IMMUNOGLOBULINS STRUCTURE AND FUNCTION IMMUNOGLOBULINS Definition Glycoprotein molecules

IMMUNOGLOBULINS STRUCTURE AND FUNCTION
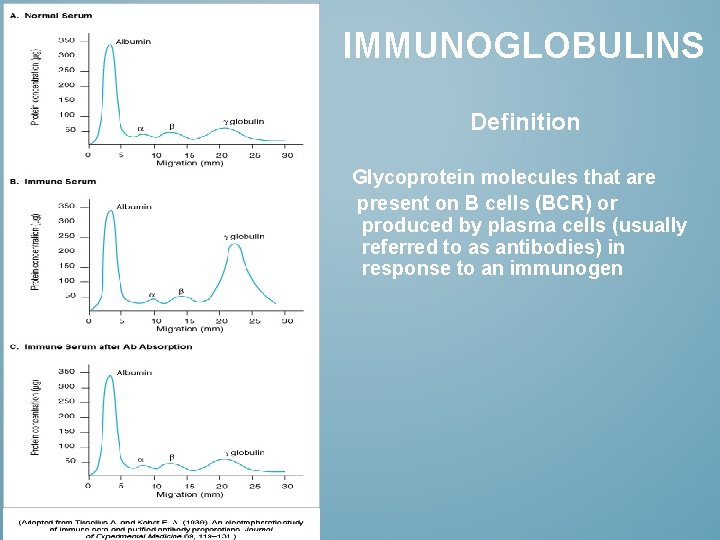
IMMUNOGLOBULINS Definition Glycoprotein molecules that are present on B cells (BCR) or produced by plasma cells (usually referred to as antibodies) in response to an immunogen

Ø Antibodies production is the sole function of the B cells Ø Not toxic or destructive, bind the pathogen tightly and target destructive components of the immune system Ø Antibodies are useful in the defense against extracellular pathogens Ø Antibodies are secreted in the secondary lymphoid organs and in bone marrow and find their way to the extracellular spaces Ø During the course of an infection antibody effectiveness improves steadily
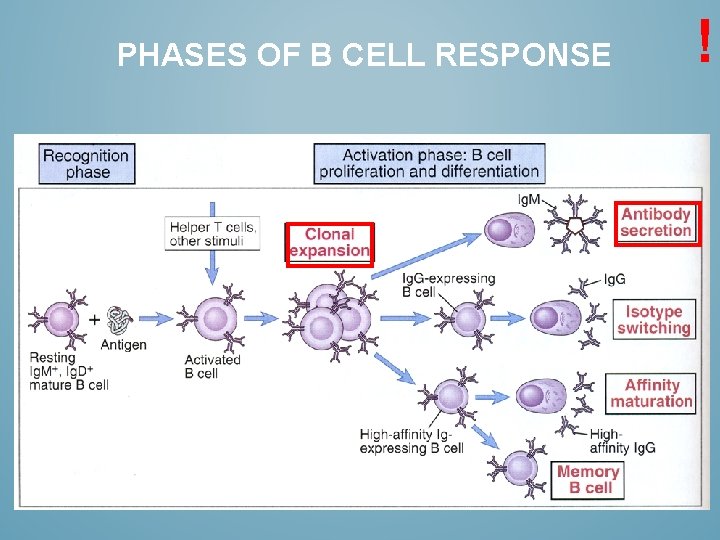
PHASES OF B CELL RESPONSE !
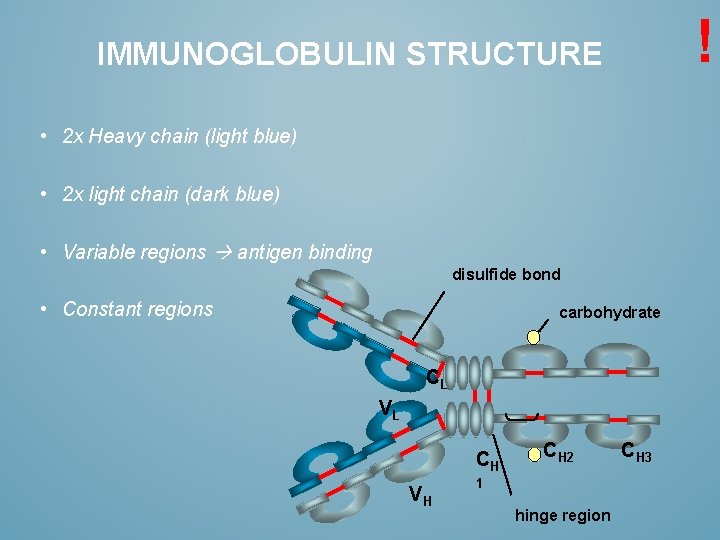
! IMMUNOGLOBULIN STRUCTURE • 2 x Heavy chain (light blue) • 2 x light chain (dark blue) • Variable regions antigen binding disulfide bond • Constant regions carbohydrate CL VL CH VH CH 2 1 hinge region CH 3
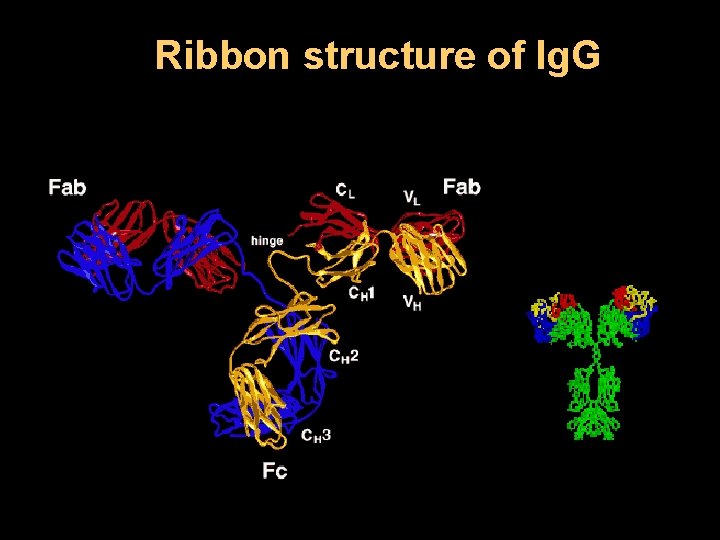
Ribbon structure of Ig. G
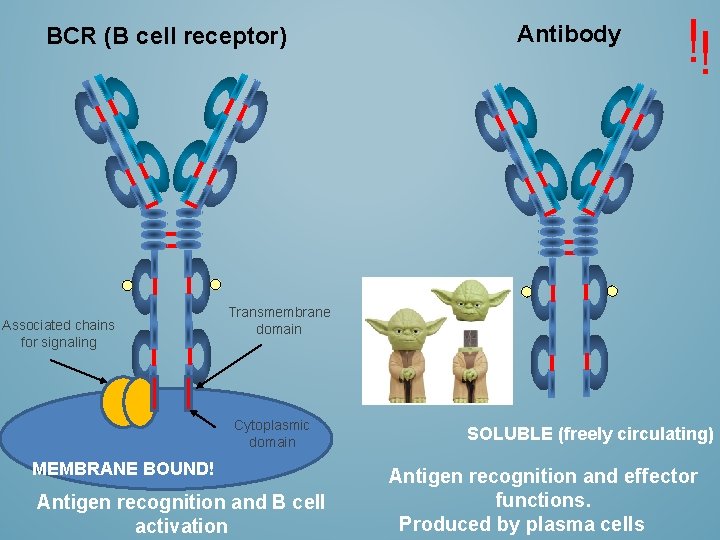
BCR (B cell receptor) Associated chains for signaling Antibody !! Transmembrane domain Cytoplasmic domain MEMBRANE BOUND! Antigen recognition and B cell activation SOLUBLE (freely circulating) Antigen recognition and effector functions. Produced by plasma cells
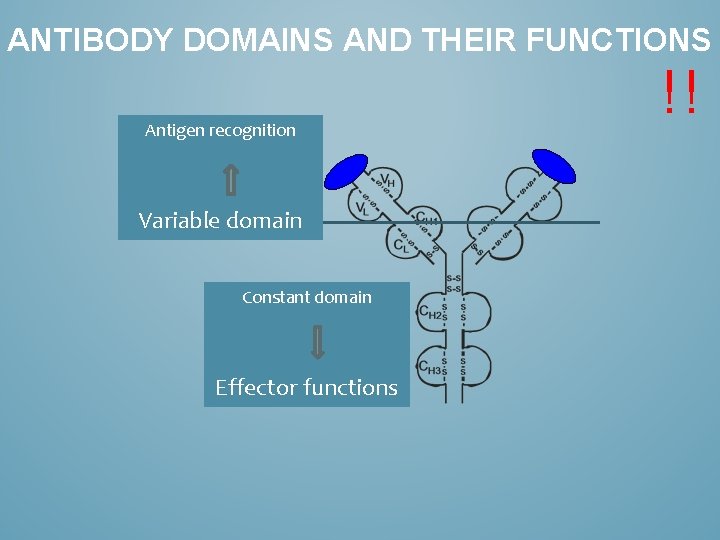
ANTIBODY DOMAINS AND THEIR FUNCTIONS Antigen recognition Variable domain Constant domain Effector functions !!
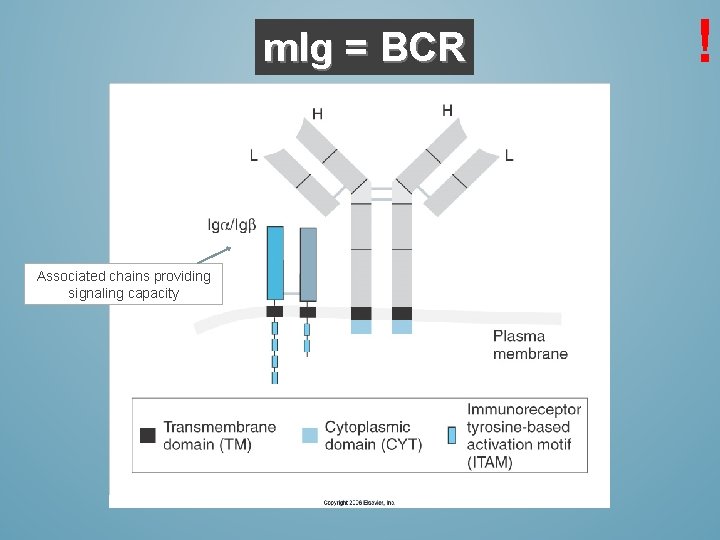
m. Ig = BCR Associated chains providing signaling capacity !
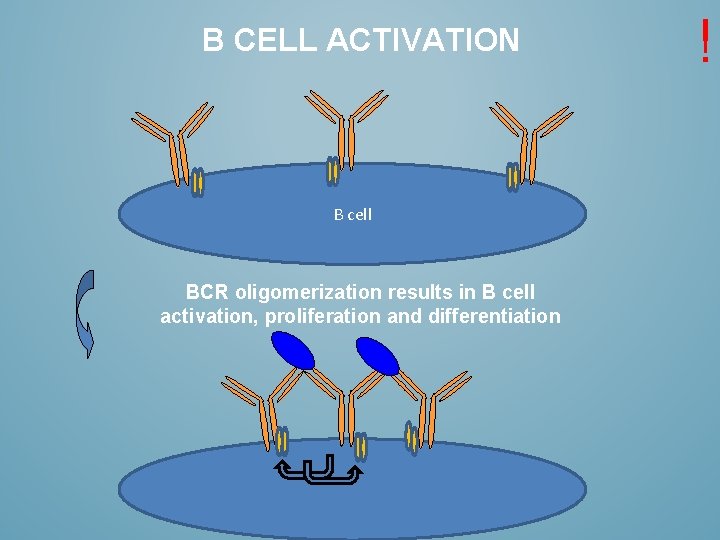
B CELL ACTIVATION B cell BCR oligomerization results in B cell activation, proliferation and differentiation !
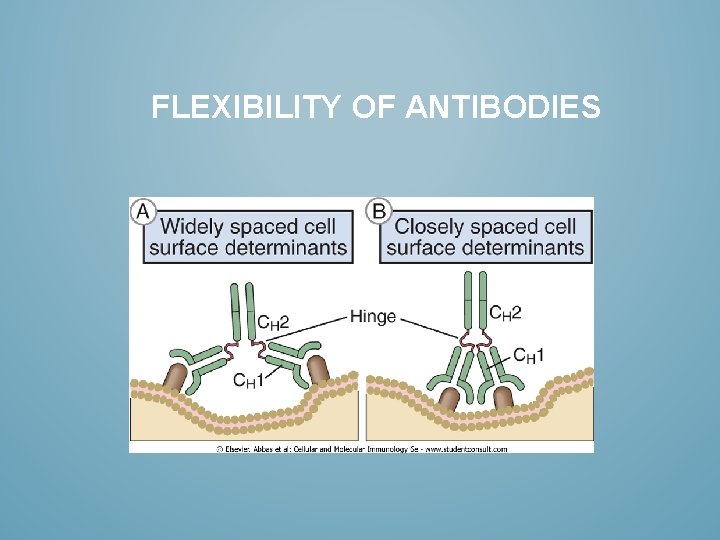
FLEXIBILITY OF ANTIBODIES
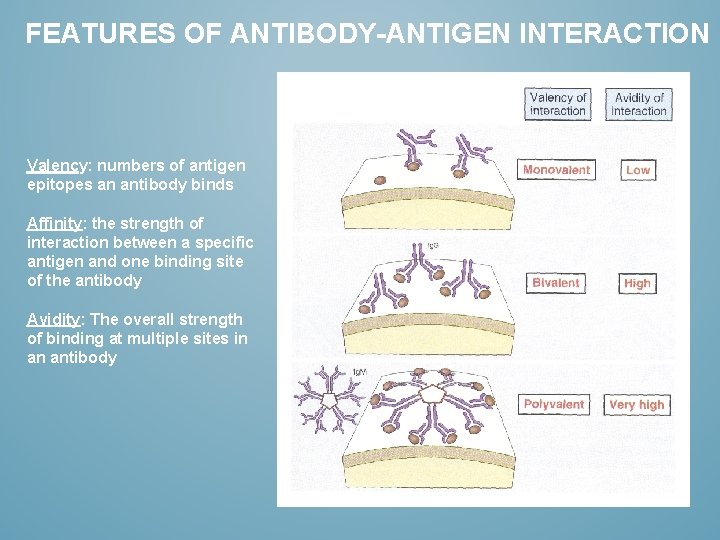
FEATURES OF ANTIBODY-ANTIGEN INTERACTION Valency: numbers of antigen epitopes an antibody binds Affinity: the strength of interaction between a specific antigen and one binding site of the antibody Avidity: The overall strength of binding at multiple sites in an antibody
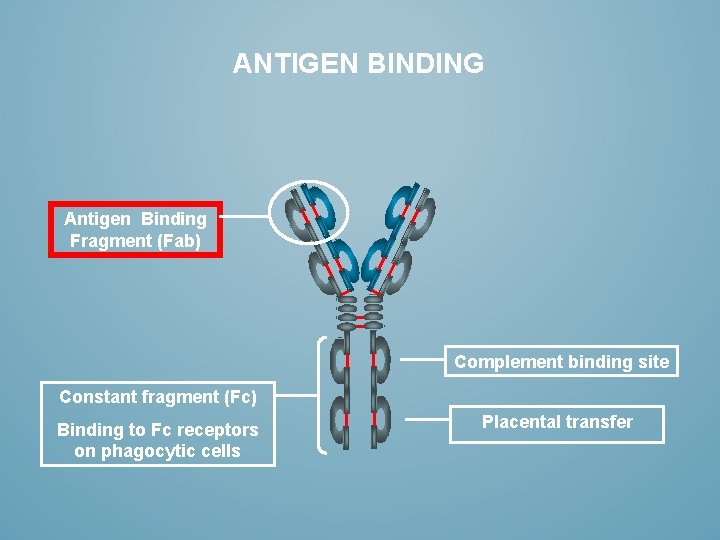
ANTIGEN BINDING Antigen Binding Fragment (Fab) Complement binding site Constant fragment (Fc) Binding to Fc receptors on phagocytic cells Placental transfer
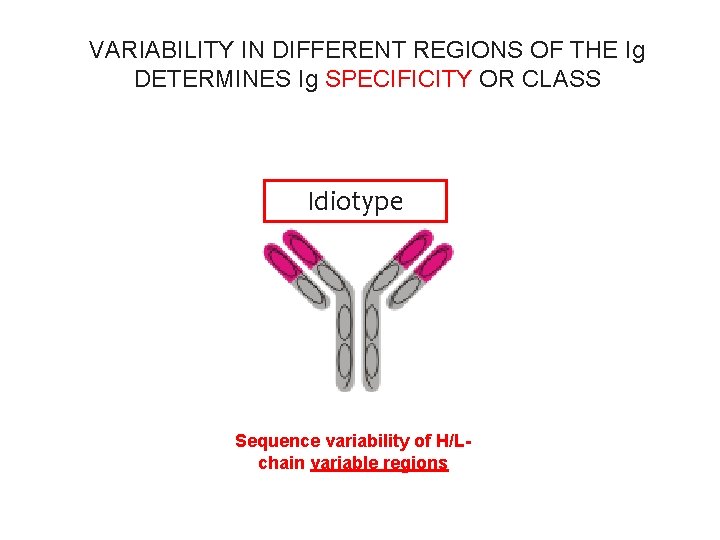
VARIABILITY IN DIFFERENT REGIONS OF THE Ig DETERMINES Ig SPECIFICITY OR CLASS isotype Idiotype Sequence variability of H/Lchain constant regions Sequence variability of H/Lchain variable regions
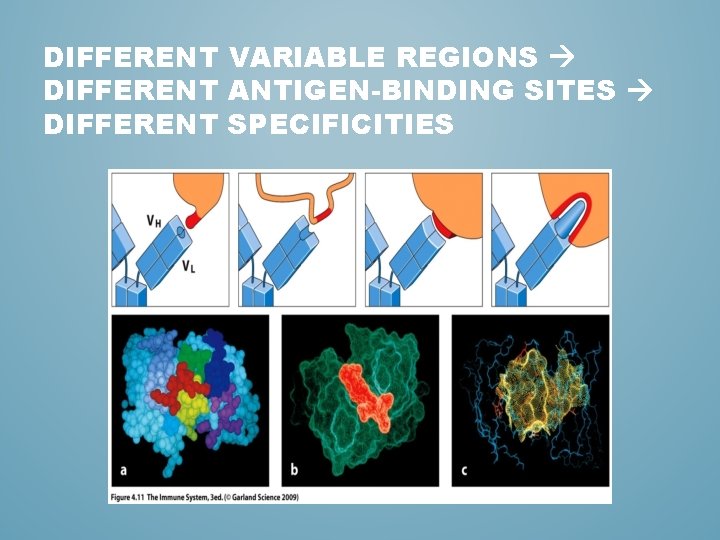
DIFFERENT VARIABLE REGIONS DIFFERENT ANTIGEN-BINDING SITES DIFFERENT SPECIFICITIES
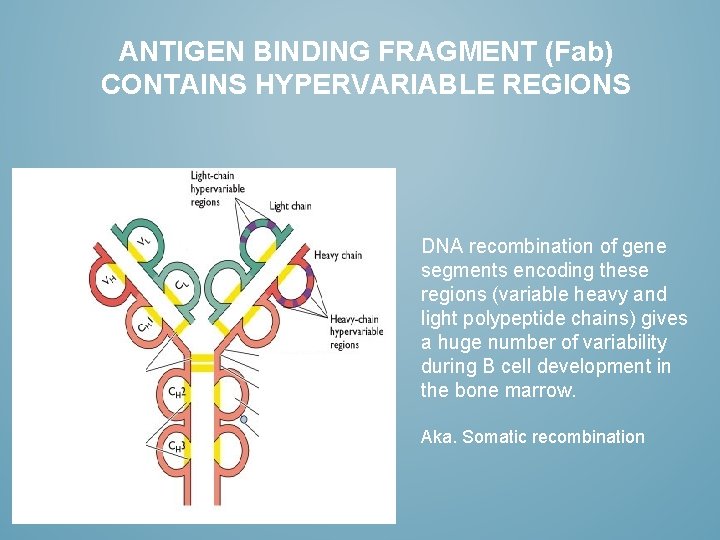
ANTIGEN BINDING FRAGMENT (Fab) CONTAINS HYPERVARIABLE REGIONS DNA recombination of gene segments encoding these regions (variable heavy and light polypeptide chains) gives a huge number of variability during B cell development in the bone marrow. Aka. Somatic recombination
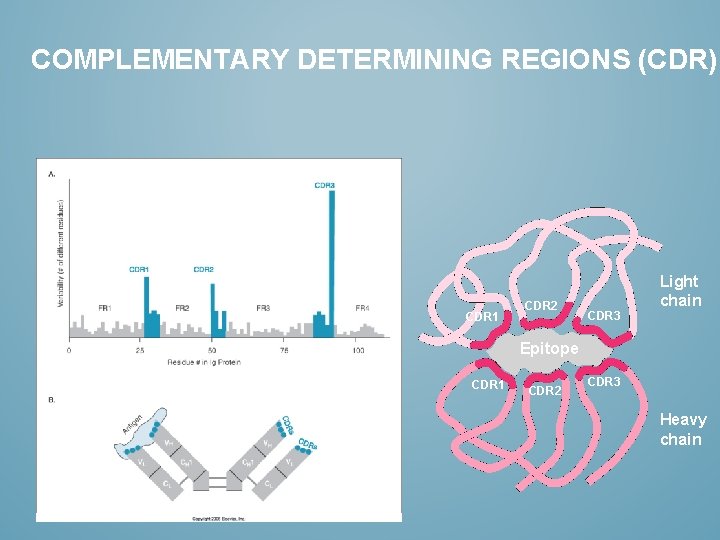
COMPLEMENTARY DETERMINING REGIONS (CDR) CDR 1 CDR 2 CDR 3 Light chain Epitope CDR 1 CDR 2 CDR 3 Heavy chain
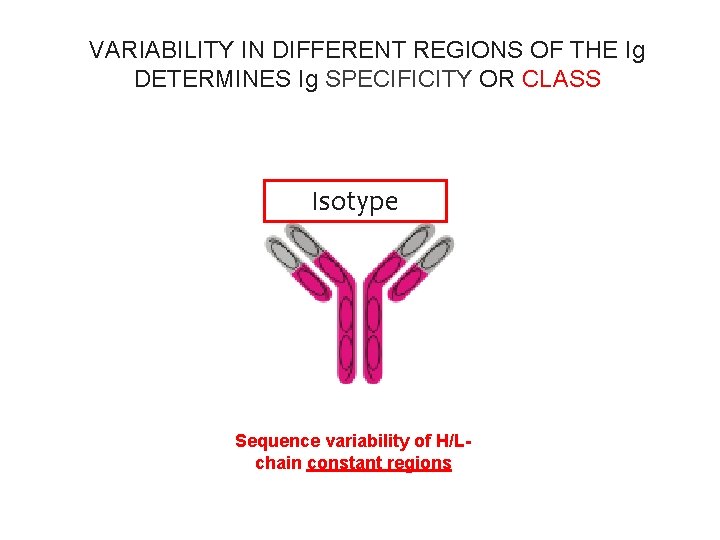
VARIABILITY IN DIFFERENT REGIONS OF THE Ig DETERMINES Ig SPECIFICITY OR CLASS Isotype Sequence variability of H/Lchain constant regions

HUMAN IMMUNOGLOBULIN CLASSES ! ENCODED BY DIFFERENT STRUCTURAL GENE SEGMENTS (ISOTYPES) Heavy chain types: • • • Ig. G - gamma (γ) heavy chains Ig. M - mu (μ) heavy chains Ig. A - alpha (α) heavy chains Ig. D - delta (δ) heavy chains Ig. E - epsilon (ε) heavy chains Light chain types: • kappa (κ) • lambda (λ)
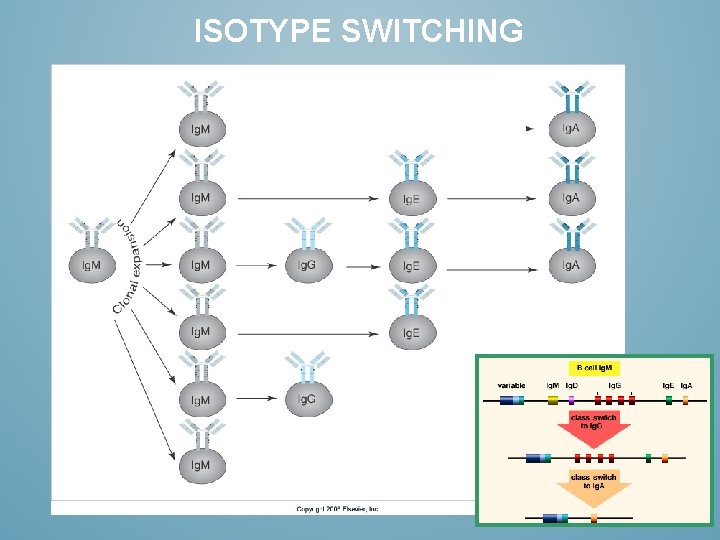
ISOTYPE SWITCHING
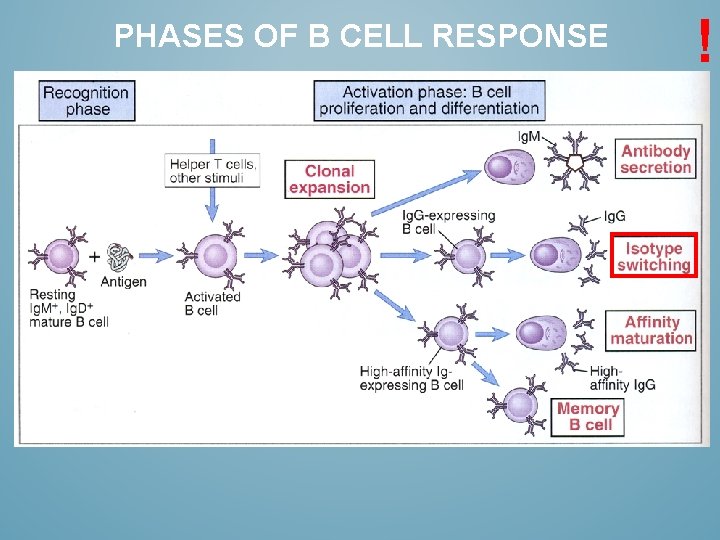
PHASES OF B CELL RESPONSE !
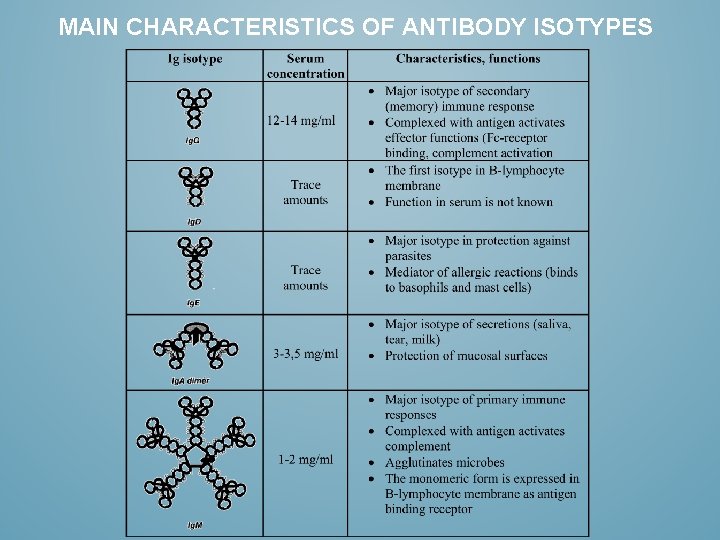
MAIN CHARACTERISTICS OF ANTIBODY ISOTYPES
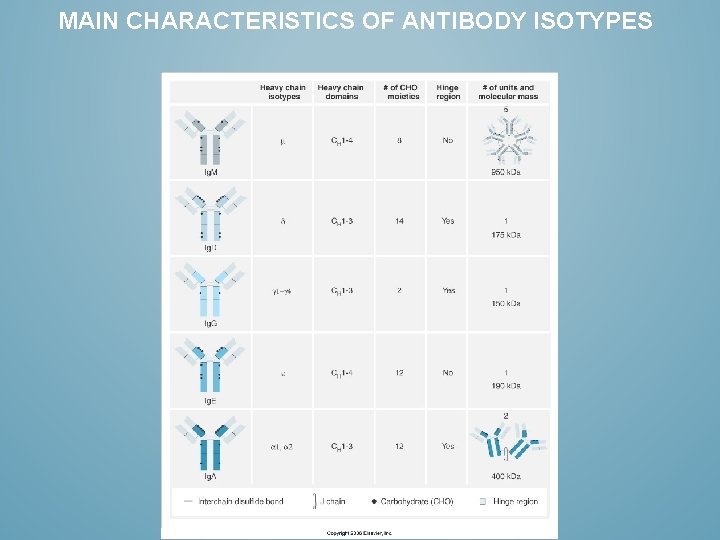
MAIN CHARACTERISTICS OF ANTIBODY ISOTYPES
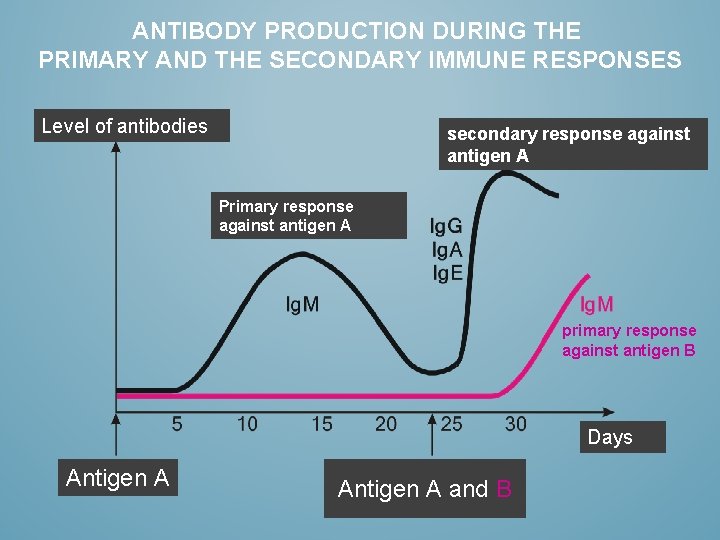
ANTIBODY PRODUCTION DURING THE PRIMARY AND THE SECONDARY IMMUNE RESPONSES Level of antibodies secondary response against antigen A Primary response against antigen A primary response against antigen B Days napok Antigen A and B
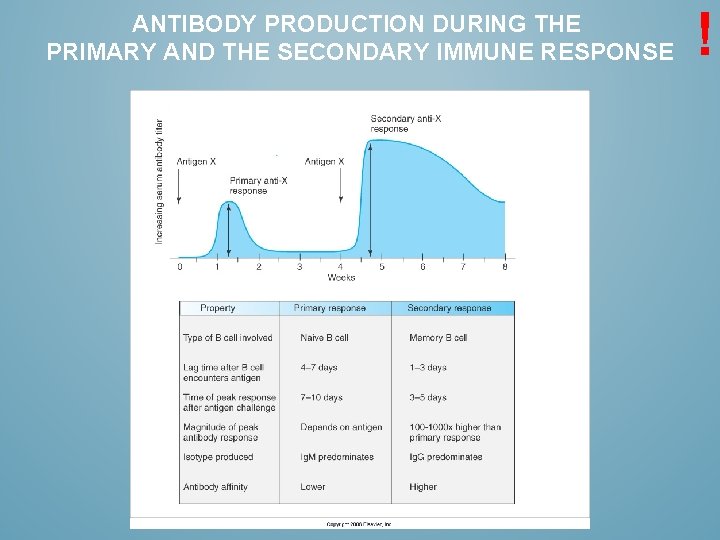
ANTIBODY PRODUCTION DURING THE PRIMARY AND THE SECONDARY IMMUNE RESPONSE !

EFFECTOR FUNCTIONS OF ANTIBODIES Antibody-mediated immune responses • NEUTRALIZATION • OPSONIZATION ADCC MAST CELL DEGRANULATION • COMPLEMENT FIXATION !!
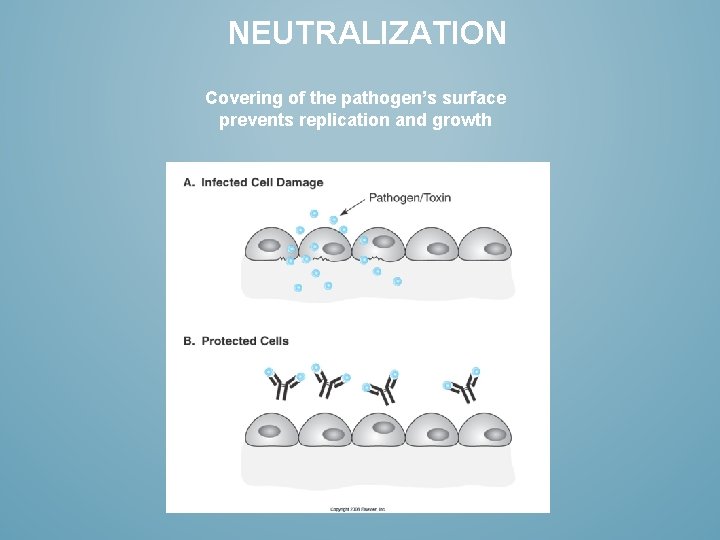
NEUTRALIZATION Covering of the pathogen’s surface prevents replication and growth
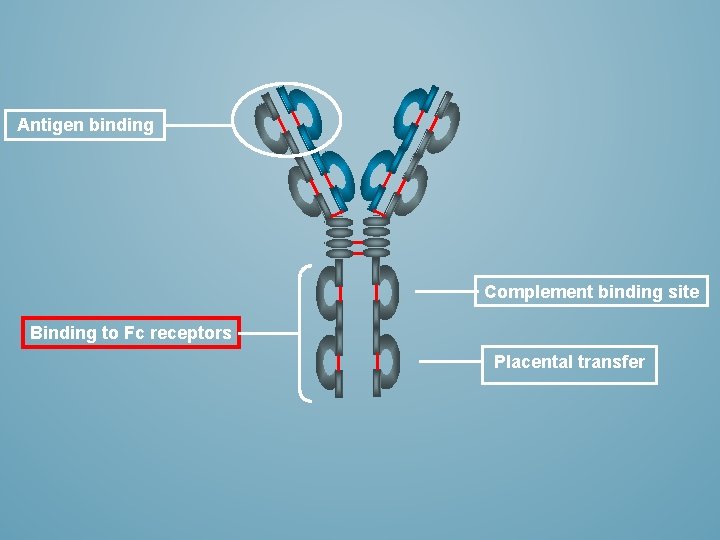
Antigen binding Complement binding site Binding to Fc receptors Placental transfer
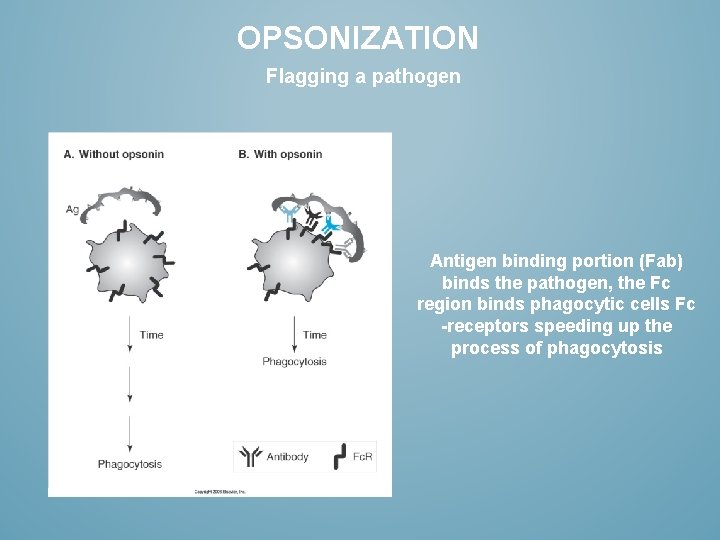
OPSONIZATION Flagging a pathogen Antigen binding portion (Fab) binds the pathogen, the Fc region binds phagocytic cells Fc -receptors speeding up the process of phagocytosis
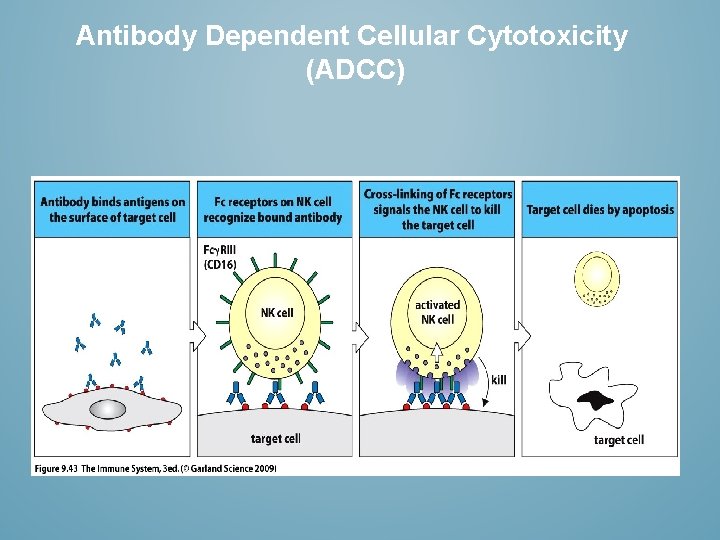
Antibody Dependent Cellular Cytotoxicity (ADCC)
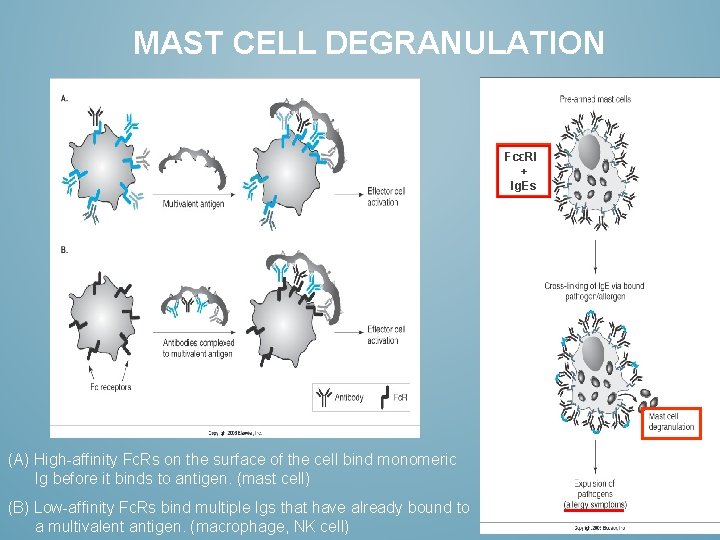
MAST CELL DEGRANULATION FcεRI + Ig. Es (A) High-affinity Fc. Rs on the surface of the cell bind monomeric Ig before it binds to antigen. (mast cell) (B) Low-affinity Fc. Rs bind multiple Igs that have already bound to a multivalent antigen. (macrophage, NK cell)
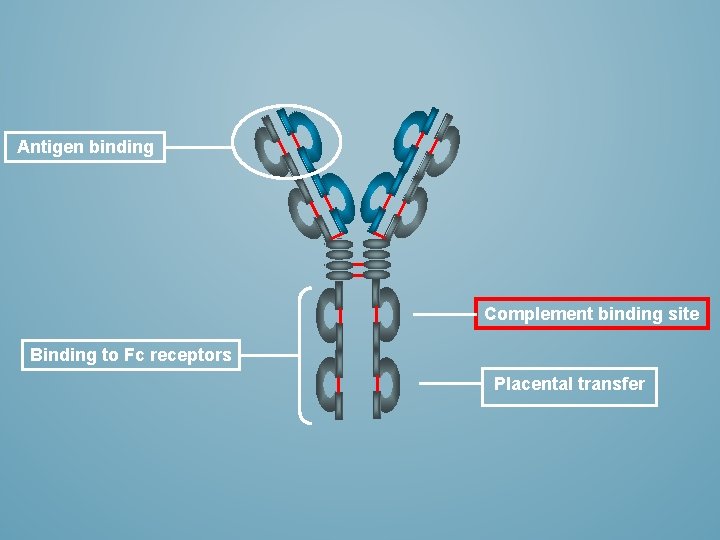
Antigen binding Complement binding site Binding to Fc receptors Placental transfer

COMPLEMENT FIXATION Ig. M and Ig. Gs activate the classical pathway of the complement system
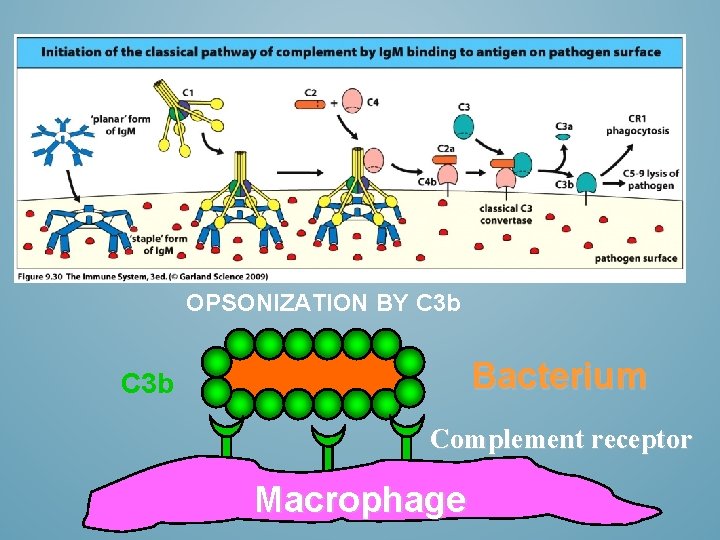
OPSONIZATION BY C 3 b Bacterium C 3 b Complement receptor Macrophage
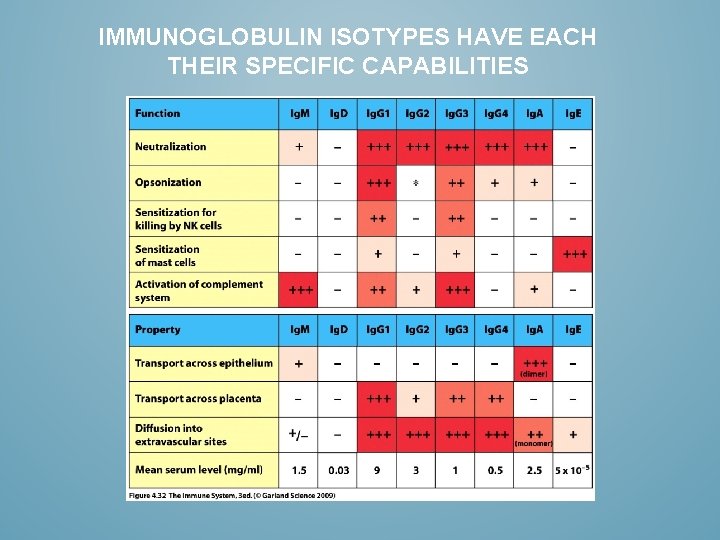
IMMUNOGLOBULIN ISOTYPES HAVE EACH THEIR SPECIFIC CAPABILITIES
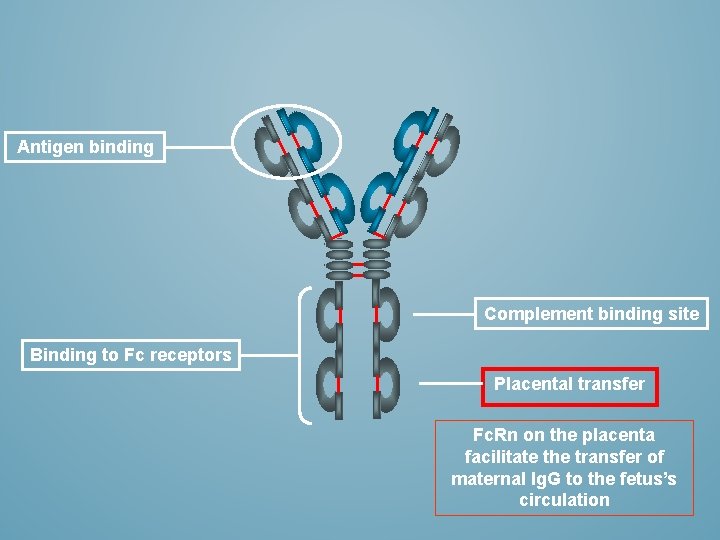
Antigen binding Complement binding site Binding to Fc receptors Placental transfer Fc. Rn on the placenta facilitate the transfer of maternal Ig. G to the fetus’s circulation
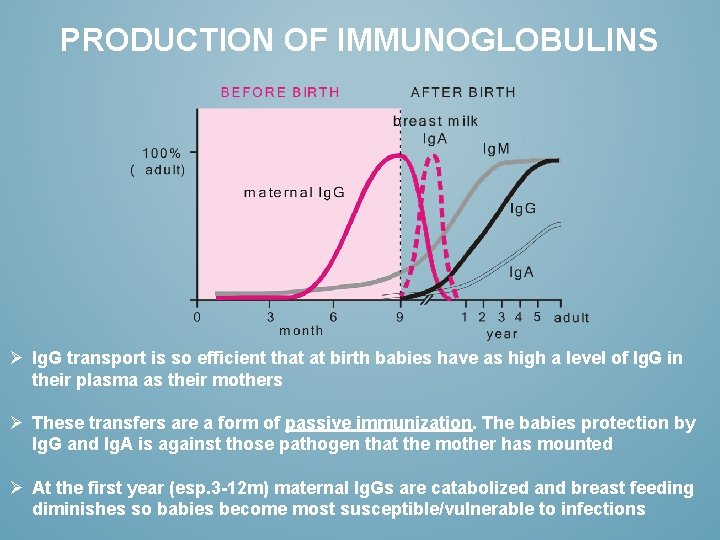
PRODUCTION OF IMMUNOGLOBULINS Ø Ig. G transport is so efficient that at birth babies have as high a level of Ig. G in their plasma as their mothers Ø These transfers are a form of passive immunization. The babies protection by Ig. G and Ig. A is against those pathogen that the mother has mounted Ø At the first year (esp. 3 -12 m) maternal Ig. Gs are catabolized and breast feeding diminishes so babies become most susceptible/vulnerable to infections
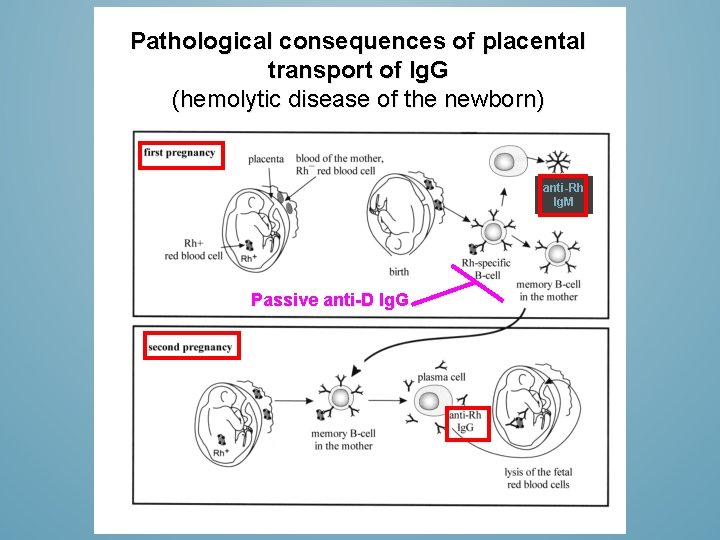
Pathological consequences of placental transport of Ig. G (hemolytic disease of the newborn) anti-Rh Ig. M Passive anti-D Ig. G
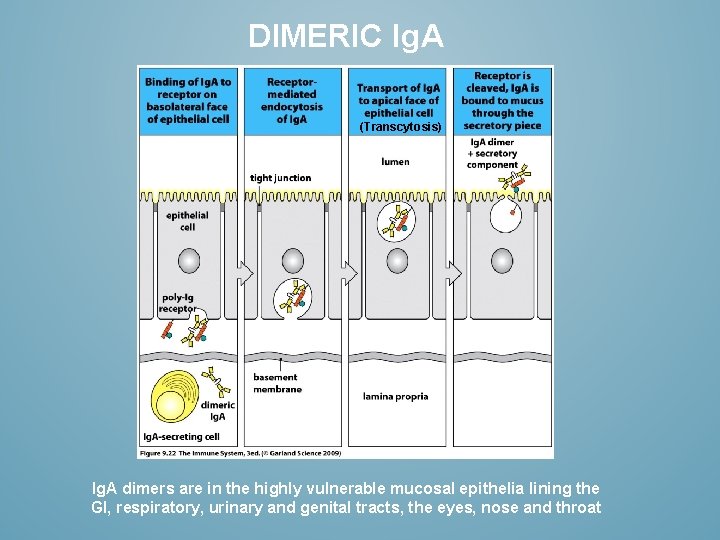
DIMERIC Ig. A (Transcytosis) Ig. A dimers are in the highly vulnerable mucosal epithelia lining the GI, respiratory, urinary and genital tracts, the eyes, nose and throat
- Slides: 39