Immunoglobulin Collage of Medical Henan University Contents Introduction

Immunoglobulin Collage of Medical, Henan University

Contents Introduction SectionⅠ Molecular Structure of Ig SectionⅡ Characteristics and Functions of the 5 Classes of Ig SectionⅢ Fc Receptors for Ab Molecules SectionⅣ Biological Activity of Ab SectionⅤ Immunogenicity of Ig SectionⅥ Artificial Ab

Concepts Antibody (Ab): Glycoprotein molecules that are produced by plasma cells and can combine with the corresponding Ag specifically are called Ab. Ab is produced by B cells in the response to a stimulation of Ag. Ab possesses a high degree of specificity and affinity

• Immunoglobulin(Ig): It refers to all globulins that possess the activity of Ab or show a similar structure to Ab • Therefore, All Abs are Igs, but not all Igs possess the functions of Abs

Other Concepts γ- Globulin Antiserum Humoral Immunity s. Ig and m. Ig(BCR)

SectionⅠ Molecular Structure of Ig
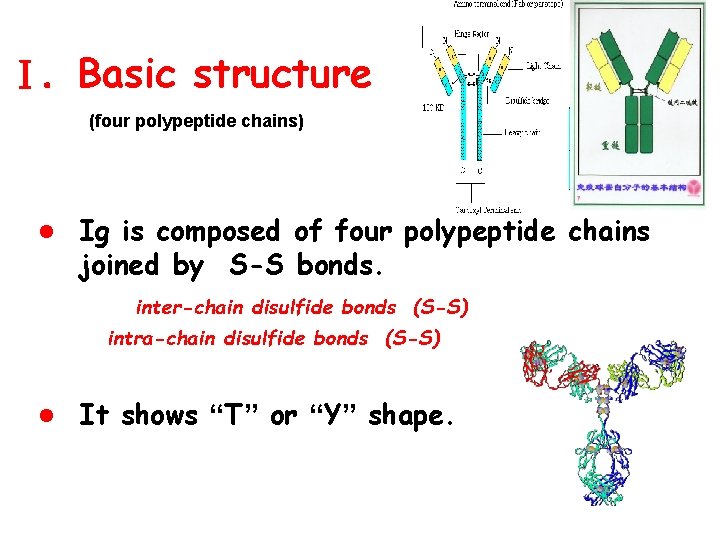
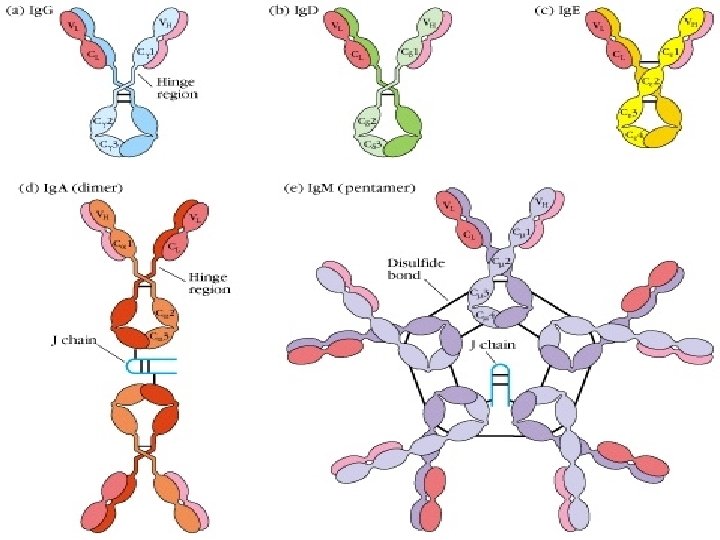
Ⅰ. Basic structure (four polypeptide chains) l Ig is composed of four polypeptide chains joined by S-S bonds. inter-chain disulfide bonds (S-S) intra-chain disulfide bonds (S-S) l It shows “T” or “Y” shape.
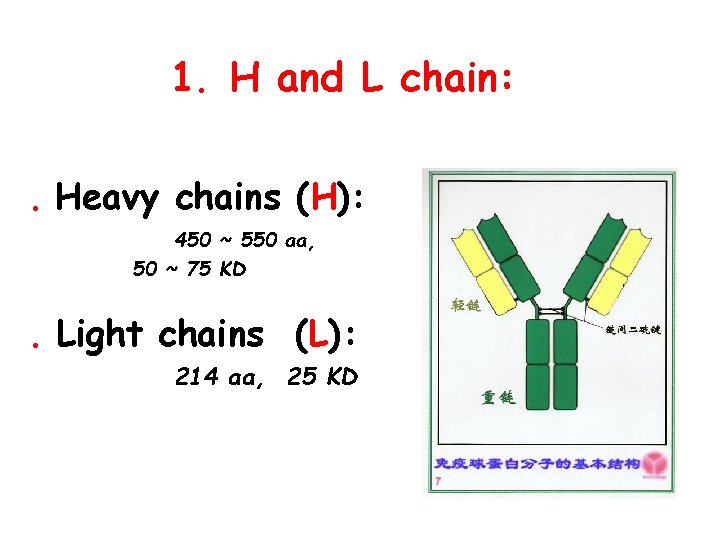
1. H and L chain: . Heavy chains (H): 450 ~ 550 aa, 50 ~ 75 KD . Light chains (L): 214 aa, 25 KD
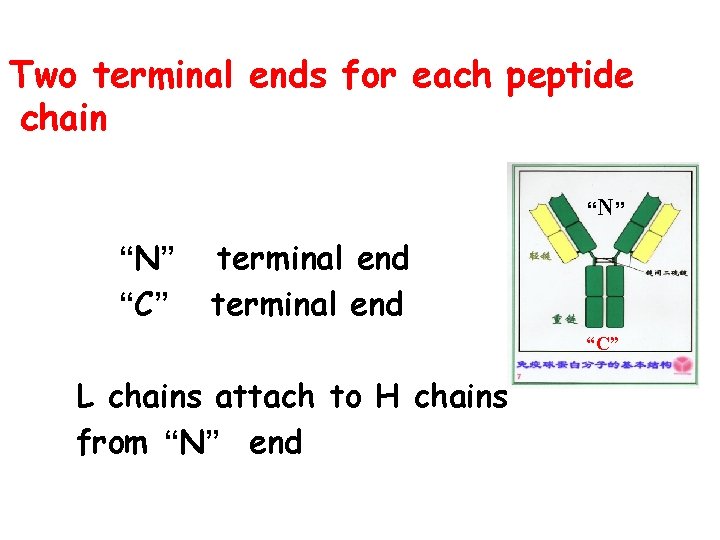
Two terminal ends for each peptide chain “N” “C” terminal end “C” L chains attach to H chains from “N” end

2. classes and types of Ig (1) According to the differences of H chains (amino acid composition, sequence) Igs can be divided into 5 classes subclasses • Five classes of H Chain: • Five classes of Igs: Ig. G Ig. A Ig. M Ig. D Ig. E Ig. G 1~ Ig. G 4 Ig. A 1, Ig. A 2
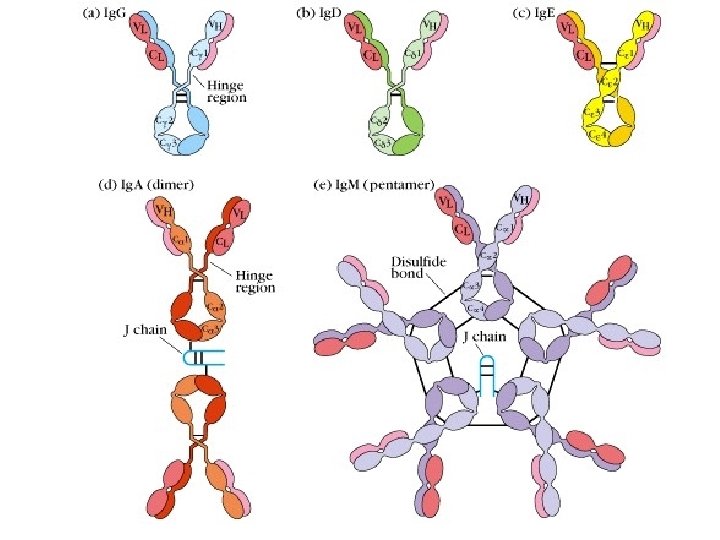
(2) According to the differences of L chains • Two types of L chain: , : human) 1~ 4 subtypes 20: 1 (in mice); 2: 1 (in
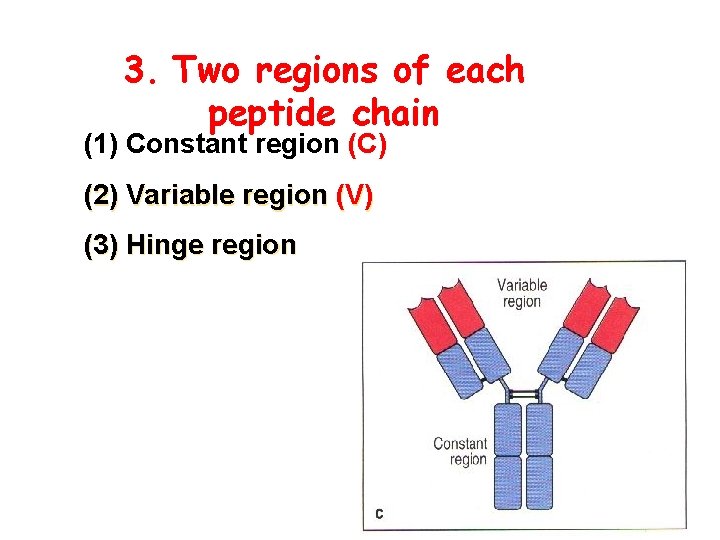
3. Two regions of each peptide chain (1) Constant region (C) (2) Variable region (V) (3) Hinge region
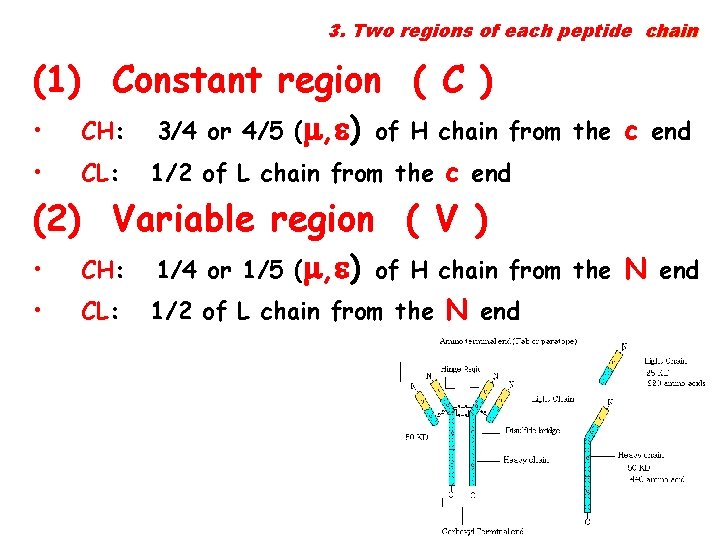
3. Two regions of each peptide chain (1) Constant region ( C ) • CH: 3/4 or 4/5 ( , ) of H chain from the c end • CL: 1/2 of L chain from the c end (2) Variable region ( V ) • CH: 1/4 or 1/5 ( , ) of H chain from the N end • CL: 1/2 of L chain from the N end
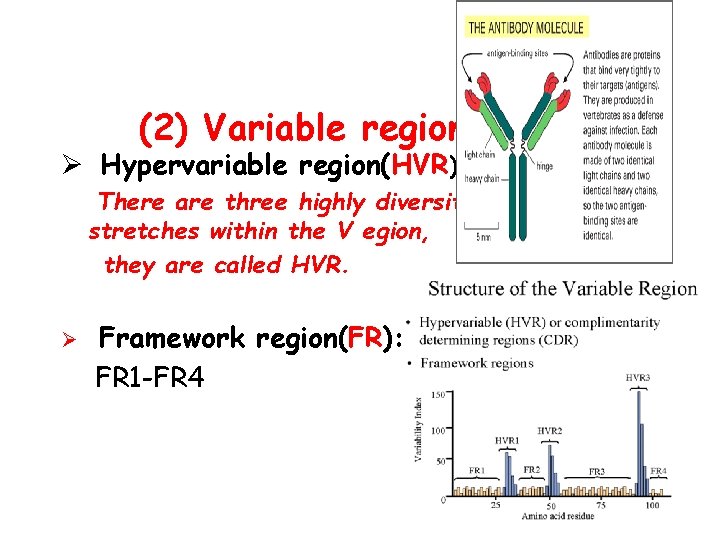
(2) Variable region ( V ): Ø Hypervariable region(HVR) There are three highly diversity stretches within the V egion, they are called HVR. Ø Framework region(FR): FR 1 -FR 4
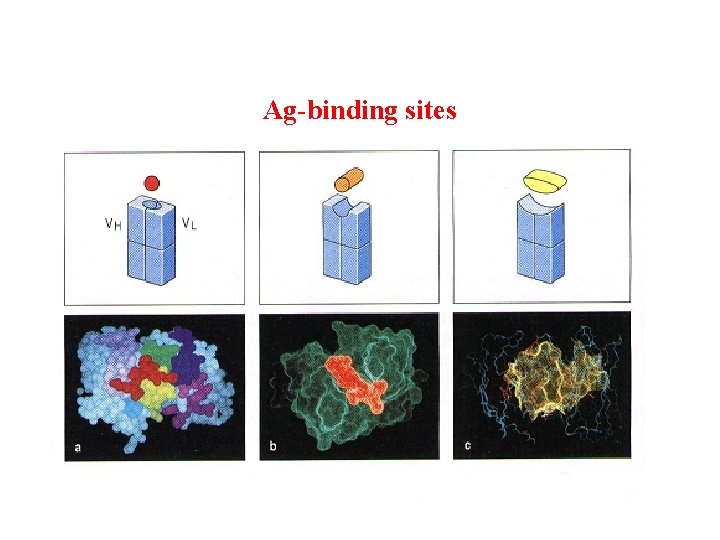
Ag-binding sites
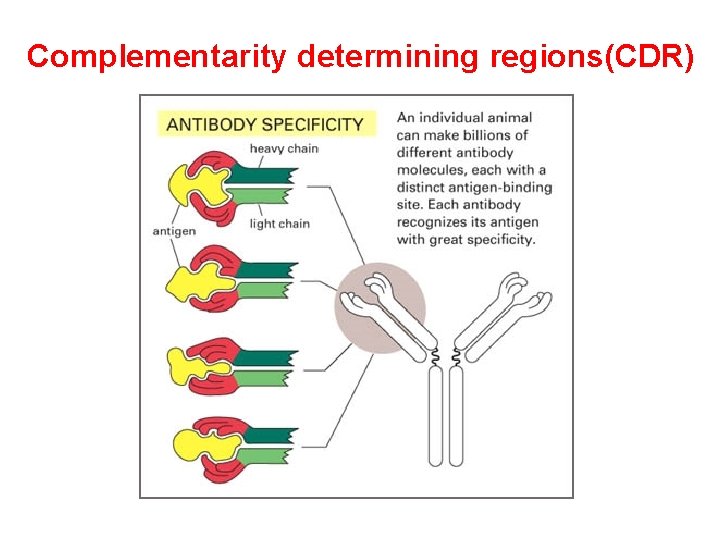
Complementarity determining regions(CDR)
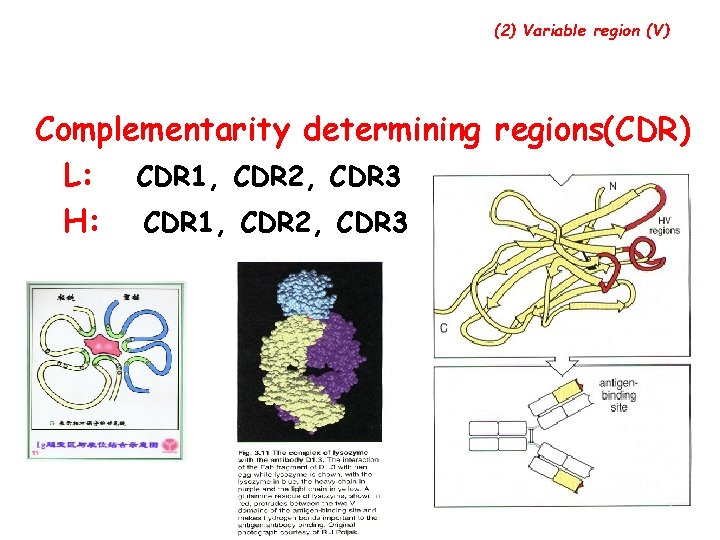
(2) Variable region (V) Complementarity determining regions(CDR) L: CDR 1, CDR 2, CDR 3 H: CDR 1, CDR 2, CDR 3
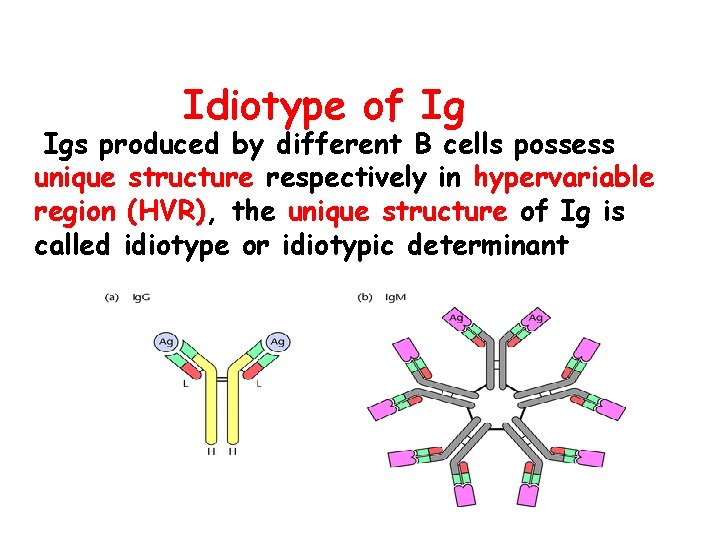
Idiotype of Ig Igs produced by different B cells possess unique structure respectively in hypervariable region (HVR), the unique structure of Ig is called idiotype or idiotypic determinant
In fact: HVR CDR Idiotype are in the same sites of Ig
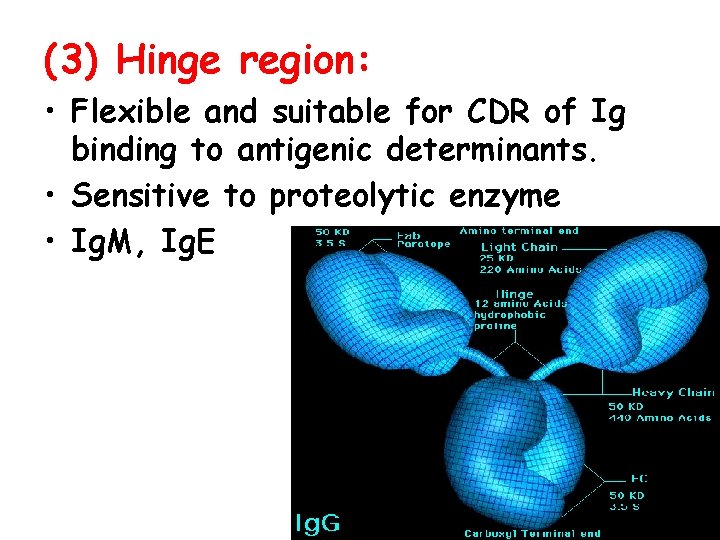
(3) Hinge region: • Flexible and suitable for CDR of Ig binding to antigenic determinants. • Sensitive to proteolytic enzyme • Ig. M, Ig. E
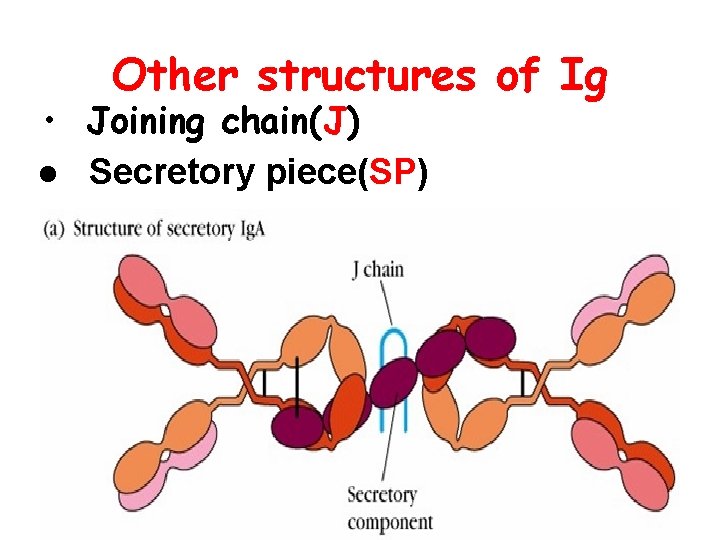
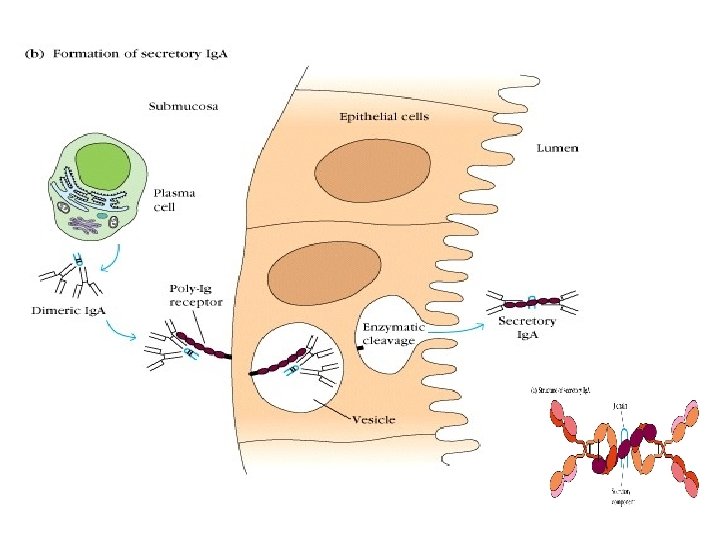
Other structures of Ig • Joining chain(J) l Secretory piece(SP)
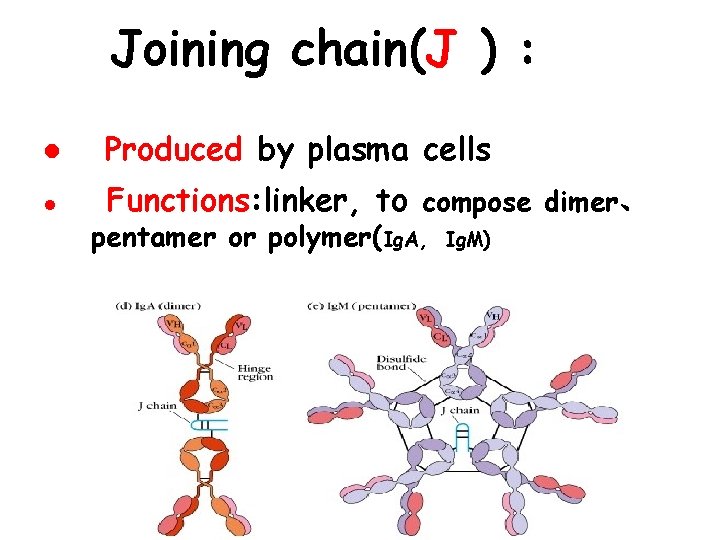
Joining chain(J ) : l Produced by plasma cells l Functions: linker, to compose dimer、 pentamer or polymer(Ig. A, Ig. M)
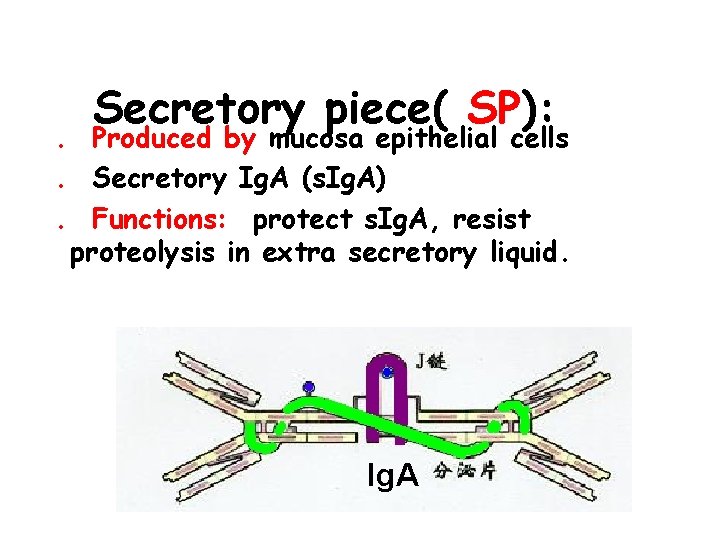
Secretory piece( SP): . Produced by mucosa epithelial cells. Secretory Ig. A (s. Ig. A). Functions: protect s. Ig. A, resist proteolysis in extra secretory liquid. Ig. A
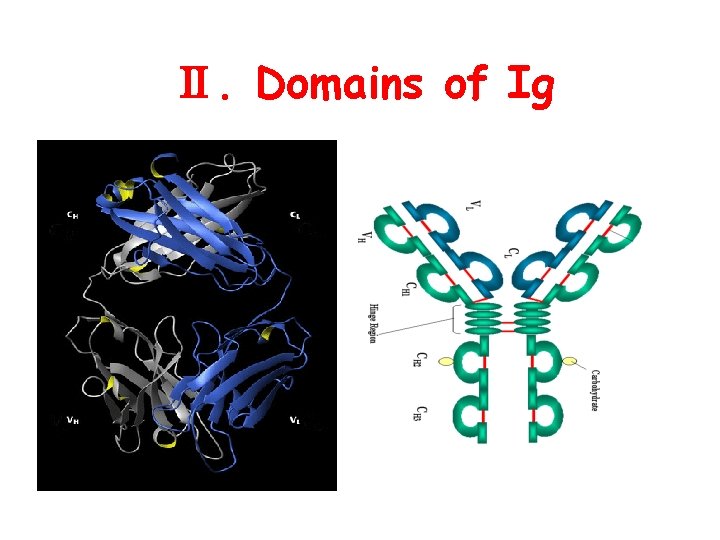
Ⅱ. Domains of Ig

1. Domain : Polypeptide chains of Ig are folded into a globular structure by intra chain s-s bond within each 110 aa region which is called a domain
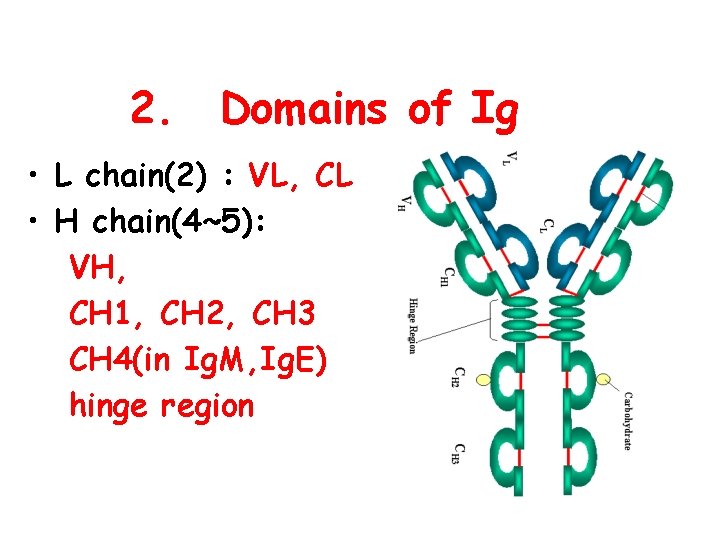
2. Domains of Ig • L chain(2) : VL, CL • H chain(4~5): VH, CH 1, CH 2, CH 3 CH 4(in Ig. M, Ig. E) hinge region
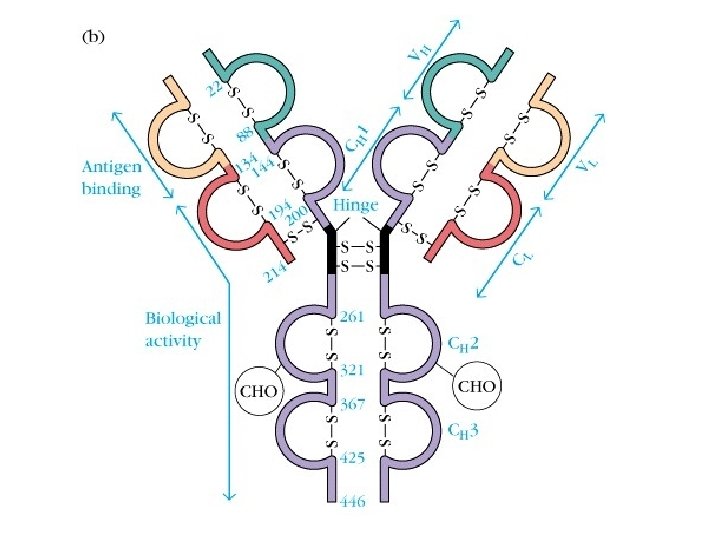

3. Function of each domain • VH, VL: antigen-binding site • CH 1, CL: allogeneic marker • CH 2/CH 3: complement-fixing site, permeate placenta(Ig. G) • CH 3/CH 4: cell-binding site l Hinge region : flexible and suitable for CDR of Ig binding to antigenic determinants
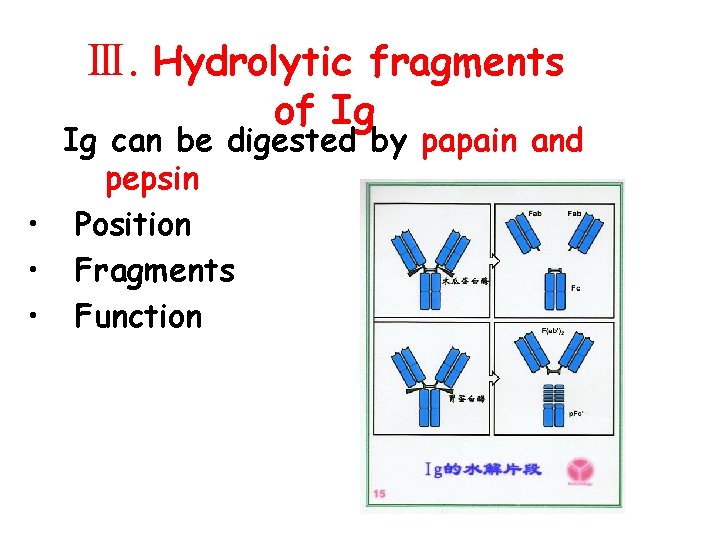
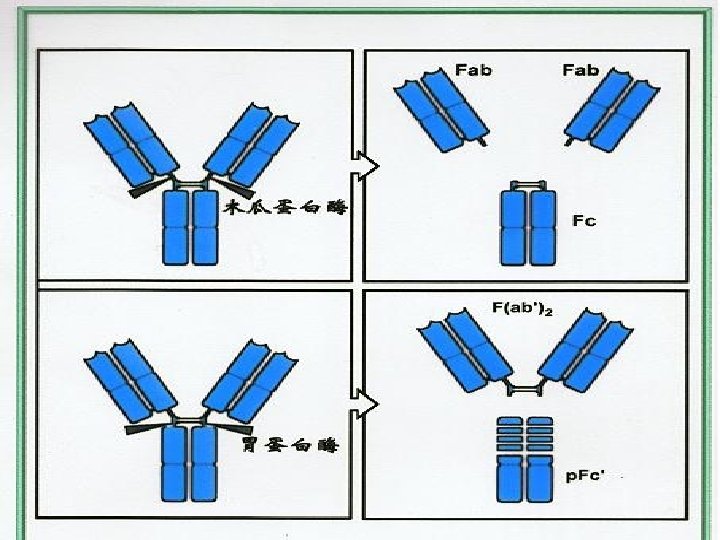
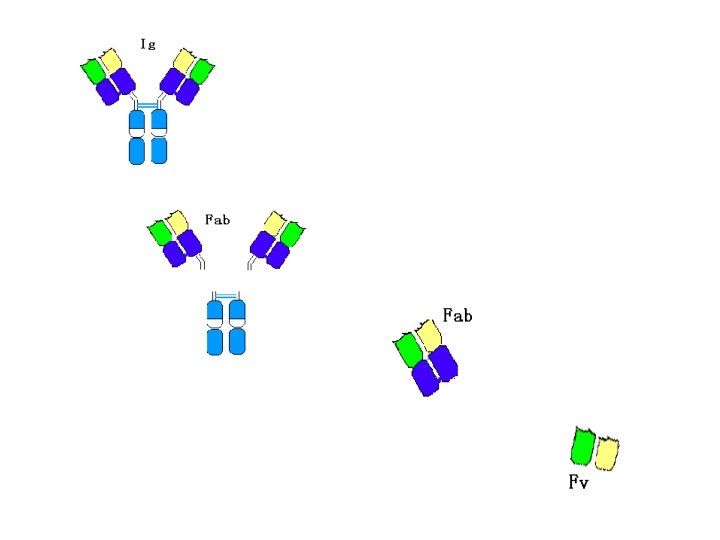
Ⅲ. Hydrolytic fragments of Ig Ig can be digested by papain and pepsin • Position • Fragments • Function
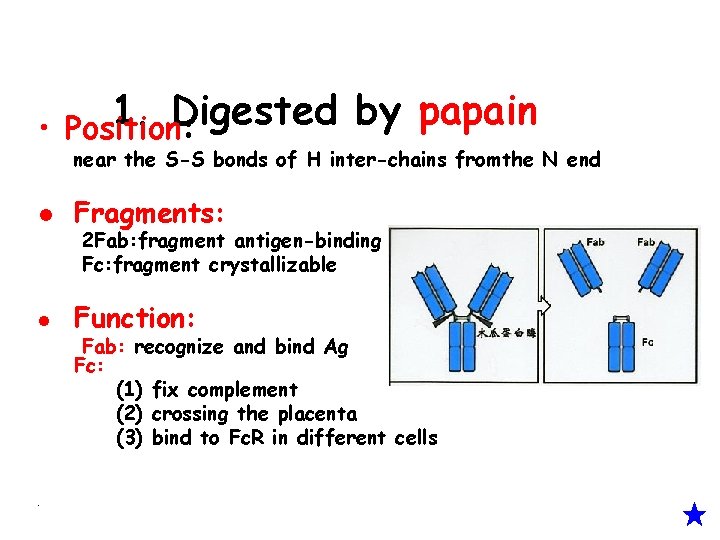
1. Digested by papain • Position: near the S-S bonds of H inter-chains fromthe N end l Fragments: l Function: • 2 Fab: fragment antigen-binding Fc: fragment crystallizable Fab: recognize and bind Ag Fc: (1) fix complement (2) crossing the placenta (3) bind to Fc. R in different cells
Ag: Ab ratio

2. Digested by pepsin • Position: near the S-S bond of H inter-chains from the C end • Fragments and function : F(ab′)2: bind antigen(2 valence) p. Fc′: no function

Significance l Elucidating the relationships between the structure and function of Igs l Decrease the immunogenicity of Ig for clinical treatment
SectionⅡ Characteristics and Functions of the 5 Classes of Igs
Ⅰ. Ig. G 1. Highest concentration in serum(75% of total Ig)
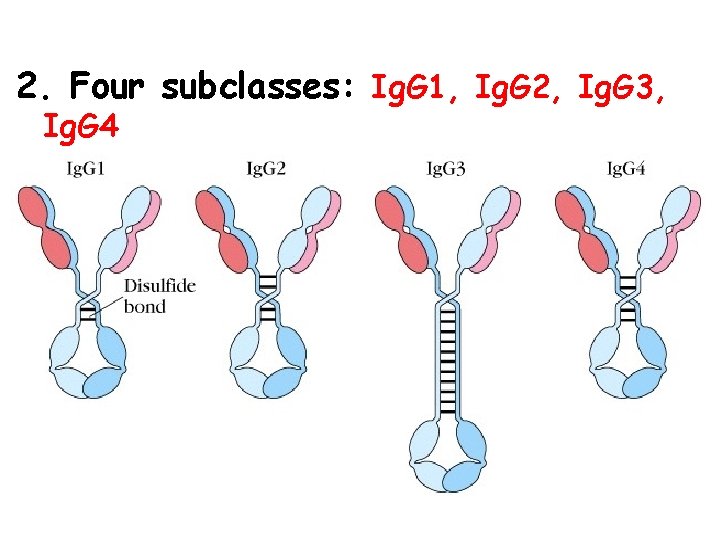
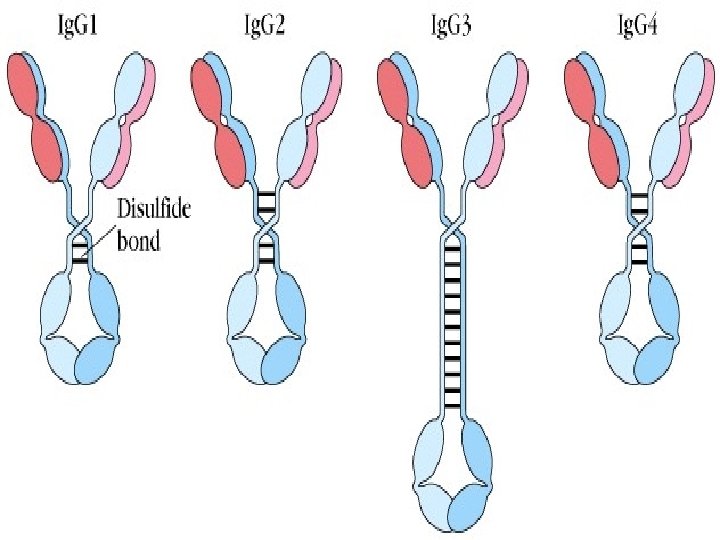
2. Four subclasses: Ig. G 1, Ig. G 2, Ig. G 3, Ig. G 4
3. Unique Ig that can pass through placenta 4. Half-life is longer( 16 -24 days ) 5. Starts to be produced at 2 -3 month after birth and reach the level of adult at 5 years old

6. Functions of Ig. G: • • • Against bacteria and virus, neutralize toxin Combine with the Fc receptor(FcγR) Activate complement Combine with SPA Some belong to the auto-antibodies • Take part in type Ⅱ and Ⅲ hypersensitivity
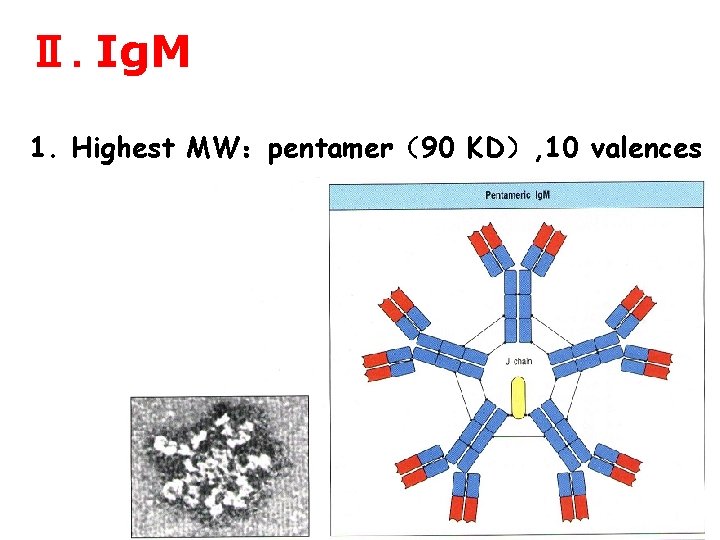
Ⅱ. Ig. M 1. Highest MW:pentamer(90 KD), 10 valences

2. Half-life is shorter(4~5 days) 3. The first Ig to be synthesized • Appear in the early stage after infection • Be produced during fetus • The first m. Ig of the B cells, act as the antigen receptors(BCR)

4. Functions: • Ig. M is more effective in binding Ag and activating C, and play an important role in anti-infection • Natural Ab for blood-type antigen • Auto-antibody: rheumatoid factor(RF) • Take part in type Ⅱ and Ⅲ hypersensitivity
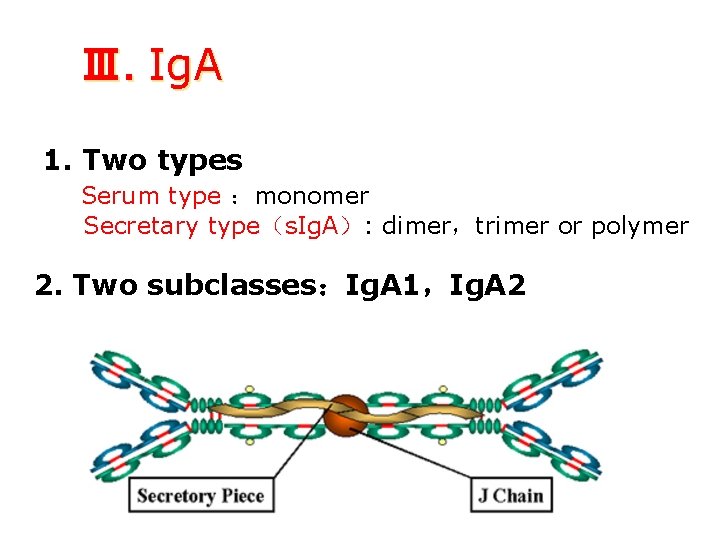
Ⅲ. Ig. A 1. Two types Serum type :monomer Secretary type(s. Ig. A): dimer,trimer or polymer 2. Two subclasses:Ig. A 1,Ig. A 2
3. To be produced at 4 months after birth 4. Exist in almost all body fluid

6. Local mucosal immunity • • Immune barrier Neutralize virus/toxin Rich in colostrum Activate C by alternative pathway • Take part in type Ⅲ hypersensitivity

Ⅳ. Ig. D 1. The concentration in serum is low and sensitive to proteinase 2. Act as the antigen receptor on B cells (m. Ig. D): Regulate the differentiation of B cells

Ⅴ. Ig. E 1. Concerntration of Ig. E in serum is the lowest in normal individual, but is very high in some patients. 2. Related to typeⅠpersensitivity FcεRⅠ: mast cell, basophil
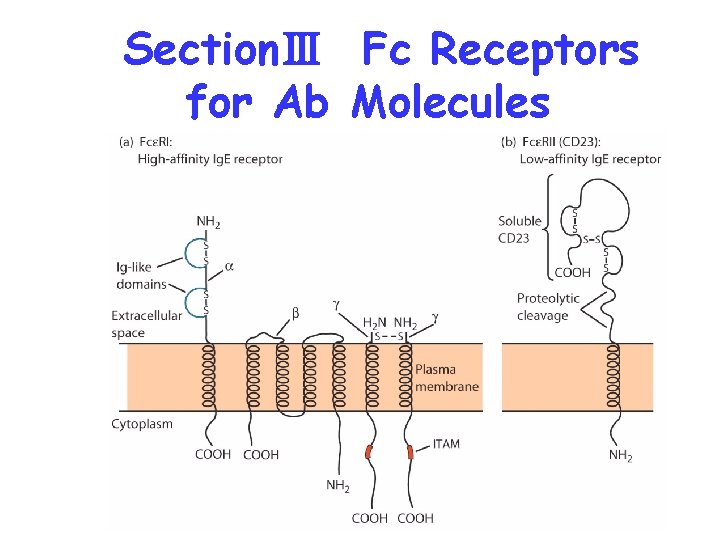
SectionⅢ Fc Receptors for Ab Molecules

Ig. G---Fc R: Fc RⅠ(CD 64)---phagocyte Fc RⅡ(CD 32)---immune complex Fc RⅢ(CD 16)--NK, macrophage, T cell Ig. E---Fc R: Fc RⅠ--- mast cell, basophil Fc RⅡ--- macrophage, B cell Ig. A---FcαR(CD 89)---phagocyte, neutrophil
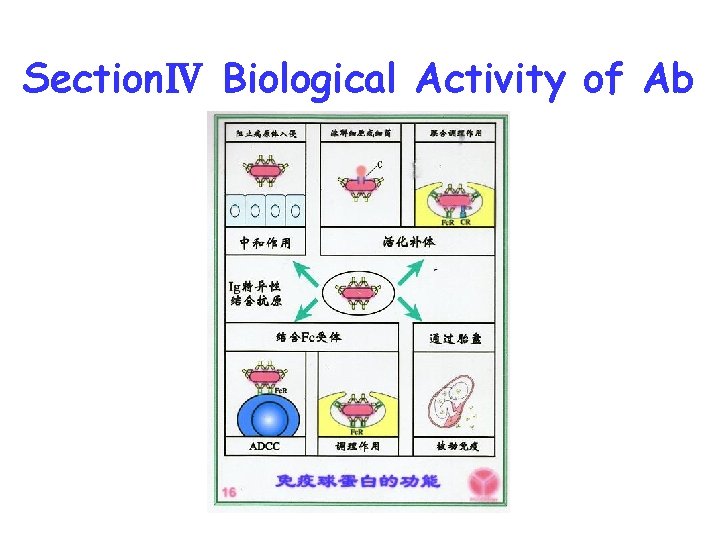
SectionⅣ Biological Activity of Ab

1. Recognize and bind to antigen specifically 2. Fix complement 3. Bind to Fc receptor on some cells 4. Transfer selectively : . Planceta transfer (Ig. G). Mucosa transfer (s. Ig. A)
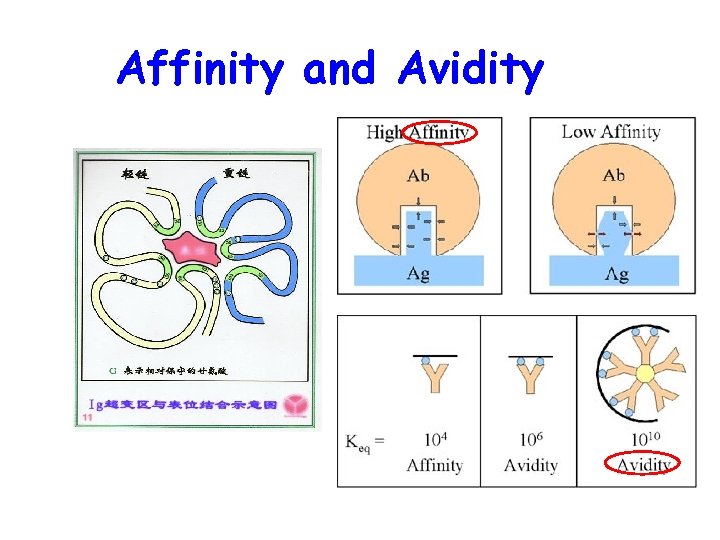
Affinity and Avidity
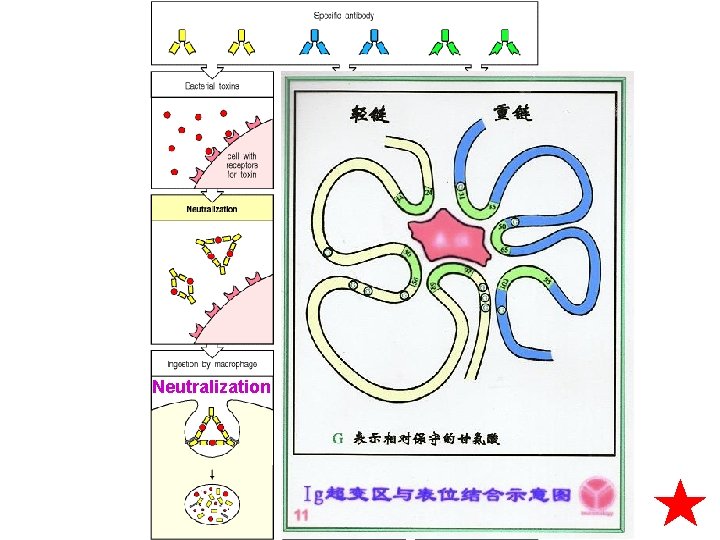
Neutralization
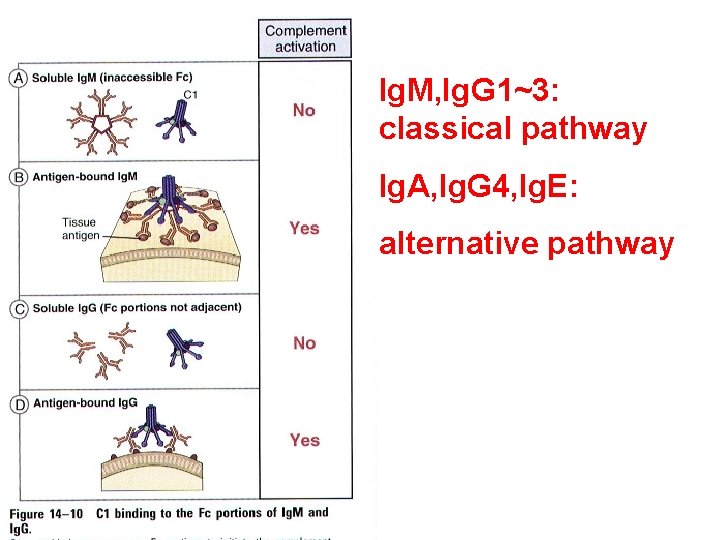
Ig. M, Ig. G 1~3: classical pathway Ig. A, Ig. G 4, Ig. E: alternative pathway
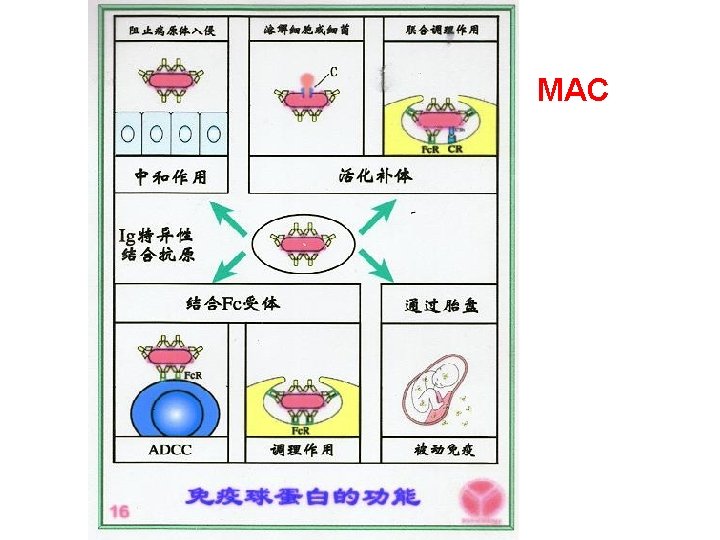
MAC
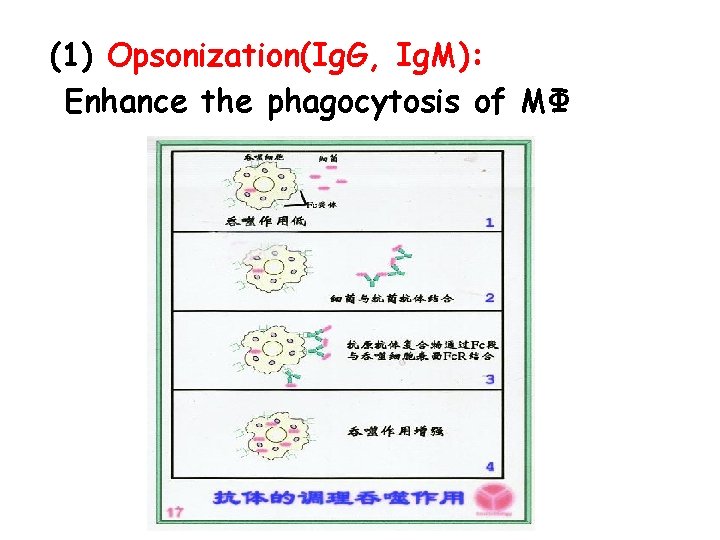
(1) Opsonization(Ig. G, Ig. M): Enhance the phagocytosis of MΦ
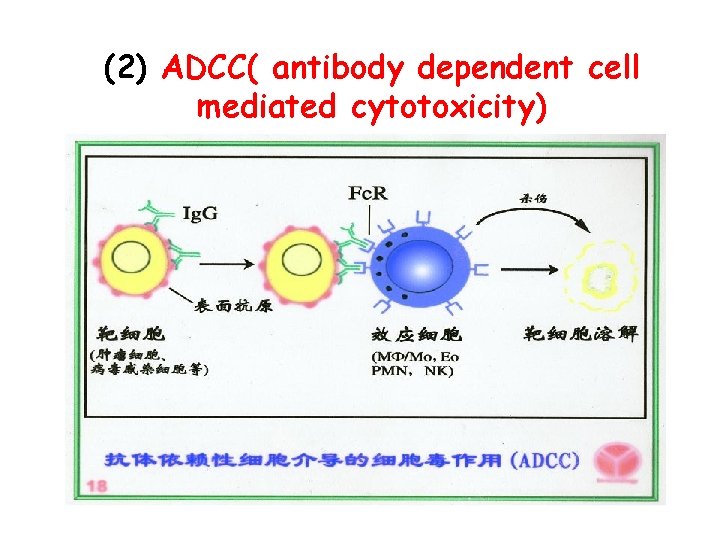
(2) ADCC( antibody dependent cell mediated cytotoxicity)
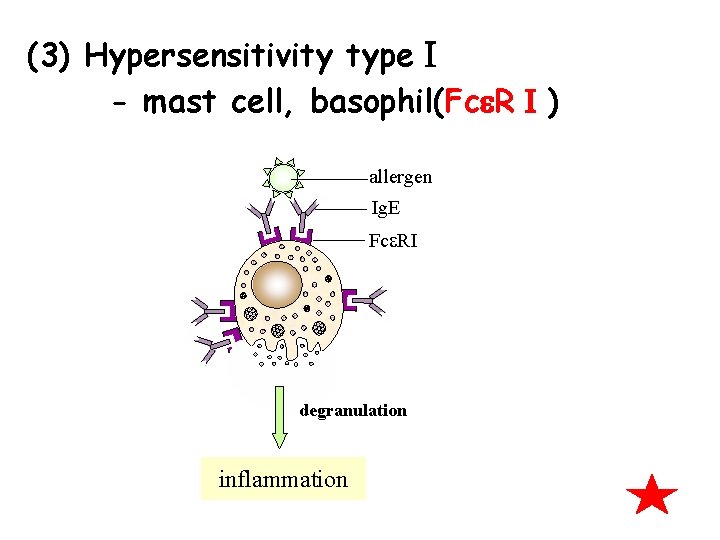
(3) Hypersensitivity typeⅠ - mast cell, basophil(Fc RⅠ) allergen Ig. E Fce. RI degranulation inflammation
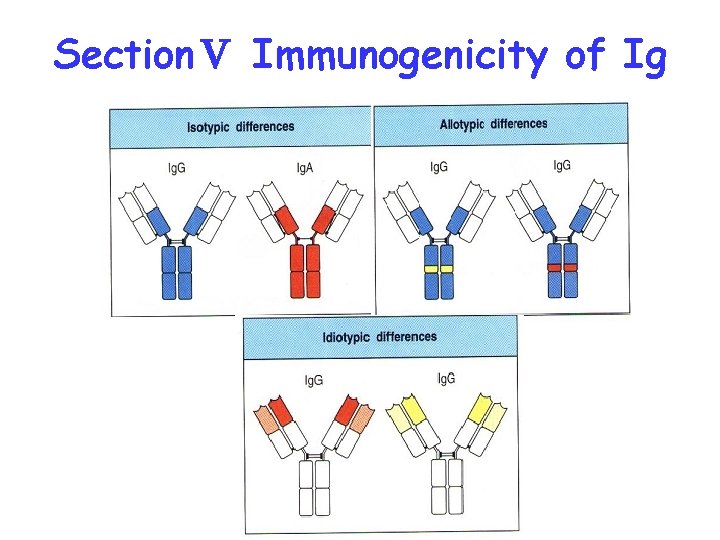
SectionⅤ Immunogenicity of Ig
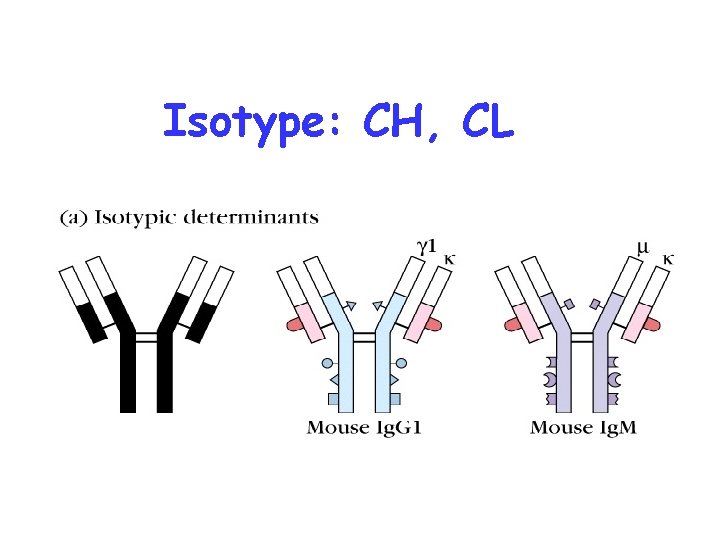
Isotype: CH, CL
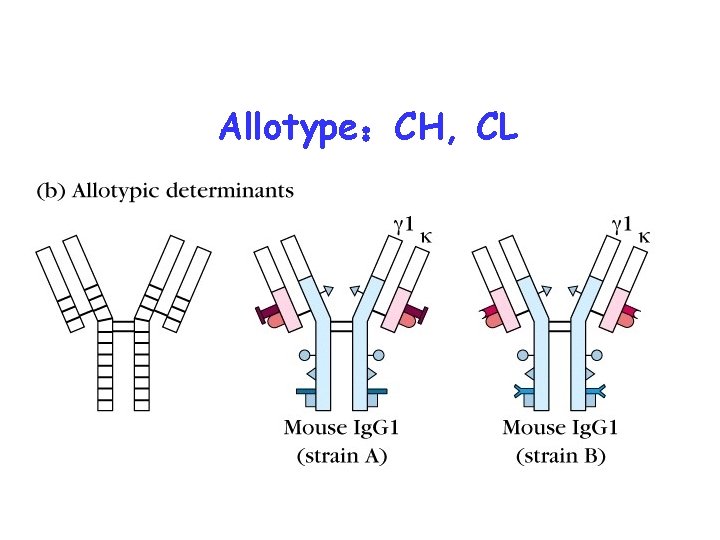
Allotype:CH, CL
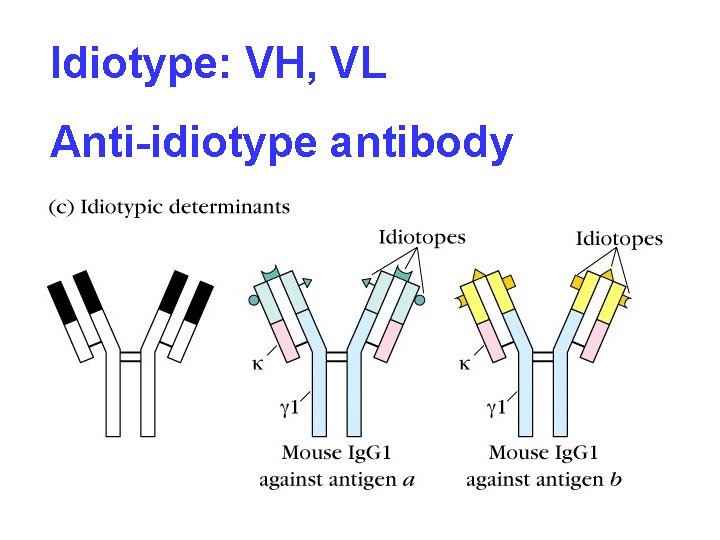
Idiotype: VH, VL Anti-idiotype antibody

SectionⅥ Artificial Ab Ø Ø Ø Polyclonal Ab Monoclonal Ab Gene engineering Ab

1. Polyclonal Ab l A mixture Ab with different specificities and affinities l Generate in a natural response or artificial immunization l Cross reaction

Cross-reactivity: if two antibody antigens share recognizes an chemically similar, epitope. an epitope unrelated, an but

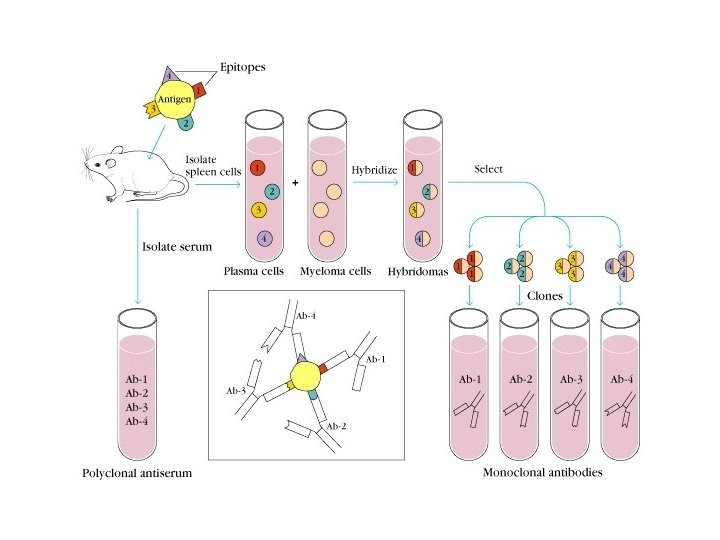
2. Monoclonal Ab (m. Ab) Ab produced by single clone (or one hybridomas clone ) and having a single specificity

m. Ab / Mc. Ab u Prepared by hybridomas technique: Immunized spleen cells(B) hybride with myeloma cells----hybridomas

Artificial antibodies POLYCLONAL. MONOCLONAL. Derived from different B Lymphocytes cell lines Derived from a single B cell clone Batch to Batch variation affecting Ab reactivity & titre m. Ab offer Reproducible, Predictable & Potentially inexhaustible supply of Ab with exquisite specificity NOT Powerful tools for clinical diagnostic tests Enable the development of secure immunoassay systems.
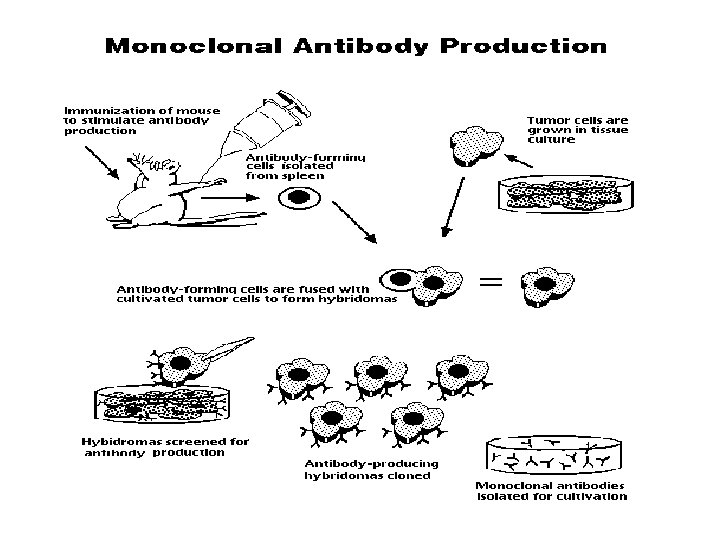
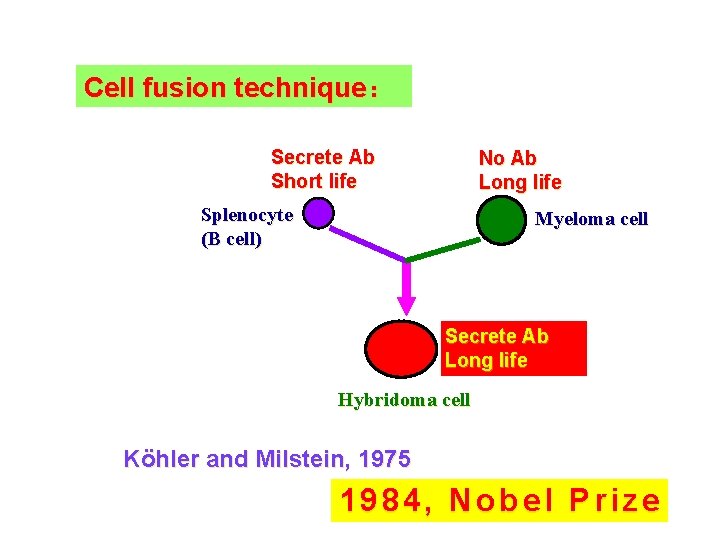
Cell fusion technique: Secrete Ab Short life No Ab Long life Splenocyte (B cell) Myeloma cell Secrete Ab Long life Hybridoma cell Köhler and Milstein, 1975 1984, Nobel Prize
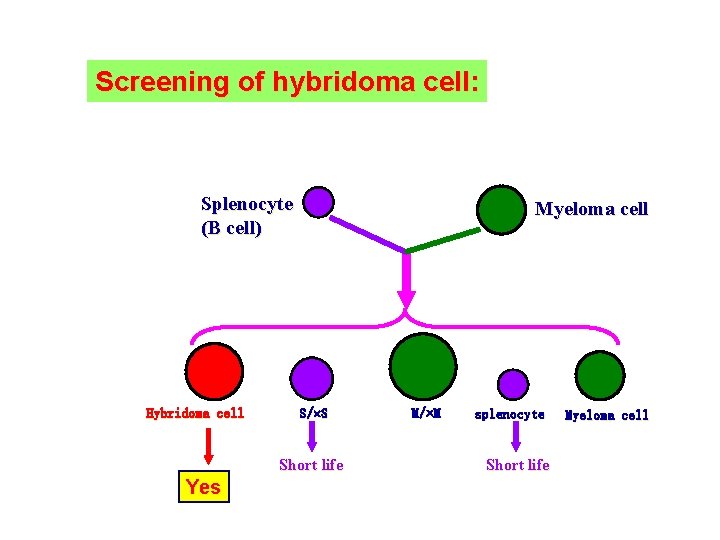
Screening of hybridoma cell: Splenocyte (B cell) Hybridoma cell Myeloma cell S/ S Short life Yes M/ M splenocyte Short life Myeloma cell
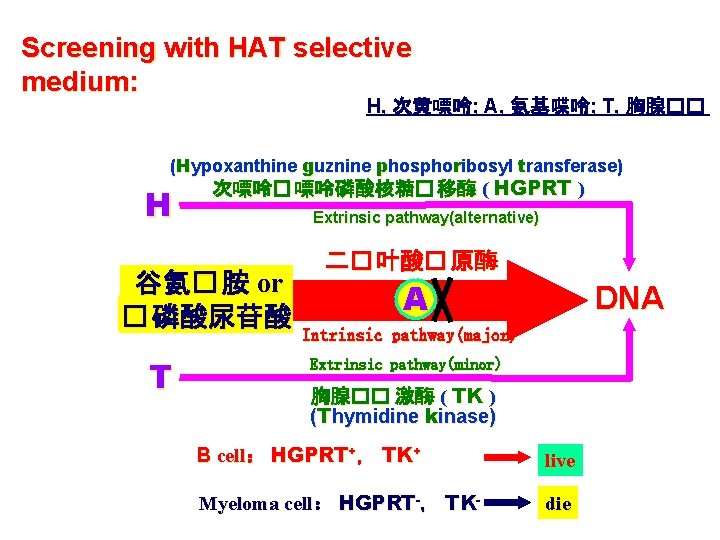
Screening with HAT selective medium: H, 次黄嘌呤; A, 氨基喋呤; T, 胸腺�� (Hypoxanthine guznine phosphoribosyl transferase) H 次嘌呤� 嘌呤磷酸核糖� 移酶 ( HGPRT ) Extrinsic pathway(alternative) 谷氨� 胺 or � 磷酸尿苷酸 T 二� 叶酸� 原酶 A DNA Intrinsic pathway(major) Extrinsic pathway(minor) 胸腺�� 激酶 ( TK ) (Thymidine kinase) B cell: HGPRT+, TK+ live Myeloma cell: HGPRT-, TK- die
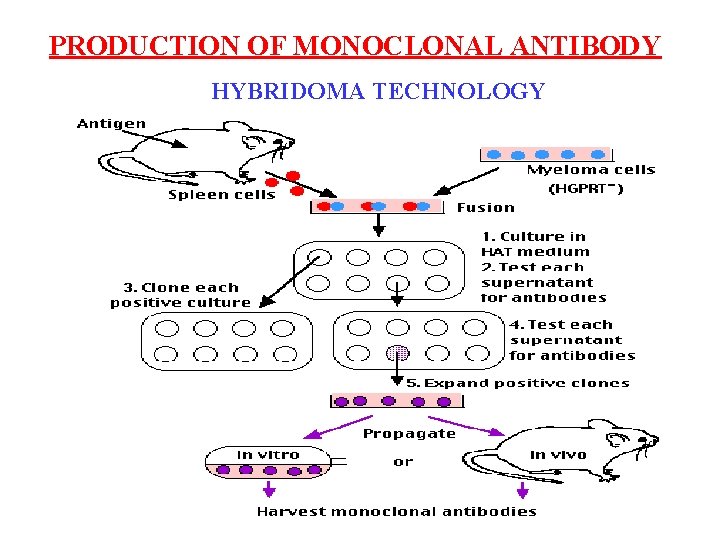
PRODUCTION OF MONOCLONAL ANTIBODY HYBRIDOMA TECHNOLOGY

Applications of Monoclonal Antibodies • Diagnostic Applications Biosensors & Microarrays • Therapeutic Applications Transplant rejection Muronomab-CD 3 Cardiovascular disease Abciximab Cancer Rituximab Infectious Diseases Palivizumab Inflammatory disease Infliximab • Clinical Applications Purification of drugs, Imaging the target • Future Applications Fight against Bioterrorism
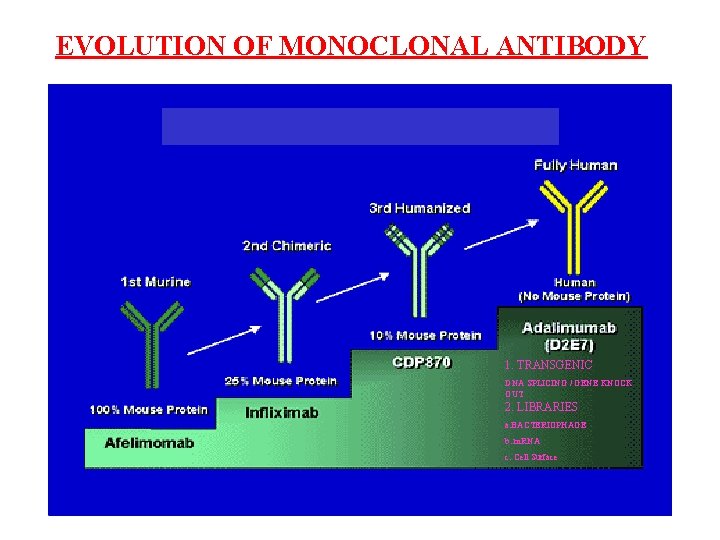
EVOLUTION OF MONOCLONAL ANTIBODY 1. TRANSGENIC DNA SPLICING / GENE KNOCK OUT 2. LIBRARIES a. BACTERIOPHAGE b. m. RNA c. Cell Surface

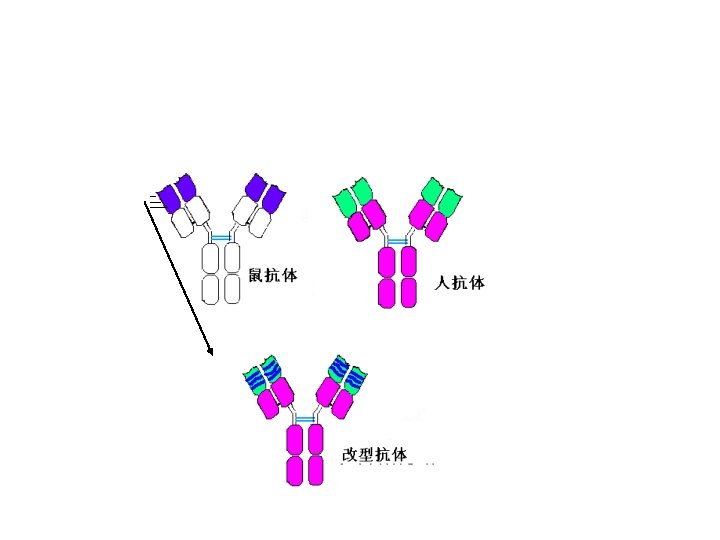
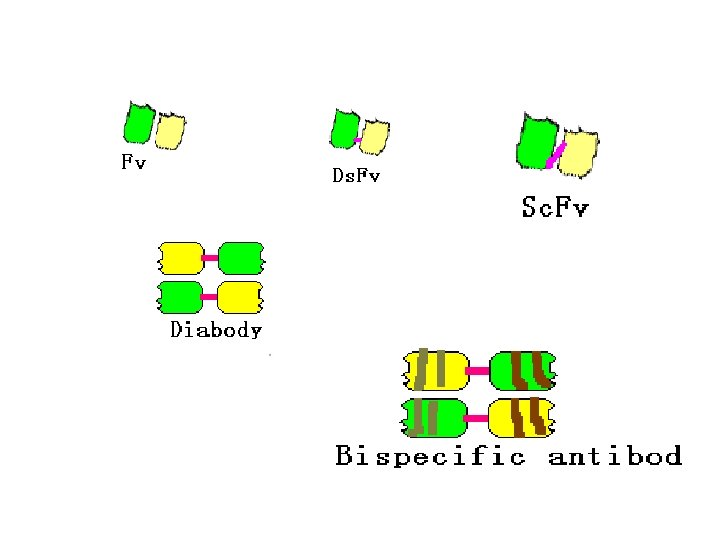
3. Gene engineering Ab • Abs prepared by the method of gene recombination • Chimeric Ab: human Fc bind with mice Fab • Recombinant single chain Ab: VH-linker-VL
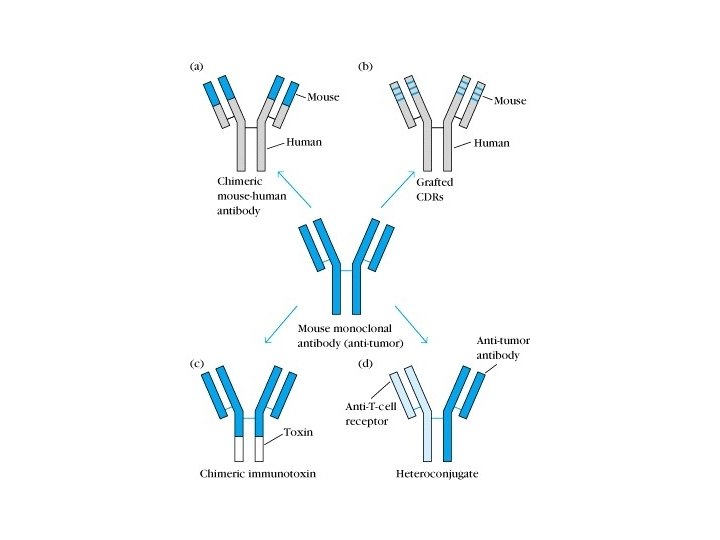
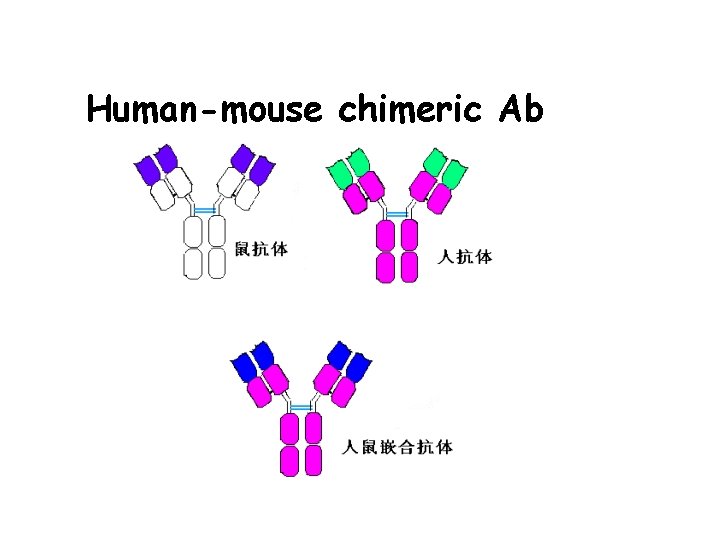
Human-mouse chimeric Ab

Niels K Jerne Denmark Basel Institute for Immunology Basel, Switzerland J. Kohler Germany Basel Institute for Immunology Basel, Switzerland S. Milstein Argentina MRC Laboratory of Molecular Biology Cambridge,
- Slides: 89