Immunosuppressive agents Biologic agents2 Outlines Current therapeutic agents

Immunosuppressive agents. Biologic agents-2 三軍總醫院 風濕免疫過敏科

Outlines Current therapeutic agents using in rheumatoid arthritis: B cell target therapy The Janus Kinases (JAK) inhibitor Current therapeutic agents using in SLE: Belimumab
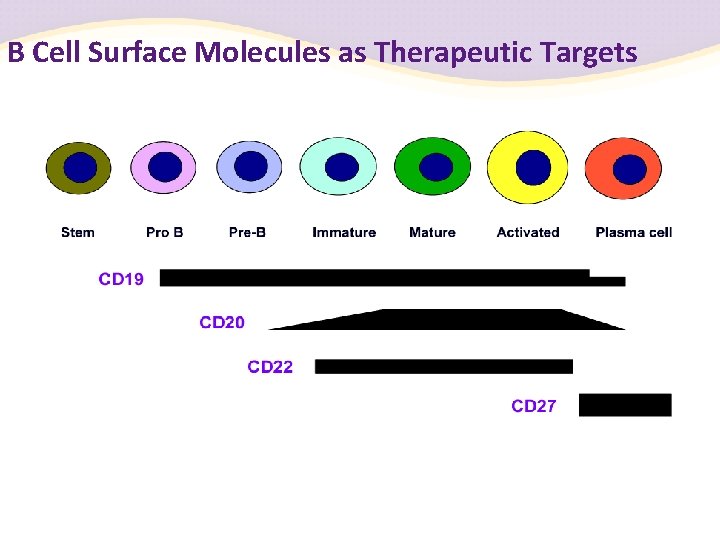
B Cell Surface Molecules as Therapeutic Targets

Rituximab (Mab. Thera®) A chimeric murine/ human monoclonal antibody against CD 20 that is expressed on B cells, developed in 1994 Rituximab in combination with methotrexate for RA refractory to anti. TNF-a therapy (Arthritis & Rheumatism 2006; 54: 2793– 2806) Dosage: 500 to 1000 mg intravenous infusion slowly on Day 1 and Day 15 B cells are markedly depleted for a prolonged time (> 6 months) Adverse events: infusion reaction

More Biological Agents for the RA Treatment by Targeting B Cell Ocrelizumab: a second generation chimeric murine/ human monoclonal antibody against CD 20, more “humanized” Ofatumumab (Hu. Max-CD 20): a fully human monoclonal antibody against CD 20 Belimumab (Lympho. Stat-B): Monoclonal Anti-BLy. S Antibody Atacicept: TACI-Ig for anti-BLy. S therapy Epratuzumab: a humanized anti-CD 22 antibody

More Biological Agents for the RA Treatment by Targeting B Cell Ocrelizumab: a second generation chimeric murine/ human monoclonal antibody against CD 20, more “humanized” Ofatumumab (Hu. Max-CD 20): a fully human monoclonal antibody against CD 20 Belimumab (Lympho. Stat-B): Monoclonal Anti-BLy. S Antibody Atacicept: TACI-Ig for anti-BLy. S therapy Epratuzumab: a humanized anti-CD 22 antibody

The Janus Kinases (JAK) inhibitor

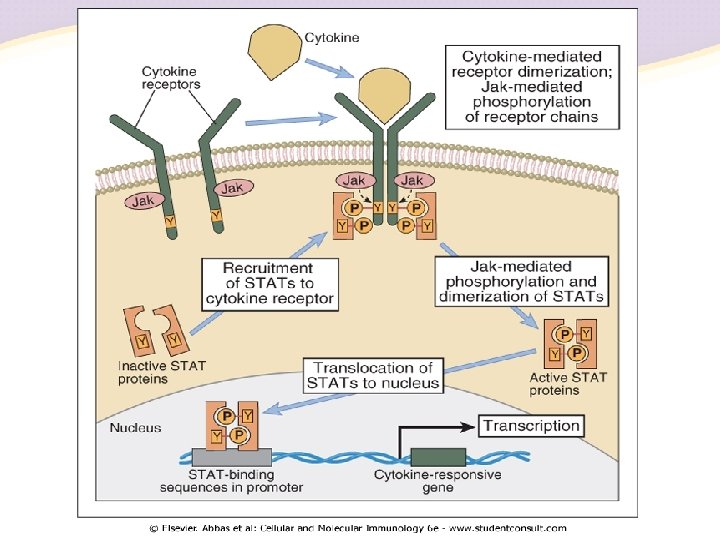
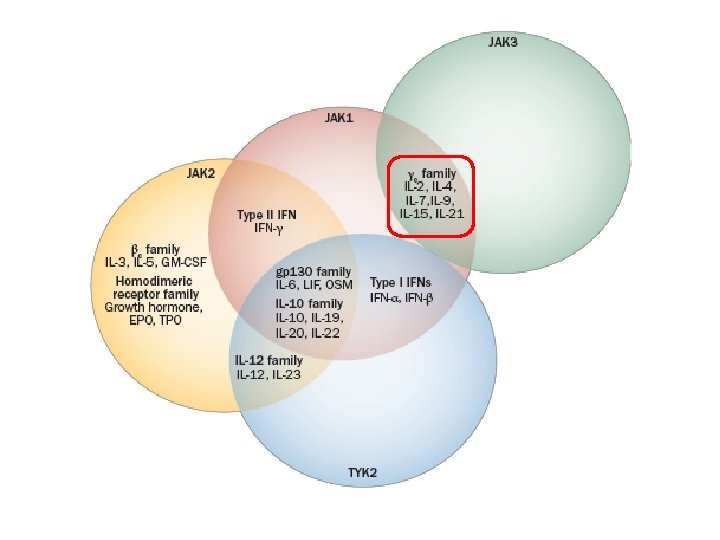
Janus kinase inhibitors • The Janus Kinases (JAK) are a family of tyrosine kinases that mediate intracellular signal transduction of cytokines in immune cells • Four Jaks have been identified in mammals: Jak 1, Jak 2, Jak 3 and Tyk 2 • Only Jak 3 shows restricted expression being confined predominately to cells of hematopoietic origin

Janus kinase inhibitors • JAK 3, in combination with JAK 1, is critical for signal transduction from the common γ-chain of receptors for IL-2, IL-4, IL- 7, IL- 9, IL- 15, and IL- 21 • These γ-chain interleukins are critical to lymphocyte activation, function and proliferation • JAK 3 expression is primarily limited to hematopoietic cells, including NK cells, thymocytes and activated T and B lymphocytes, but not in resting T cells • JAK 3 -deficient mice have defects in T, B and NK cells
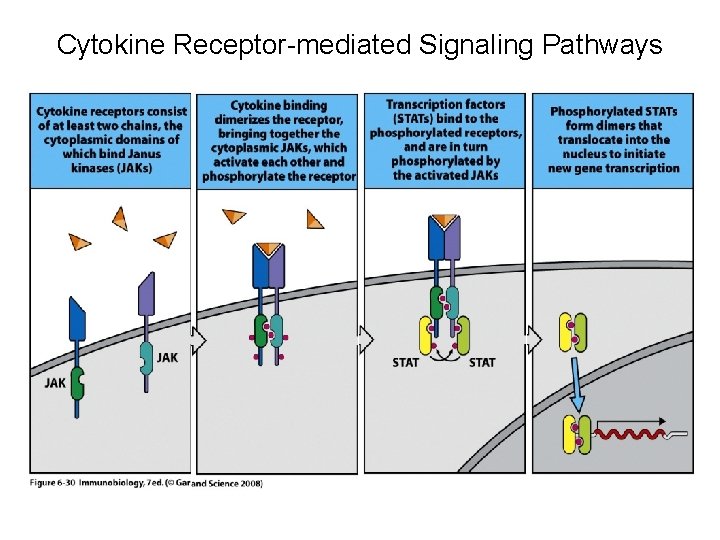
Cytokine Receptor-mediated Signaling Pathways
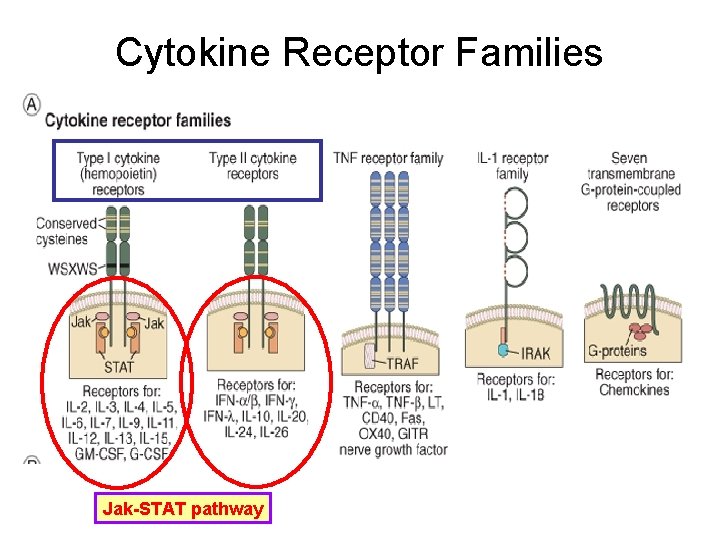
Cytokine Receptor Families Jak-STAT pathway

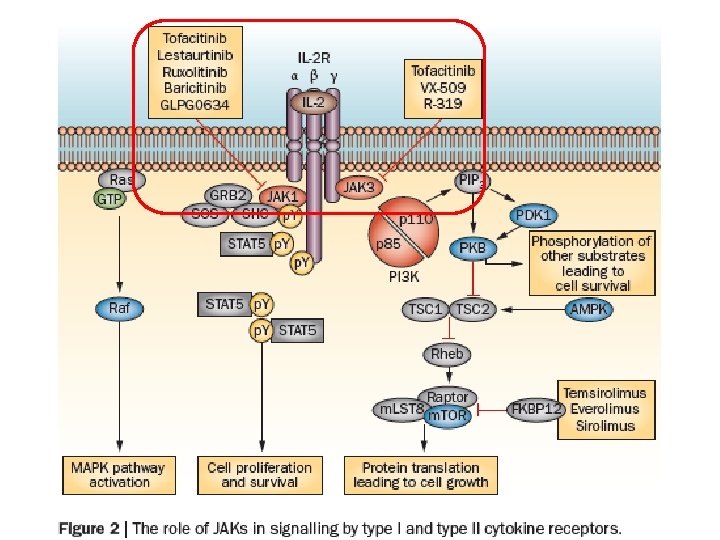
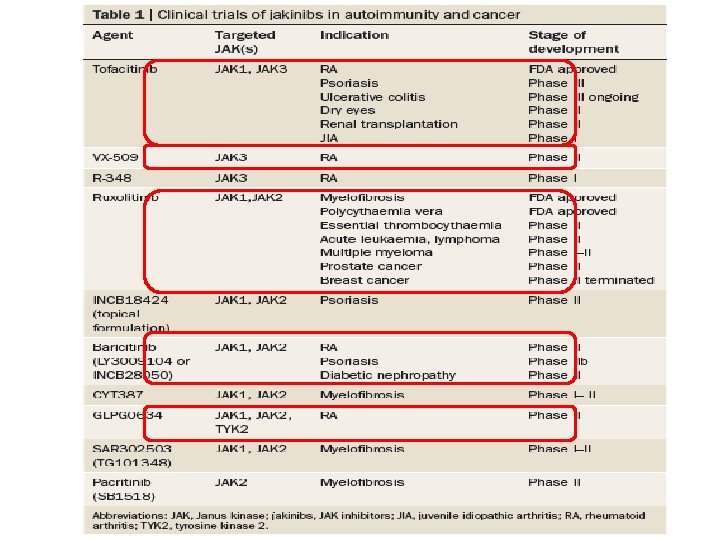
Janus kinase inhibitors • The first selective JAK inhibitor to be tested in humans was tofacitinib (formerly designated CP‑ 690, 550) • Tofacitinib inhibits JAK 1, JAK 3 and, to a lesser extent, JAK 2, this compound has limited effect on TYK 2 and on kinases other than JAKs • Efficacious in a variety of preclinical models ranging from transplant to arthritis models in multiple clinical trials in RA, inflammatory bowel disease, psoriasis and renal transplantation


Antibodies as Biological Agents Down-regulation of cell function Molecular blockade (‘coating’) Modulation of target antigen from the cell surface Cell depletion Clearance from the circulation Complement-dependent cytotoxicity (CDC) Antibody-dependent cell-mediated cytotoxicity (ADCC) Induction of apoptosis

The Future Need in RA Therapy Many patients are partial responders True remission is achieved by only a minority Destructive process cannot be halted in all patients ‘Cure’ remains an elusive goal Repair of previous damage remains elusive Adverse effects of biologic therapies might not be entirely predictable

Current therapeutic agents using in SLE: Belimumab
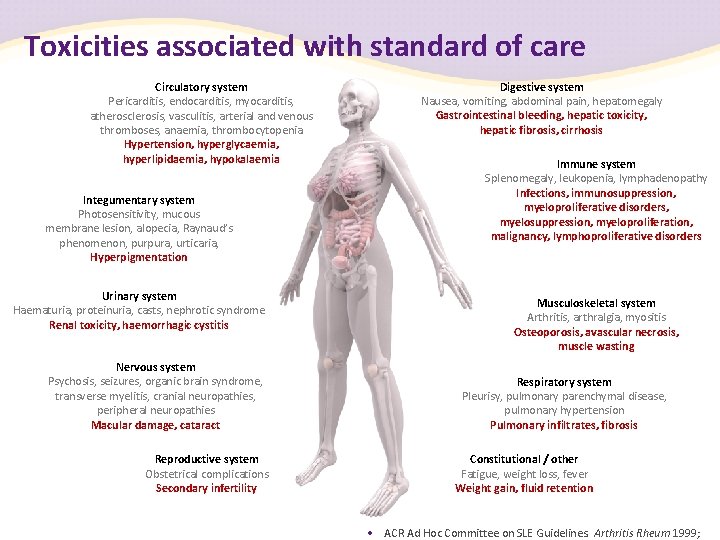
Toxicities associated with standard of care Circulatory system Pericarditis, endocarditis, myocarditis, atherosclerosis, vasculitis, arterial and venous thromboses, anaemia, thrombocytopenia Hypertension, hyperglycaemia, hyperlipidaemia, hypokalaemia Integumentary system Photosensitivity, mucous membrane lesion, alopecia, Raynaud’s phenomenon, purpura, urticaria, Hyperpigmentation Urinary system Haematuria, proteinuria, casts, nephrotic syndrome Renal toxicity, haemorrhagic cystitis Nervous system Psychosis, seizures, organic brain syndrome, transverse myelitis, cranial neuropathies, peripheral neuropathies Macular damage, cataract Reproductive system Obstetrical complications Secondary infertility Digestive system Nausea, vomiting, abdominal pain, hepatomegaly Gastrointestinal bleeding, hepatic toxicity, hepatic fibrosis, cirrhosis Immune system Splenomegaly, leukopenia, lymphadenopathy Infections, immunosuppression, myeloproliferative disorders, myelosuppression, myeloproliferation, malignancy, lymphoproliferative disorders Musculoskeletal system Arthritis, arthralgia, myositis Osteoporosis, avascular necrosis, muscle wasting Respiratory system Pleurisy, pulmonary parenchymal disease, pulmonary hypertension Pulmonary infiltrates, fibrosis Constitutional / other Fatigue, weight loss, fever Weight gain, fluid retention • ACR Ad Hoc Committee on SLE Guidelines. Arthritis Rheum 1999;
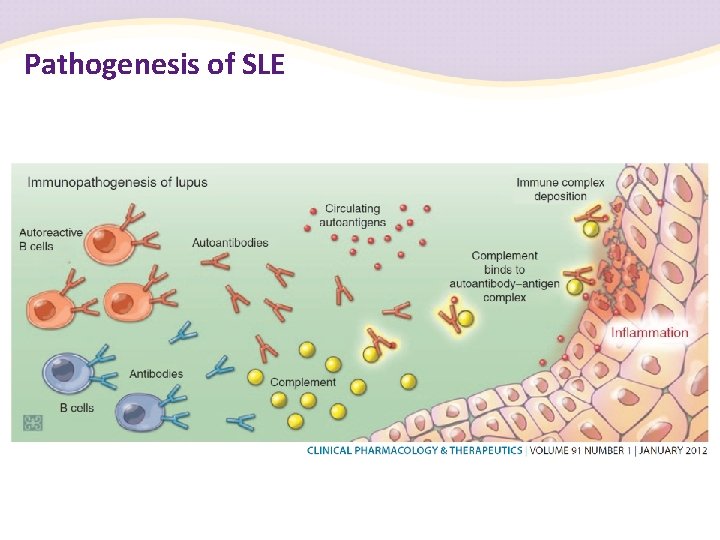
Pathogenesis of SLE
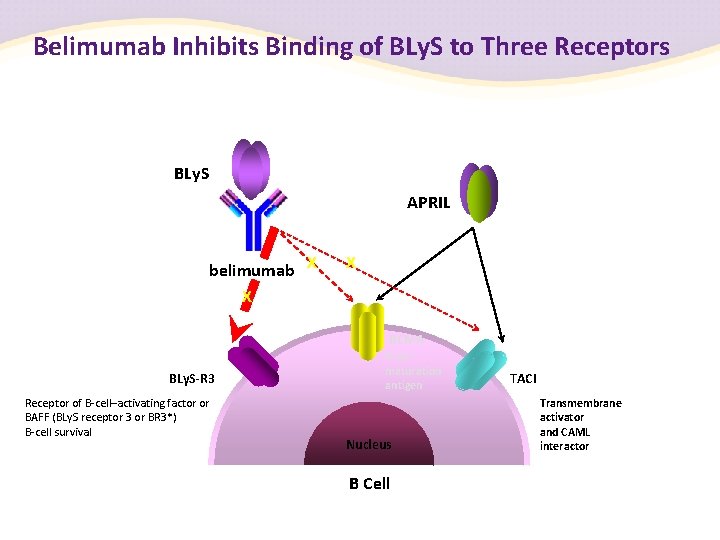
Belimumab Inhibits Binding of BLy. S to Three Receptors BLy. S APRIL belimumab X X X BCMA BLy. S-R 3 Receptor of B-cell–activating factor or BAFF (BLy. S receptor 3 or BR 3*) B-cell survival B cell maturation antigen Nucleus B Cell TACI Transmembrane activator and CAML interactor
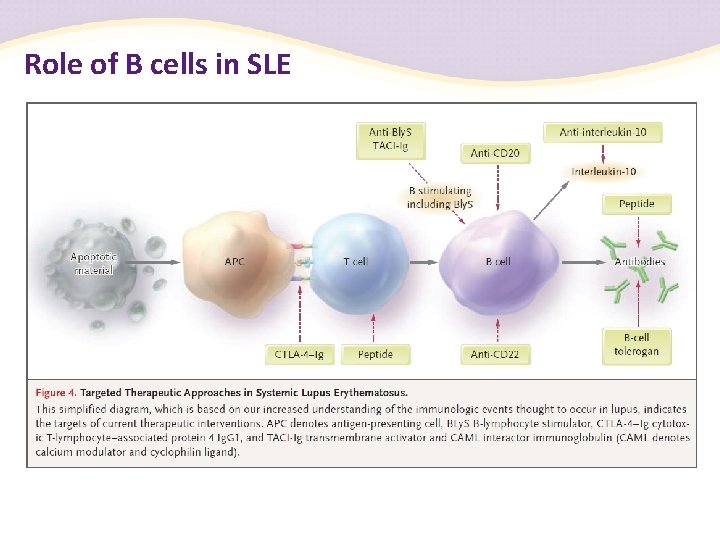
Role of B cells in SLE

Goals of SLE treatment 1. Tailor treatment to the extent and severity of organ involvement 2. Adequately control inflammatory disease activity while minimising drug side-effects 3. Prevent organ damage 4. Maintain the patient’s daily function and improve quality of life (Qo. L) • Kalunian K & Merrill JT. Curr Med Res Opin 2009; 25: 1501– 1514.

Summary of BLy. S • B-lymphocyte stimulator (BLy. S)1 – Expressed by monocytes, activated neutrophils, T cells and dendritic cells – Has been shown to bind to three receptors on the surface of B cells (BR 3, TACI and BCMA) – Has been shown to promote B-cell survival and isotype switching (Ig. M/Ig. D to Ig. G) • BLy. S overexpression induced SLE-like disease in multiple mouse strains 2, 3 • BLy. S blockade in a mouse model of SLE prevented disease onset and led to remission 4 BCMA = B-cell maturation antigen; BR 3 = B-cell activation factor receptor-3; TACI = trans-membrane activator and calcium-modulator and cytophilin ligand interactor. 1. Cancro MP, et al. J Clin Invest 2009; 119: 1066– 1073; 2. Mackay F, et al. J Exp Med 1999; 190: 1697– 1710; 3. Khare SD, et al. Proc Natl Acad Sci USA 2000; 97: 3370– 3375; 4. Ramanujam M, et al. Arthritis Rheum 2010; 62: 1457– 1468.
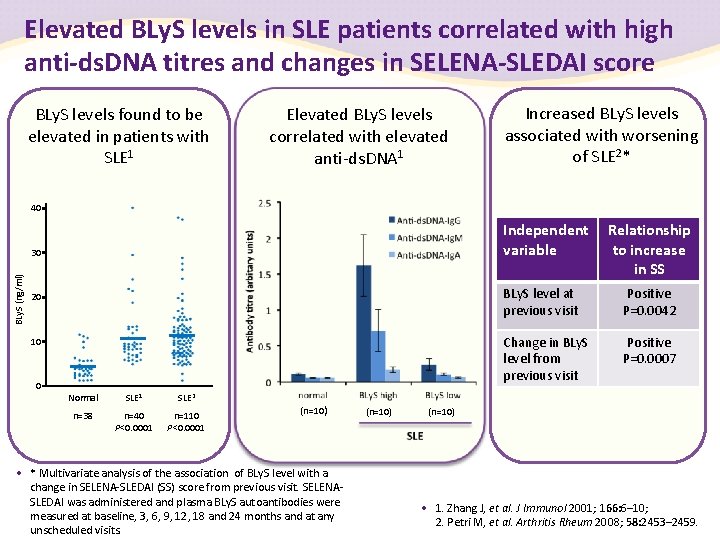
Elevated BLy. S levels in SLE patients correlated with high anti-ds. DNA titres and changes in SELENA-SLEDAI score BLy. S levels found to be elevated in patients with SLE 1 Elevated BLy. S levels correlated with elevated anti-ds. DNA 1 Increased BLy. S levels associated with worsening of SLE 2* 40 Independent variable BLy. S (ng/ml) 30 20 10 0 Normal SLE 1 SLE 2 n=38 n=40 P<0. 0001 n=110 P<0. 0001 (n=10) • * Multivariate analysis of the association of BLy. S level with a change in SELENA-SLEDAI (SS) score from previous visit. SELENASLEDAI was administered and plasma BLy. S autoantibodies were measured at baseline, 3, 6, 9, 12, 18 and 24 months and at any unscheduled visits. (n=10) Relationship to increase in SS BLy. S level at previous visit Positive P=0. 0042 Change in BLy. S level from previous visit Positive P=0. 0007 (n=10) SLE • 1. Zhang J, et al. J Immunol 2001; 166: 6– 10; 2. Petri M, et al. Arthritis Rheum 2008; 58: 2453– 2459.

Belimumab · Fully human Ig. G 1 l monoclonal antibody · Selectively targets and inhibits biologic activity of soluble BLy. S · Inhibition of BLy. S may result in autoreactive B-cell apoptosis Baker KP et al. Arthritis Rheum. 2003; 48: 3253 -65.
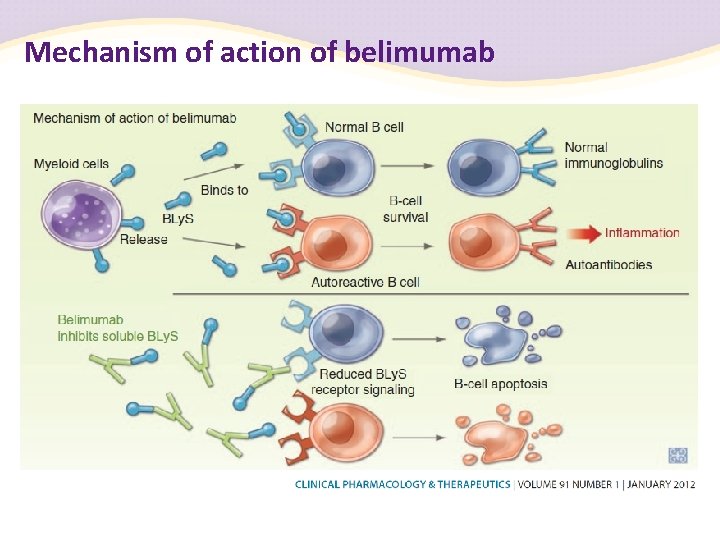
Mechanism of action of belimumab

Summary of Belimumab Clinical Studies in SLE* Phase Study Identifier 1 2 3 3 Randomized Patient Population N Treatment Duration (doses) LBSL 01 (NCT 00657007)1 SLE with stable disease for ≥ 2 mo prior to screening 70 21 d (1, 4, 10, or 20 mg/kg; 1 or 2 IV, 21 d apart) LBSL 02 (NCT 00071487)2, 3 Active SLE defined as SELENASLEDAI ≥ 4 at screening, stable SLE treatment, history of autoantibodies 449 76 wk (1, 4, 10 mg/kg IV; 52 -wk placebo-controlled treatment phase and 24 -wk extension) BLISS-52 (NCT 00424476)4 Active SLE defined as SELENASLEDAI ≥ 6, stable SLE treatment, and + ANA/ds. DNA at 2 independent time points 865 52 wk (1 or 10 mg/kg IV, placebo controlled) BLISS-76 (NCT 00410384)5 Active SLE defined as SELENASLEDAI ≥ 6, stable SLE treatment, +ANA/ds. DNA at 2 independent time points 819 76 wk (1 or 10 mg/kg IV, placebo controlled); primary endpoint at 52 wk *Excludes continuation clinical trials. 1. Furie R et al. Arthritis Res Ther. 2008; 10: R 109. 2. Wallace DJ et al. Arthritis Rheum. 2009; 61: 1168 -78. 3. Petri MA et al. ACR 2009. Abstr 2069. 4. Navarra S et al. ACR 2009. Abstr LB 1. 5. Human Genome Sciences. BENLYSTA™ (belimumab). Available at: http: //www. hgsi. com/belimumab. html.
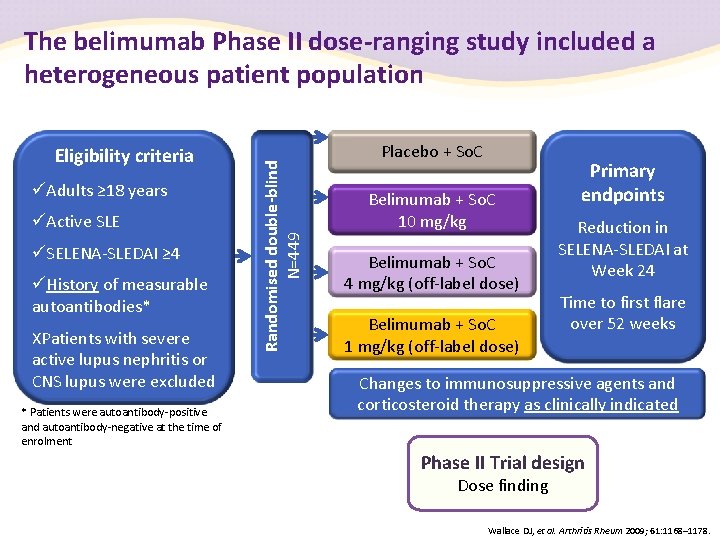
Eligibility criteria Adults ≥ 18 years Active SLE SELENA-SLEDAI ≥ 4 History of measurable autoantibodies* XPatients with severe active lupus nephritis or CNS lupus were excluded * Patients were autoantibody-positive and autoantibody-negative at the time of enrolment Randomised double-blind N=449 The belimumab Phase II dose-ranging study included a heterogeneous patient population Placebo + So. C Belimumab + So. C 10 mg/kg Belimumab + So. C 4 mg/kg (off-label dose) Belimumab + So. C 1 mg/kg (off-label dose) Primary endpoints Reduction in SELENA-SLEDAI at Week 24 Time to first flare over 52 weeks Changes to immunosuppressive agents and corticosteroid therapy as clinically indicated Phase II Trial design Dose finding Wallace DJ, et al. Arthritis Rheum 2009; 61: 1168– 1178.
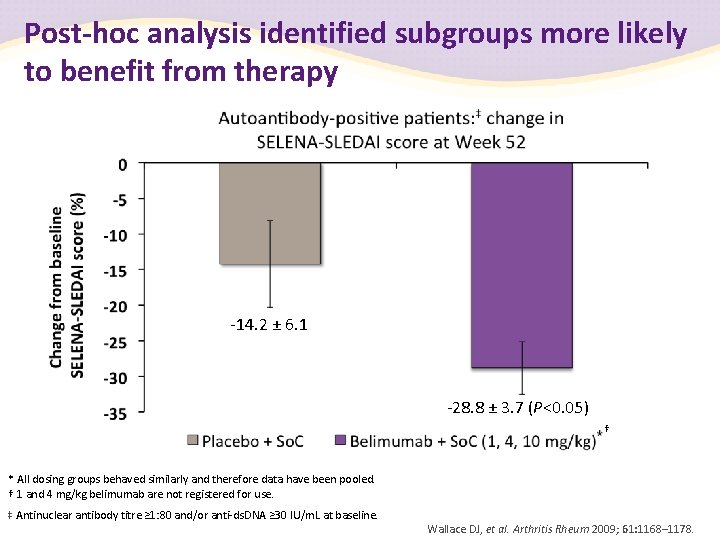
Post-hoc analysis identified subgroups more likely to benefit from therapy -14. 2 ± 6. 1 -28. 8 ± 3. 7 (P<0. 05) † * All dosing groups behaved similarly and therefore data have been pooled. † 1 and 4 mg/kg belimumab are not registered for use. ‡ Antinuclear antibody titre ≥ 1: 80 and/or anti-ds. DNA ≥ 30 IU/m. L at baseline. Wallace DJ, et al. Arthritis Rheum 2009; 61: 1168– 1178.
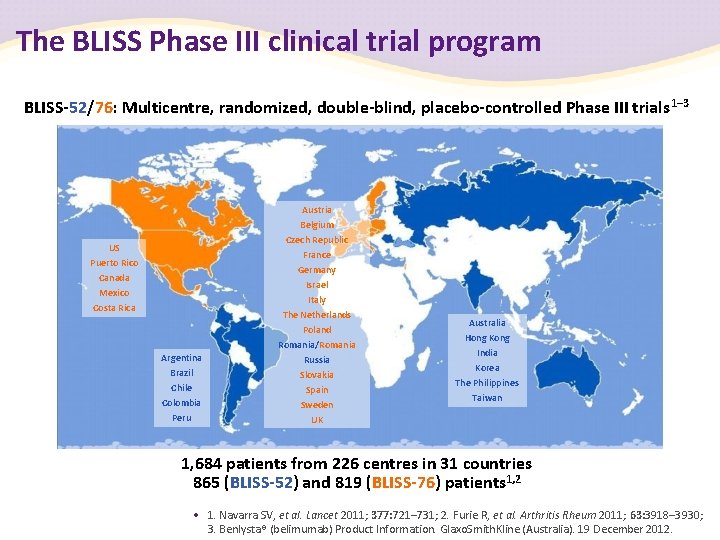
The BLISS Phase III clinical trial program BLISS-52/76: Multicentre, randomized, double-blind, placebo-controlled Phase III trials 1– 3 Austria US Puerto Rico Canada Mexico Costa Rica Argentina Brazil Chile Colombia Peru Belgium Czech Republic France Germany Israel Italy The Netherlands Poland Romania/Romania Russia Slovakia Spain Sweden UK Australia Hong Kong India Korea The Philippines Taiwan 1, 684 patients from 226 centres in 31 countries 865 (BLISS-52) and 819 (BLISS-76) patients 1, 2 • 1. Navarra SV, et al. Lancet 2011; 377: 721– 731; 2. Furie R, et al. Arthritis Rheum 2011; 63: 3918– 3930; 3. Benlysta® (belimumab) Product Information. Glaxo. Smith. Kline (Australia). 19 December 2012.

Surrogate markers of high disease activity were selected to define subgroup for further analyses Parameters associated with greater clinical benefit SELENA-SLEDAI ≥ 10 Not routine in all clinical practice Anti-ds. DNA ≥ 30 IU/m. L Routine laboratory assessment Low C 3 <90 mg/d. L Routine laboratory assessment Low C 4 <16 mg/d. L Routine laboratory assessment Steroid use (yes/no) Depends on physician judgement and access to treatment The high disease activity subgroup was defined using laboratoryassessed disease parameters routinely used in clinical practice 1. Low complement: C 3 <90 mg/d. L and/or C 4 <16 mg/d. L at baseline 2. Anti-ds. DNA-positive: anti-ds. DNA ≥ 30 IU/m. L at baseline van Vollenhoven RF, et al. Ann Rheum Dis 2012; 71: 1343– 1349.
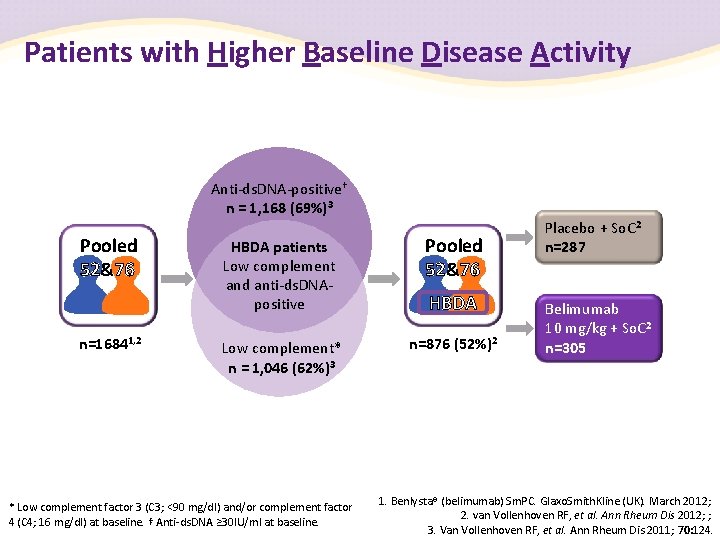
Patients with Higher Baseline Disease Activity Anti-ds. DNA-positive† n = 1, 168 (69%)3 Pooled 52&76 n=16841, 2 HBDA patients Low complement and anti-ds. DNApositive Pooled 52&76 Low complement* n = 1, 046 (62%)3 n=876 (52%)2 * Low complement factor 3 (C 3; <90 mg/dl) and/or complement factor 4 (C 4; 16 mg/dl) at baseline. † Anti-ds. DNA ≥ 30 IU/ml at baseline. HBDA Placebo + So. C 2 n=287 Belimumab 10 mg/kg + So. C 2 n=305 1. Benlysta® (belimumab) Sm. PC. Glaxo. Smith. Kline (UK). March 2012; 2. van Vollenhoven RF, et al. Ann Rheum Dis 2012; ; 3. Van Vollenhoven RF, et al. Ann Rheum Dis 2011; 70: 124.
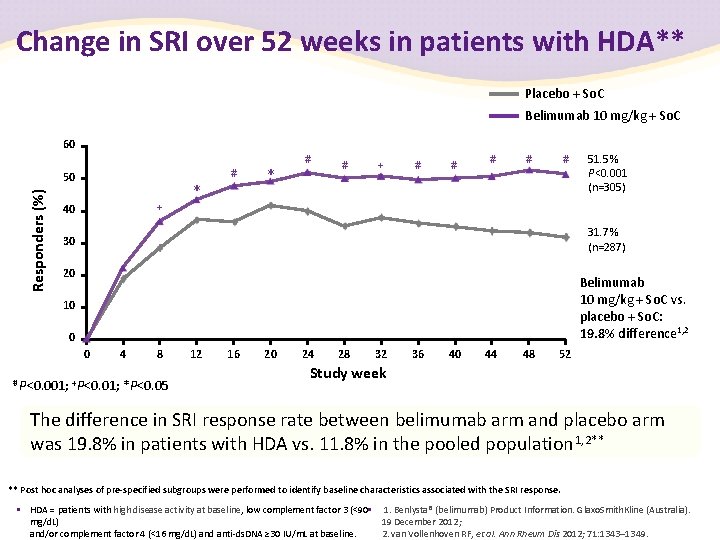
Change in SRI over 52 weeks in patients with HDA** Placebo + So. C Belimumab 10 mg/kg + So. C 60 # Responders (%) 50 + 40 * * # # + # # # 51. 5% P<0. 001 (n=305) 31. 7% (n=287) 30 20 Belimumab 10 mg/kg + So. C vs. placebo + So. C: 19. 8% difference 1, 2 10 0 0 4 8 # P<0. 001; +P<0. 01; *P<0. 05 12 16 20 24 28 32 36 40 44 48 52 Study week The difference in SRI response rate between belimumab arm and placebo arm was 19. 8% in patients with HDA vs. 11. 8% in the pooled population 1, 2** ** Post hoc analyses of pre-specified subgroups were performed to identify baseline characteristics associated with the SRI response. • HDA = patients with high disease activity at baseline, low complement factor 3 (<90 • 1. Benlysta® (belimumab) Product Information. Glaxo. Smith. Kline (Australia). 19 December 2012; mg/d. L) 2. van Vollenhoven RF, et al. Ann Rheum Dis 2012; 71: 1343– 1349. and/or complement factor 4 (<16 mg/d. L) and anti-ds. DNA ≥ 30 IU/m. L at baseline.
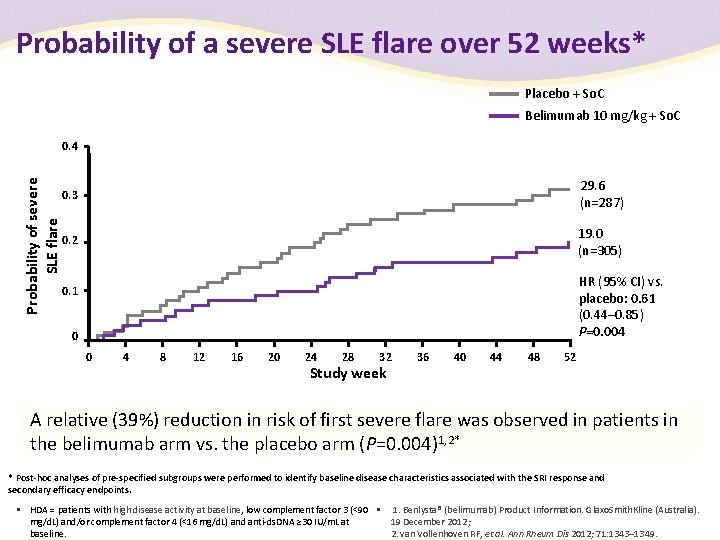
Probability of a severe SLE flare over 52 weeks* Placebo + So. C Belimumab 10 mg/kg + So. C Probability of severe SLE flare 0. 4 0. 3 29. 6 (n=287) 0. 2 19. 0 (n=305) HR (95% CI) vs. placebo: 0. 61 (0. 44– 0. 85) P=0. 004 0. 1 0 0 4 8 12 16 20 24 28 32 Study week 36 40 44 48 52 A relative (39%) reduction in risk of first severe flare was observed in patients in the belimumab arm vs. the placebo arm (P=0. 004)1, 2* * Post-hoc analyses of pre-specified subgroups were performed to identify baseline disease characteristics associated with the SRI response and secondary efficacy endpoints. • HDA = patients with high disease activity at baseline, low complement factor 3 (<90 • 1. Benlysta® (belimumab) Product Information. Glaxo. Smith. Kline (Australia). mg/d. L) and/or complement factor 4 (<16 mg/d. L) and anti-ds. DNA ≥ 30 IU/m. L at 19 December 2012; baseline. 2. van Vollenhoven RF, et al. Ann Rheum Dis 2012; 71: 1343– 1349.
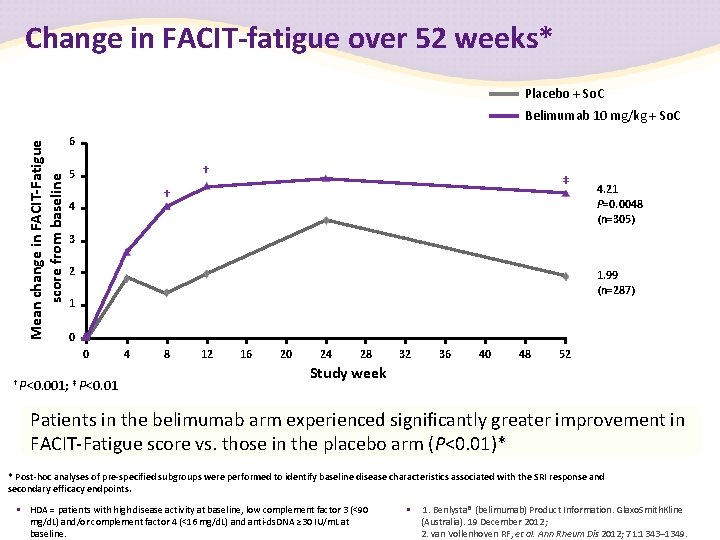
Change in FACIT-fatigue over 52 weeks* Placebo + So. C Mean change in FACIT-Fatigue score from baseline Belimumab 10 mg/kg + So. C 6 † 5 ‡ † 4 4. 21 P=0. 0048 (n=305) 3 2 1. 99 (n=287) 1 0 0 † P<0. 001; ‡ P<0. 01 4 8 12 16 20 24 28 32 36 40 48 52 Study week Patients in the belimumab arm experienced significantly greater improvement in FACIT-Fatigue score vs. those in the placebo arm (P<0. 01)* * Post-hoc analyses of pre-specified subgroups were performed to identify baseline disease characteristics associated with the SRI response and secondary efficacy endpoints. • HDA = patients with high disease activity at baseline, low complement factor 3 (<90 mg/d. L) and/or complement factor 4 (<16 mg/d. L) and anti-ds. DNA ≥ 30 IU/m. L at baseline. • 1. Benlysta® (belimumab) Product Information. Glaxo. Smith. Kline (Australia). 19 December 2012; 2. van Vollenhoven RF, et al. Ann Rheum Dis 2012; 71: 1343– 1349.
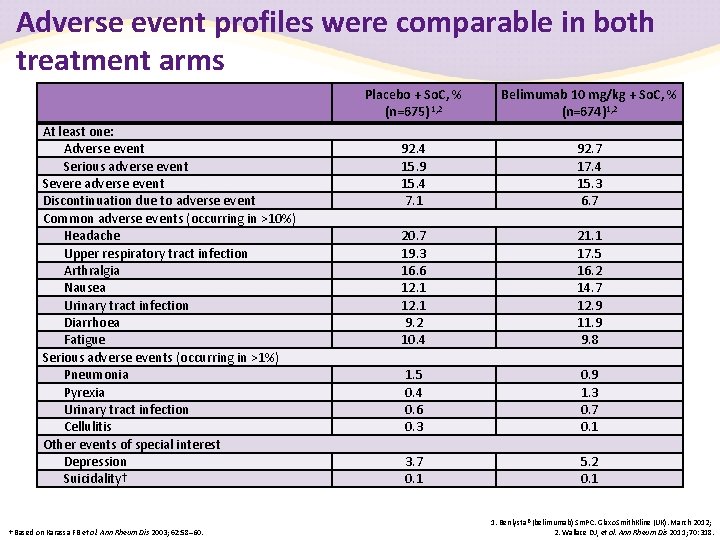
Adverse event profiles were comparable in both treatment arms At least one: Adverse event Serious adverse event Severe adverse event Discontinuation due to adverse event Common adverse events (occurring in >10%) Headache Upper respiratory tract infection Arthralgia Nausea Urinary tract infection Diarrhoea Fatigue Serious adverse events (occurring in >1%) Pneumonia Pyrexia Urinary tract infection Cellulitis Other events of special interest Depression Suicidality† † Based on Karassa FB et al. Ann Rheum Dis 2003; 62: 58– 60. Placebo + So. C, % (n=675) 1, 2 Belimumab 10 mg/kg + So. C, % (n=674)1, 2 92. 4 15. 9 15. 4 7. 1 92. 7 17. 4 15. 3 6. 7 20. 7 19. 3 16. 6 12. 1 9. 2 10. 4 21. 1 17. 5 16. 2 14. 7 12. 9 11. 9 9. 8 1. 5 0. 4 0. 6 0. 3 0. 9 1. 3 0. 7 0. 1 3. 7 0. 1 5. 2 0. 1 1. Benlysta® (belimumab) Sm. PC. Glaxo. Smith. Kline (UK). March 2012; 2. Wallace DJ, et al. Ann Rheum Dis 2011; 70: 318.
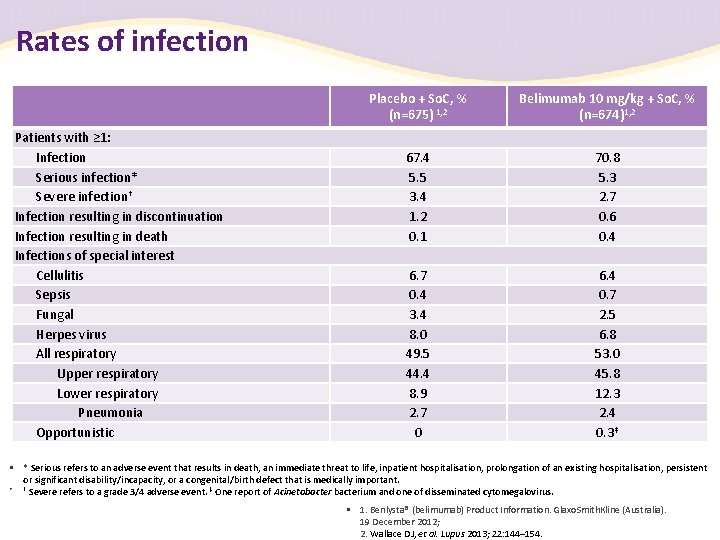
Rates of infection Patients with ≥ 1: Infection Serious infection* Severe infection† Infection resulting in discontinuation Infection resulting in death Infections of special interest Cellulitis Sepsis Fungal Herpes virus All respiratory Upper respiratory Lower respiratory Pneumonia Opportunistic Placebo + So. C, % (n=675) 1, 2 Belimumab 10 mg/kg + So. C, % (n=674)1, 2 67. 4 5. 5 3. 4 1. 2 0. 1 70. 8 5. 3 2. 7 0. 6 0. 4 6. 7 0. 4 3. 4 8. 0 49. 5 44. 4 8. 9 2. 7 0 6. 4 0. 7 2. 5 6. 8 53. 0 45. 8 12. 3 2. 4 0. 3‡ • * Serious refers to an adverse event that results in death, an immediate threat to life, inpatient hospitalisation, prolongation of an existing hospitalisation, persistent or significant disability/incapacity, or a congenital/birth defect that is medically important. • † Severe refers to a grade 3/4 adverse event. ‡ One report of Acinetobacterium and one of disseminated cytomegalovirus. • 1. Benlysta® (belimumab) Product Information. Glaxo. Smith. Kline (Australia). 19 December 2012; 2. Wallace DJ, et al. Lupus 2013; 22: 144– 154.
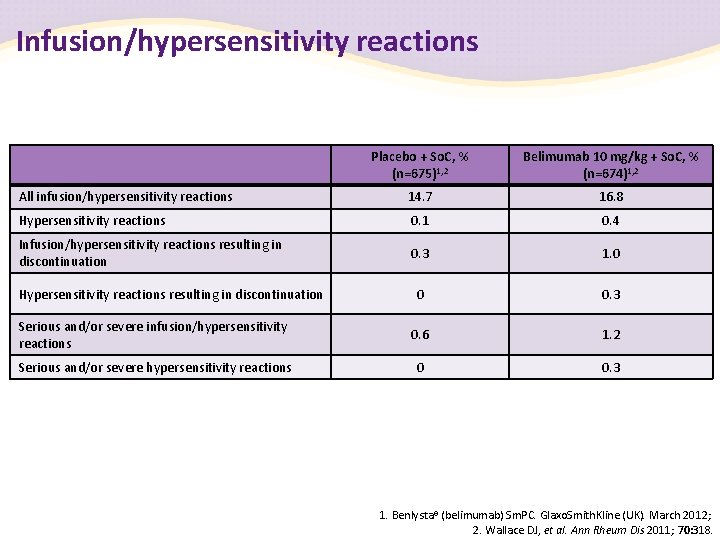
Infusion/hypersensitivity reactions Placebo + So. C, % (n=675)1, 2 Belimumab 10 mg/kg + So. C, % (n=674)1, 2 All infusion/hypersensitivity reactions 14. 7 16. 8 Hypersensitivity reactions 0. 1 0. 4 Infusion/hypersensitivity reactions resulting in discontinuation 0. 3 1. 0 0 0. 3 Serious and/or severe infusion/hypersensitivity reactions 0. 6 1. 2 Serious and/or severe hypersensitivity reactions 0 0. 3 Hypersensitivity reactions resulting in discontinuation 1. Benlysta® (belimumab) Sm. PC. Glaxo. Smith. Kline (UK). March 2012; 2. Wallace DJ, et al. Ann Rheum Dis 2011; 70: 318.

Summary: from primary efficacy endpoint to licensing Primary efficacy endpoint met 1, 2 Benlysta is indicated in Post-hoc analysis to identify parameters associated with greater clinical benefit Parameters associated with high disease activity 2 • SELENA-SLEDAI ≥ 10 • Anti-ds. DNA ≥ 30 IU/m. L • Low C 3 and/or Low C 4 • Corticosteroid use Surrogate marker of high disease activity agreed with regulators • Anti-ds. DNA ≥ 30 IU/m. L • Low C 3 and/or Low C 4 adult patients with active, autoantibody-positive SLE with a high degree of disease activity, desperate So. C. High disease activity classifies as (must meet all three criterion listed below): (1). Positive anti-ds. DNA (2). Low complement (3). SELENA SLEDAI≧ 10 Outcomes in patients with Higher Baseline Disease Activity 2 • SRI • Flares • Steroid sparing • Fatigue SELENA-SLEDAI = Safety of Estrogens in Lupus Erythematosus National Assessment– Systemic Lupus Erythematosus Disease Activity Index. So. C = standard of care. 1. Benlysta® (belimumab) Sm. PC. Glaxo. Smith. Kline (UK). March 2012; 2. van Vollenhoven RF, et al. Ann Rheum Dis 2012; [Epub ahead of print].

Approval of belimumab (Benlysta®) • Benlysta® was approved for use by US FDA in March 2011 • Benlysta® is indicated: – For use as an add-on therapy for reducing disease activity in adult patients with active, autoantibody-positive SLE with a high degree of disease activity (e. g. ANA titre ≥ 1: 80 and/or anti-ds. DNA titre ≥ 30 IU/m. L) despite standard therapy) – Approved dosage: 10 mg/kg given intravenously at 2 -week interval for the first 3 doses, then by 4 -week intervals • Benlysta® is contraindicated: – In patients who have demonstrated anaphylaxis to belimumab or to any of the excipients

Thank you �
- Slides: 44