Immunization Dr Bhavesh Patel Principal V P and

Immunization Dr. Bhavesh Patel Principal V. P. and R. P. T. P. Science College, Vallabh Vidyanagar Email- bhavesh 1968@rediffmail. com

Milestones in immunization u 3000 BC u Evidence of sniffing powdered small pox crust in Egypt u 2000 BC u Sniffing of small pox crust in China u 1500 BC u Turks introduce variolation u 1700 AD u. Introduction of variolation in England later in the US 2

Introduction of variolation The wife of the British Ambassador in Turkey, in March 1717 wrote, following the variolation of her son, to a friend in England: “The small pox, so fatal, so general amongst us, is entirely harmless here by the invention of ingrafting…. I am patriot enough to bring this invention into fashion in England. 3

Milestones in immunization u 1780 A. D. u Edward Jenner discovers small pox vaccine 4

Edward Jenner Discovery of small pox vaccine 5

Edward Jenner Among patients awaiting small pox vaccination 6

Modern era of the vaccine 1885 Rabies vaccine (Pasteur) 1920 s Diphtheria and Tetanus 1934 Pertussis 1955 Salk polio 7

Modern era of the vaccine 1960 s 1985 Mumps measles and rubella virus Sabin polio Haemoph ilus 1990 s Hepatitis and varicella 8
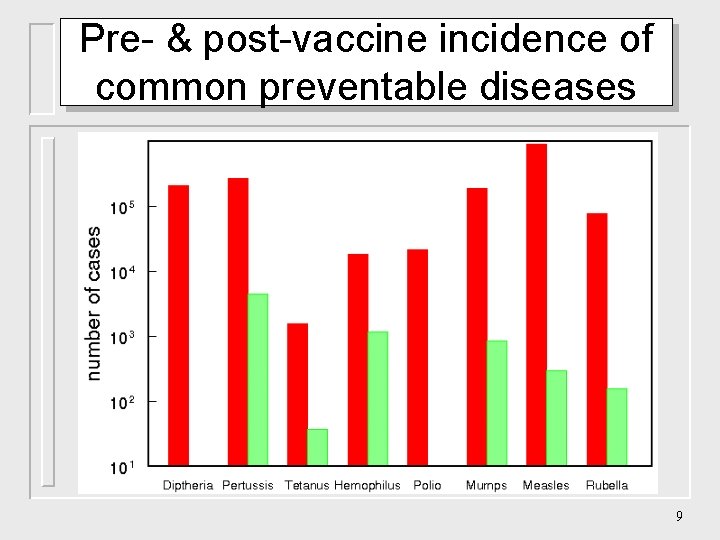
Pre- & post-vaccine incidence of common preventable diseases 9
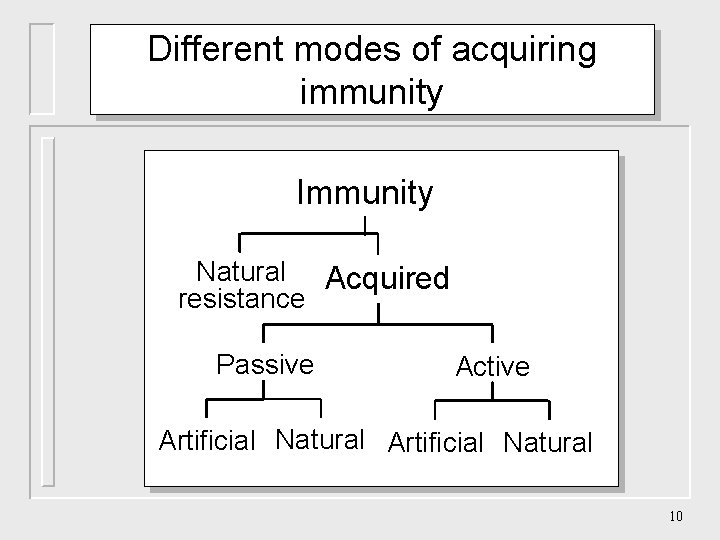
Different modes of acquiring immunity Immunity Natural Acquired resistance Passive Active Artificial Natural 10
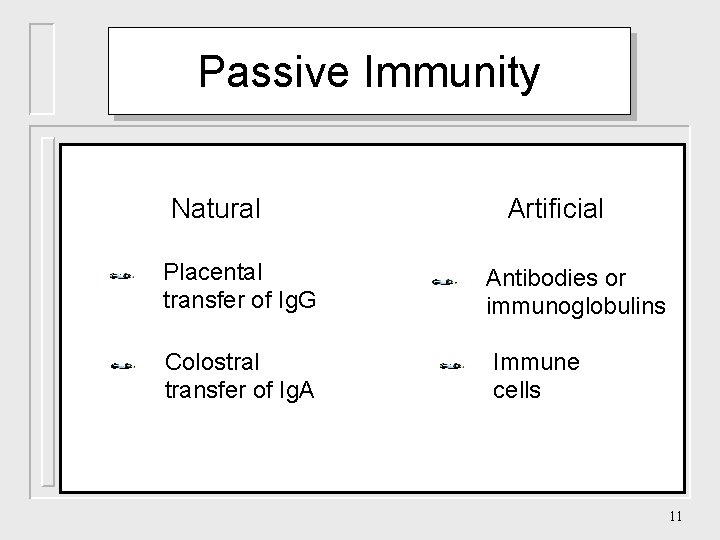
Passive Immunity Natural Placental transfer of Ig. G Colostral transfer of Ig. A Artificial Antibodies or immunoglobulins Immune cells 11

Passive Immunization disease antibody source indication diphtheria, tetanus human, horse prophylaxis, therapy vericella zoster human immunodeficiencies gangrene, botulism, snake bite, scorpion sting horse post-exposure rabies, human post-exposure hypogammaglobulinemia human prophylaxis 12

Advantages and Disadvantages of Passive Immunization Advantages immediate protection Disadvantages no long term protection serum sickness risk of hepatitis and Aids graft vs. host disease (cell graft only) 13
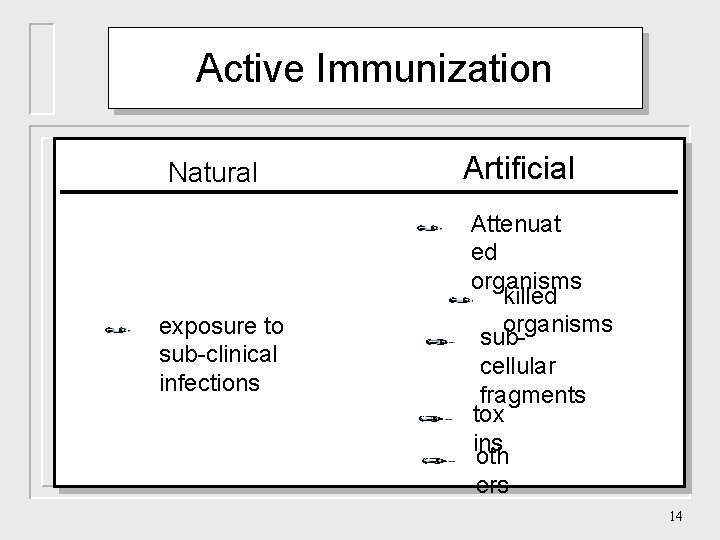
Active Immunization Natural exposure to sub-clinical infections Artificial Attenuat ed organisms killed organisms subcellular fragments tox ins oth ers 14

Live Attenuated Vaccines polio* not used in std. schedule measles, mumps & rubella Varicella zoster children with no history of chicken pox hepatitis A standard yellow fever 2006 Military and travelers Influenza selected age group (5 -49) tuberculosis not used in this country 15
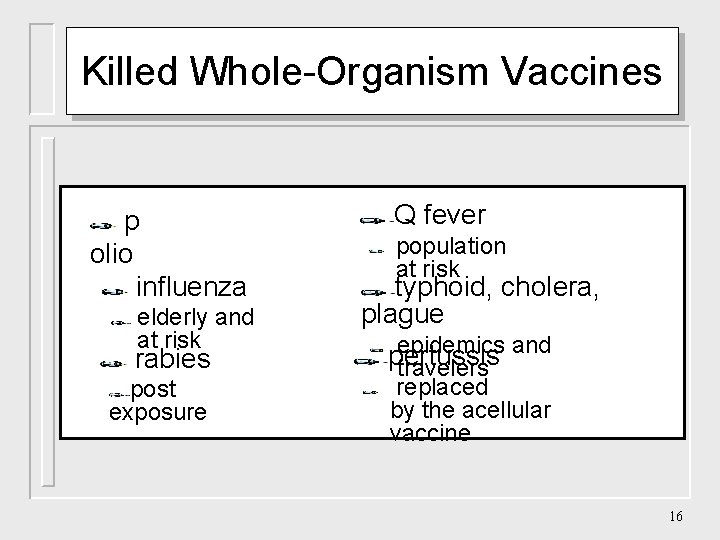
Killed Whole-Organism Vaccines p olio influenza elderly and at risk rabies post exposure Q fever population at risk typhoid, cholera, plague epidemics and pertussis travelers replaced by the acellular vaccine 16

Microbial Fragment Vaccines Bordetella. Pertussis virulence factor Haemophilus protein influenzae B protein conjugated Streptococcus polysaccharide pneumoniae Polysaccharide mixture Neisseria meningitidis polysaccharide 17

Microbial Fragment Vaccines Clostridium tetani (tetanus) inactivated toxin (toxoid) Corynebacterium diphtheriae inactivated toxin (toxoid) Vibrio cholerae toxin subunits Hepatitis B virus cloned in yeast 18
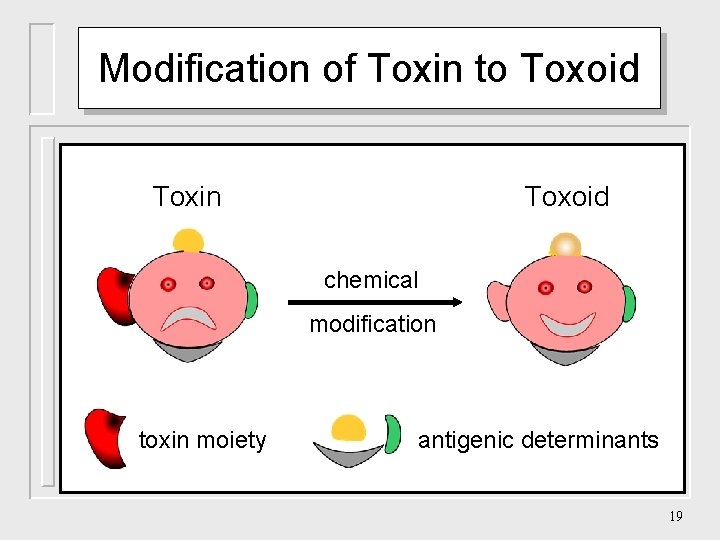
Modification of Toxin to Toxoid Toxin Toxoid chemical modification toxin moiety antigenic determinants 19

Future Vaccines anti-Idiotype Vaccine DNA Immuno-dominant peptide 20
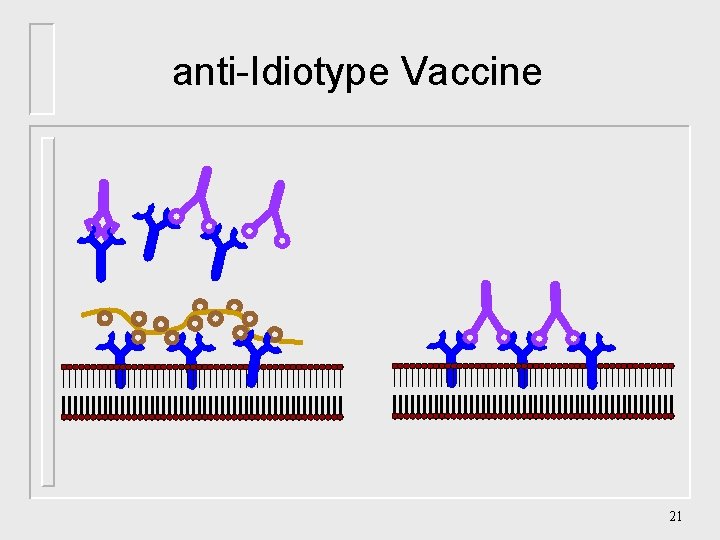
anti-Idiotype Vaccine 21
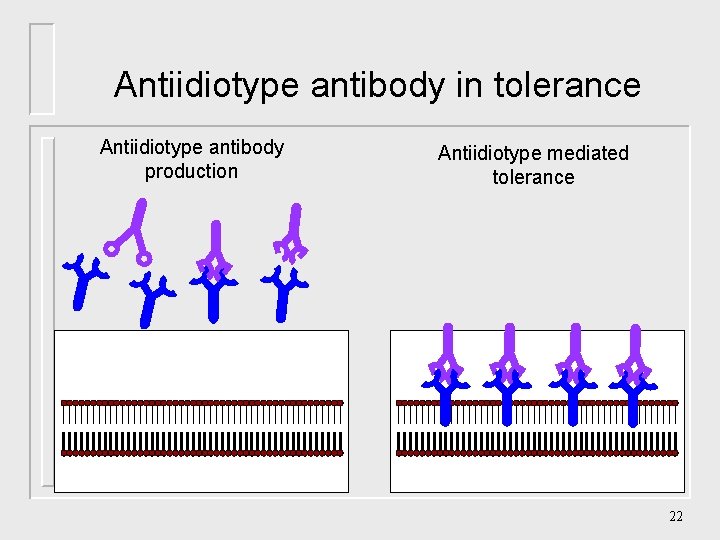
Antiidiotype antibody in tolerance Antiidiotype antibody production Antiidiotype mediated tolerance 22
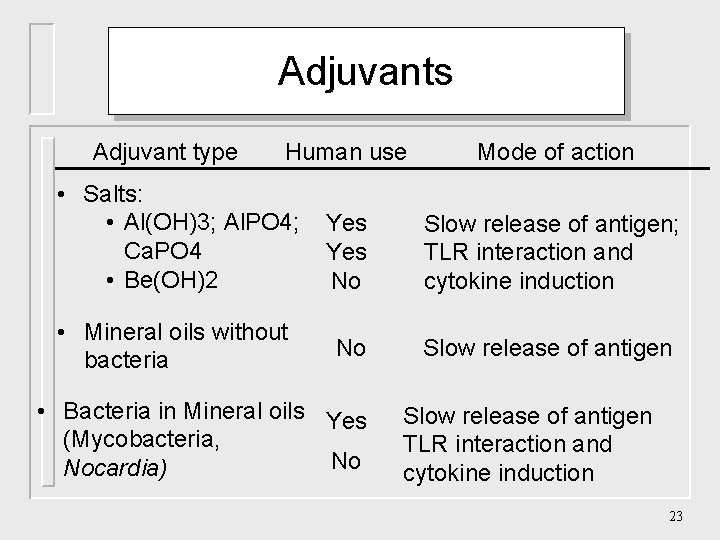
Adjuvants Adjuvant type Human use • Salts: • Al(OH)3; Al. PO 4; Yes Ca. PO 4 Yes • Be(OH)2 No • Mineral oils without bacteria No • Bacteria in Mineral oils Yes (Mycobacteria, No Nocardia) Mode of action Slow release of antigen; TLR interaction and cytokine induction Slow release of antigen TLR interaction and cytokine induction 23
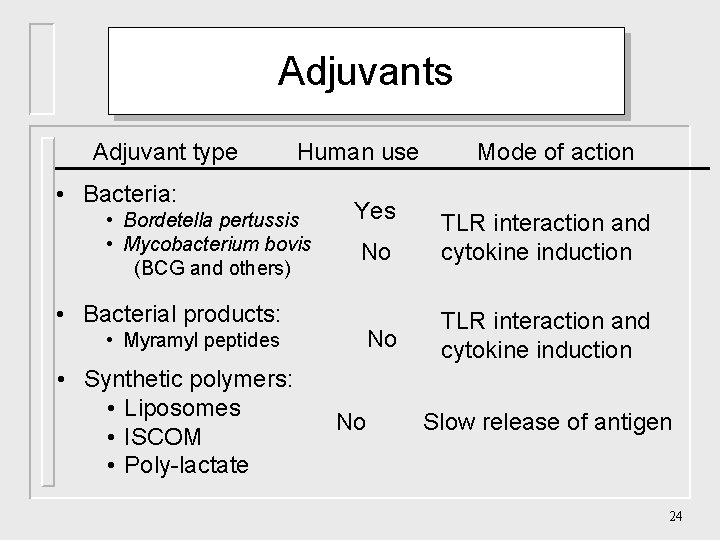
Adjuvants Adjuvant type Human use • Bacteria: • Bordetella pertussis • Mycobacterium bovis (BCG and others) Yes No TLR interaction and cytokine induction • Bacterial products: • Myramyl peptides • Synthetic polymers: • Liposomes • ISCOM • Poly-lactate Mode of action No Slow release of antigen 24
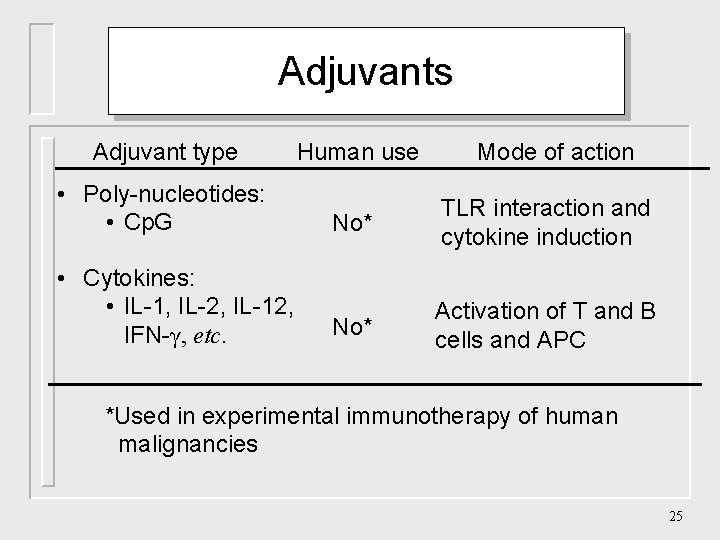
Adjuvants Adjuvant type Human use Mode of action • Poly-nucleotides: • Cp. G No* TLR interaction and cytokine induction No* Activation of T and B cells and APC • Cytokines: • IL-1, IL-2, IL-12, IFN-γ, etc. *Used in experimental immunotherapy of human malignancies 25
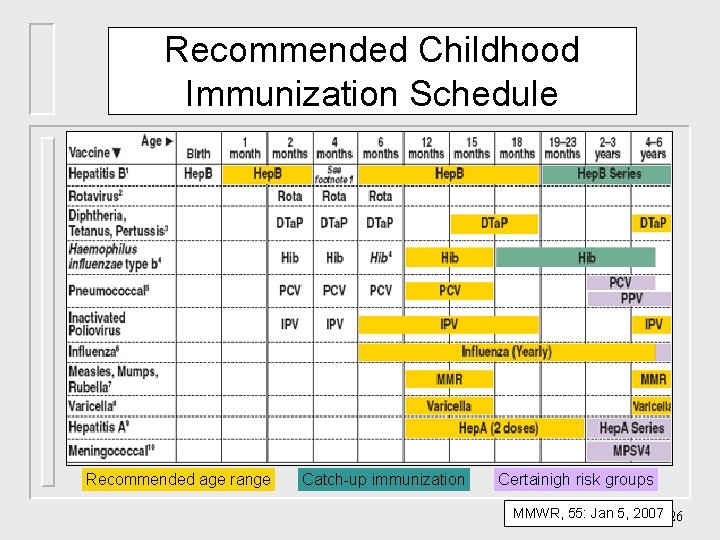
Recommended Childhood Immunization Schedule Recommended age range Catch-up immunization Certainigh risk groups MMWR, 55: Jan 5, 2007 26
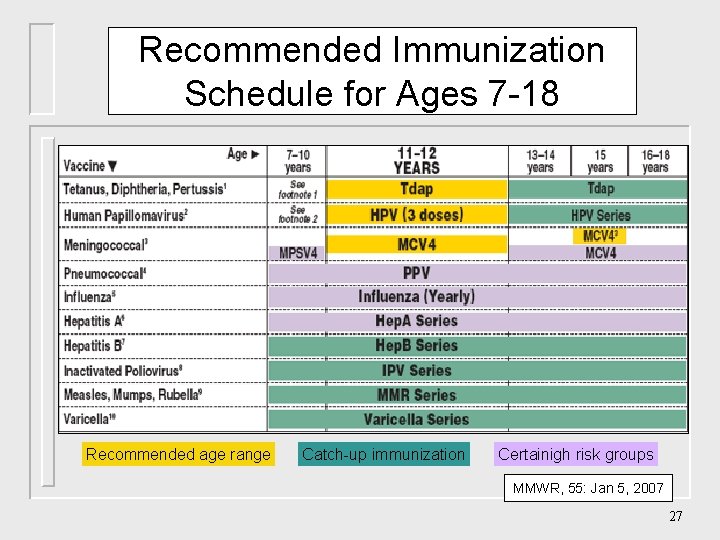
Recommended Immunization Schedule for Ages 7 -18 Recommended age range Catch-up immunization Certainigh risk groups MMWR, 55: Jan 5, 2007 27
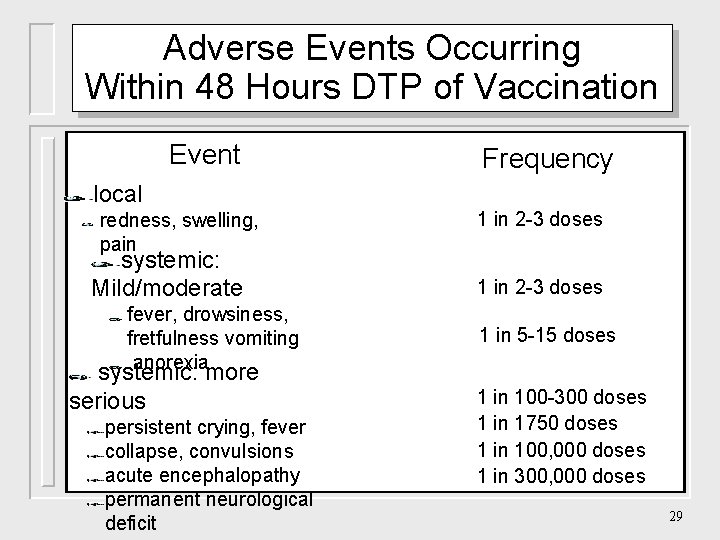
Adverse Events Occurring Within 48 Hours DTP of Vaccination Event Frequency local redness, swelling, pain systemic: Mild/moderate fever, drowsiness, fretfulness vomiting anorexia systemic: more serious persistent crying, fever collapse, convulsions acute encephalopathy permanent neurological deficit 1 in 2 -3 doses 1 in 5 -15 doses 1 in 100 -300 doses 1 in 1750 doses 1 in 100, 000 doses 1 in 300, 000 doses 29
- Slides: 28