IMMUNOFLUORESCENCE Lab 6 Immunofluorescence It is a technique

IMMUNOFLUORESCENCE Lab. 6

Immunofluorescence • It is a technique that uses a fluorescent compound (fluorophore or fluorochrome) to indicate a specific antigen-antibody reaction • If antibody molecules are tagged with a fluorescent dye and then binds to an antigen, this immune fluorescently labeled complex can be detected by colored light emission when excited by light of the appropriate wavelength UV 2
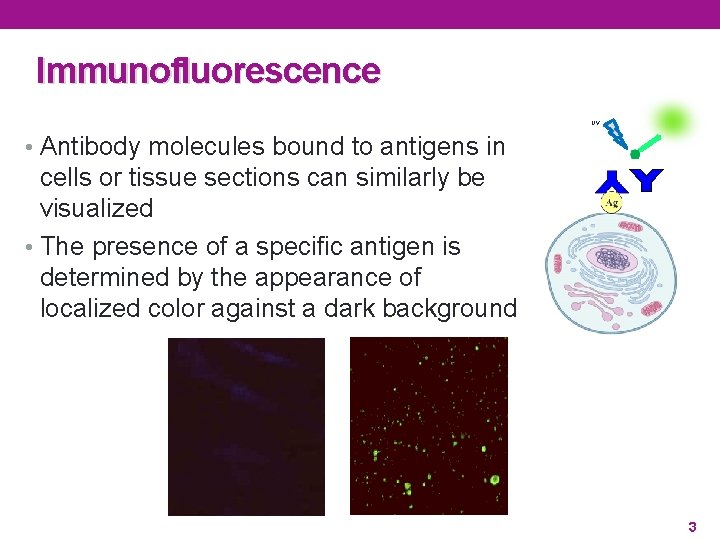
Immunofluorescence UV • Antibody molecules bound to antigens in cells or tissue sections can similarly be visualized • The presence of a specific antigen is determined by the appearance of localized color against a dark background 3

Applications of IF • This method is used for: • Rapid identification of microorganisms in cell culture or infected tissue • Antigens on neoplastic tissue & inside cells • and CD antigens on T and B cells through the use of cell flow cytometry • Detection of different proteins inside cells 4
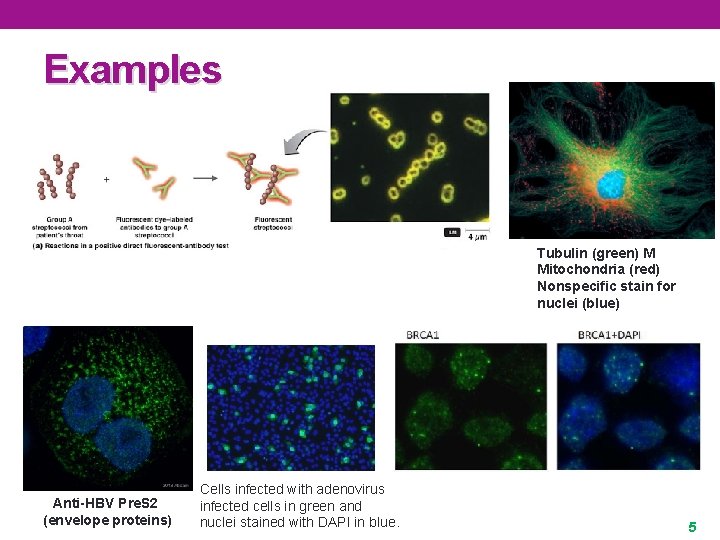
Examples Tubulin (green) M Mitochondria (red) Nonspecific stain for nuclei (blue) Anti-HBV Pre. S 2 (envelope proteins) Cells infected with adenovirus infected cells in green and nuclei stained with DAPI in blue. 5

Fluorophores • Fluorophores are typically organic molecules with a ring structure • They absorb light energy over a range of wavelengths that is characteristic for that compound • This absorption of light causes an electron in the fluorescent compound to be raised to a higher energy level • The excited electron quickly decays to its ground state, emitting the excess energy as a photon of light, which has a longer wavelength and lower energy • This transition of energy is called fluorescence 6
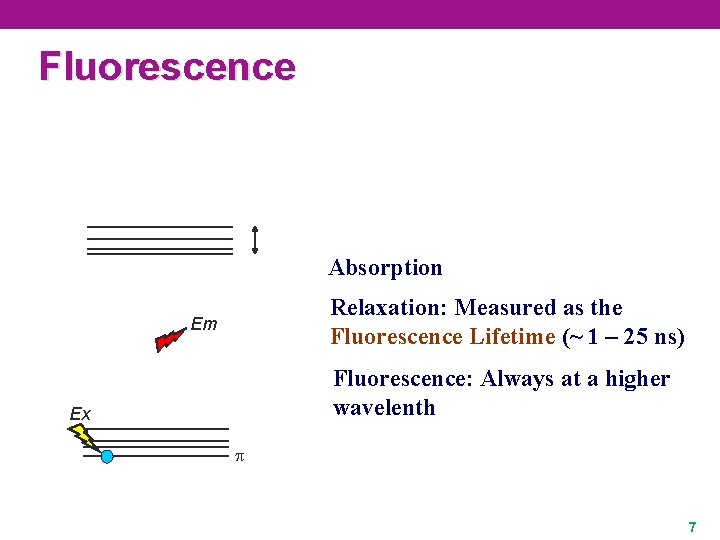
Fluorescence Absorption Relaxation: Measured as the Fluorescence Lifetime (~ 1 – 25 ns) Em Fluorescence: Always at a higher wavelenth Ex 7
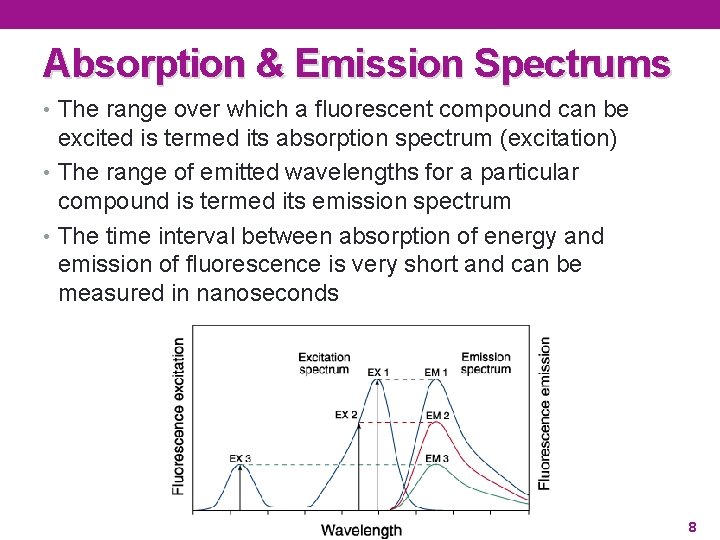
Absorption & Emission Spectrums • The range over which a fluorescent compound can be excited is termed its absorption spectrum (excitation) • The range of emitted wavelengths for a particular compound is termed its emission spectrum • The time interval between absorption of energy and emission of fluorescence is very short and can be measured in nanoseconds 8

Factors Affecting Fluorescence • Increase fluorescence • Structure • Aromatic groups • Rigidity • rigid structures have lower probability of collisions • Decrease fluorescence • Temperature increase • Heavy atoms in solvent ( Intersystem crossing) • Dissolved O 2 ( Intersystem crossing)
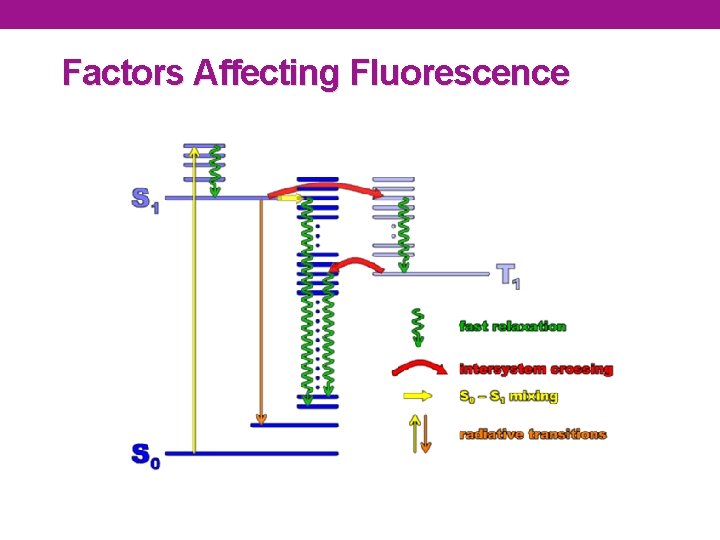
Factors Affecting Fluorescence

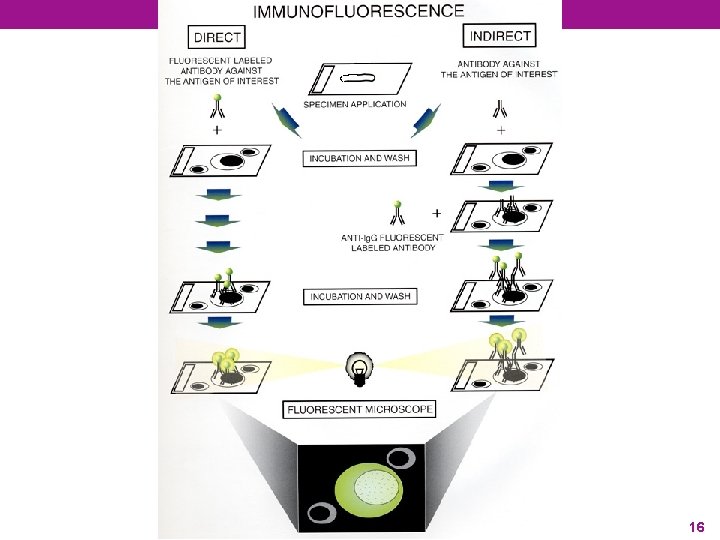
Types of Immunofluorescence • Fluorescent staining can be categorized as direct or indirect, depending on whether the original antibody has a fluorescent tag attached • Direct IF • Indirect IF 11

Direct Immunofluorescence • The antibody to the tissue antigen is conjugated with the fluorochrome and applied directly • The antibody used is usually monoclonal antibody • For example, to show the presence of virus antigens in tissue, fluorescence labeled antibodies are applied directly to the tissue • When viewed with the fluorescence microscope, the tissue will be brightly stained 12
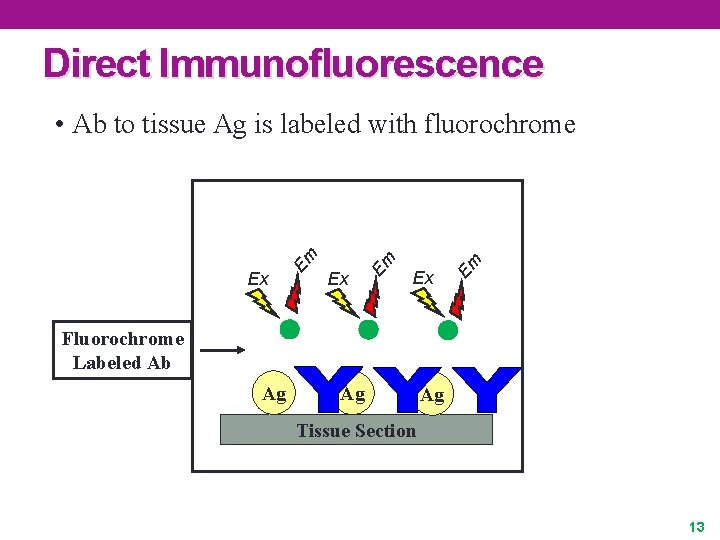
Direct Immunofluorescence Ex Em • Ab to tissue Ag is labeled with fluorochrome Fluorochrome Labeled Ab Ag Ag Ag Tissue Section 13

Indirect Immunofluorescence • In this double-layer technique, • the unlabeled antibody (primary Ab) is applied directly to the tissue • and visualized by treatment with a fluorochromeconjugated to anti-antibody (secondary antibody) • The secondary antibody is anti-species antibody which is raised against the species where the primary antibody was produced • It is a polyclonal antibody 14
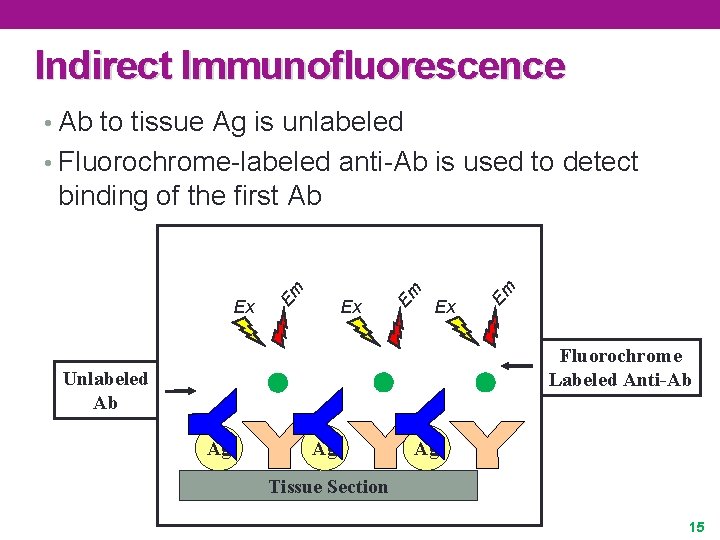
Indirect Immunofluorescence • Ab to tissue Ag is unlabeled • Fluorochrome-labeled anti-Ab is used to detect Ex Em binding of the first Ab Fluorochrome Labeled Anti-Ab Unlabeled Ab Ag Ag Ag Tissue Section 15
16

Direct & Indirect IF Direct Indirect • Fix specimen on slide • Add labeled antibody • Add primary antibody, specific for the desired antigen • Look for fluorescence specific for the desired antigen • Add secondary labeled antibody • Look for fluorescence 17

Advantages of Indirect IF • The fluorescence is brighter than with the direct test • since several fluorescent anti-immunoglobulins bind on to each of the antibody molecules present in the first layer • Even when many sera have to be screened for specific antibodies it is only necessary to purchase a single labeled reagent • The primary antibody does not need to be conjugated with a fluorochrome • Because the supply of primary antibody is often a limiting factor, indirect methods avoid the loss of antibody that usually occurs during the conjugation reaction 18

Controls • • • Reagent and tissue controls are necessary for the validation of immunofluorescence staining results Without their use, interpretation of staining would be haphazard and the results of doubtful value More specifically, controls determine if the staining protocols were: • followed correctly • whether day-to-day and worker-to-worker variations have occurred • and that reagents remain in good working order 19

Controls • In carrying out an immunofluorescence experiment one has to be confident that: • the reaction is specific and that the Ab is in fact binding selectively to the target Ag and not to other components of the cell or other closely related Ags • In addition if no fluorescence is observed with the probe does this mean that: • the Ag is not present • or it mean that there may be a problem with preparation or with the tissue itself • If the correct controls are included in the experiment we can, with high certainty, answer these questions 20

Positive and Negative Controls • Negative Tissue Controls: • Specimens serving as negative controls must be processed (fixed, embedded) identically to the unknown, but do not contain the target antigen • If a signal is detected then this suggests that a problem exists within your technique or protocol • Positive Tissue Controls: • Again, these controls must be processed identically to the specimen but contain the target antigen • If a signal is not detected then this suggests the problem exists within your technique, protocol or reagent 21

DETECTION OF SIGNAL 22

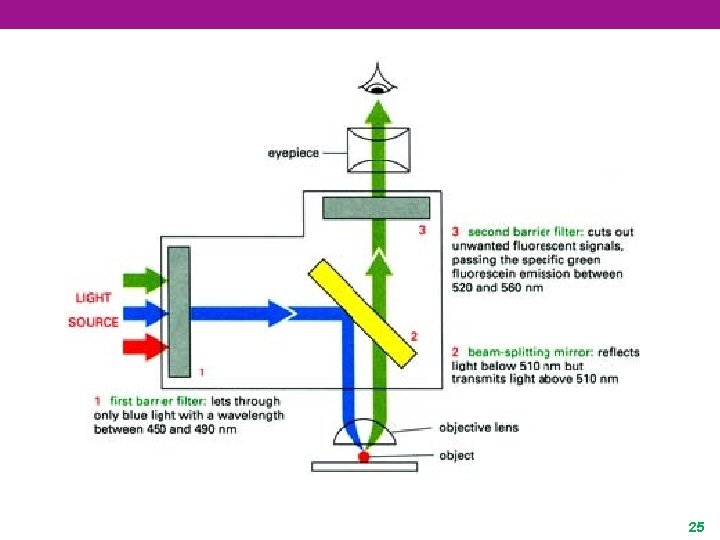
Fluorescence Instrument • Instrument for detection of Xenon Arc Lamp fluorescence consists of: • Light source • Xenon Arc Lamp or mercury vapor lamp • Laser • Wavelength selector • Excitation filter • Emission filter • Detector • Signal processor Emission filter 23
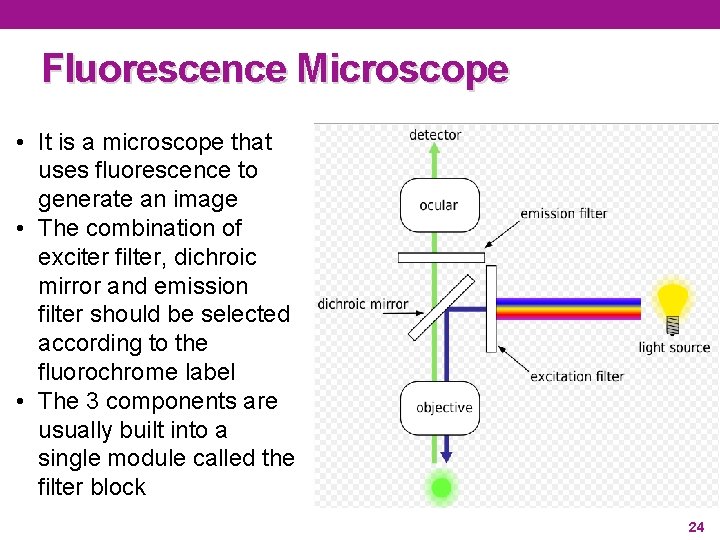
Fluorescence Microscope • It is a microscope that uses fluorescence to generate an image • The combination of exciter filter, dichroic mirror and emission filter should be selected according to the fluorochrome label • The 3 components are usually built into a single module called the filter block 24
25

Fluorescence Microscope 26

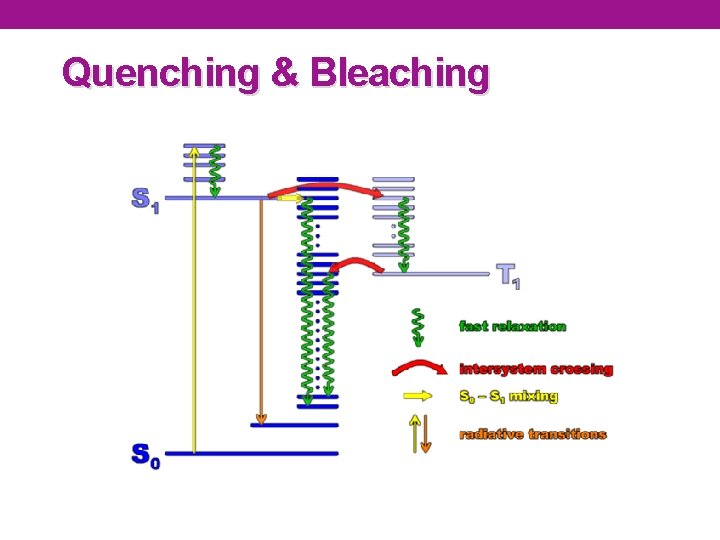
Quenching & Bleaching • Quenching is when excited molecules relax to ground states via nonradiative pathways avoiding fluorescence emission (vibration, collision, intersystem crossing) • Molecular oxygen quenches by increasing the probability of intersystem crossing • Photobleaching is defined as the irreversible destruction of an excited fluorophore
Quenching & Bleaching

Indirect Fluorescence Assay For Mumps Virus Ig. G Antibody 29

Introduction and Summary of Test Procedures • Mumps, an acute, contagious disease, is generally characterized clinically by parotitis • Invasion of the central nervous system, testes, ovaries, and other visceral organs can accompany the infection • The introduction of a mumps virus vaccine in 1967 has resulted in a decline in the incidence of mumps • But because of the vaccine's restricted use, mumps will remain a common worldwide problem 30

Introduction and Summary of Test Procedures • Laboratory confirmation of mumps infection is usually not required in those patients with characteristic parotitis • However, the two most common complications, meningoencephalitis and orchitis, can occur without the classic parotitis • In these cases, laboratory detection is necessary to confirm mumps infection 31

Principle of the Test • Fluorescent antibody assays use the indirect method of antibody detection and titer determination • Patient serum or plasma samples are applied to cultured cells containing inactivated viral antigens provided on wells on glass microscope slides • During a 30 minute incubation, antibody specific for mumps virus antigens forms an antigen/antibody complex with the mumps virus antigens in the infected cells 32

Principle of the Test • In a brief washing step, nonspecific antibody and other unreacted serum proteins are eliminated • Fluorescein-conjugated goat antihuman Ig. G is then applied to the wells of the glass slide • The anti-Ig. G conjugate combines with human Ig. G, if present, during a 30 minute incubation • After a brief wash to remove unreacted conjugate, the slides are viewed by fluorescence microscopy • A positive antibody reaction is denoted by bright green fluorescence at the antigen sites 33

Controls • Mumps Virus Ig. G Positive Control: • Each vial contains 0. 5 ml mumps virus Ig. G antibody positive human control • This component is a ready for use liquid at a 1: 10 working dilution • Mumps Virus Ig. G Negative Control: • Each vial contains 0. 5 ml mumps virus Ig. G antibody negative human control • This component is a ready for use liquid at a 1: 10 working dilution 34
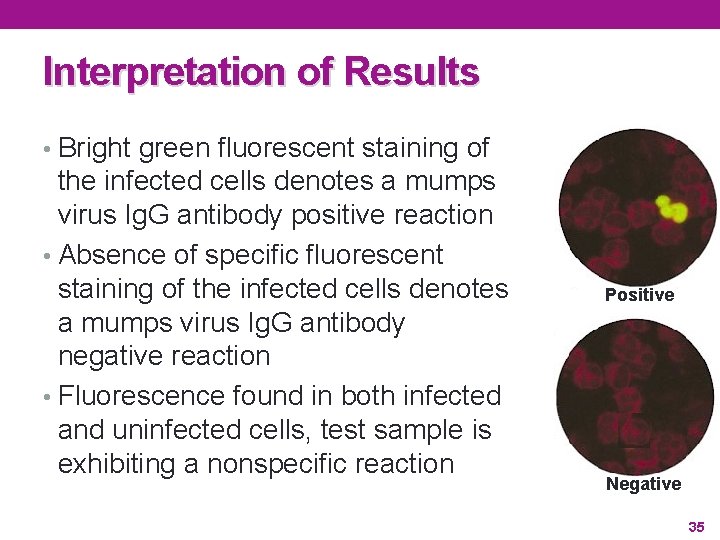
Interpretation of Results • Bright green fluorescent staining of the infected cells denotes a mumps virus Ig. G antibody positive reaction • Absence of specific fluorescent staining of the infected cells denotes a mumps virus Ig. G antibody negative reaction • Fluorescence found in both infected and uninfected cells, test sample is exhibiting a nonspecific reaction Positive Negative 35
- Slides: 35