Immunology Biol 465 Minot State University Spring 2020

Immunology Biol 465 Minot State University Spring 2020 MWF 9 -9: 50 am Lab Tuesdays 9 am-noon

Lecture 12 February 14 th , 2020 No class Monday---Presidents’ Day No Lab Tuesday---Assessment Day Tuesday, February 18 th. Northwest Art Center. 4 PM Please consider attending a presentation and Q and A by area medical students and physician. Snacks… See Flyer.

Where we? B-cell receptors/Antibodies/Ig molecules Sketch and antibody molecule, highlighting variable and constant regions Sketch an antibody molecule cut into Fab and Fc fragments. Sketch an antibody molecule, highlighting heavy and light chains How are genes decoded to make proteins? How does DNA sequence affect protein shape and thus function?
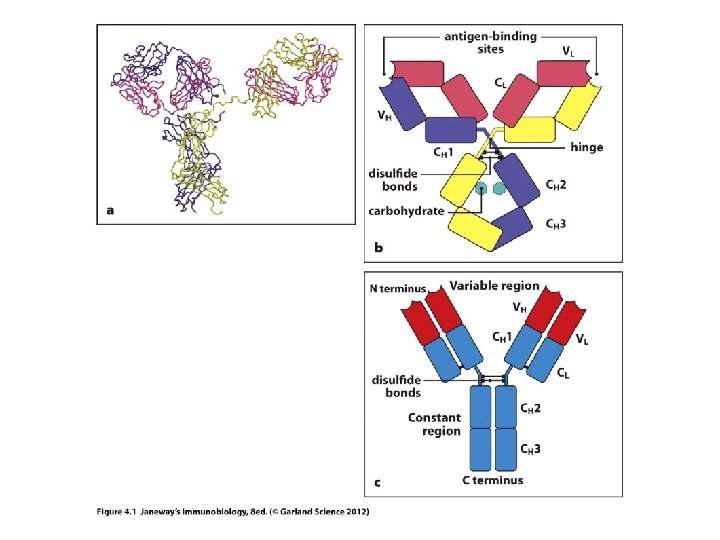
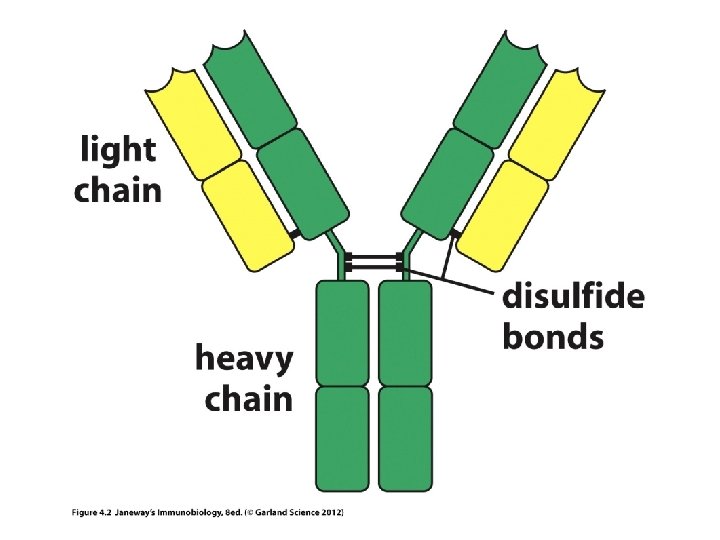

• Each chain in an Ig molecule is encoded by a different gene. – Humans have a singe H-chain gene – Humans have 2 different L-chain genes (lambda l and kappa k)

• Recall, each heavy and light chain has a variable region and a constant region. • How is each put together? (They are one continuous protein, encoded by a single gene), but the gene itself is put together in a very unusual way—no other genes are assembled like this.

Let’s review protein structure first… • N-terminus • C-terminus Levels of structure Primary---amino acid sequence Secondary—basic folded shapes (a helix or b sheet) Tertiary---interactions of different regions of secondary structure Quaternary---shape of whole protein when more than one protein is in a complex. e. g. the overall shape of an antibody
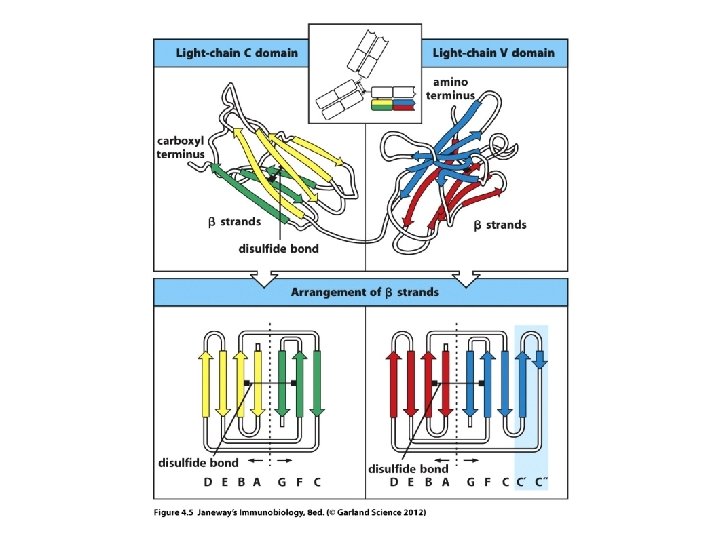

• The models show the predominant secondary structure is b-sheet. Antibodies are made of proteins that fold in a predictable way---a number of proteins fold this way and are included in the Ig superfamily (TCR proteins fold like this too. ) They fold into Ig-like domains.

• Observe carefully…each chain heavy and light, contributes to both the V-region and to the Cregion. • If you think of the primary structure of each protein, the N-terminal 110 amino acids contribute to the V-region.

Within the V-region are 3 regions of extreme variability---called hypervariable (HV) regions.

3 views of the hypervariable regions within the V-region
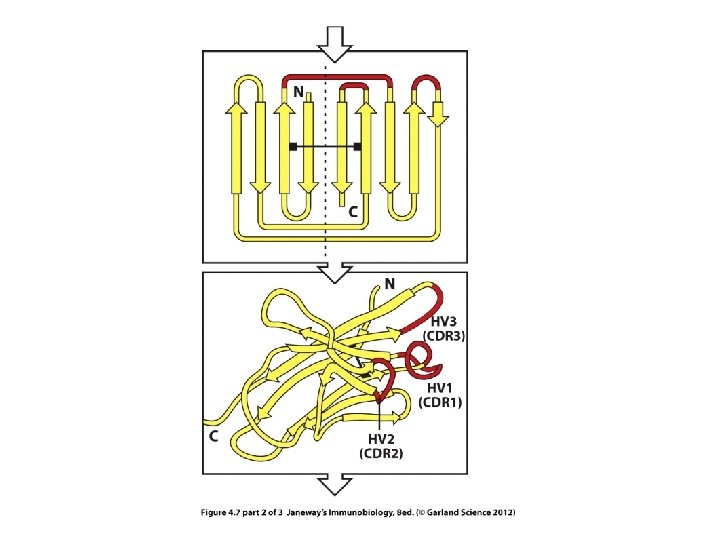
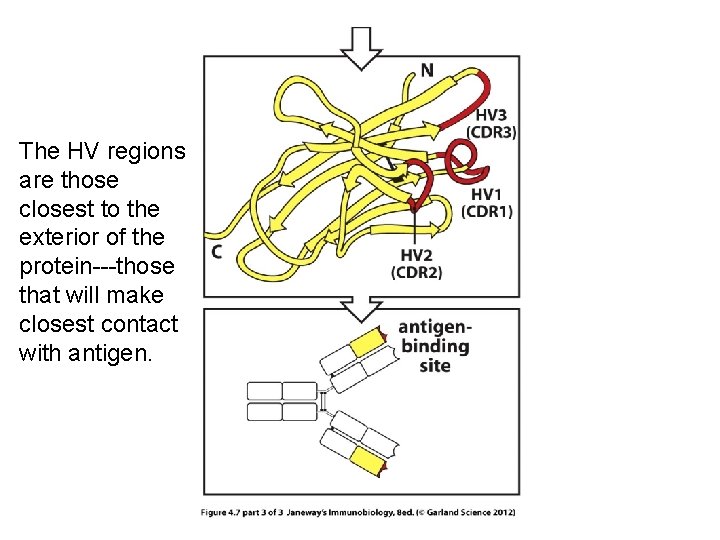
The HV regions are those closest to the exterior of the protein---those that will make closest contact with antigen.
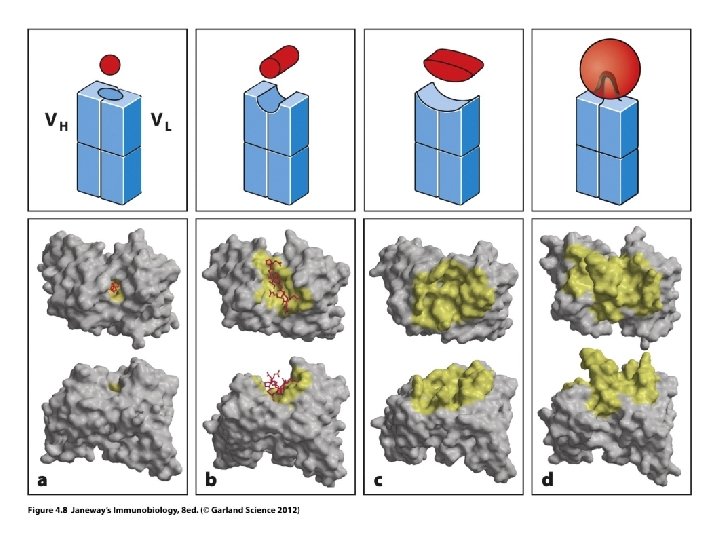

What holds antigen to V-region?
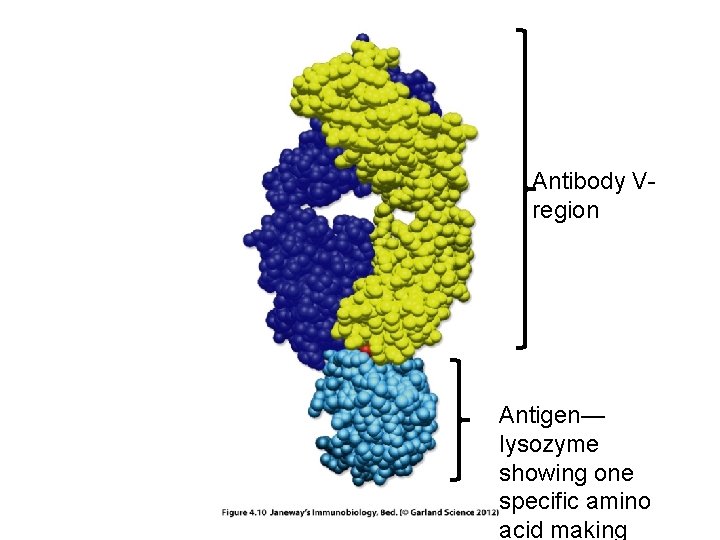
Antibody Vregion Antigen— lysozyme showing one specific amino acid making

• Recall each antigen has a specific site of recognition: • Called the epitope

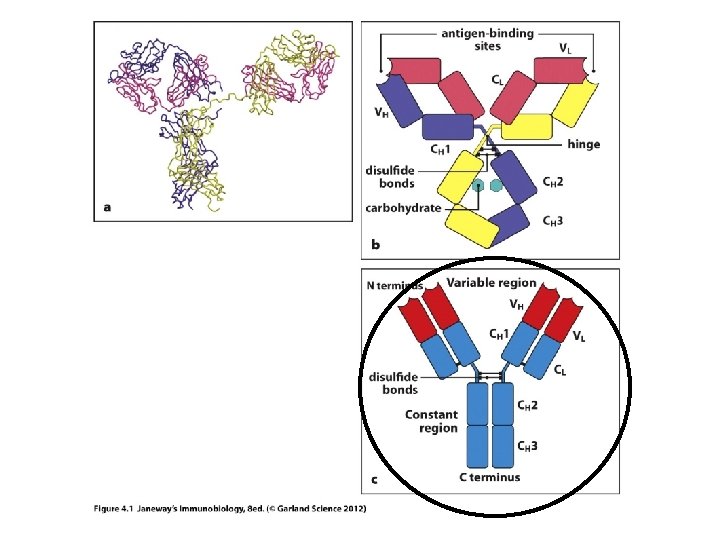
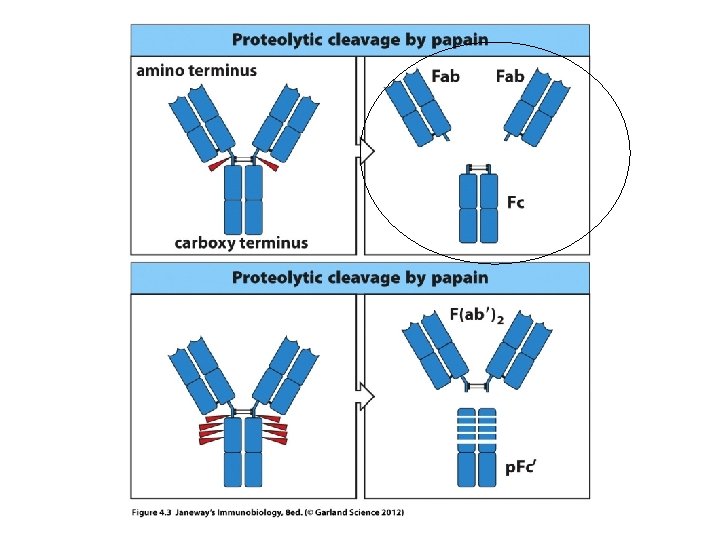
• Now we can understand the Fab and Fc fragments a bit better. – In research or medicine, the Fab fragments can be used alone to mark or detect antigens. – The smaller the fragment, the easier it is work with, dissolve etc, and to tag with something the human eye or technology can detect.
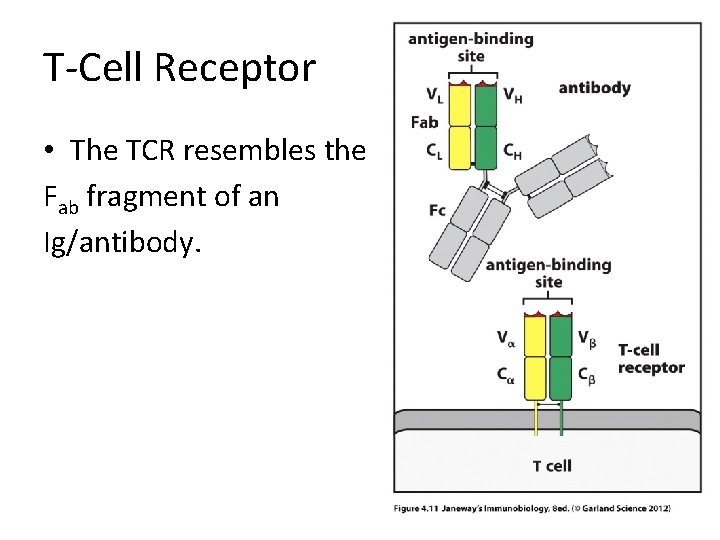
T-Cell Receptor • The TCR resembles the Fab fragment of an Ig/antibody.

• TCRs are composed of 2 protein subunits, which are never secreted. – In most T-cells, the subunits are designated TCRa, TCRb and come together to make TCR a: b heterodimer. – A small subset of T-cells have TCR made with 2 other protein subunits designated TCRg and TCRd; these make a TCR g: d heterodimer. Unless specified, we will be discussing the a: b TCR.
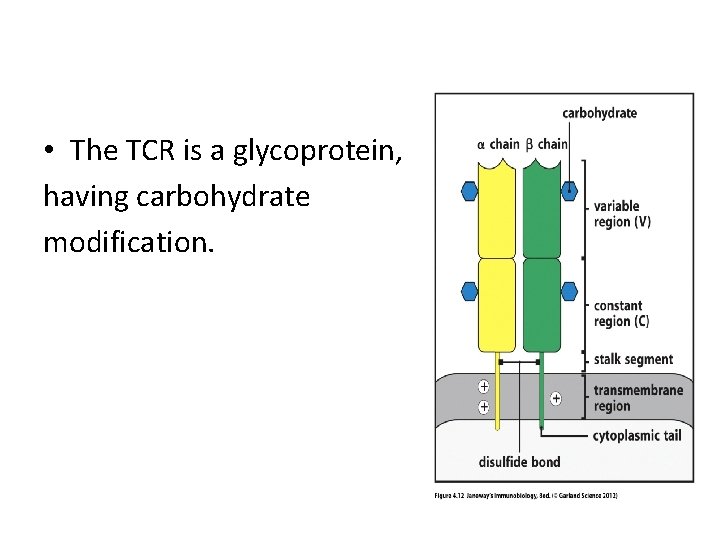
• The TCR is a glycoprotein, having carbohydrate modification.

• As you may have expected, the variability in each TCR lies in the antigen binding site at the end of the receptor. Several regions of the protein contribute to making the structure variable.

• Recall…that variable region of the TCR will bind to antigen on another host cell (not free antigen). • The antigen will be displayed in a molecule of MHC. There are 2 types/classes • Let’s look at MHC structure too.
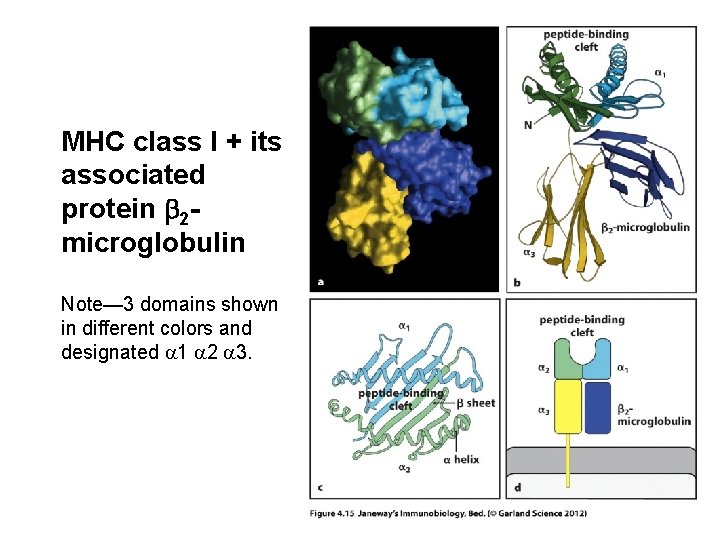
MHC class I + its associated protein b 2 microglobulin Note— 3 domains shown in different colors and designated a 1 a 2 a 3.
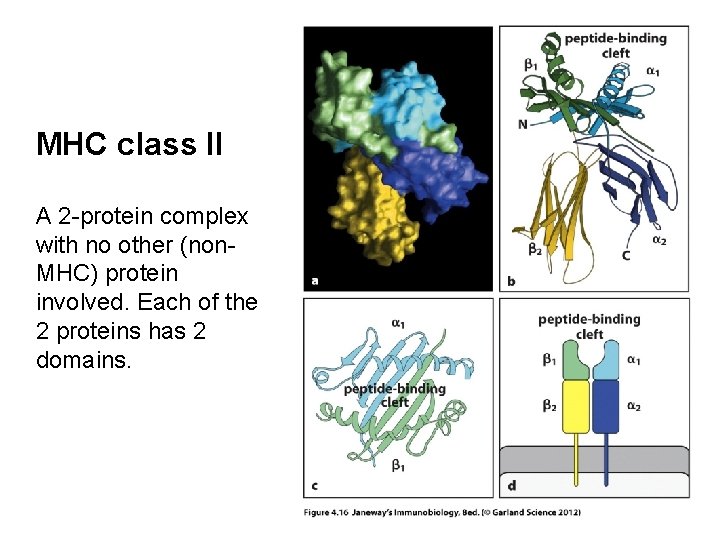
MHC class II A 2 -protein complex with no other (non. MHC) protein involved. Each of the 2 proteins has 2 domains.

• With that introduction to BCR and TCR, keep in mind we will focus first on the variable region of the antigen receptors. . • Again—the BCR and TCR genes are unique in how they generate variability…

Begin Chapter 5 Generation of lymphocyte receptors • Consider B-cells and BCR. How on earth can one heavy chain gene and 2 light chain genes encode billions of BCRs with unique antigenbinding ability? – After all…It is a firmly established fact---one gene encodes one protein.

• At this moment, you have millions of B-cells circulating and with the remaining B-cells that will develop, the ability to make an estimated 108 different BCRs. • It’s is predicted that these B-cells could recognize virtually any foreign antigen we might encounter.

• The large # of BCRs with a unique antigen binding conformations ensures that most antigens will have a shape that is complementary to the antigen binding site.

• Could this be pre-determined? Some amazing evolution of BCR to match all current pathogens? NO! • Can BCR bend/fold/change shape after they are made? NO! (Again, we don’t have millions of Ig genes— 1 Hchain gene, 2 -L-chain genes)

Mechanism for BCR variability • Each of the protein subunits making up each antibody is encoded by a gene with an unusual organization. • The Ig genes are made of individual gene segments.

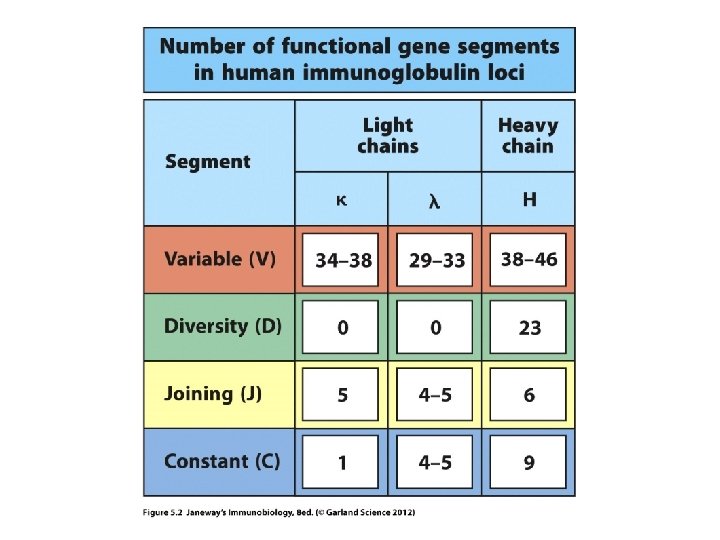
Heavy chain gene—variable region • Made of 3 different types of gene segments. – V-gene segments (don’t confuse with the whole V -region) – J-gene segments – D-gene segments

The light chain genes—variable region • Made of 2 different types of gene segments. – V-gene segments (don’t confuse with the whole V-region) – J-gene segments
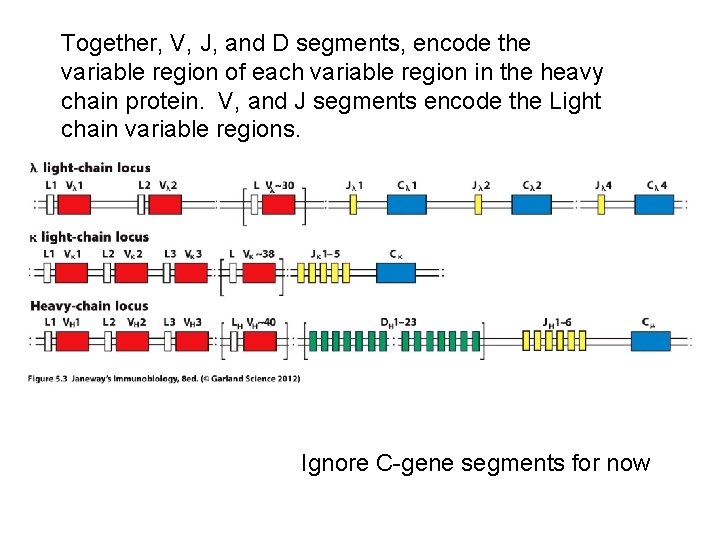
Together, V, J, and D segments, encode the variable region of each variable region in the heavy chain protein. V, and J segments encode the Light chain variable regions. Ignore C-gene segments for now
- Slides: 39