Immobilization on nicotinic Acetylcholine Receptor Subunits Ligandgated ion


Immobilization on nicotinic Acetylcholine Receptor Subunits 양 홍 석 울산대학교 의과대학 서울아산병원 마취통증의학과

Ligand-gated ion Channels • Nicotinic acetylcholine receptor (n. ACh. R) muscle type neuronal type • 5 -hydroxytryptopane type 3 receptor • -aminobutylic acid type (GABAA) • -aminobutylic acid type (GABAC) • Glycine receptor • Invertebrate Glutamate and Histidine receptor

Nicotinic Acetylcholine Receptor Subunits in Skeletal Muscle • Pentameric membrane protein containing four kinds of subunit Type - embrionic : , , , adult : , , , • Cationic selective, ligand-gated ion channels • Fast excitatory synaptic transmission
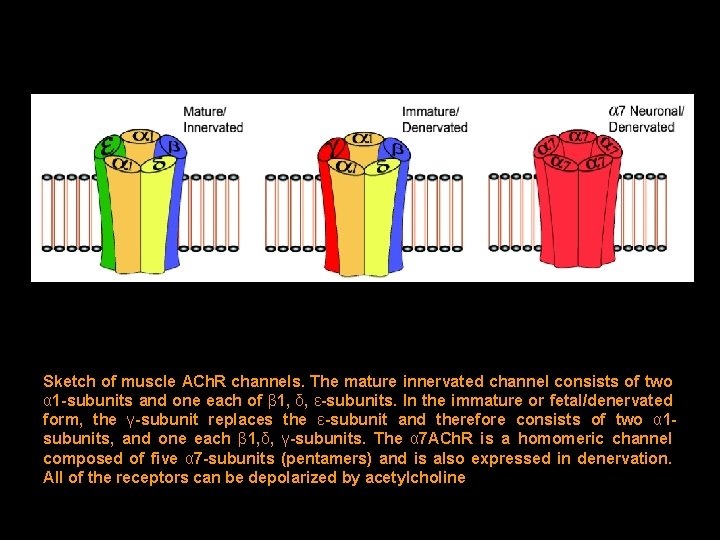
Sketch of muscle ACh. R channels. The mature innervated channel consists of two α 1 -subunits and one each of β 1, δ, ε-subunits. In the immature or fetal/denervated form, the γ-subunit replaces the ε-subunit and therefore consists of two α 1 subunits, and one each β 1, δ, γ-subunits. The α 7 ACh. R is a homomeric channel composed of five α 7 -subunits (pentamers) and is also expressed in denervation. All of the receptors can be depolarized by acetylcholine

7 -nicotinic Acetylcholine Receptor Subunit • Homomeric ligand-gated ion channel • Expressed throughout mammarian brain • Implicated ; sensory gating, cognition, inflammation, neuroprotection

nicotinic Acetylcholine Receptor : 7 - Subunit • Expression in embryonic and adult muscle • Demonstrate by i) muscle cell culture ii) double labelling study with neuronal and muscle cell marker iii) studies with denervated adult muscle • Electrophysiological measurement ; activated by 7 agonist, choline. • Several weeks after chronic denervation ; role in nerve-muscle synapse formation • Specific steps of muscle development ; myoblast fusion and/or migration • Pseudo-metatropic properties; high Ca+2 permeability
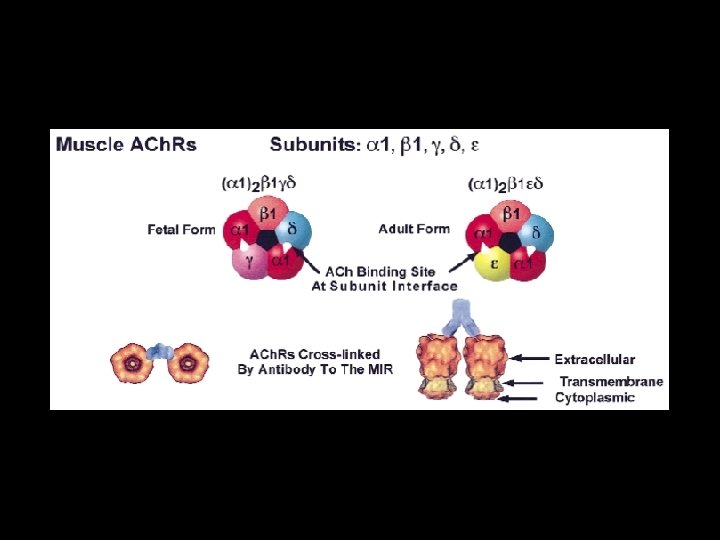
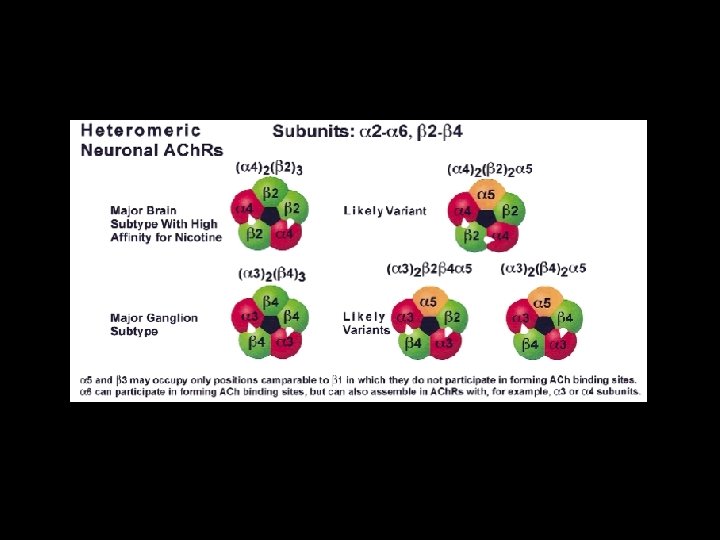

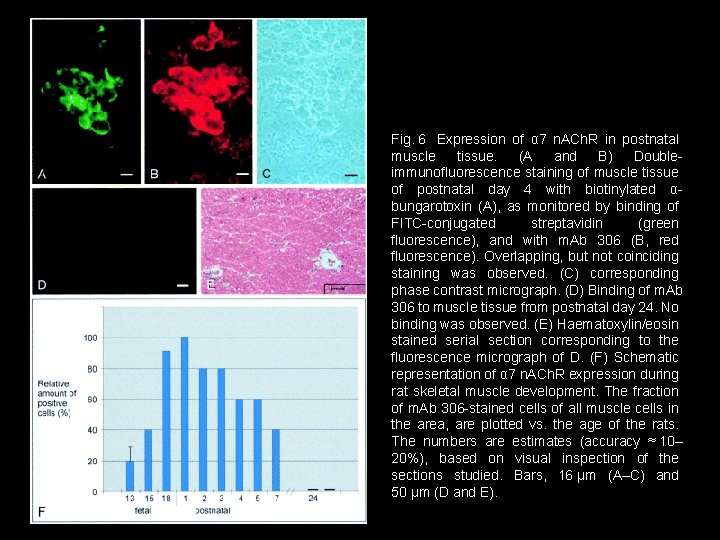
Fig. 6 Expression of α 7 n. ACh. R in postnatal muscle tissue. (A and B) Doubleimmunofluorescence staining of muscle tissue of postnatal day 4 with biotinylated αbungarotoxin (A), as monitored by binding of FITC-conjugated streptavidin (green fluorescence), and with m. Ab 306 (B, red fluorescence). Overlapping, but not coinciding staining was observed. (C) corresponding phase contrast micrograph. (D) Binding of m. Ab 306 to muscle tissue from postnatal day 24. No binding was observed. (E) Haematoxylin/eosin stained serial section corresponding to the fluorescence micrograph of D. (F) Schematic representation of α 7 n. ACh. R expression during rat skeletal muscle development. The fraction of m. Ab 306 -stained cells of all muscle cells in the area, are plotted vs. the age of the rats. The numbers are estimates (accuracy ≈ 10– 20%), based on visual inspection of the sections studied. Bars, 16 μm (A–C) and 50 μm (D and E).
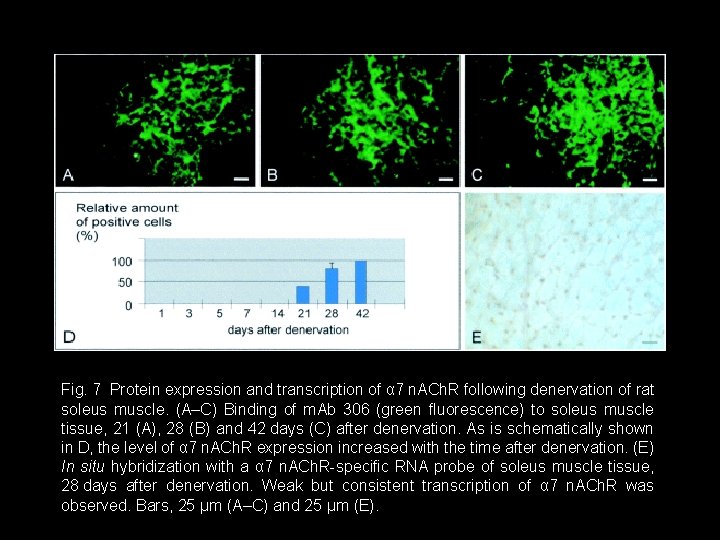
Fig. 7 Protein expression and transcription of α 7 n. ACh. R following denervation of rat soleus muscle. (A–C) Binding of m. Ab 306 (green fluorescence) to soleus muscle tissue, 21 (A), 28 (B) and 42 days (C) after denervation. As is schematically shown in D, the level of α 7 n. ACh. R expression increased with the time after denervation. (E) In situ hybridization with a α 7 n. ACh. R-specific RNA probe of soleus muscle tissue, 28 days after denervation. Weak but consistent transcription of α 7 n. ACh. R was observed. Bars, 25 μm (A–C) and 25 μm (E).

Waglerine-1 Trimeresurus (Tropidolaemus) Wagleri 1. 2. 3. 4. Disulfide bond in lethal peptide no signs of hemorrhage or necrosis Heat stable at 125 ℃ for 15 mins. Not be neutralized by antivenins directed against other snakes venom. 5. Pre- and post-synapse NM junction. = -subunit selective inhibition
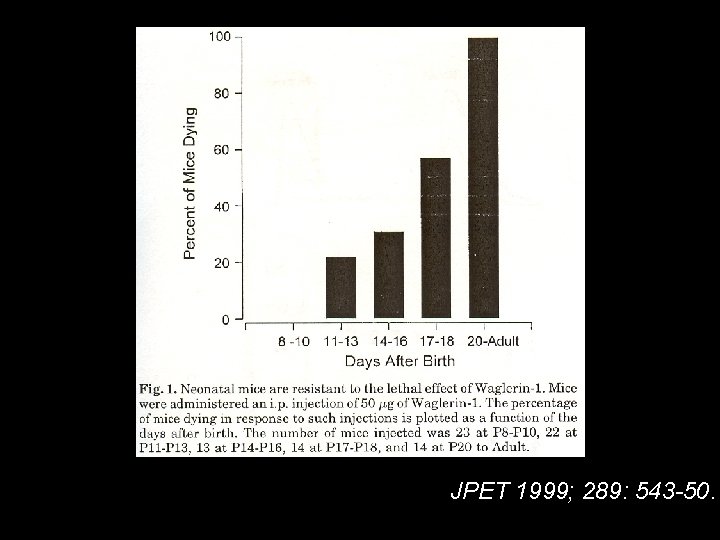
JPET 1999; 289: 543 -50.

-Conotoxin • Conus species: toxin small, disulfide-bonded peptide target: voltage-gated ion channel ligand-gated ion channel G-protein-coupled receptor antagonized n. ACh. R: characterize n. ACh. R subtypes

Ten different cone snails. Shown are the shells of ten Conus species including, top row: the cloth-of-gold cone, Conus textile; the cone of the magi, Conus magus. Second row, left to right: the circumcision cone, Conus circumcisus; the geography cone, Conus geographus; Dusavel's cone, Conus dusaveli. Third row, left to right: the glory-of-the-sea cone, Conus gloriamaris, the glory-of-India cone, Conus milneedwardsi. Bottom row, left to right: the admiral cone, Conus ammiralis; the banded marble cone, Conus bandanus vidua; Hirasei's cone, Conus hirasei. The geography cone, C. geographus is responsible for the majority of human fatalities. This species, along with C. magus, C. circumcisus, and C. dusaveli, are all likely to be fish hunting. The other Conus shown all hunt other gastropod molluscs, except for C. hirasei, which is probably a vermivorous species, although its biology has not been studied.
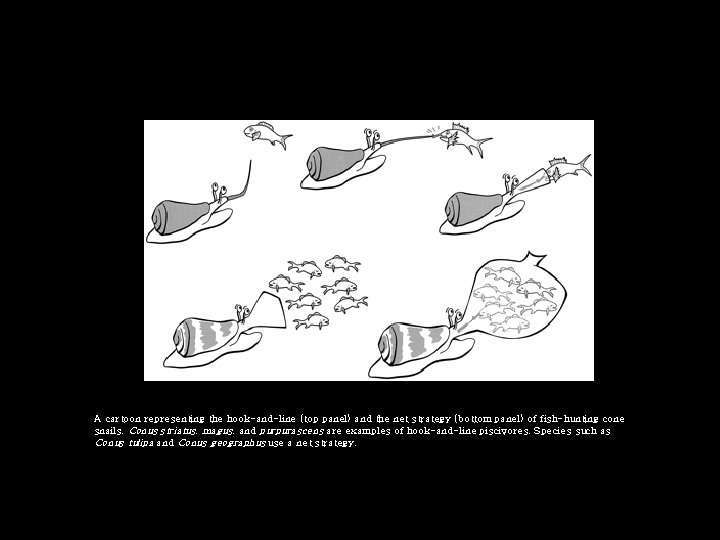
A cartoon representing the hook-and-line (top panel) and the net strategy (bottom panel) of fish-hunting cone snails. Conus striatus, magus, and purpurascens are examples of hook-and-line piscivores. Species such as Conus tulipa and Conus geographus use a net strategy.

Biochemistry 2006; 45: 1304 -12.
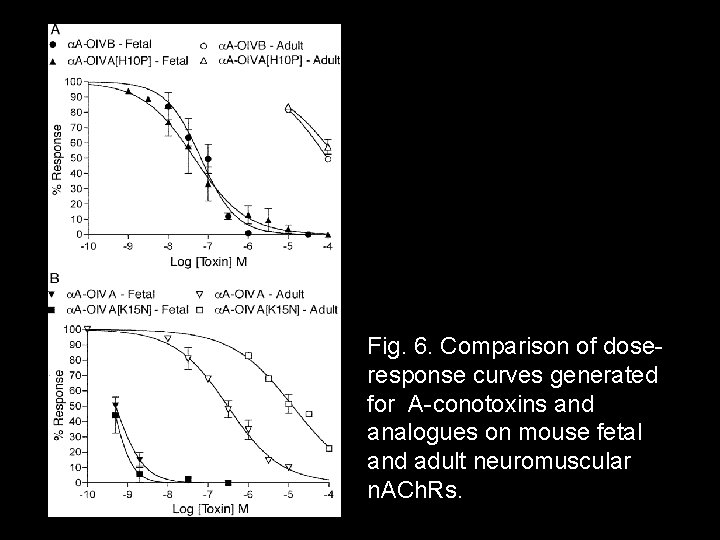
Fig. 6. Comparison of doseresponse curves generated for A-conotoxins and analogues on mouse fetal and adult neuromuscular n. ACh. Rs.

Effects of Immobilization • Methods: a. plaster caster b. application of anesthetics or toxin to the nerve or muscle c. pinning of the joints • Physiologic state a. no direct damage to cord or nerve roots b. muscle fibers remain innervated; by morphological criteria, MEEP, distal segment of nerve and muscle function

Immobilization: Comparable with Denervation • Muscle atrophy • Enlargement & Proliferation of ACh-sensitive area • Shift to the left in Dose-response curve to ACh or SCh • Decreased cholinesterase activity • Sprouting of terminal branches • Revert to normal within 20 -50 days

Immobilization: Neuromuscular Responses • 10 fold shift to right in dose requirement Resistance to NDMR ; 4 days after immobilization subjected to bed rest • May affected the other limb not of the diaphragm
Immobilization : Clinical Implication • SCh ; harzadous acute hyperkalemia 1. in total hip spica or bed rest in ICU 2. dependent on degree and duration 3. only after > 10 days
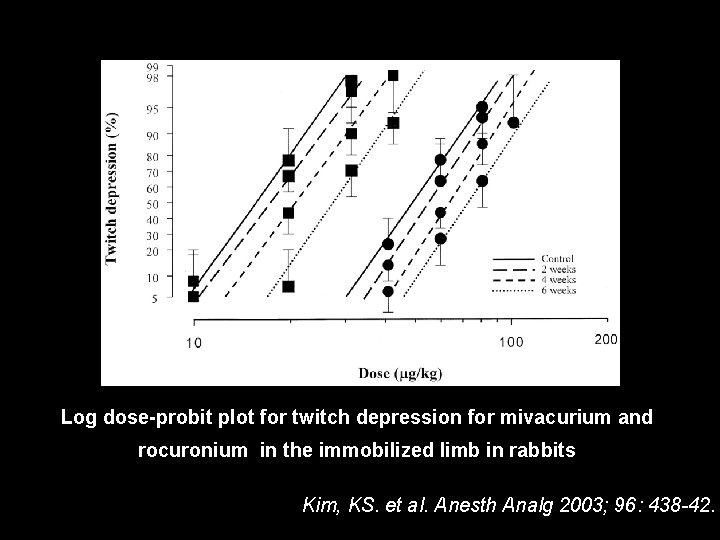
Log dose-probit plot for twitch depression for mivacurium and rocuronium in the immobilized limb in rabbits Kim, KS. et al. Anesth Analg 2003; 96: 438 -42.
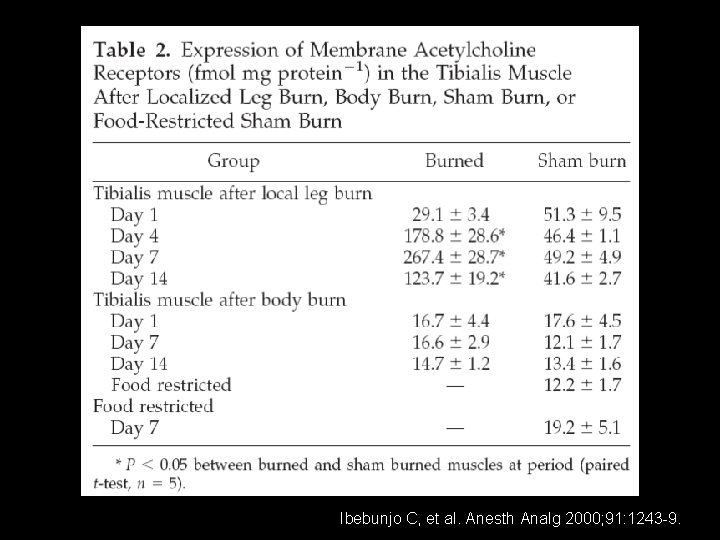
Ibebunjo C, et al. Anesth Analg 2000; 91: 1243 -9.
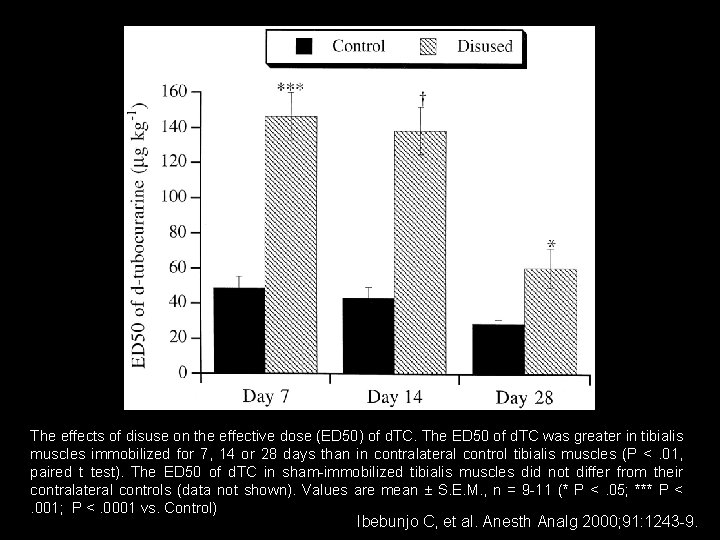
The effects of disuse on the effective dose (ED 50) of d. TC. The ED 50 of d. TC was greater in tibialis muscles immobilized for 7, 14 or 28 days than in contralateral control tibialis muscles (P < . 01, paired t test). The ED 50 of d. TC in sham-immobilized tibialis muscles did not differ from their contralateral controls (data not shown). Values are mean ± S. E. M. , n = 9 -11 (* P < . 05; *** P < . 001; P <. 0001 vs. Control) Ibebunjo C, et al. Anesth Analg 2000; 91: 1243 -9.
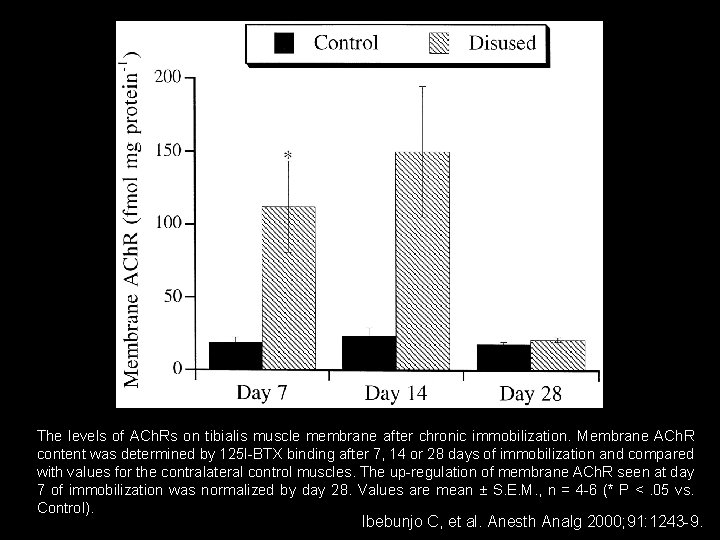
The levels of ACh. Rs on tibialis muscle membrane after chronic immobilization. Membrane ACh. R content was determined by 125 I-BTX binding after 7, 14 or 28 days of immobilization and compared with values for the contralateral control muscles. The up-regulation of membrane ACh. R seen at day 7 of immobilization was normalized by day 28. Values are mean ± S. E. M. , n = 4 -6 (* P <. 05 vs. Control). Ibebunjo C, et al. Anesth Analg 2000; 91: 1243 -9.

Materials and Methods I • Mice: C 57 BL/6 J, α 7 Knock out. • Specific receptor subunit blockers : Waglerin-1, Ar 1. 5, α-Conotoxin Pe. IVA, α-Conotoxin OIVA.
Material and Methods II • Immobilization: at knee and ankle joint with pin. duration : 1, 7, 14, and 21 days. • Muscle function test: tibialis anterior • Muscle histochemical study: tibialis anterior, soleus, gastrocunemious




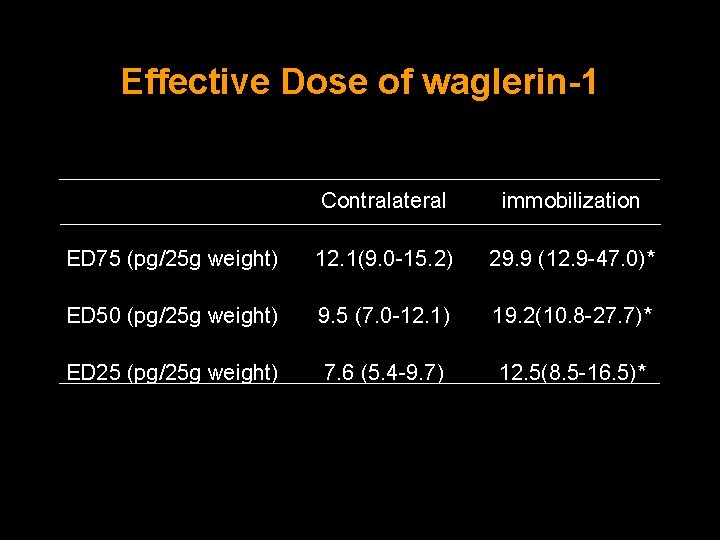
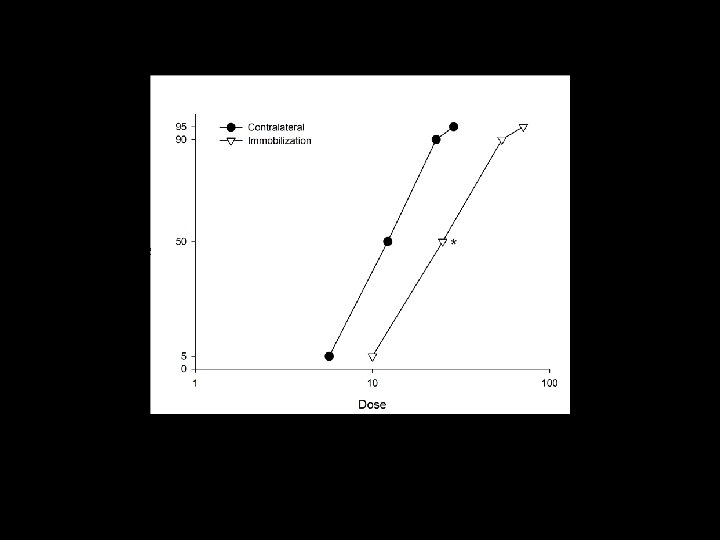
Effective Dose of waglerin-1 Contralateral immobilization ED 75 (pg/25 g weight) 12. 1(9. 0 -15. 2) 29. 9 (12. 9 -47. 0)* ED 50 (pg/25 g weight) 9. 5 (7. 0 -12. 1) 19. 2(10. 8 -27. 7)* ED 25 (pg/25 g weight) 7. 6 (5. 4 -9. 7) 12. 5(8. 5 -16. 5)*
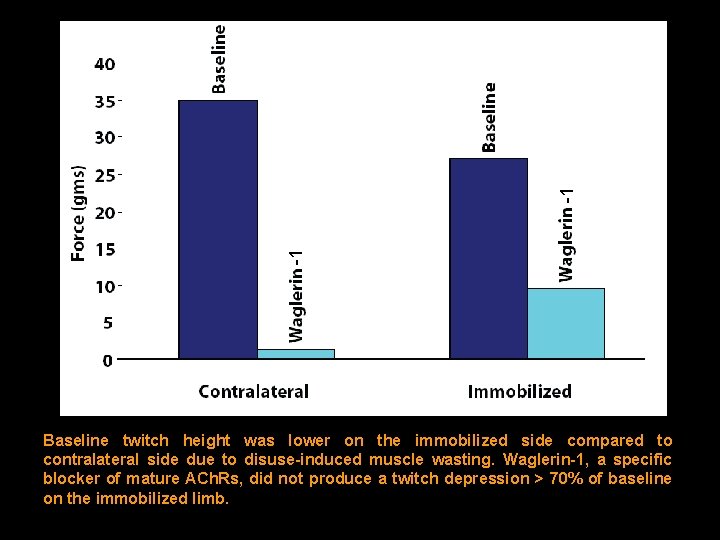
-1 -1 Baseline twitch height was lower on the immobilized side compared to contralateral side due to disuse-induced muscle wasting. Waglerin-1, a specific blocker of mature ACh. Rs, did not produce a twitch depression > 70% of baseline on the immobilized limb.
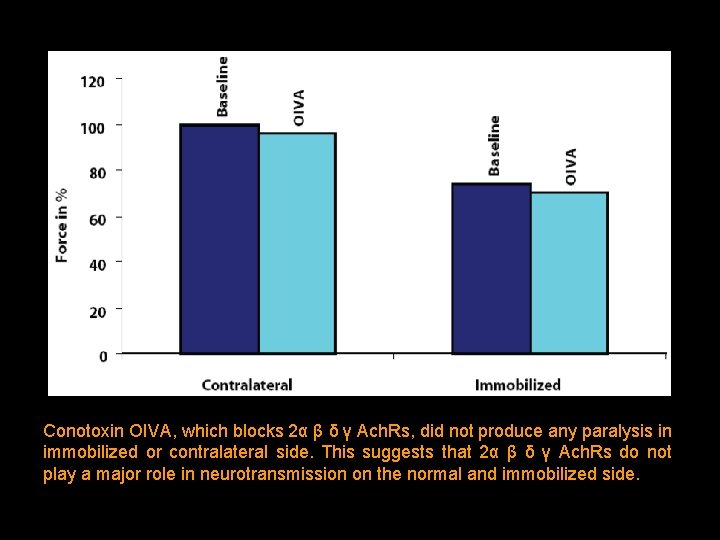
Conotoxin OIVA, which blocks 2α β δ γ Ach. Rs, did not produce any paralysis in immobilized or contralateral side. This suggests that 2α β δ γ Ach. Rs do not play a major role in neurotransmission on the normal and immobilized side.
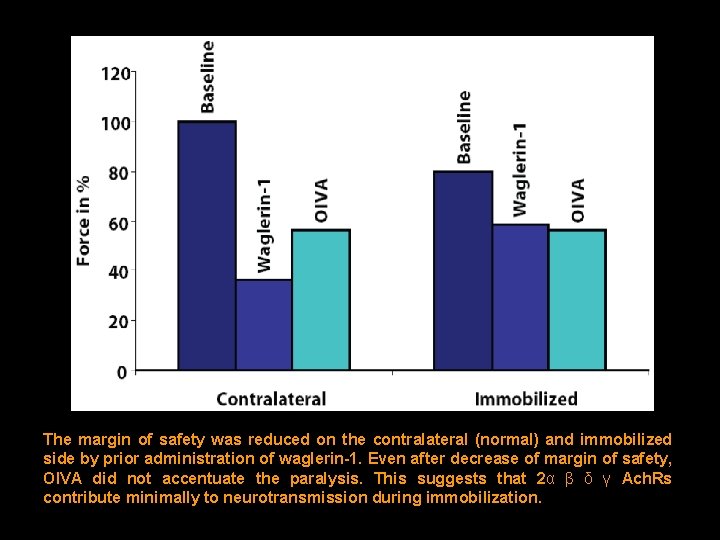
The margin of safety was reduced on the contralateral (normal) and immobilized side by prior administration of waglerin-1. Even after decrease of margin of safety, OIVA did not accentuate the paralysis. This suggests that 2α β δ γ Ach. Rs contribute minimally to neurotransmission during immobilization.
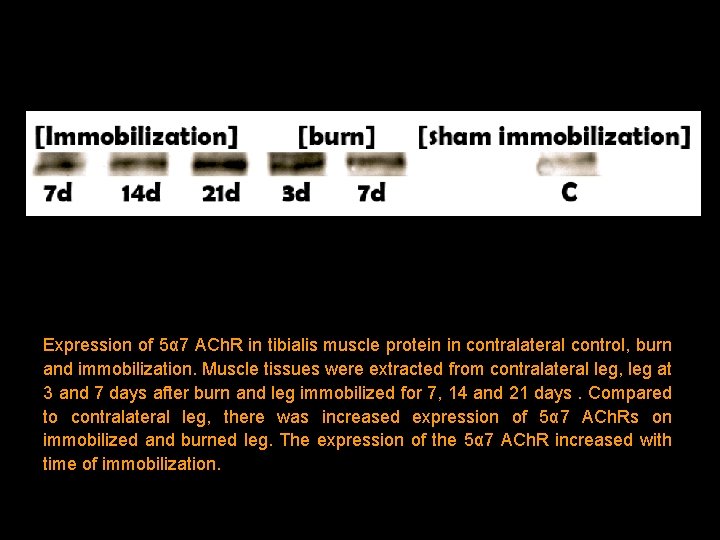
Expression of 5α 7 ACh. R in tibialis muscle protein in contralateral control, burn and immobilization. Muscle tissues were extracted from contralateral leg, leg at 3 and 7 days after burn and leg immobilized for 7, 14 and 21 days. Compared to contralateral leg, there was increased expression of 5α 7 ACh. Rs on immobilized and burned leg. The expression of the 5α 7 ACh. R increased with time of immobilization.
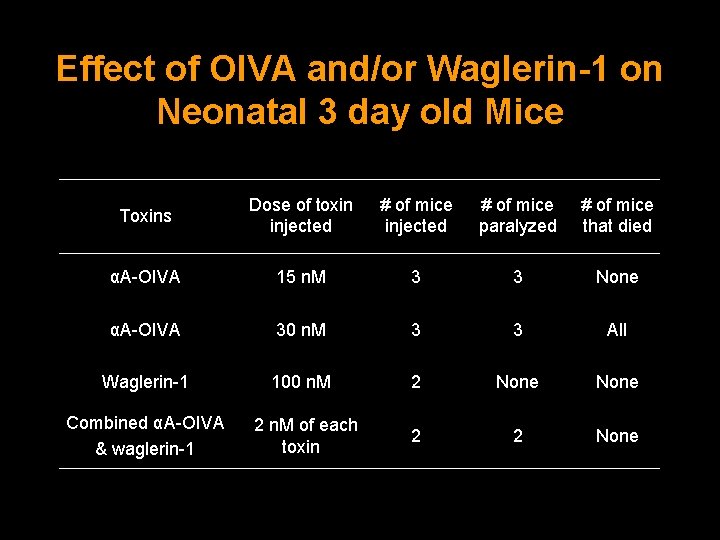
Effect of OIVA and/or Waglerin-1 on Neonatal 3 day old Mice Toxins Dose of toxin injected # of mice injected # of mice paralyzed # of mice that died αA-OIVA 15 n. M 3 3 None αA-OIVA 30 n. M 3 3 All Waglerin-1 100 n. M 2 None 2 2 None Combined αA-OIVA 2 n. M of each toxin & waglerin-1

Discussion • Our findings in adult immobilized mice suggest that the contribution to neurotransmission by mature (ε) ACh. Rs decreases during immobilization. • ACh. Rs other than 2α β δ ε or 2α β δ γ, play a role and contribute to neurotransmission during immobilization. The most likely candidate is 5 α 7 ACh. Rs.

- Slides: 44