Immunisation against 21 0 2 meningococcal B disease

Immunisation against 21 0 2 meningococcal B disease for y l u J infants aged from two months n w a r d h t i An update for healthcare professionals September 2015 v 2 W PHE Publications gateway number: 2015165

Key Messages • Meningococcal disease is caused by invasive infection with the bacterium Neisseria meningitides also known as meningococcus • Although there are 12 capsular groups of meningococcus, group B accounts for approximately 80% of all laboratory confirmed cases reported to Public Health England • • 2 1 2 0 2 y l u Invasive meningococcal disease most. Jcommonly presents as meningitis or septicaemia and affects children under 2 years, particularly infants aged 5 n months and older adolescents w a r Routinely immunising infants against meningococcal B disease reduces the d thinvasive meningococcal disease in the UK by burden and severity of i protecting those at Wincreased risk Immunisation against meningococcal B disease for infants aged from two months

Meningococcal B programme Routine cohort: • 1 2 Starting on the 1 September 2015 all infants born on or after the 1 July 2015 will be eligible for the meningococcal B vaccine at 2, 4 and 12 months 0 2 y l u Catch-up cohort: • J n A catch-up programme will also commence on the 1 September 2015 for infants born from 1 May to 30 June 2015 w a r d th The JCVI agreed that these infants would be offered the meningococcal B vaccine when they attend for their remaining primary immunisation appointments from 1 September 2015 i W 3 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… Cohort Dates of birth Recommended immunisation schedule Those born on or after 2, 4 and 12 months (2+1) 1 July 2015 Those born on or after If second routine primary 1 May to the 30 th June immunisation appointment due on 2015 or after 1 st September then follow this schedule: Routine 1 2 0 2 y l u Catch-up w a W 4 Those born before 1 May 2015 are NOT eligible to receive the vaccine r d th i J n 3, 4 and 12 months (2+1) If third routine primary immunisation appointment due on or after 1 st September then follow this schedule: 4 and 12 months (1+1) Immunisation against meningococcal B disease for infants aged from two months

Aim of resource • To raise awareness of invasive meningococcal disease (IMD) epidemiology and the impact of IMD on infants and adolescents • To support and educate healthcare professionals involved in discussing immunisation against meningococcal B disease with parents • 5 1 2 0 2 To promote the uptake of meningococcal l. Byvaccine through increasing u awareness in healthcare professionals involved in immunisation J n w a r d h it W Immunisation against meningococcal B disease for infants aged from two months

Learning outcomes After completing this training, healthcare professionals will be able to: • • • 6 1 2 Describe the aetiology and epidemiology of meningococcal capsular group B disease 0 2 y Be aware of the most common types of meningococci in the UK and their l u relationship in causing invasive meningococcal disease J n Advise and inform parents of thew importance of introducing a meningococcal B a based information vaccine in England, providingrevidence d h Understand the healthcare it professionals role in supporting the implementation of the meningococcal WB immunisation programme Identify sources of additional information and resources Immunisation against meningococcal B disease for infants aged from two months

Contents 1. 2. 3. 4. 5. 7 1 2 Why routinely immunise infants at 2 months of age? 0 2 Immunisation against meningococcal B disease and the use of Bexsero® y l u The role of the health care professionals J n Resources w a r d h it W What is meningococcal B disease Immunisation against meningococcal B disease for infants aged from two months

1 2 0 What is meningococcal B disease 2 y l u J n w a r d h it W 8 Immunisation against meningococcal B disease for infants aged from two months

What is meningococcal B disease? • Meningococcal disease occurs as a result of an invasive bacterial infection caused by Neisseria meningitidis, which is commonly known as the meningococcus 1 2 • • • 9 0 2 There are 12 known meningococcal groups, each possessing a distinct y l outer polysaccharide (sugar) capsule. In England, capsular groups B, W and u Y are responsible for nearly all meningococcal infections across all age J groups n w a conjugate vaccination introduced in 1999 Routine meningococcal C (Men. C) r d has nearly eliminated invasive Men. C disease in England h t i Men. B now accounts for ~80% of all laboratory confirmed IMD cases in W England >90% of IMD cases in children and adolescents Immunisation against meningococcal B disease for infants aged from two months
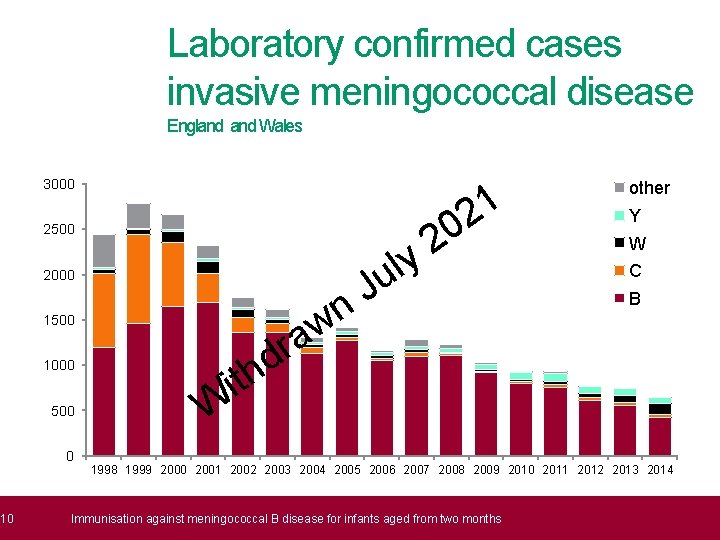
10 Laboratory confirmed cases invasive meningococcal disease Outline England Wales 3000 1 2 0 2 y l u 2500 2000 w a 1500 1000 500 0 r d th J n other Y W C B i W 1998 1999 2000 2001 2002 2003 2004 2005 2006 2007 2008 2009 2010 2011 2012 2013 2014 Immunisation against meningococcal B disease for infants aged from two months
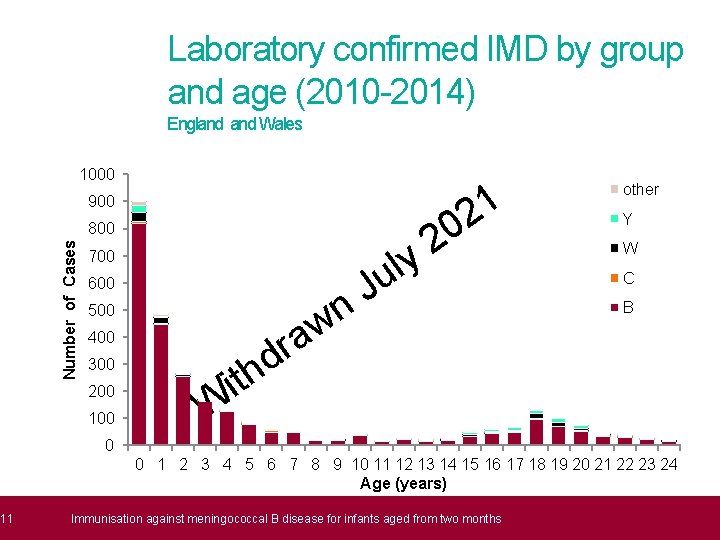
11 Laboratory confirmed IMD by group and age (2010 -2014) England Wales 1000 1 2 900 0 2 y l u Number of Cases 800 700 600 500 w a r d th 400 300 200 100 J n other Y W C B i W 0 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Age (years) Immunisation against meningococcal B disease for infants aged from two months
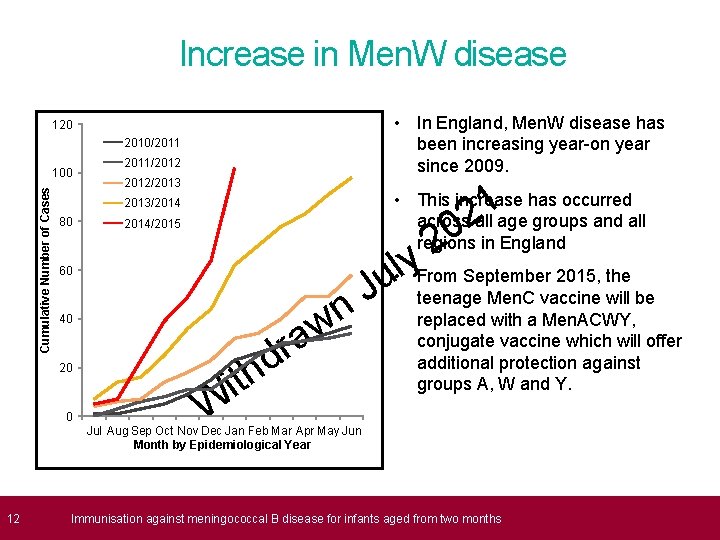
Increase in Men. W disease • In England, Men. W disease has been increasing year-on year since 2009. 120 2010/2011 Cumulative Number of Cases 100 2011/2012/2013 1 2 • This increase has occurred across all age groups and all regions in England 2013/2014 80 0 2 y l • u From September 2015, the 2014/2015 60 w a 40 20 0 r d th J n i W teenage Men. C vaccine will be replaced with a Men. ACWY, conjugate vaccine which will offer additional protection against groups A, W and Y. Jul Aug Sep Oct Nov Dec Jan Feb Mar Apr May Jun Month by Epidemiological Year 12 Immunisation against meningococcal B disease for infants aged from two months

Clinical presentation of IMD Babies and toddlers Children and young adults Fever with poor peripheral perfusion Poor feeding, refusing food or vomiting Vomiting w a Neck stiffness r d th W i 0 2 y l u J n Tense, bulging fontanelle and photophobia Fretful, unusual cry, moaning or rapid breathing 1 2 Severe headache and photophobia Confusion and irritability Neck stiffness and muscle pain Pale blotchy complexion &/or non blanching rash that does not fade when a glass is rolled over it Pale blotchy complexion &/or non blanching rash that does not fade when a Neck stiffness & muscle pain glass is rolled over it Drowsy & loss of consciousness Symptoms can appear in any order, some may not appear at all. Immunisation against meningococcal B disease for infants aged from two months

The meningococcal rash • A distinctive red rash can appear anywhere on the body • The rash is formed of tiny “pinpricks” also known as petechiae and appears red in colour. The rash may later develop into purple bruising of the skin • The meningococcal rash can be distinguished from other rashes by pressing a glass tumbler against it • • 14 1 2 0 2 y l u w a J n r d hnot fade when a A meningococcal rash will t i glass tumbler is rolled over it W A febrile illness and rash that does not fade is a sign of meningococcal septicaemia The ‘tumbler’ test picture courtesy of Meningitis Research Foundation http: //www. meningitis. org/symptoms Immunisation against meningococcal B disease for infants aged from two months

Transmission, infectivity, incubation and carriage • • 1 2 Transmission is through person to person spread from respiratory aerosols, droplets or by direct close contact with respiratory secretions of someone who is carrying the bacteria 0 2 y l Infectivity of meningococcal is relatively low and requires prolonged close u contact, for example, those living in the same household or through direct J n contact with nose and respiratory secretions such as intimate “wet” kissing w a 2 to 7 days with the onset of disease ranging Incubation period ranges from r d from severe with overwhelming features to insidious mild prodromal h it symptoms W Carriage in the nose and throat (without any signs or symptoms) is uncommon in infants and young children but increases to 25% in adolescents 15 Immunisation against meningococcal B disease for infants aged from two months

Potential complications of meningococcal disease • • 1 2 Meningococcal disease is associated with significant case-fatality, ranging from around 5% in infants and young children to 25% in older adults. 0 2 Around a quarter of survivors of meningococcal disease will suffer serious y l long-term complications after recoveringu from the infection J Complications can vary in severity n and can either be temporary or permanent. w The more severe the disease, athe greater the risk of complications r d Complications can include h t i • Loss of hearing, loss of vision, loss of memory and/or concentration, W and balance, epilepsy, cerebral palsy, limb difficulties in coordination amputations and may result in death 16 Immunisation against meningococcal B disease for infants aged from two months

1 2 Why routinely immunise infants at 2 0 2 y months of age l u J n w a r d h it W 17 Immunisation against meningococcal B disease for infants aged from two months

Why immunise infants from 2 months? • • • 18 1 2 Meningococcal disease can affect all age groups but the highest rates of disease are highest in the year of life 0 2 Cases of invasive meningococcal disease increase from birth and peak at y l around 5 months of age before decliningugradually over subsequent months J In considering the epidemiological n and economic evidence as well as the w vaccine safety and efficacy, the JCVI decided to prioritise young infants a r aim of providing optimal protection as early from 2 months of age withd the h peak increase in disease as possible and before itthe W Immunisation against meningococcal B disease for infants aged from two months
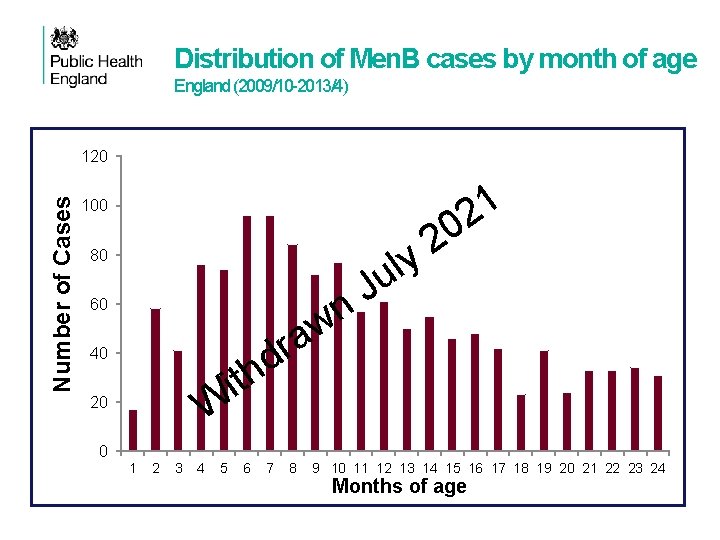
Distribution of Men. B cases by month of age England (2009/10 -2013/4) Number of Cases 120 1 2 100 0 2 y l u 80 60 w a 40 r d th J n i W 20 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 Changes to Men. C conjugate vaccine schedule Months of age

1 2 Immunisation against meningococcal 0 2 y B disease l u J n w a -The use of Bexsero® vaccine r d h it W 20 Immunisation against meningococcal B disease for infants aged from two months

The recommended vaccine Brand name: Bexsero® 1 2 Multi-component inactivated vaccine marketed by Glaxo. Smith. Kline • • • 21 0 2 Available through General Practitioner (GP)ly services from 1 September 2015 u J Routinely recommended for infants at 2 months of age as part of the primary n immunisation schedule at 2, 4 and 12 months w a 4 months of age as part of the primary r “Catch-Up” for infants at 3 and d immunisation scheduleth i Schedule and interval W dependant on child’s age on 1 September 2015 Licensed for use from 2 months of age Immunisation against meningococcal B disease for infants aged from two months

The recommended vaccine: Bexsero® • Bexsero® has been shown to be immunogenic in infants and toddlers • Because the incidence of meningococcal disease is so low, there have been no clinical trials to demonstrate vaccine effectiveness against invasive disease • • • 22 1 2 0 2 ly In laboratory tests, antibodies induced byuvaccination have been shown to kill J at least 73 -88% of Men. B strains causing meningococcal disease in England n The UK is the first country in thew world to introduce Bexsero® into the a national infant immunisationrprogramme d h The UK will, therefore, itbe the first country to evaluate vaccine effectiveness against meningococcal W disease at a population level Immunisation against meningococcal B disease for infants aged from two months

The recommended vaccine: Bexsero® • • • Bexsero® is the recommended vaccine for the routine infant immunisation programme and is the only market authorised meningococcal B vaccine in the UK Bexsero® will be centrally supplied through Imm. Form in packs of 10 It is important immunisers familiarise themselves with the vaccine and its product information to avoid administration errors 0 2 y l u J n i W 23 r d th w a 1 2 Image courtesy of Glaxo. Smith. Kline (GSK) Please note: Bexsero® is now owned and supplied by GSK. Initial stocks of Bexsero® will be supplied in Novartis packaging until 2016 Immunisation against meningococcal B disease for infants aged from two months

Composition of Bexsero® Composition 1. Recombinant Neisseria meningitidis group B NHBA fusion protein 2. Recombinant Neisseria meningitidis group B Nad. A protein 3. Recombinant Neisseria meningitidis group B f. Hbp fusion protein 4. Outer membrane vesicles (OMV) from Neisseria meningitidis group B strain NZ 98/254 measured as amount of total protein containing the Por. A w a r d th 1 2 0 2 y l u J n i Excipients W Sodium chloride, Histidine Sucrose, Water for injections 24 Immunisation against meningococcal B disease for infants aged from two months Image courtesy of Glaxo. Smith. Kline

How is Bexsero® administered ? • Bexsero® is a newly licensed vaccine that is subject to additional monitoring under the black triangle labelling scheme (MHRA) • Recommended that Bexsero® be administered via intramuscular injection (IM) ideally on its own in the infants left thigh (antereolataral aspect) so that any local reactions can be accurately monitored • For older infants aged 12 months, Bexsero® should be administered via IM injection (IM) ideally on its own in the child's left thigh • • 1 2 0 2 y l u J n w ais given should be noted in the infants The site at which each vaccine r d health records h t i Where it is not practically possible to administer the vaccine on its own i. e. W at 12 months, other vaccines can be administered in the left thigh at the same time rather than delay immunisation 25 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • • 26 Healthcare professionals are reminded that some infants may receive additional vaccines as part of a selective immunisation programme at around 12 months of age 1 2 0 2 Selective vaccines can include Hepatitis B and BCG y l u not be administered in the same It is important to note that vaccines should J limb as the BCG vaccine for a period of 3 months n w Healthcare professionals should discuss any recent immunisations with the a r appointment parent at the 12 month booster d ith W Immunisation against meningococcal B disease for infants aged from two months

Administration and Supply • The vaccines are supplied in packs containing 10 pre-filled syringes each with a volume of 0. 5 mls of suspension per syringe • During storage, the contents of the syringe may settle with off-white deposits being noticeable • • • 1 2 0 2 y l Before use, the pre-filled syringe must be shaken well forming an u homogenous suspension that should be administered immediately J n The vaccine should not be administered where there are variations in w physical appearance (i. e. notra an homogenous suspension) or signs of d foreign particulate are observed after shaking h t i Bexsero® has a shelf life of two years when stored in its original W packaging in a refrigerator at the recommended temperatures of +2°C and +8°C 27 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • • Initial vaccine supplies will have a short-shelf life (April 2016). It is important not to over-order vaccines and only order what is needed for the 2 -4 week period 1 2 0 2 y l u Healthcare professionals are encouraged to familiarise themselves with Public Health England’s protocol for ordering, storing and handling of vaccines to ensure vaccines are stored and monitored as per national recommendations w a r d th J n i W 28 Immunisation against meningococcal B disease for infants aged from two months

Administration of Bexsero® should only be administered: 1 2 • Against a prescription written manually or electronically by a registered medical practitioner or other authorised prescriber • Against a Patient Specific Direction • Against a Patient Group Direction w a r d th 0 2 y l u J n i W 29 Immunisation against meningococcal B disease for infants aged from two months

Contraindications Bexsero® should not be administered to those who have had: 1. 2. • • 30 1 2 A confirmed anaphylaxis to a previous dose of the vaccine OR 0 2 y l There are very few infants who cannot receive meningococcal u J vaccines n Where there is doubt, appropriate advice should be sought rather than w withholding immunisation ra d h it W A confirmed anaphylaxis to any constituent or excipient of the vaccine Immunisation against meningococcal B disease for infants aged from two months

Precautions • • • 31 Minor illnesses without fever or systemic upset are not valid reasons to postpone immunisation 1 2 0 2 Meningococcal vaccines may be given to pregnant women when clinically y l indicated. There is no evidence of risk from vaccinating pregnant women or u J those who are breast-feeding with inactivated virus or bacterial vaccines or n toxoids w a r Premature infants d h t infants have their immunisations at the It is important that premature i appropriate chronological W age, according to the schedule Pregnancy and breast-feeding Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • Immunosuppression and HIV infection 1 2 Individuals with immunosuppression and human immunodeficiency virus (HIV) infection (regardless of CD 4 count) should be given meningococcal vaccines in accordance with the routine schedule 0 2 y l u w a r d th J n i W 32 Immunisation against meningococcal B disease for infants aged from two months

Possible adverse reactions (up to 10 years of age) 1 2 Most commonly reported • Less commonly reported • r d th w a J n Fever (>40ºC), eczema, urticaria (hives; itching), Kawasaki syndrome, seizures and pallor i W 33 0 2 y l u Fever (>38ºC), tenderness at the injection site (including severe tenderness), rash, swelling or induration at the injection site, irritability, change in feeding/eating, sleepiness and unusual crying Immunisation against meningococcal B disease for infants aged from two months

Bexsero® and Fever • • 34 The most common adverse reaction observed in infants and toddlers was fever (>38ºC) when Bexsero® was administered with the other routine infant vaccines 1 2 0 2 y l u In one clinical trial 1, fever (>38ºC) was reported in 51 -62% of infants receiving Bexsero® and routine vaccines administered together, although high fever (>39ºC) was less common (6 -12%) J Overall, fever (>38ºC) after any immunisation was reported in 76% of n infants receiving Bexsero® andw routine vaccines together, compared to a immunisations alone 51% in infants receiving routine r d h t only 6 out of the 1885 recruited infants In the same study, however, i attended hospital. W because of fever within 2 days after immunisation with Bexsero® Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • In a subsequent study, 70% of infants receiving Bexsero® had fever (>38. 5ºC) at least once in the first 3 days after the primary dose • Fever was less common (39%) in infants receiving prophylactic paracetamol just before or at the time of immunisation followed by 2 further administrations at 4 -6 hourly intervals after immunisation • • 1 2 0 2 y l u Of note only ~5% of infants receiving paracetamol had fever >39ºC J n The frequency of medically-attended fever within 3 days of immunisation w was <2% for any immunisation visit, irrespective of whether Bexsero was a r administered alone or at the same time as other routine immunisations d h t i This study was important as it showed that responses to Bexsero® and W were not affected by administering paracetamol other routine immunisations 1 • at the time of immunisation 35 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • • • 36 In another vaccine study that did not include Bexsero®, 2 infants receiving three doses of paracetamol (at immunisation and 6 -8 hourly intervals) were half as likely to develop post immunisation fever (>38 o. C) including high fever (>39ºC) compared with infants receiving two doses (first dose 6 -8 hours after immunisation and another dose 6 -8 hours later) 1 2 0 2 y l u J n The greatest benefit in reducing post-immunisation fever appears to come from the paracetamol dose administered at the time of immunisation w arecommended because it does not prevent Prophylactic ibuprofen is not r post-immunisation fever d ith W Immunisation against meningococcal B disease for infants aged from two months

Guidance on the use of prophylactic paracetamol • Given that fever has been a very common adverse reaction in trials, the 1 2 at the time of JCVI recommended the use of prophylactic paracetamol 0 immunisation with Bexsero® 2 lyinformed about the change in • Parents and health professionals need toube J advice regarding the use of prophylactic paracetamol and the reactogencity n of Bexsero® when administered with other routine vaccines w a • This is a change to previousradvice whereby the prophylactic use of d antipyretics was not recommended as there was some evidence that h t i vaccine immune responses in infants antipyretics could lower W • Parents will be advised to give 2. 5 ml (120 mg/5 ml) to their babies around the time of immunisation and two additional doses at 4 -6 hourly intervals 37 Immunisation against meningococcal B disease for infants aged from two months

Dosage and timing of infant Paracetamol suspension (120 mg/5 ml) for the routine immunisation programme at 2 and 4 months 1 2 Age of baby Dose 1 Dose 2 2 months/4 months One 2. 5 ml as soon as possible after vaccination One 2. 5 ml 4 -6 hours after 1 st dose w a 0 2 y l u J n Dose 3 One 2. 5 ml 4 -6 hours after 2 nd dose r d If baby still febrile after the first three doses of paracetamol but is h t i can continue giving paracetamol at otherwise well, parents W recommended intervals up to 48 hours post-vaccination. Do not exceed four doses in a day. If any concern at all speak to GP or call NHS 111. 38 Immunisation against meningococcal B disease for infants aged from two months
Ordering paracetamol and syringes • • • Until the programme becomes established, GP practices will be able to order single 5 ml sachets of liquid paracetamol (120 mg/5 ml) and accompanying 2. 5 ml syringes via Imm. Form 1 2 0 2 Sachets of liquid paracetamol should only be y offered to those attending for their first Menu. Bl vaccine and only to parents who do not J have n timely access to over-the-counter medicines w Parents should be instructed tora buy some d infant strength liquid paracetamol to complete h t i the two remaining recommended doses of Wand in preparation for paracetamol at home the child’s second primary Men B vaccine 39 Immunisation against meningococcal B disease for infants aged from two months

Should parents be worried about fever after vaccination? 40 • Fever after vaccination with or without Bexsero® is common and nearly always <39 o. C • Fever is a normal and expected response of the immune system against the vaccine antigens and not harmful • Parents are often concerned about the risk of febrile seizures or “fever fits” • Parents should be reassured that febrile seizures generally occur in infants from 6 months to 5 years of age and are very uncommon in younger age groups • It is important that parents are reassured and are advised of the importance of administering prophylactic paracetamol to reduce the risk and intensity of post-immunisation fever 1 2 0 2 y l u w a r d th J n i W Immunisation against meningococcal B disease for infants aged from two months

Paracetamol recommendation • • • The Commission on Human Medicines (CHM) has been consulted regarding the licencing restriction on Pharmacy (P) and General Sales List (GSL) paracetamol products 1 2 0 2 y l u The current licensure advises consulting a GP or pharmacist if more than 2 doses are required for a 2 month old infant post-immunisation to ensure early diagnosis of systemic bacterial infection J The CHM supported PHE’s recommendations for 3 doses of paracetamol n post-immunisation with Men. B w a r The CHM also supportedd use of paracetamol (every 4 -6 hours) for up to 48 hours after immunisations ith if needed W is based on the likelihood that fever is due to This recommendation immunisation 41 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • • This recommendation does not extend to fever at any other time and, if the infant is unwell, parents should trust their instincts and not delay seeking medical attention 1 2 0 2 y l u It is hoped that infant paracetamol suspension manufacturers will update product packaging and literature in due course J n “Healthcare professionals are reminded that in some circumstances the recommendations regarding vaccines given in the Green Book chapters may differ from those in the Summary of Product Characteristics (SPC). When this occurs, the recommendations in the Green Book are based on current expert advice received from the JCVI and should be followed” w a r d th i W 42 Immunisation against meningococcal B disease for infants aged from two months

Do nurses need a PGD to supply or administer paracetamol • • 1 2 A PGD is not a legal requirement for the supply or administration of overthe-counter medicines and is therefore not required by nurses 0 2 y To enable nursing colleagues to practice in accordance with the NMC l u. PHE will make available a Homely Standards for Medicines Management, J Remedy Protocol n w Homely Remedy Protocols r are a not prescriptions but protocols that enable the supply and administration of general sales list (GSL) and pharmacy only d h (P) listed medicines in itcommunity settings W a written instruction that has been developed and The protocol includes agreed upon in consultation with relevant qualified professionals (medical practitioner/nurse/pharmacist) 43 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… • The protocol will clarify what medicinal product may be administered, its indication for administration and its dose and frequency • Nursing colleagues may wish to familiarise themselves with the Nursing and Midwifery Councils (NMC) Standards for Medicines Management 1 2 0 2 y l u w a r d th J n i W 44 Immunisation against meningococcal B disease for infants aged from two months

Reporting suspected adverse reactions Yellow card scheme 45 1 2 • Bexsero® is a newly licensed vaccine and is subject to additional monitoring under the black triangle labelling scheme • All suspected adverse reactions should be reported to the MHRA using the yellow card scheme • Success depends on early, complete and accurate reporting • Report even if uncertain about whether vaccine caused condition • http: //mhra. gov. uk/yellowcard • See chapter 8 of Green Book for details 0 2 y l u w a r d th J n i W Immunisation against meningococcal B disease for infants aged from two months

The role of healthcare professionals 1 2 To provide clear, concise and accurate information to parents of infants from 2 months of age receiving Bexsero® as part of their routine primary immunisations 0 2 y l u J n Every effort should be made by healthcare professionals to maximise the uptake of the meningococcal B vaccine and to ensure that parents are fully informed about the importance of ensuring protection against meningococcal B disease for their child w a r d th i W 46 Immunisation against meningococcal B disease for infants aged from two months

Useful links Public Health England/ NHS England. Introduction of meningococcal B immunisation for infants https: //www. gov. uk/government/publications/menb -vaccination-introduction-from-1 -september-2015 1 2 0 2 y l u Public Health England. Immunisation against infectious diseases: meningococcal chapter 22. https: //www. gov. uk/government/publications/meningococcal-the-green-book -chapter-22 w a r d th J n Public Health England. JCVI recommendation to introduce new Men. B vaccine if available at a low price will protect young babies and children. [internet] https: //www. gov. uk/government/news/phe-welcomes-prospect-of-newmeningitis-b-vaccine i W Meningitis Research Foundation: http: //www. meningitis. org/ 47 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… Meningitis Now. https: //www. meningitisnow. org/ 1 2 0 2 Joint Committee on Vaccination and Immunisation. y l u https: //www. gov. uk/government/groups/joint-committee-on-vaccination-and. J immunisation n w a r d h it W NHS Choices. http: //www. nhs. uk/conditions/Meningitis/Pages/Introduction. aspx 48 Immunisation against meningococcal B disease for infants aged from two months

Key Message • • 49 Meningococcal disease is caused by invasive infection with the bacterium Neisseria meningitides also known as meningococcus 1 2 group B Although there are 12 capsular groups of meningococcus, 0 2 accounts for approximately 80% of all laboratory confirmed cases reported y l to Public Health England u J Invasive meningococcal disease most commonly presents as meningitis or n septicaemia and affects children under 2 years, particularly infants aged 5 w a months and older adolescents r d h Routinely immunising tinfants against meningococcal B disease reduces the i burden and severity of invasive meningococcal disease in the UK by W protecting those at increased risk Immunisation against meningococcal B disease for infants aged from two months

References 1. Clinical. Trials. gov (2014). Study assessing life effect of medications to prevent fever on Prevenar 13 (outcomes 18 -21). [internet] accessed on 29 April 2015. https: //clinicaltrials. gov/ct 2/show/results/NCT 01392378? term=paracetamol+vacci ne&rank=3§=X 01256#all 1 2 0 2 2. Nursing and Midwifery Council (2008) Standards for Medicines Management. y [internet] accessed 11 June 2015. http: //www. nmc. org. uk/standards/additionall u standards/standards-for-medicines-management/ J 3. Gossger N, Snape MD, Yu LM, Finn A, n Bona G, Esposito S, Principi N, Diezw Domingo J, Sokal E, Becker B, Kieninger D, Prymula R, Dull P, Ypma E, a r Toneatto D, Kimura A, Pollard AJ; European Men. B Vaccine Study Group ( 2012). d h of recombinant serogroup B meningococcal Immunogenicity and tolerability t i vaccine administered with or without routine infant vaccinations according to W different immunization schedules: a randomized controlled trial. JAMA. 2012 Feb 8; 307(6): 573 -82. doi: 10. 1001/jama. 2012. 85. 50 Immunisation against meningococcal B disease for infants aged from two months

Cont’d… 4. Prymula R 1, Esposito S, Zuccotti GV, Xie F, Toneatto D, Kohl I, Dull PM (2014). 1 2 A phase 2 randomized controlled trial of a multicomponent meningococcal serogroup B vaccine. Hum Vaccin Immunother. 2014; 10(7): 1993 -2004. doi: 10. 4161/hv. 28666 0 2 y 5. Novartis Vaccines (2015). Bexsero Meningococcal Group B vaccine for injection l in pre-filled syringe. [internet] accessed. J onu 11 June 2015. https: //www. medicines. org. uk/emc/medicine/28407/SPC/Bexsero+Meningococc n al+Group+B+vaccine+for+injection+in+pre-filled+syringe/ w a r 6. Centre for Disease Control d (CDC) 2012. Frequently Asked Questions about hthe Immune System. [internet] accessed 11 June Multiple Vaccinations iand t 2015. http: //www. cdc. gov/vaccinesafety/Vaccines/multiplevaccines. html W 51 Immunisation against meningococcal B disease for infants aged from two months
- Slides: 51