Immunotherapy the good news and the bad Associate

Immunotherapy – the good news and the bad Associate Professor Philip Beale Chair ANZGOG Clinical Trials Group Medical Oncologist Sydney ◦ Concord Hospital ◦ Chris O’Brien Lifehouse ◦ Royal Prince Alfred Hospital

Outline v. Background to immunotherapy v. Immunotherapy in gynaecological cancer v. Ongoing trials v. Future directions

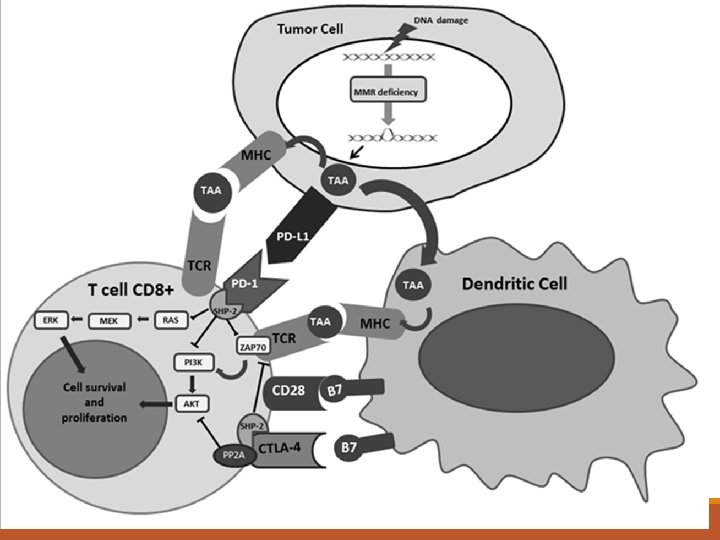
What is Immune therapy 3 types 1. Immune boosting therapy 2. Immunotherapy – drugs that have been designed to unlock the immune system to allow targeted destruction of cancer cells 3. Vaccine type therapy – preventative and treatment

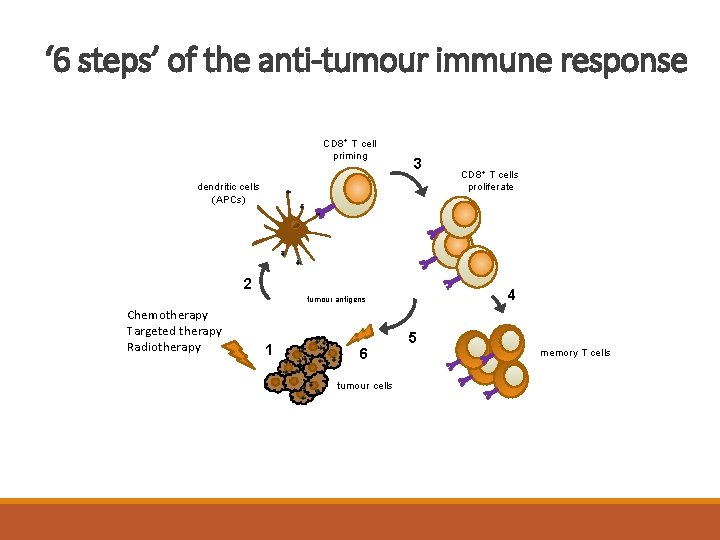
‘ 6 steps’ of the anti-tumour immune response CD 8+ T cell priming 3 dendritic cells (APCs) 2 4 tumour antigens Chemotherapy Targeted therapy Radiotherapy 1 6 tumour cells CD 8+ T cells proliferate 5 memory T cells
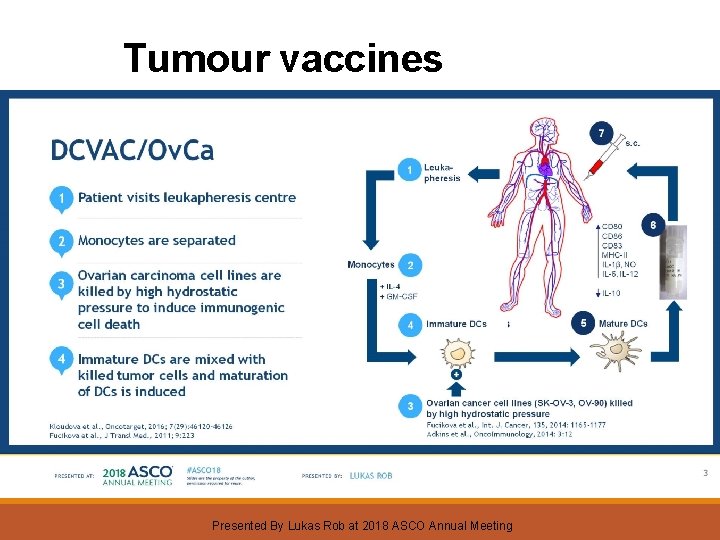
Tumour vaccines Presented By Lukas Rob at 2018 ASCO Annual Meeting

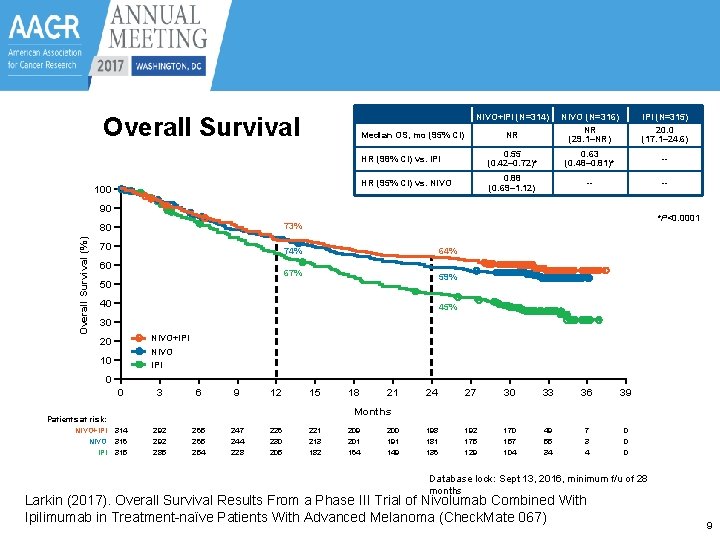
Overall Survival NIVO+IPI (N=314) NIVO (N=316) IPI (N=315) NR NR (29. 1–NR) 20. 0 (17. 1– 24. 6) HR (98% CI) vs. IPI 0. 55 (0. 42– 0. 72)* 0. 63 (0. 48– 0. 81)* -- HR (95% CI) vs. NIVO 0. 88 (0. 69– 1. 12) -- -- Median OS, mo (95% CI) 100 Overall Survival (%) Percentage of PFS 90 *P<0. 0001 80 73% 70 74% 67% 59% 60 50 40 45% 30 NIVO+IPI NIVO IPI 20 10 0 0 3 6 9 12 15 21 24 27 30 33 36 39 198 181 136 192 175 129 170 157 104 49 55 34 7 3 4 0 0 0 Months Patients at risk: NIVO+IPI NIVO IPI 18 314 316 315 292 285 265 254 247 244 228 226 230 205 221 213 182 209 201 164 200 191 149 Database lock: Sept 13, 2016, minimum f/u of 28 months Larkin (2017). Overall Survival Results From a Phase III Trial of Nivolumab Combined With Ipilimumab in Treatment-naïve Patients With Advanced Melanoma (Check. Mate 067) 9
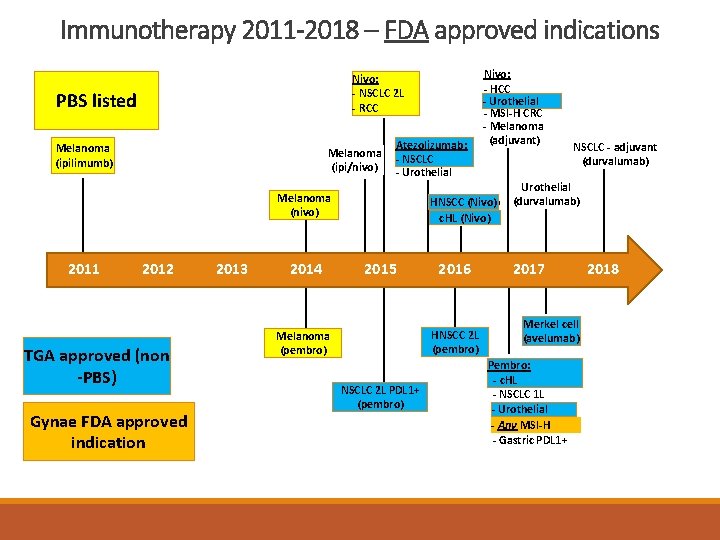
Immunotherapy 2011 -2018 – FDA approved indications Nivo: --NSCLC 2 L 2 L --RCC PBS listed Melanoma (ipilimumb) Melanoma (ipi/nivo) Atezolizumab: - NSCLC - Urothelial Melanoma (nivo) 2011 2012 TGA approved (non -PBS) Gynae FDA approved indication 2013 2014 Nivo: - HCC - Urothelial - MSI-H CRC - Melanoma (adjuvant) HNSCC(Nivo) HNSCC c. HL (Nivo) 2015 2016 HNSCC 2 L (pembro) Melanoma (pembro) NSCLC 2 L 2 LPDL 1+ (pembro) NSCLC - adjuvant (durvalumab) Urothelial (durvalumab) 2017 Merkel cell (avelumab) Pembro: c. HL -- c. HL - NSCLC 1 L - Urothelial Any MSI-H -- Any - Gastric PDL 1+ 2018

Immunotherapy in Cervical Cancer
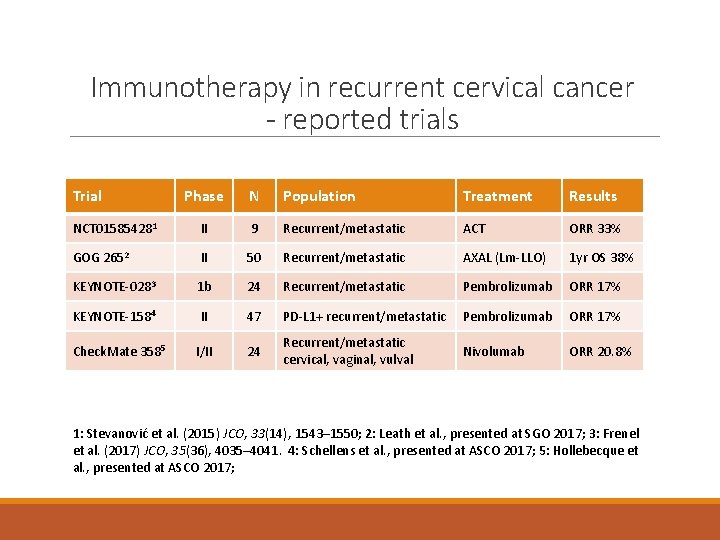
Immunotherapy in recurrent cervical cancer - reported trials Trial Phase N Population Treatment Results NCT 015854281 II 9 Recurrent/metastatic ACT ORR 33% GOG 2652 II 50 Recurrent/metastatic AXAL (Lm-LLO) 1 yr OS 38% KEYNOTE-0283 1 b 24 Recurrent/metastatic Pembrolizumab ORR 17% KEYNOTE-1584 II 47 PD-L 1+ recurrent/metastatic Pembrolizumab ORR 17% I/II 24 Recurrent/metastatic cervical, vaginal, vulval Nivolumab ORR 20. 8% Check. Mate 3585 1: Stevanović et al. (2015) JCO, 33(14), 1543– 1550; 2: Leath et al. , presented at SGO 2017; 3: Frenel et al. (2017) JCO, 35(36), 4035– 4041. 4: Schellens et al. , presented at ASCO 2017; 5: Hollebecque et al. , presented at ASCO 2017;
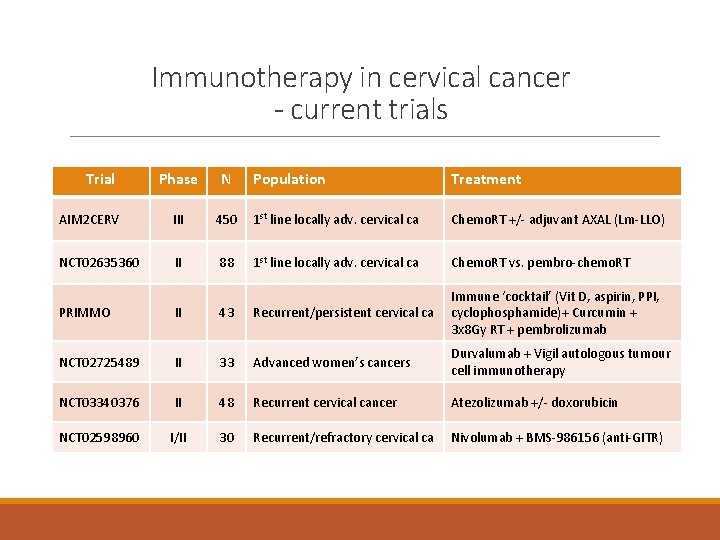
Immunotherapy in cervical cancer - current trials Trial Phase N Population Treatment AIM 2 CERV III 450 1 st line locally adv. cervical ca Chemo. RT +/- adjuvant AXAL (Lm-LLO) NCT 02635360 II 88 1 st line locally adv. cervical ca Chemo. RT vs. pembro-chemo. RT PRIMMO II 43 Recurrent/persistent cervical ca Immune ‘cocktail’ (Vit D, aspirin, PPI, cyclophosphamide)+ Curcumin + 3 x 8 Gy RT + pembrolizumab NCT 02725489 II 33 Advanced women’s cancers Durvalumab + Vigil autologous tumour cell immunotherapy NCT 03340376 II 48 Recurrent cervical cancer Atezolizumab +/- doxorubicin NCT 02598960 I/II 30 Recurrent/refractory cervical ca Nivolumab + BMS-986156 (anti-GITR)

Immunotherapy in Uterine Cancer
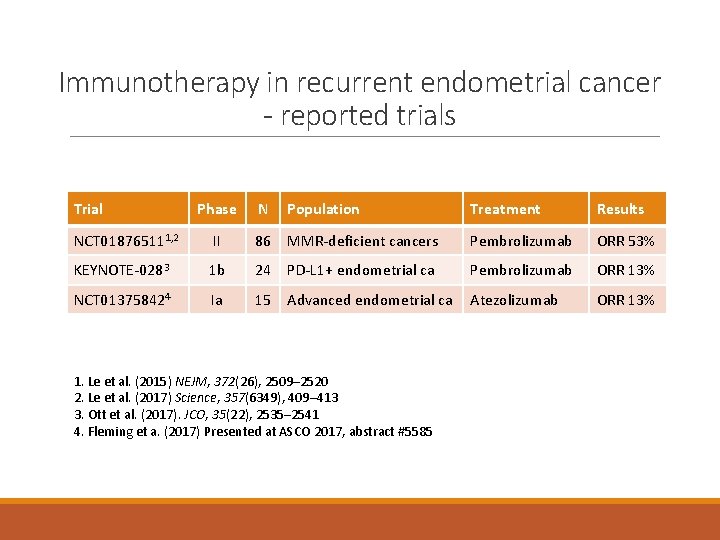
Immunotherapy in recurrent endometrial cancer - reported trials Trial Phase N Population Treatment Results NCT 018765111, 2 II 86 MMR-deficient cancers Pembrolizumab ORR 53% KEYNOTE-0283 1 b 24 PD-L 1+ endometrial ca Pembrolizumab ORR 13% NCT 013758424 Ia 15 Advanced endometrial ca Atezolizumab ORR 13% 1. Le et al. (2015) NEJM, 372(26), 2509– 2520 2. Le et al. (2017) Science, 357(6349), 409– 413 3. Ott et al. (2017). JCO, 35(22), 2535– 2541 4. Fleming et a. (2017) Presented at ASCO 2017, abstract #5585
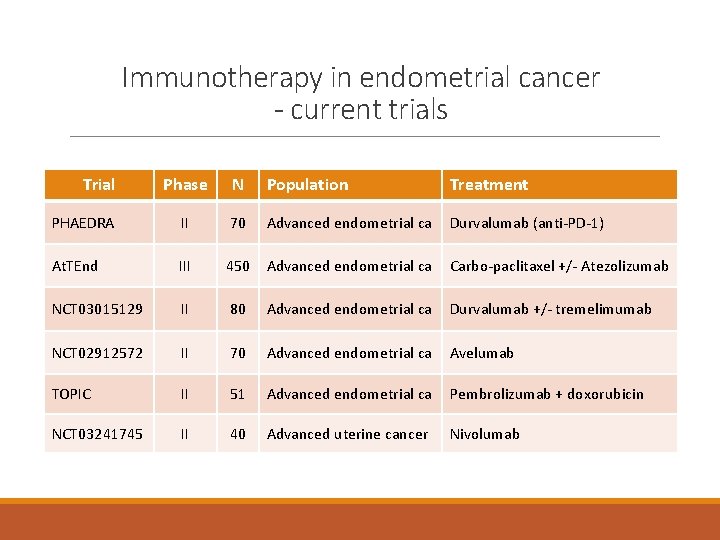
Immunotherapy in endometrial cancer - current trials Trial Phase N Population Treatment PHAEDRA II 70 Advanced endometrial ca Durvalumab (anti-PD-1) At. TEnd III 450 Advanced endometrial ca Carbo-paclitaxel +/- Atezolizumab NCT 03015129 II 80 Advanced endometrial ca Durvalumab +/- tremelimumab NCT 02912572 II 70 Advanced endometrial ca Avelumab TOPIC II 51 Advanced endometrial ca Pembrolizumab + doxorubicin NCT 03241745 II 40 Advanced uterine cancer Nivolumab

What about Tumour biology? Markers on the cancer cells can predict for response to treatment Mismatch repair deficiency PD-L 1 expression on tumour cells Tumour mutational burden
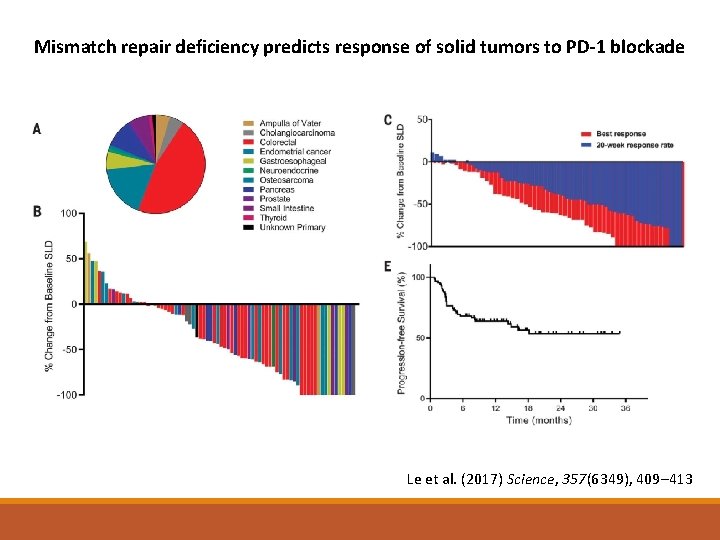
Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade Le et al. (2017) Science, 357(6349), 409– 413

to l a v o r p p a d e t a r le e nts acc a r g A D F : 7 1 0 2 , 3 n 2 io t a May ic d in ic t s o n g a e sue/sit is t t s r fi r fo b a m u z li pembro Piulats, J. M. , & Matias-Guiu, X. (2016). Immunotherapy in endometrial cancer: In the nick of time. Clinical Cancer Research, 22(23), 5623– 5625

Immunotherapy in Ovarian Cancer
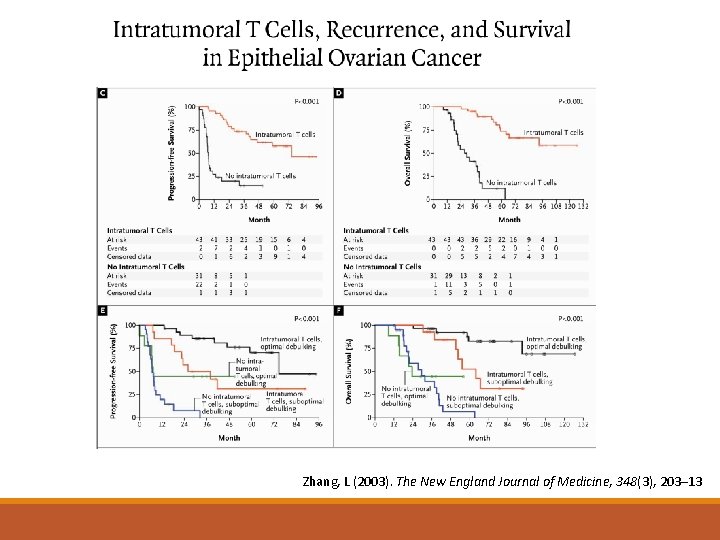
Zhang, L (2003). The New England Journal of Medicine, 348(3), 203– 13
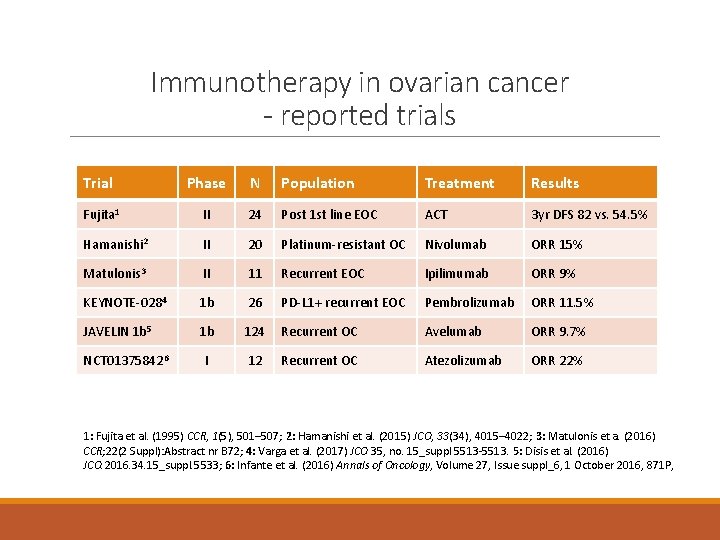
Immunotherapy in ovarian cancer - reported trials Trial Phase N Population Treatment Results Fujita 1 II 24 Post 1 st line EOC ACT 3 yr DFS 82 vs. 54. 5% Hamanishi 2 II 20 Platinum-resistant OC Nivolumab ORR 15% Matulonis 3 II 11 Recurrent EOC Ipilimumab ORR 9% KEYNOTE-0284 1 b 26 PD-L 1+ recurrent EOC Pembrolizumab ORR 11. 5% JAVELIN 1 b 5 1 b 124 Recurrent OC Avelumab ORR 9. 7% I 12 Recurrent OC Atezolizumab ORR 22% NCT 013758426 1: Fujita et al. (1995) CCR, 1(5), 501– 507; 2: Hamanishi et al. (2015) JCO, 33(34), 4015– 4022; 3: Matulonis et a. (2016) CCR; 22(2 Suppl): Abstract nr B 72; 4: Varga et al. (2017) JCO 35, no. 15_suppl 5513 -5513. 5: Disis et al. (2016) JCO. 2016. 34. 15_suppl. 5533; 6: Infante et al. (2016) Annals of Oncology, Volume 27, Issue suppl_6, 1 October 2016, 871 P,

Nivolumab (anti-PD-1) in Patients with Platinum-Resistant Ovarian Cancer Hamanishi, J (2015. Journal of Clinical Oncology, 33(34), 4015– 4022.

KEYNOTE-100 (NCT 02674061): Phase 2, Two-Cohort Study of Pembrolizumab for Recurrent Advanced Ovarian Cancer Presented By Ursula Matulonis at 2018 ASCO Annual Meeting
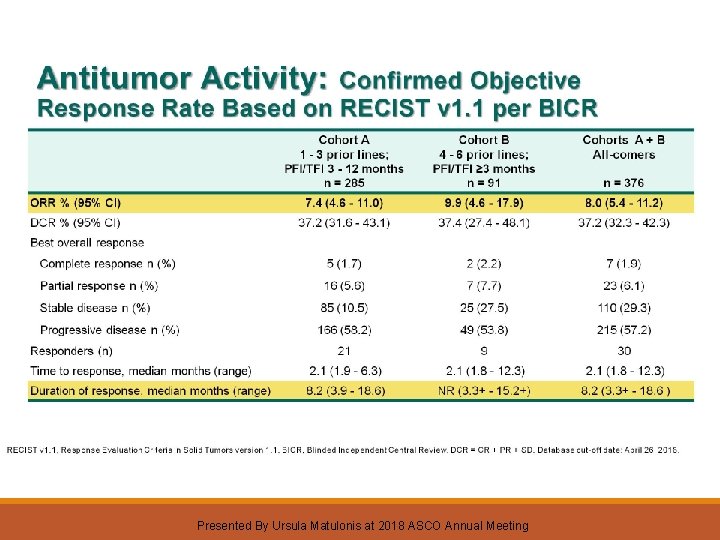
Presented By Ursula Matulonis at 2018 ASCO Annual Meeting
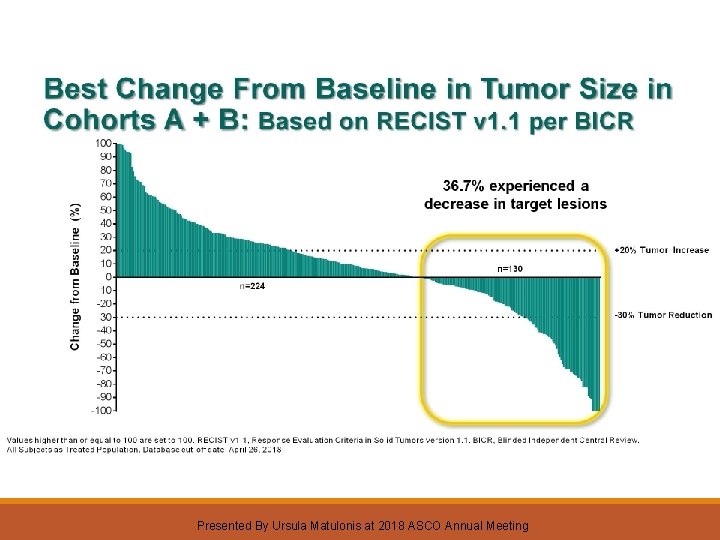
Best Change From Baseline in Tumor Size in Cohorts A + B: Based on RECIST v 1. 1 per BICR Presented By Ursula Matulonis at 2018 ASCO Annual Meeting
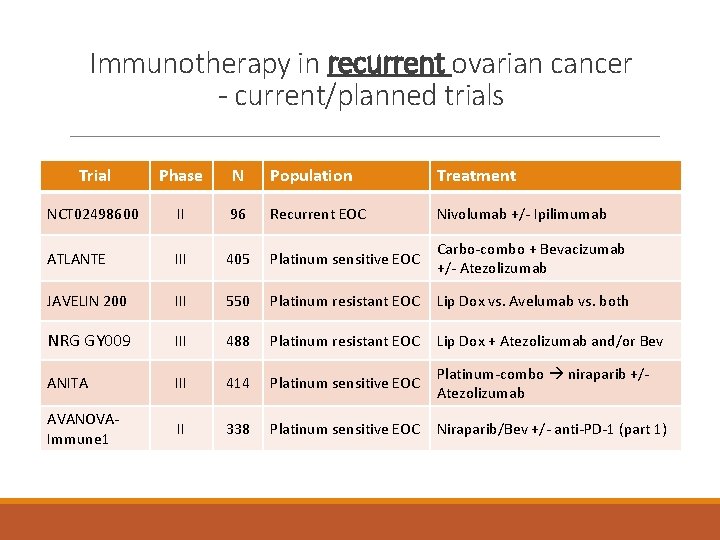
Immunotherapy in recurrent ovarian cancer - current/planned trials Trial Phase N Population Treatment NCT 02498600 II 96 Recurrent EOC Nivolumab +/- Ipilimumab ATLANTE III 405 Platinum sensitive EOC Carbo-combo + Bevacizumab +/- Atezolizumab JAVELIN 200 III 550 Platinum resistant EOC Lip Dox vs. Avelumab vs. both NRG GY 009 III 488 Platinum resistant EOC Lip Dox + Atezolizumab and/or Bev ANITA III 414 Platinum sensitive EOC Platinum-combo niraparib +/- Atezolizumab AVANOVAImmune 1 II 338 Platinum sensitive EOC Niraparib/Bev +/- anti-PD-1 (part 1)

Immunotherapy in newly diagnosed ovarian cancer - current/planned trials Trial Phase N Population Treatment i. PRIME II 75 Fronline EOC NACT +/- Durvalumab + Tremelimumab JAVELIN 100 III 951 Frontline EOC CP +/- Avelumab IMa. GYN 050 III 1300 Frontline EOC CP + Bev +/- Atezolizumab ATHENA III 1000 Frontline EOC CP Rucaparib and/or Nivolumab FIRST III 700 Frontline EOC CP +/- TSR-042 + niraparib ENGOT ov 43 III 1500 Frontline EOC CP +/- pembrolizumab + olaparib

Dendritic Cell Vaccine With Chemotherapy In Patients With Epithelial Ovarian Carcinoma After Primary Debulking Surgery Presented By Lukas Rob at 2018 ASCO Annual Meeting

DCVAC/Ov. Ca Presented By Lukas Rob at 2018 ASCO Annual Meeting
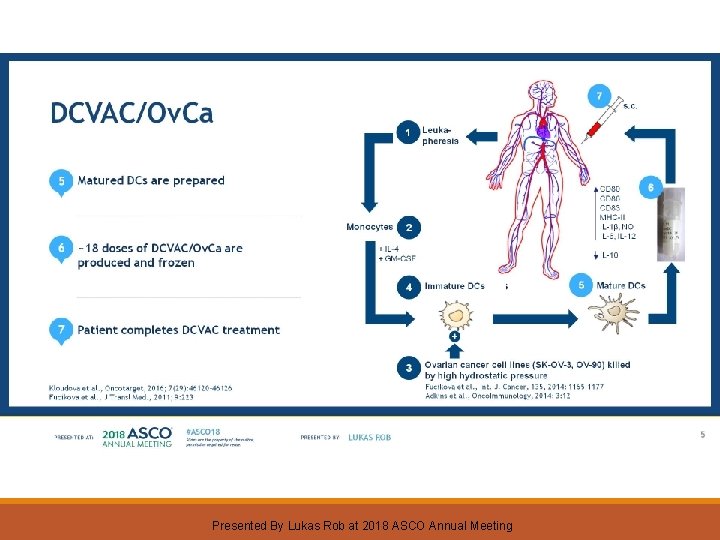
DCVAC/Ov. Ca Presented By Lukas Rob at 2018 ASCO Annual Meeting
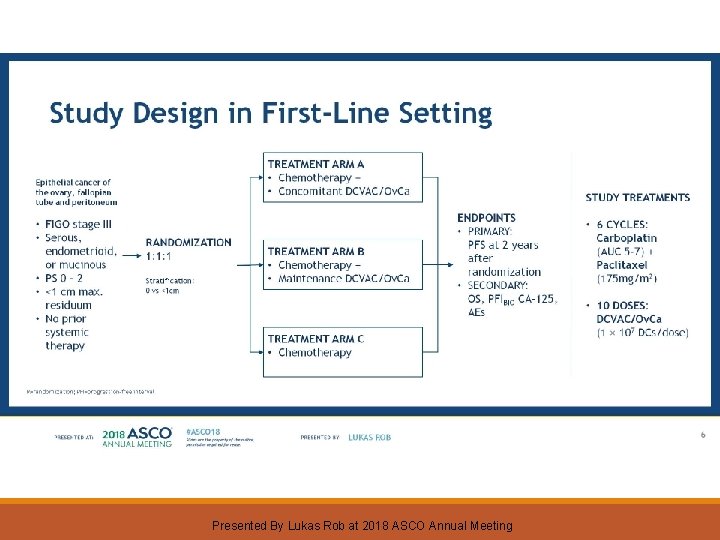
Study Design in First-Line Setting Presented By Lukas Rob at 2018 ASCO Annual Meeting
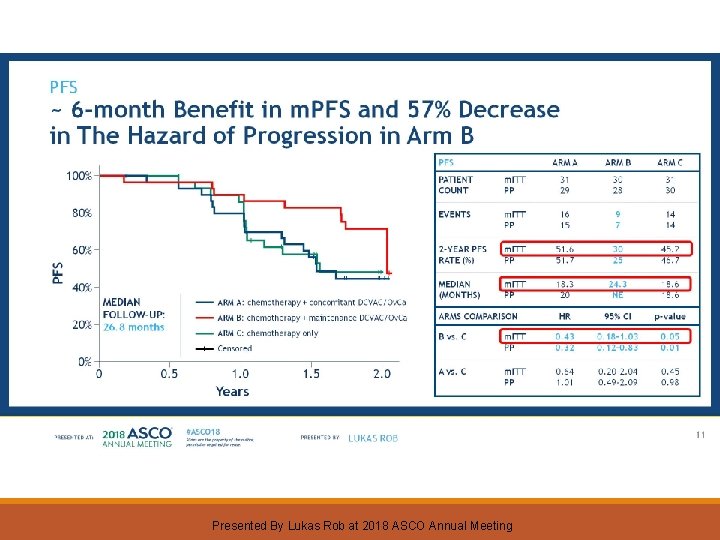
PFS ~ 6 -month Benefit in m. PFS and 57% Decrease in The Hazard of Progression in Arm B Presented By Lukas Rob at 2018 ASCO Annual Meeting
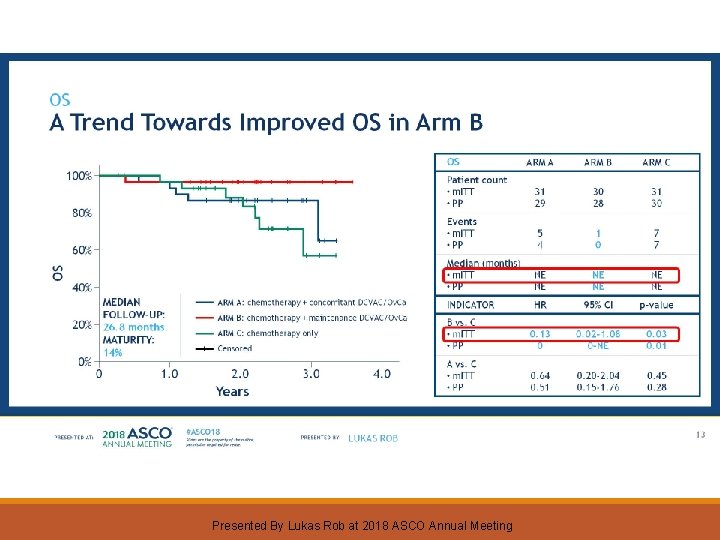
OS A Trend Towards Improved OS in Arm B Presented By Lukas Rob at 2018 ASCO Annual Meeting

Future directions: ‘closing’ the immune cycle
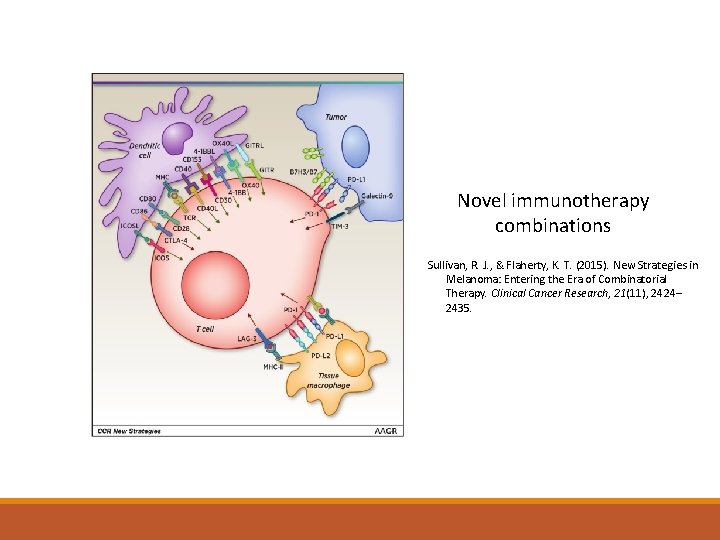
Novel immunotherapy combinations Sullivan, R. J. , & Flaherty, K. T. (2015). New Strategies in Melanoma: Entering the Era of Combinatorial Therapy. Clinical Cancer Research, 21(11), 2424– 2435.

New ideas Combination therapy 1. Adding to chemotherapy (already being trialled successfully in lung cancer 2. Combination immunotherapy – successful in melanoma treatment 3. Adding immunotherapy to other targeted agents (PARPi in ovarian cancer) 4. Better understanding of which treatment works for which patient
Willis, J. C. D (2015). Nature Reviews Immunology, 15(March), 1– 7

Can we afford to wait for all these trials of promising drugs? Can we afford not to? April 2016 cover (reproduced without permission)

Immunotherapy in gynaeoclogical cancers…. both good and bad news

Conclusion v. Good news : vwe have identified a group of patients with endometrial cancer who definitely respond to immunotherapy v. Many new trials are underway or reporting v. New ideas are being generated every month v. Not so good news: v. Response rates - so far - are generally modest, 10 -20%, but can last for some time v. Need to be smarter in picking the right patients and the right time in treatment to get the best results v. Bad news v. Immunotherapy doesn’t work for many patient with ovarian cancer v. Costs are high v. Approval is difficult
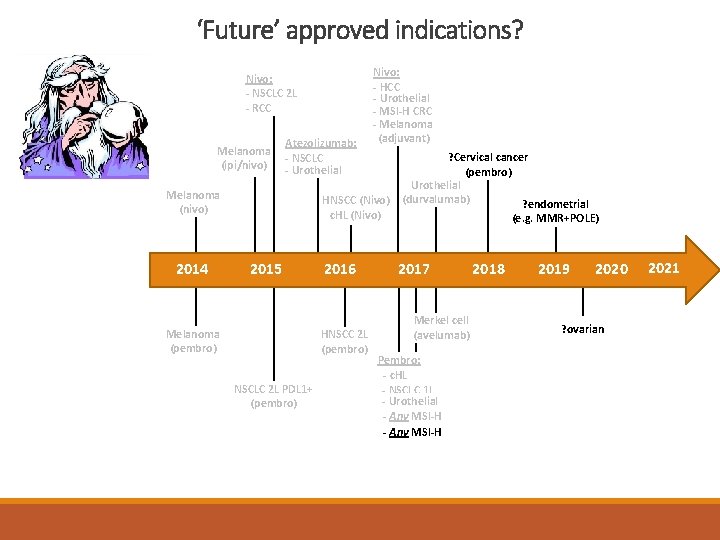
‘Future’ approved indications? Nivo: - NSCLC 2 L - RCC Melanoma (ipi/nivo) Atezolizumab: - NSCLC - Urothelial Melanoma (nivo) 2014 Nivo: - HCC - Urothelial - MSI-H CRC - Melanoma (adjuvant) HNSCC (Nivo) c. HL (Nivo) 2015 Melanoma (pembro) 2016 HNSCC 2 L (pembro) NSCLC 2 L PDL 1+ (pembro) ? Cervical cancer (pembro) Urothelial (durvalumab) ? endometrial (e. g. MMR+POLE) 2017 Merkel cell (avelumab) Pembro: - c. HL - NSCLC 1 L - Urothelial - Any MSI-H 2018 2019 2020 ? ovarian 2021
- Slides: 42