immunology an organism that causes disease Bacteria virus

immunology

an organism that causes disease: Bacteria, virus, protoctist, fungi in tissues recognisable symptoms specific to pathogen

natural barriers to infection in humans • Skin: a tough physical barrier • Lysozyme: in tears, saliva & sweat, Antibacterial hydrolyses bacterial cell walls • HCl in stomach: kills most pathogens – denatures enzymes • Epithelial lining in respiratory tract covered in mucus, traps pathogens & prevents them reaching the cells beneath. Cilia sweep mucus & pathogen to trachea.


C EM H L PLASMA A C I

Infection causes an inflammatory response • pathogen enters the tissues and releases chemicals • blood flow to the area increases • & capillaries become more leaky and plasma moves in to the surrounding tissue • phagocytes are attracted to the site & leave the blood by squeezing through the capillary walls: polymorphs arrive first in greatest numbers, followed by macrophages (made from monocytes) which are larger and longer lived.

• phagocyte membrane invaginates and encloses around the pathogen • which is engulfed and forms a vesicle, called a phagosome. • lysosomes move to the phagosome and their membranes fuse • releasing hydrolytic enzymes which digest the pathogen • the soluble products are absorbed into the cytoplasm. • phagocytes, dead pathogens and cell debris form pus which results in swelling • temperature increases at the site of infection helping to denature pathogenic enzymes

Diagram page 20 A 2 text book

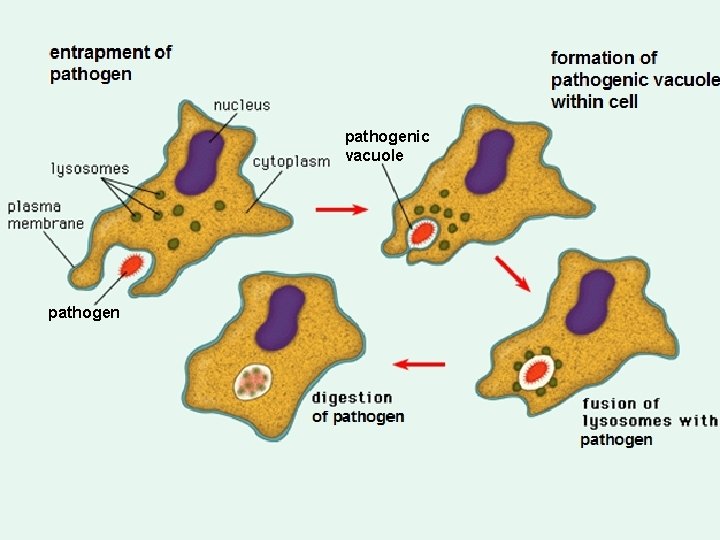
pathogenic vacuole pathogen

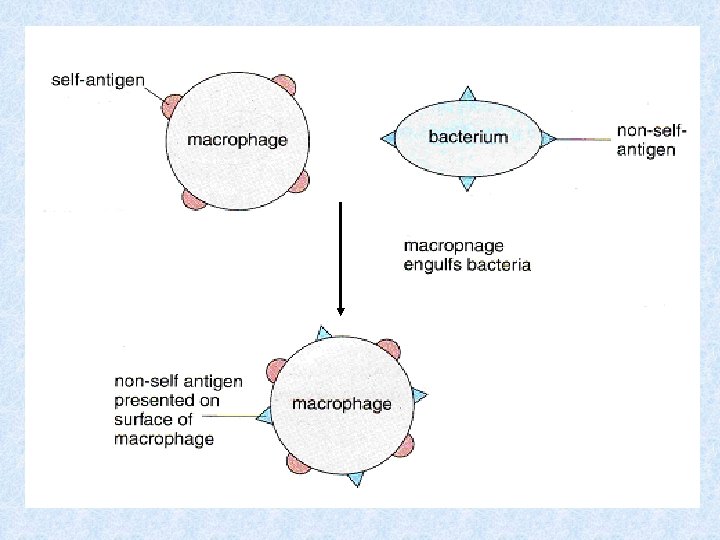
This relies on the ability of lymphocytes to recognise self and non-self tissue. All cells have polysaccharides, glycoproteins and glycolipids on their surface membrane that form antigens. These antigens can trigger an immune response. Antigens present on the membrane our own cells are called self antigens. Antigens present on the membrane of cells from other organisms, including other people, animals, bacteria, fungi, plants and virus’ are called non-self antigens. These non-self antigens may trigger an immune response.

Different pathogens have different antigens; therefore, the immune response needs to be specific to each. Lymphocytes have receptors on their cell surface membranes. These are complementary in shape to antigens allowing antigens and receptors to fit together. The lymphocytes with receptors to self antigens are switched off in the developing foetus, leaving only those that are not complementary to self cells. There are many millions of different lymphocytes, but only a few of each. Consequently this form of response is slow.

B Lymphocyte T Lymphocyte Stem cells in the bone marrow Bone marrow Stem cells in the bone marrow Thymus gland Type of response Antibody mediated Cell mediated How they respond Produces antibodies in response to non-self antigens, usually on bacteria and virus’ in body fluids such as blood and tissue fluid Produces a range of cells in response to non-self antigens, usually from viral infections, attached to body cells Where formed Mature

The non-self antigen attaches to the complementary receptor on a specific B or T lymphocyte. The lymphocyte is sensitised and becomes activated. It divides by mitosis to produce clones. B lymphocytes produce B clones that will form antibodies. T lymphocytes produce T clones, with different cells carrying out different functions. This is often called clonal expansion.

non-self antigen receptor Macrophage or antigen T or B lymphocyte Division + differentiation
This response is triggered by the body’s own cells that have been changed due to the presence of non-self material within them. Non-self antigens are presented on the cell surface membrane, marking them as different to the other body cells. Examples include: Macrophages – after engulfing and breaking down a pathogen they cut out and present the pathogen non-self antigens on their own surface membrane. Body cells that have been invaded by a virus present non-self viral antigens on the body cell membrane. Cancer (tumour) cells present abnormal antigens on their cellsurface membrane.

• Clone cells differentiate (sub-divide) into: • Killer T-cells (cytotoxic T-cells) – destroy virus infected cells – attach to the antigens on the cell-surface membrane & produce chemicals – e. g perforin (punches holes in cell-surface membrane) & nitric acid – Destroying the cell

• Helper T-cells – stimulate B to divide to increase antibody production – promote and speed up the process of phagocytosis – Attach opsonins to pathogens, marking them for attention of phagocytes – Secrete interferon that limits the ability of viruses to replicate • Memory T-cells – Circulate in body fluids & respond to future infection by the same pathogen. – As they are already sensitised they rapidly produce a large clone of T lymphocytes.
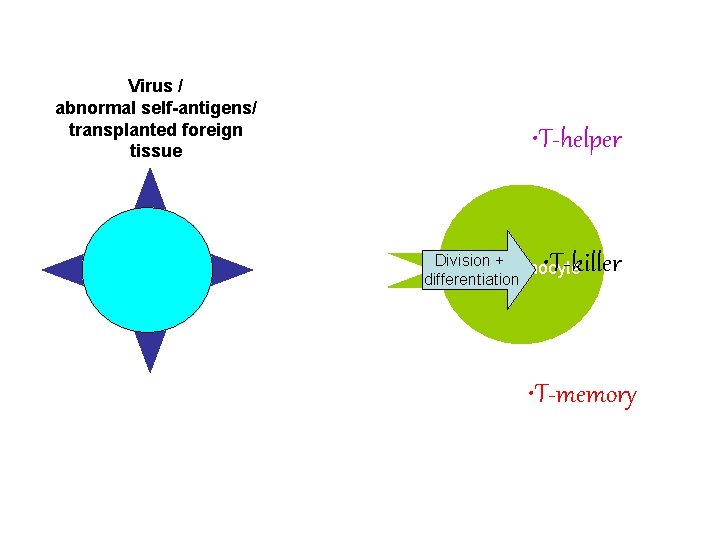
Virus / abnormal self-antigens/ transplanted foreign tissue • T-helper • T-killer Division + T-lymphocyte differentiation • T-memory

are globular proteins which are complementary to specific antigens and which can react with the antigens leading to their destruction

• B lymphocytes that have complementary receptors to a pathogen’s antigens are sensitised. • The specific B lymphocyte divides by mitosis and clones • to produce large numbers of plasma cells and a small quantity of memory cells. • Plasma cells are short lived (a few days) but each produce millions of antibodies. • Antibodies neutralise pathogens by carrying out antigen-antibody reactions.
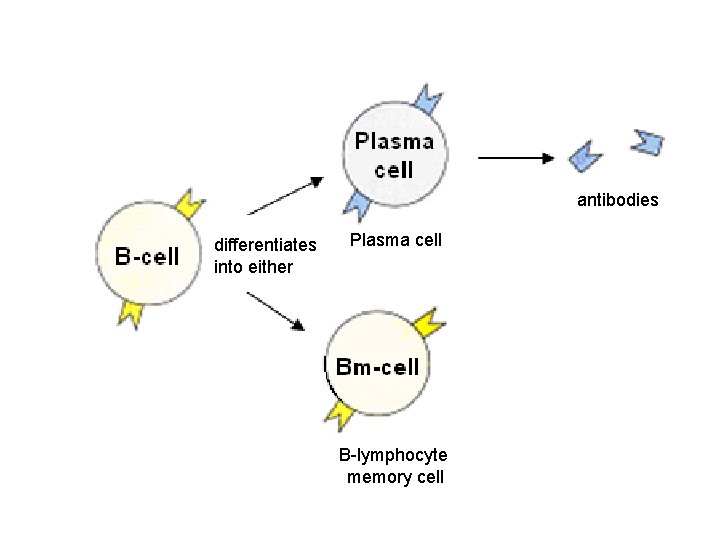
antibodies differentiates into either Plasma cell B-lymphocyte memory cell

The antibodies produced by a specific antibody mediated response will be complementary in shape to the antigens on the invading bacterium. The antibodies attach to the antigens and cause the antigens to clump together (agglutinate), forming an antigen-antibody complex. This immobilises the pathogen and eventually the complex will be engulfed by polymorphs and other phagocytes.
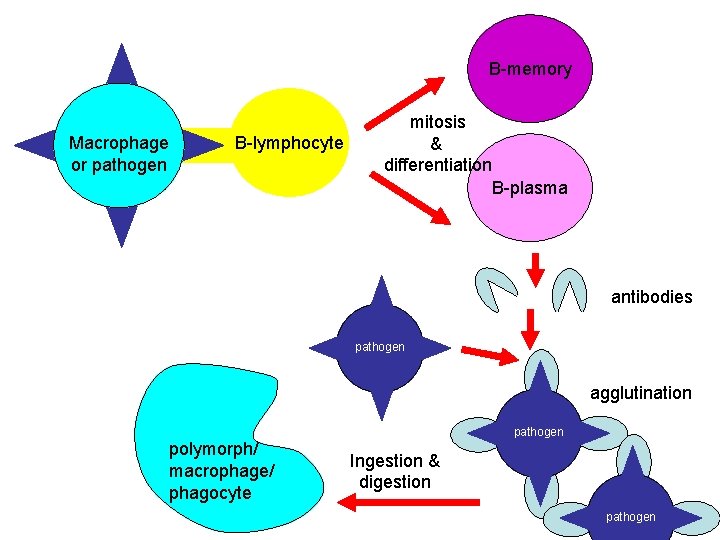
B-memory Macrophage or pathogen B-lymphocyte mitosis & differentiation B-plasma antibodies pathogen agglutination pathogen polymorph/ macrophage/ phagocyte Ingestion & digestion pathogen

The antibodies can also use other methods to defend against infection:

B memory cells can live for many years in the body fluids. They remain inactive until stimulated by the same antigen again. They are able to divide rapidly, as they are already sensitised and produce vast numbers of plasma cells. This is possible because there are more memory cells than there were correct B cells initially.

The plasma cells produce the antibodies to destroy the pathogen, the memory cells guarantee a long term protection. The initial response to the antigen when meeting it for the first time is called the primary immune response. The production of plasma cells and antibodies from memory cells on subsequent encounters with the antigen is called the secondary immune response.

Primary response • Production of antibodies by B-plasma cells in response to pathogen entering the body. • Delay in their production allows the pathogen to reproduce and damage the body, producing the characteristic symptoms associated with the pathogen • As antibodies destroy the antigens, fewer B-cells are made and their level falls.

secondary response • When a person encounters the pathogen at a later date B-memory cells rapidly divide to produce plasma cells. • The response is rapid as the B memory cells are already activated. • Larger numbers of plasma cells are produced, so larger numbers of antibodies. • There is not enough time for symptoms of the disease to develop
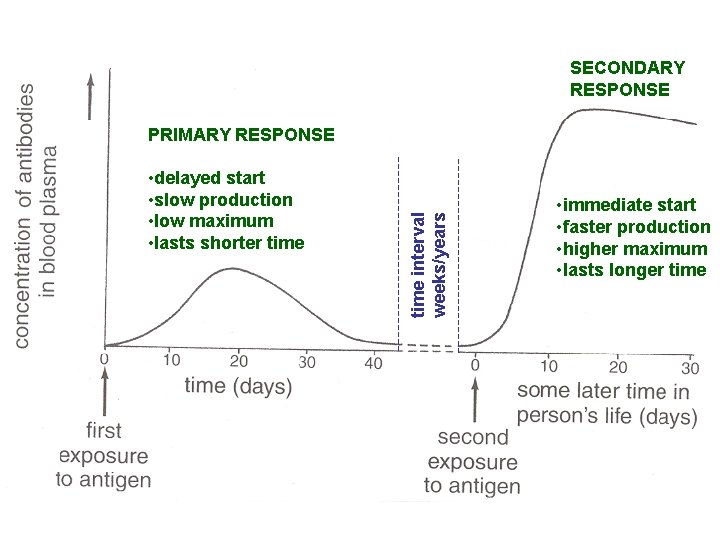
SECONDARY RESPONSE • delayed start • slow production • low maximum • lasts shorter time interval weeks/years PRIMARY RESPONSE • immediate start • faster production • higher maximum • lasts longer time
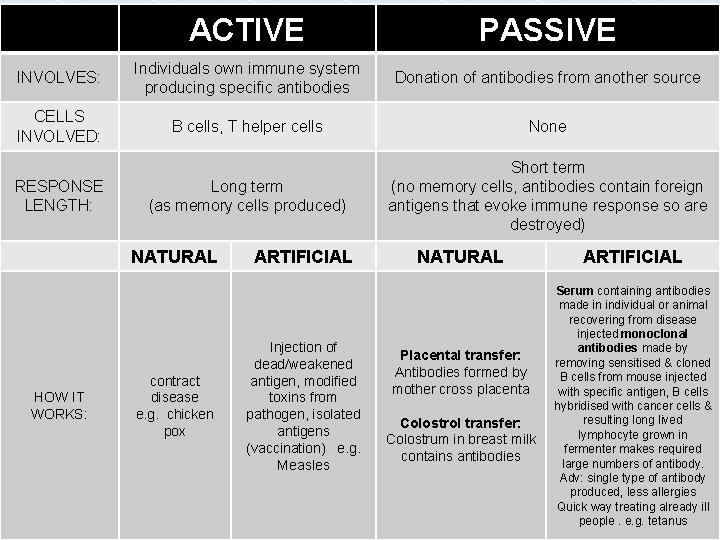
ACTIVE PASSIVE INVOLVES: Individuals own immune system producing specific antibodies Donation of antibodies from another source CELLS INVOLVED: B cells, T helper cells None Long term (as memory cells produced) Short term (no memory cells, antibodies contain foreign antigens that evoke immune response so are destroyed) RESPONSE LENGTH: NATURAL HOW IT WORKS: contract disease e. g. chicken pox ARTIFICIAL Injection of dead/weakened antigen, modified toxins from pathogen, isolated antigens (vaccination) e. g. Measles NATURAL Placental transfer: Antibodies formed by mother cross placenta Colostrol transfer: Colostrum in breast milk contains antibodies ARTIFICIAL Serum containing antibodies made in individual or animal recovering from disease injected monoclonal antibodies made by removing sensitised & cloned B cells from mouse injected with specific antigen, B cells hybridised with cancer cells & resulting long lived lymphocyte grown in fermenter makes required large numbers of antibody. Adv: single type of antibody produced, less allergies Quick way treating already ill people. e. g. tetanus
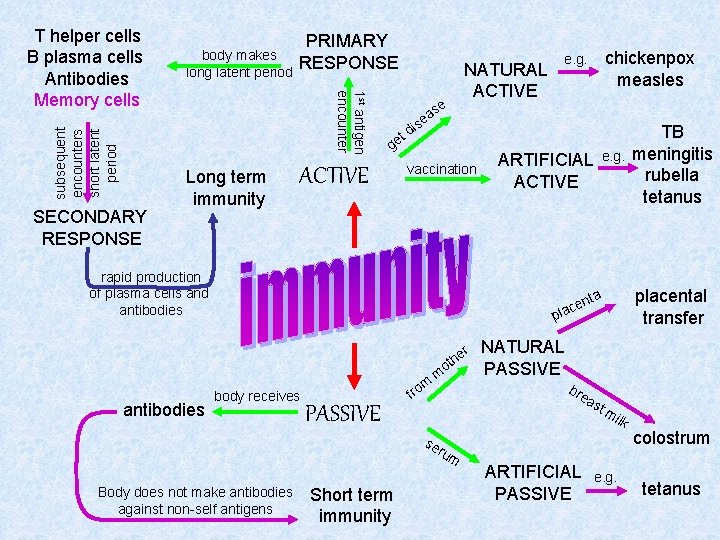
subsequent encounters short latent period SECONDARY RESPONSE body makes long latent period PRIMARY RESPONSE 1 st antigen encounter T helper cells B plasma cells Antibodies Memory cells Long term immunity e t ge ACTIVE NATURAL ACTIVE as e is d vaccination rapid production of plasma cells and antibodies e. g. ARTIFICIAL ACTIVE r body receives PASSIVE m fro m NATURAL PASSIVE bre as tm ilk se rum Body does not make antibodies against non-self antigens Short term immunity TB meningitis rubella tetanus placental transfer ta cen a l p he ot antibodies chickenpox measles e. g. ARTIFICIAL PASSIVE e. g. colostrum tetanus

• • • Tissue transplants The immune response usually occurs as a result of infection. Transplanted tissues and organs contain non-self antigens, which also initiate an immune response (unless from within same person e. g. skin grafts or between identical twins). Transplant rejection is the main reason for failing transplants. T lymphocytes are sensitised by the nonself antigens in the transplanted tissue. These clone producing Killer T-cells that destroy the transplanted cells.

Flowchart Immune response to Transplanted tissue
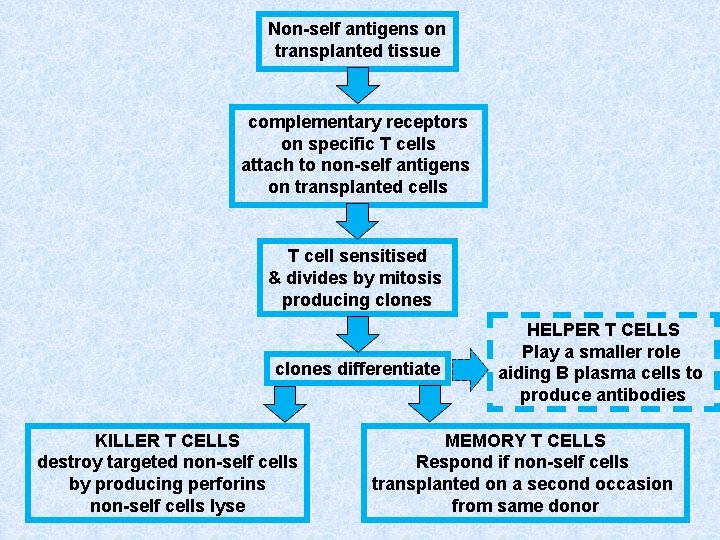
Non-self antigens on transplanted tissue complementary receptors on specific T cells attach to non-self antigens on transplanted cells T cell sensitised & divides by mitosis producing clones differentiate KILLER T CELLS destroy targeted non-self cells by producing perforins non-self cells lyse HELPER T CELLS Play a smaller role aiding B plasma cells to produce antibodies MEMORY T CELLS Respond if non-self cells transplanted on a second occasion from same donor

true or false Inject foreign antigens into patients so that their immune system is too busy to recognise the new organ e s l a f

true or false inject drugs to destroy antibodies produced in the immune response e s l a f

true or false True inject drugs that prevent cell division called immunosuppressants

true or false use radiation to prevent stem cells in the bone marrow from differentiating True X-rays

true or false Inject antibodies to destroy recipients B & T lymphocytes e s l a f

true or false True Tissue typing Check donated tissue for non-self antigens

• Successful transplants require: – Tissue typing where markers on the donor and recipient tissue are checked, and that with the most matched markers will be used. Tissue matching is most likely to occur between close relatives. – X-rays to irradiate bone marrow and lymph tissue to inhibit lymphocyte production and slow down rejection. Unfortunately this increases the chances of infection during treatment and has unpleasant side effects.

– Immunosuppresion, using drugs to inhibit DNA replication, & therefore cell division and cloning of lymphocytes to delay rejection. Need to be taken for the life of the transplant. It compromises the immune system & infections are more likely as the whole immune response is depressed. Other strategies such as monoclonal antibodies, anti-viral drugs & anti-bacterial mouthwashes help fight any infections. There needs to be a delicate balance between reducing the risk of rejection and restricting side-effects from the immunosuppressant.
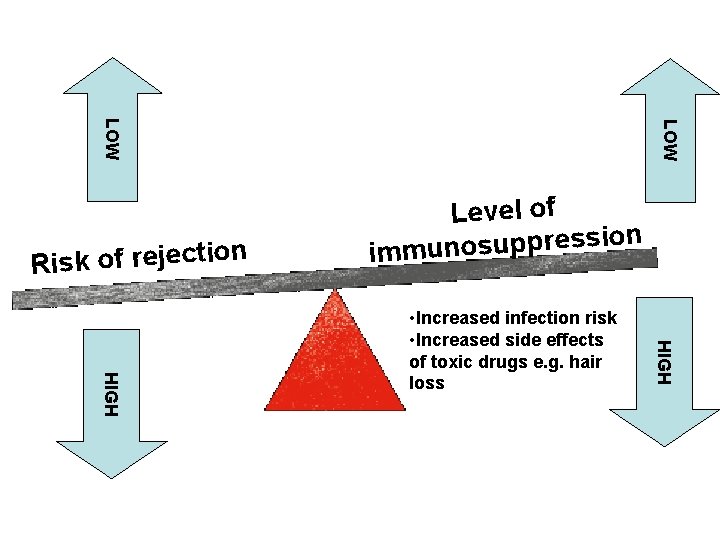
LOW n o i t c e j e r f o k s Ri Level of on i s s e r p p u s o n u imm HIGH • Increased infection risk • Increased side effects of toxic drugs e. g. hair loss

BLOOD TRANSFUSIONS

• Erythrocytes (RBCs) have antigens on their cell-surface membrane. • The blood of any one person will not have the antibodies that correspond to the antigens on their RBCs, as this would trigger an immune response. • This is because the B lymphocytes that would produce the antibodies that correspond to these self-antigens are switched off during foetal development.

• There is a variety of different antigens on the erythrocytes of different people. • In the ABO system there are 2 different antigens, but 4 different possible blood groups (known as polymorphism). • Donated blood must be compatible with the recipient to avoid an immune response. • This happens because the person getting the blood transfusion (recipient) has antibodies that will react with antigens on the red blood cells that they receive. • Recipients red blood cells are not damaged by the donors antibodies because there are not enough to cause a response.
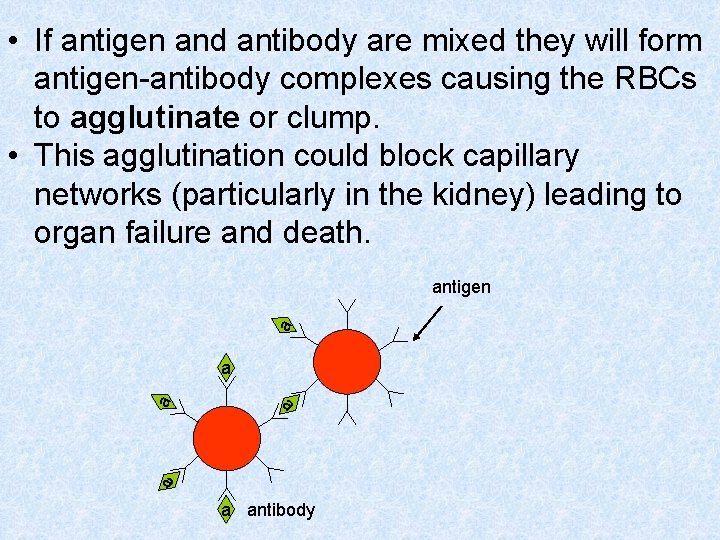
• If antigen and antibody are mixed they will form antigen-antibody complexes causing the RBCs to agglutinate or clump. • This agglutination could block capillary networks (particularly in the kidney) leading to organ failure and death. a antigen a a antibody
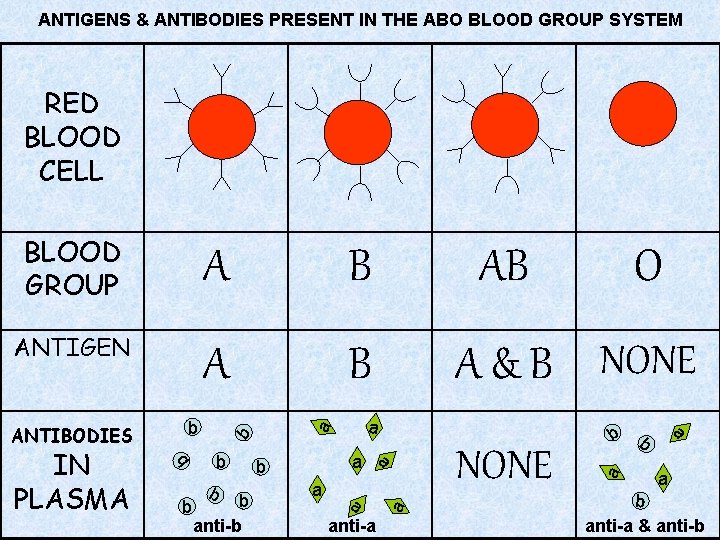
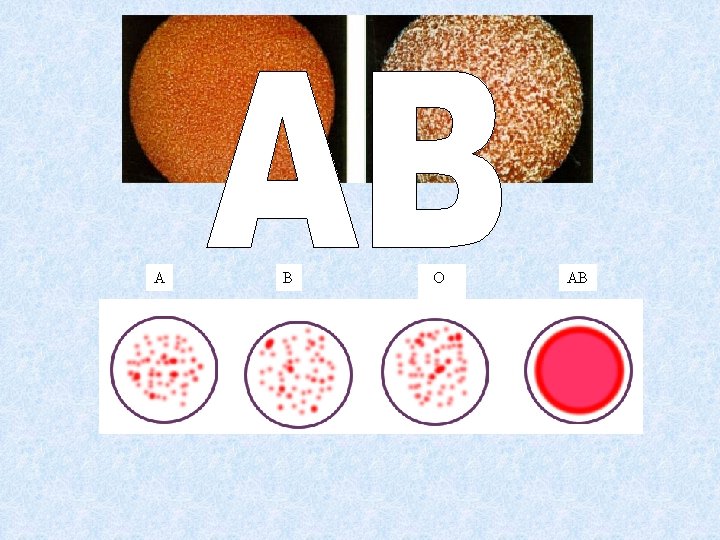
ANTIGENS & ANTIBODIES PRESENT IN THE ABO BLOOD GROUP SYSTEM RED BLOOD CELL A B ANTIGEN A B a b b b anti-b a a anti-a a b b b A & B NONE a IN PLASMA b O NONE b b a ANTIBODIES AB a BLOOD GROUP a b anti-a & anti-b
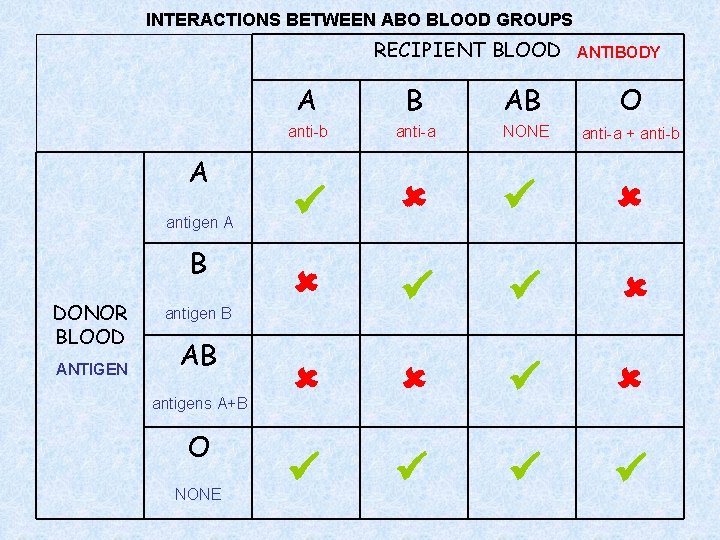
INTERACTIONS BETWEEN ABO BLOOD GROUPS RECIPIENT BLOOD A antigen A B DONOR BLOOD ANTIGEN antigen B AB antigens A+B O NONE ANTIBODY A B AB O anti-b anti-a NONE anti-a + anti-b

examples • A donor with blood group A can give blood to a recipient with blood group A because the recipient has no anti-a antibodies that correspond to the A antigens on the donor’s RBCs. • A donor with blood group A cannot give blood to a recipient with blood group B because the recipient has anti-A antibodies which will react with the A antigens on the donor’s RBCs, causing agglutination.
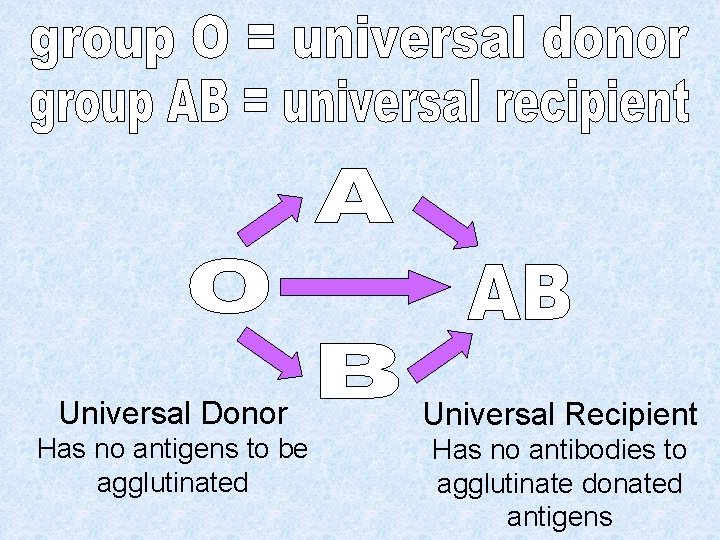
Universal Donor Universal Recipient Has no antigens to be agglutinated Has no antibodies to agglutinate donated antigens
A B O AB

The Rhesus System • The rhesus antigen (or antigen D) is either present or absent on the cell-surface membrane of erythrocytes. • Approx. 85% of individuals possess the antigen and are described as rhesus positive (Rh+) • Rhesus negative individuals (Rh-) do not possess the rhesus antigen.

• Antibodies for the rhesus antigen are not naturally produced by the body, therefore absent form the plasma. • HOWEVER, they may be produced as a result of – Rhesus positive blood being donated to a rhesus negative recipient (rare due to blood matching techniques) – A rhesus negative mother carrying a rhesus positive baby.

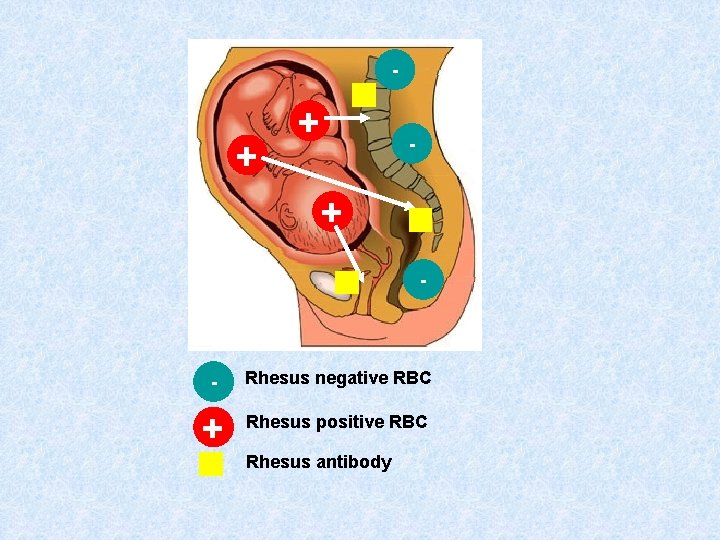
• • 1 st pregnancy Mother rhesus negative Baby rhesus positive Baby’s Rh+ RBC enter mother’s bloodstream Mother makes anti-D antibodies to destroy Rh+ antigen, a slow process, so baby born before significant numbers are produced 2 nd pregnancy • If baby is Rh+ & foetal RBCs enter maternal circulation, relevant B lymphocytes are already sensitised and large numbers of anti-D antibodies are produced immediately. • Anti-D antibodies cross the placenta & enter baby’s bloodstream • causing agglutination of foetal RBCs
- + + - - Rhesus negative RBC + Rhesus positive RBC Rhesus antibody

• • • 2 nd pregnancy This is known as haemolytic disease of the newborn Few cases occur as mothers identified as rhesus negative are given an injection of anti-D antibodies during pregnancy. These attach to any foetal RBCs that may have entered the maternal circulation before the mother’s B lymphocytes are stimulated to produce ant-D antibodies. Following birth the mother is given another injection of anti-D antibodies, if the baby is rhesus positive. If the mother is not identified as rhesus negative the baby can be treated with a blood transfusion.


- Slides: 63