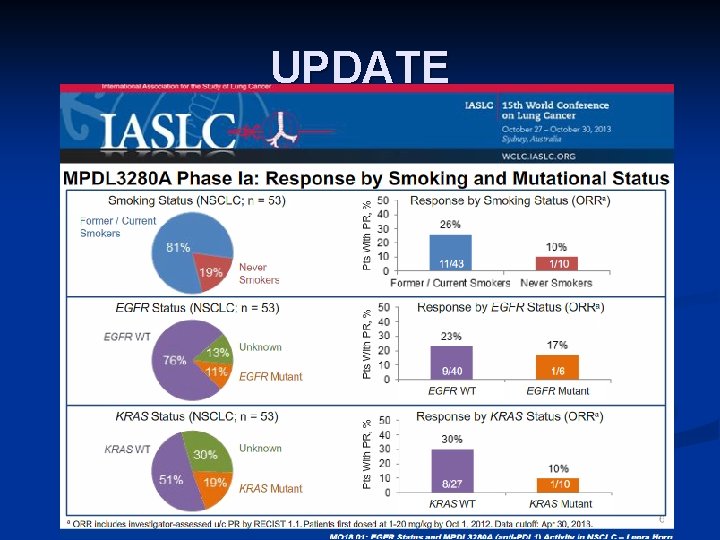
UPDATE

UPDATE

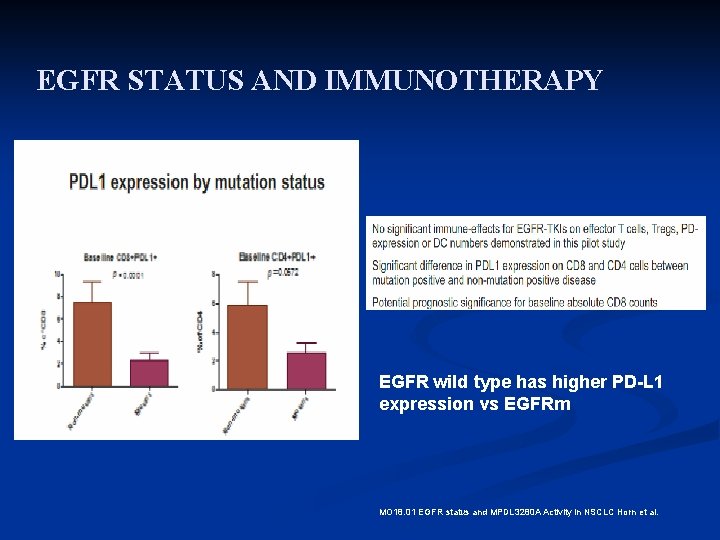
EGFR STATUS AND IMMUNOTHERAPY EGFR wild type has higher PD-L 1 expression vs EGFRm MO 18. 01 EGFR status and MPDL 3280 A Activity in NSCLC Horn et al.


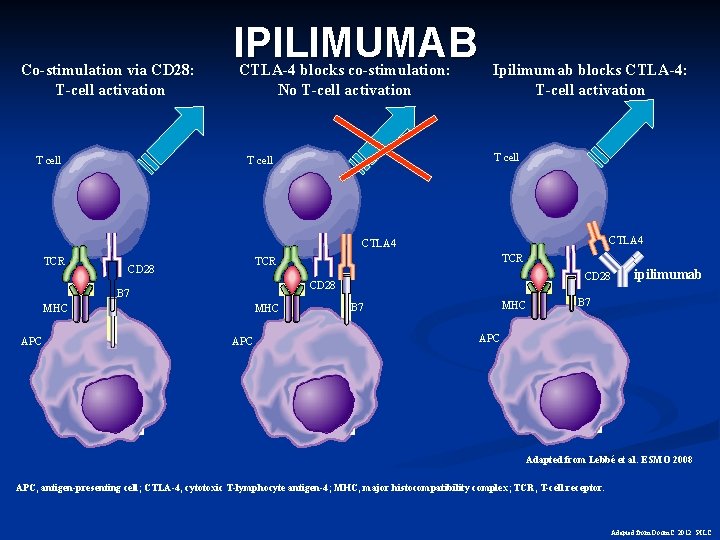
Co-stimulation via CD 28: T-cell activation T cell IPILIMUMAB CTLA-4 blocks co-stimulation: No T-cell activation Ipilimumab blocks CTLA-4: T-cell activation T cell CTLA 4 TCR CD 28 B 7 MHC APC TCR CD 28 MHC APC MHC B 7 ipilimumab B 7 APC Adapted from Lebbé et al. ESMO 2008 APC, antigen-presenting cell; CTLA-4, cytotoxic T-lymphocyte antigen-4; MHC, major histocompatibility complex; TCR, T-cell receptor. Adapted from Doom. C. 2012 PILC
![RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC TITLE Lynch TJ RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ,](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-24.jpg)
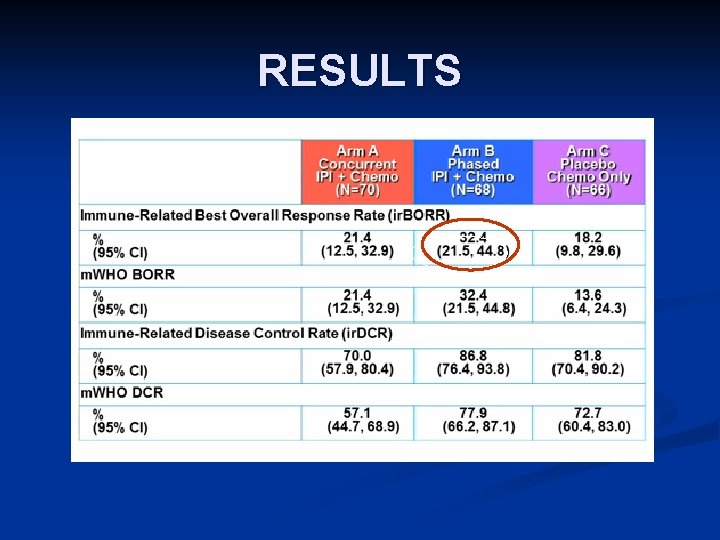
RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ, et al. phase II study. J Clin Oncol. 2012; 30: 2046 -2054. Adapted by J. Brahmer, 2013 ASCO Annual Meeting

RESULTS
![RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC TITLE Lynch TJ RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ,](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-26.jpg)
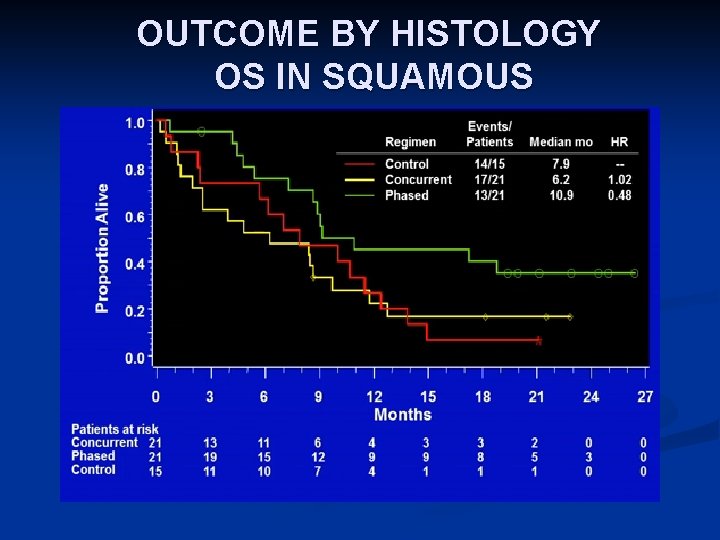
RANDOMIZED PHASE II STUDY OF IPILIMUMAB AND CT IN ADVANCED NSCLC [TITLE] Lynch TJ, et al. phase II study. J Clin Oncol. 2012; 30: 2046 -2054. Adapted by J. Brahmer, 2013 ASCO Annual Meeting

OUTCOME BY HISTOLOGY OS IN SQUAMOUS

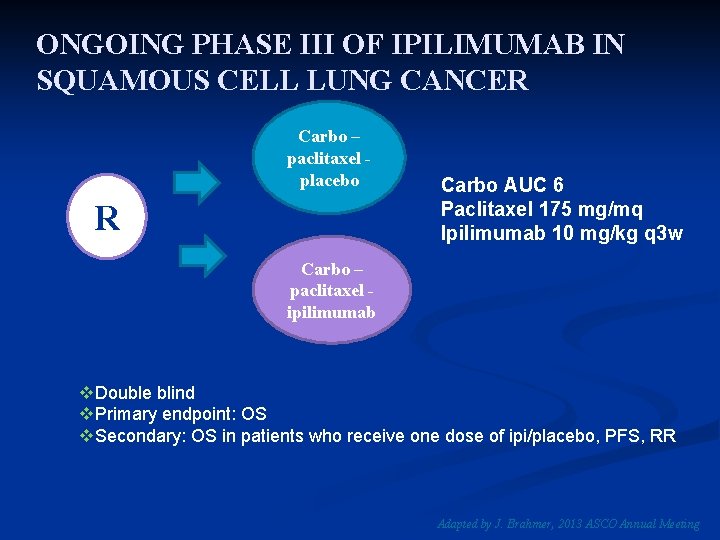
ONGOING PHASE III OF IPILIMUMAB IN SQUAMOUS CELL LUNG CANCER Carbo – paclitaxel placebo R Carbo AUC 6 Paclitaxel 175 mg/mq Ipilimumab 10 mg/kg q 3 w Carbo – paclitaxel ipilimumab v. Double blind v. Primary endpoint: OS v. Secondary: OS in patients who receive one dose of ipi/placebo, PFS, RR Adapted by J. Brahmer, 2013 ASCO Annual Meeting

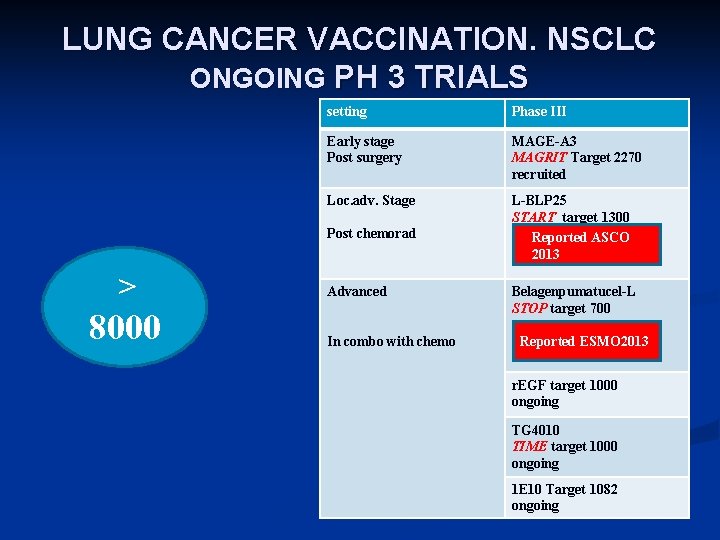
LUNG CANCER VACCINATION. NSCLC ONGOING PH 3 TRIALS setting Phase III Early stage Post surgery MAGE-A 3 MAGRIT Target 2270 recruited Loc. adv. Stage L-BLP 25 START target 1300 Reported ASCO 2013 Post chemorad > 8000 Advanced In combo with chemo Belagenpumatucel-L STOP target 700 Reported ESMO 2013 r. EGF target 1000 ongoing TG 4010 TIME target 1000 ongoing 1 E 10 Target 1082 ongoing

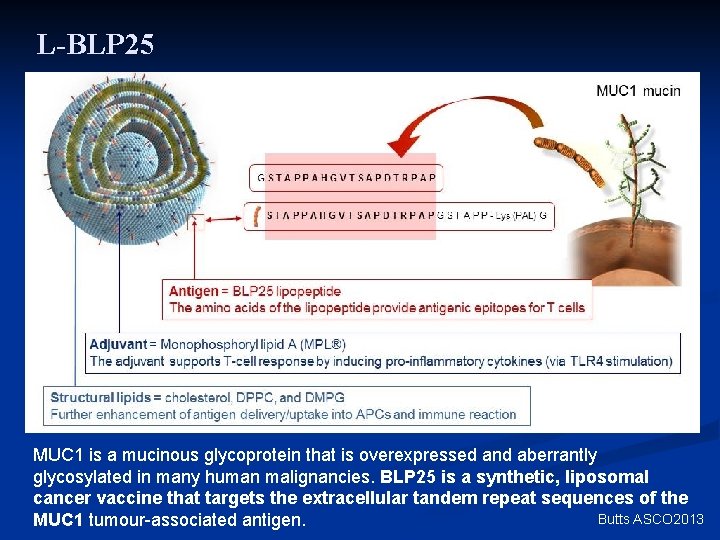
L-BLP 25 MUC 1 is a mucinous glycoprotein that is overexpressed and aberrantly glycosylated in many human malignancies. BLP 25 is a synthetic, liposomal cancer vaccine that targets the extracellular tandem repeat sequences of the Butts ASCO 2013 MUC 1 tumour-associated antigen.
![START TRIAL STUDY DESIGN stage IIIAB post CTRT DC TITLE START TRIAL – STUDY DESIGN stage IIIA/B post CT/RT (DC) [TITLE]](https://slidetodoc.com/presentation_image/1469ac6476f36cc4b6b234cbc4f6c5c1/image-31.jpg)
START TRIAL – STUDY DESIGN stage IIIA/B post CT/RT (DC) [TITLE]

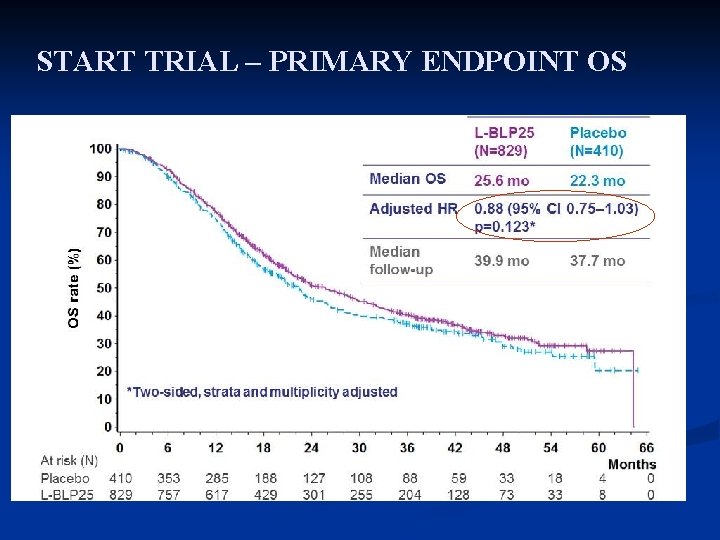
START TRIAL – PRIMARY ENDPOINT OS

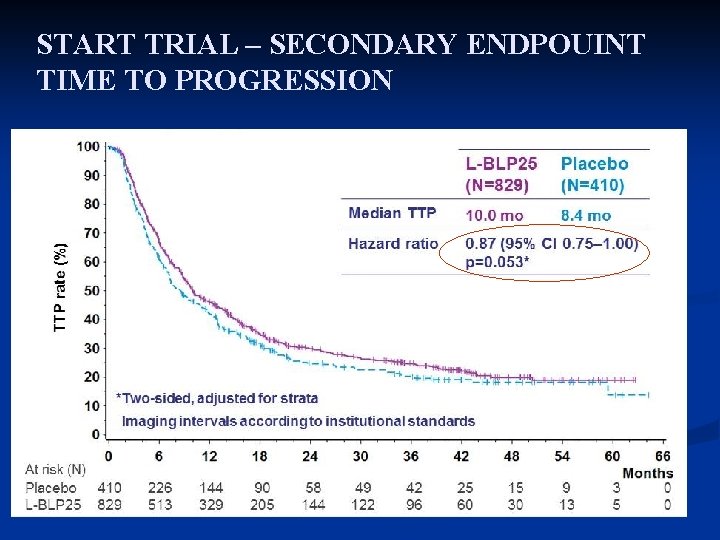
START TRIAL – SECONDARY ENDPOUINT TIME TO PROGRESSION

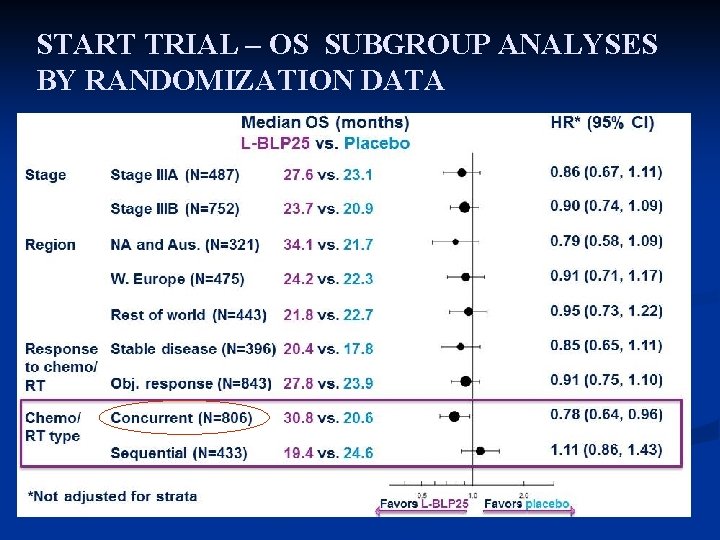
START TRIAL – OS SUBGROUP ANALYSES BY RANDOMIZATION DATA

STOP TRIAL stage III/IV in DC after 1° line belagenpumatucel-L Primary endpoint: OS Other endpoints: PFS, RR, Qo. L, CNS metastases, safety Belagenpumatucel-L(Lucanix®) uses genetically modified whole tumour cells to stimulate the patient's own immune system to attack the tumour. It is comprised of 4 transforming growth factor (TGF)-ß 2 antisense gene-modified, Giaccone et al. ESMO 2013 irradiated, allogeneic NSCLC cell lines.

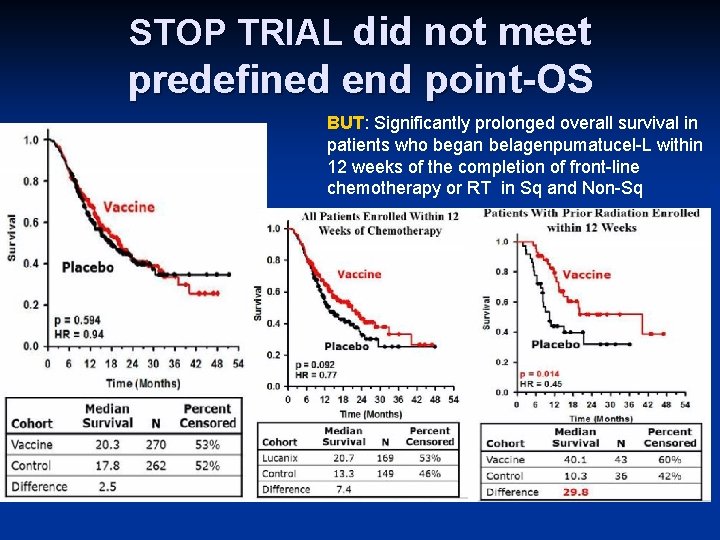
STOP TRIAL did not meet predefined end point-OS point. BUT: Significantly prolonged overall survival in patients who began belagenpumatucel-L within 12 weeks of the completion of front-line chemotherapy or RT in Sq and Non-Sq

conclusions v. Immunotherapy has promising anti-tumor activity in NSCLC and an unique set of side effects consistent with the immune mechanism of action v. Patient selection (biomarker) might be the key to further development as a single agent and in combination with otherapies v. Phase III trials are ongoing in order to make immunotherapy a reality for the treatment of NSCLC