Imaging in Oncology Clinical Trials Susan Galbraith Clinical

Imaging in Oncology Clinical Trials Susan Galbraith Clinical Discovery Bristol-Myers Squibb
What can we see? • Microvasculature – Blood volume - MRI, PET – Vessel permeability - MRI – Blood flow - PET, MRI, SPECT – Hypoxia - MRI, PET – VEGF - PET

What can we see? • • Gene expression - optical imaging, PET Enzyme activation - optical imaging, MRI Receptor expression/occupancy - PET, MRI Apoptosis - MRI, PET, SPECT Cell proliferation - PET Glucose metabolism - PET Membrane turnover - PET
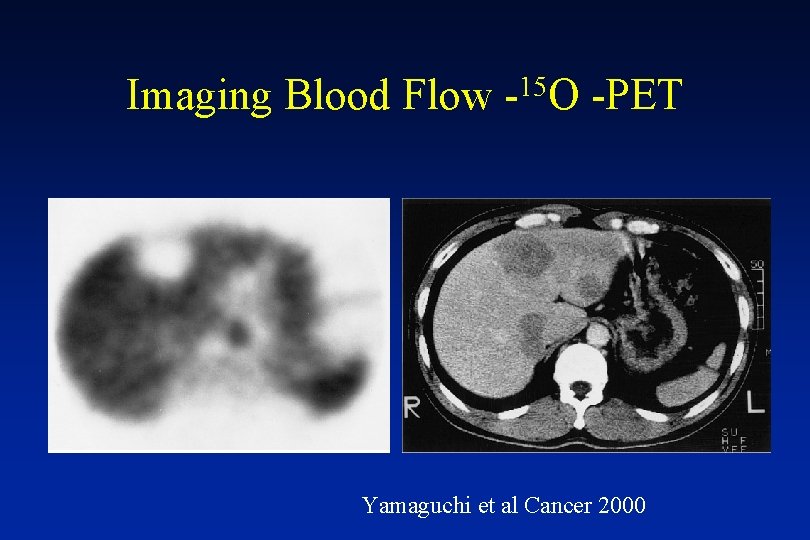
Imaging Blood Flow 15 - O -PET Yamaguchi et al Cancer 2000

CT PET -Anatomy and Glucose Metabolism Ken Krohn University of Washington
FLT PET - Imaging Proliferation • FLT (3’- deoxy-3’ – fluorothymidine) is phosphorylated by thymidine kinase 1 and trapped within cells • Since TK-1 levels increase around 10 -fold in S-phase, retention should theoretically reflect DNA synthesis Shields et al. Nature Med 1998
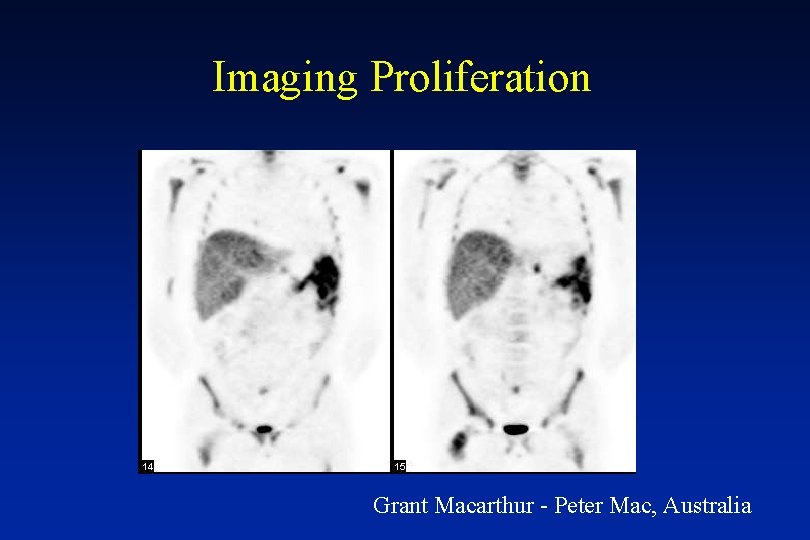
Imaging Proliferation Grant Macarthur - Peter Mac, Australia

DCE-MRI • Using Gd-DTPA - composite of vessel permeability, surface area and blood flow • Using high molecular weight contrast agents - permeability, blood volume • Need arterial input function to determine blood flow
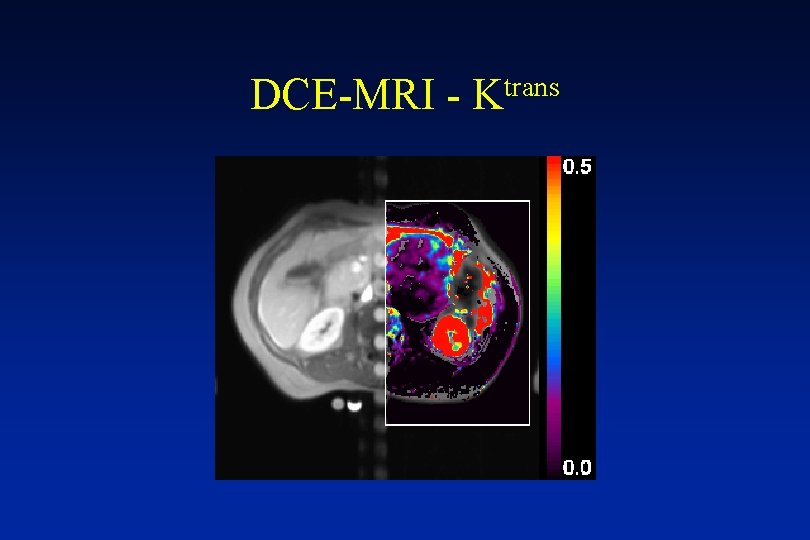
DCE-MRI - trans K

MRI - Imaging of permeability and blood volume • Need high molecular weight contrast agent – Albumin- Gd. DTPA - Overexpression of VEGF 165 drives peritumor interstitial convection and induces lymphatic drain (Dafni et al Cancer Res 2002) – Superparamagnetic iron oxide contrast agents

What can imaging do for you? …. Novel imaging technology has the potential to • assist lead compound selection • enable earlier Go/No Go decisions • have greater confidence about those decisions • save patients from treatment with drugs destined to fail • save money How to utilize this potential to truly affect decisions in drug development ?

Objectives of Phase I Oncology Trials • Safety • Pharmacokinetics • Dose selection – cytotoxics - ‘maximum tolerated dose’ – ‘targeted’ drugs - ‘optimal biological dose’

What answers would help a novel ‘targeted’ oncology drug? • Pre-clinical/Phase 1 – does the drug hit the target in the tumor – what is the exposure response / time course of response • Phase I/II – how does hitting target relate to anti-tumor efficacy – any early indicators of toxicity • Phase II/III – can tumor response be predicted by target expression/ activation – differentiation from competitors

Definition of Go/No Go • Drug does not hit target • Do not achieve desired effect size at tolerable doses • Selectivity of effect in tumor/normal tissues

Where does imaging fit in development? • SAD (if TI allows) - rapidly define single dose PK, tolerability, ability to reach exposure range for efficacy • MAD - imaging or other biomarker to demonstrate biological activity, dose response and PK/PD relationship
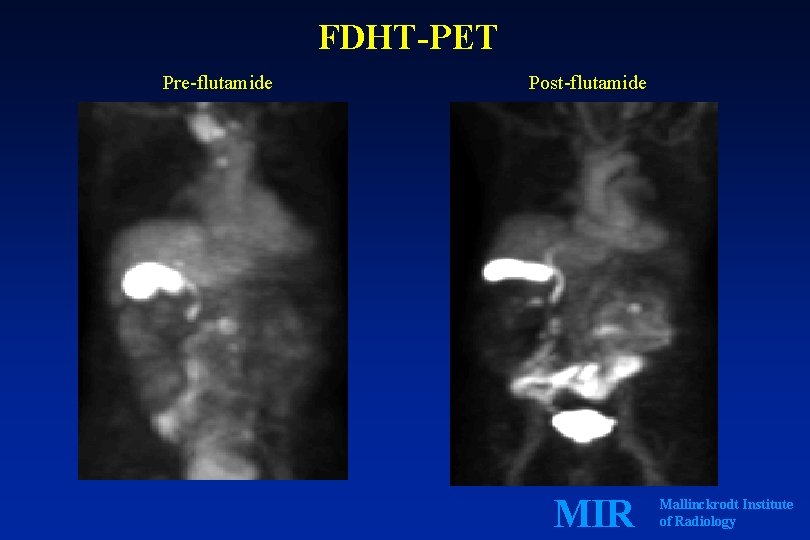
FDHT-PET Pre-flutamide Post-flutamide MIR Mallinckrodt Institute of Radiology
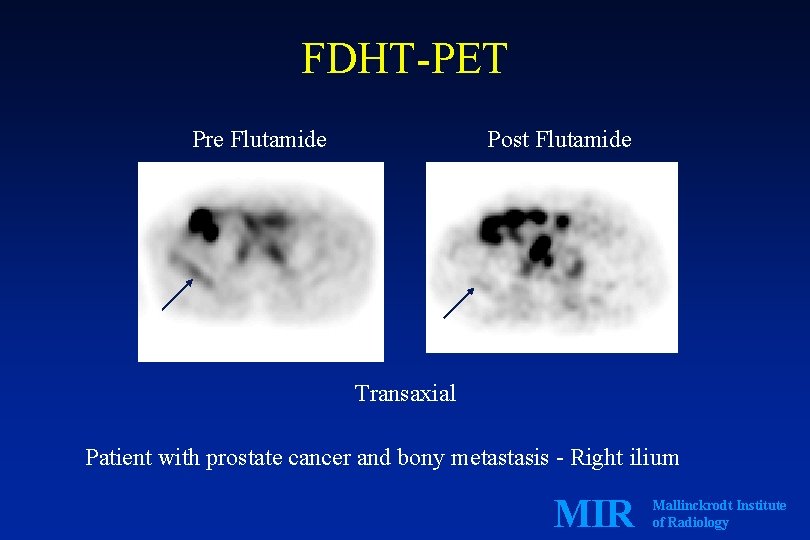
FDHT-PET Pre Flutamide Post Flutamide Transaxial Patient with prostate cancer and bony metastasis - Right ilium MIR Mallinckrodt Institute of Radiology

Phase I trial • Dose escalate ? To MTD (depends on TI) • Expand cohorts for imaging studies (n depends on reproducibility and effect size of interest) • Need same imaging protocol implemented at all sites • Quality control • Centralized data analysis

Implications • Technology used - relatively established vs ‘cutting edge’ • Definition of every stage of imaging process • Reproducibility studies needed before measurement of treatment effect • SDV as detailed as for clinical aspects of study • Site selection • Consensus on methodology e. g. EORTC FDG PET recommendations 1999

Reproducibility studies NMR in Biomed 2002, 15, p 132 -142

Reproducibility Studies • Determine 95% limits of change for individuals and for groups • Identify ‘key determinants’ of reproducibility how much is dependent on subjective definition of ‘ROI’s etc • Learning curve for technique • Project cohort size needed for measurement of treatment effect

Choice of parameter • DCE-MRI - gradient, enhancement, AUC, Ktrans, kep, ve • FDG PET - dynamic, SUV - which SUV? • Balance – reproducibility – sensitivity to treatment effect – validity of assumptions – availability – heterogeneity effect
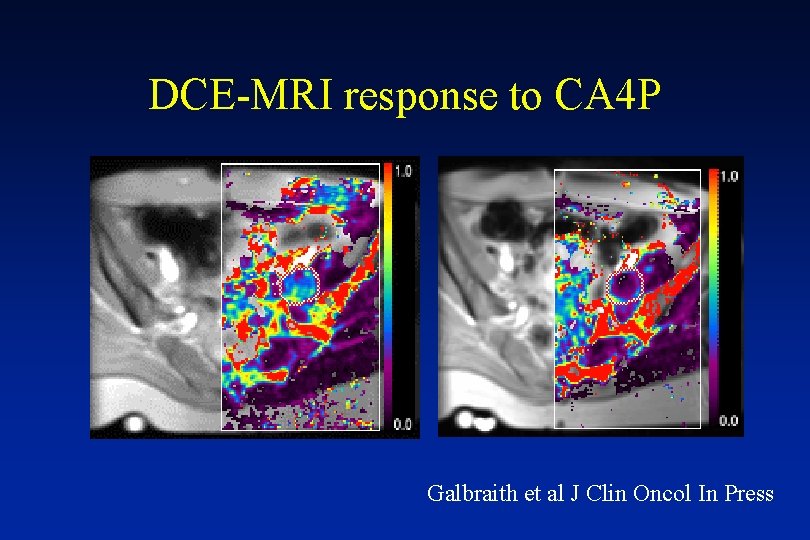
DCE-MRI response to CA 4 P Galbraith et al J Clin Oncol In Press

Choice of patient population • Homogeneous tumor type, site • Ability to obtain good quality images respiration/movement artefact • Ability to accrue trial within reasonable time

Phase II - Efficacy • Is stable disease indicative of anti-tumor efficacy? • Effects on tumor metabolism/ proliferation/ microvasculature seen before effects on tumor size • Are changes in proliferation/ metabolism seen in higher proportion of patients than proportion with PR/CR
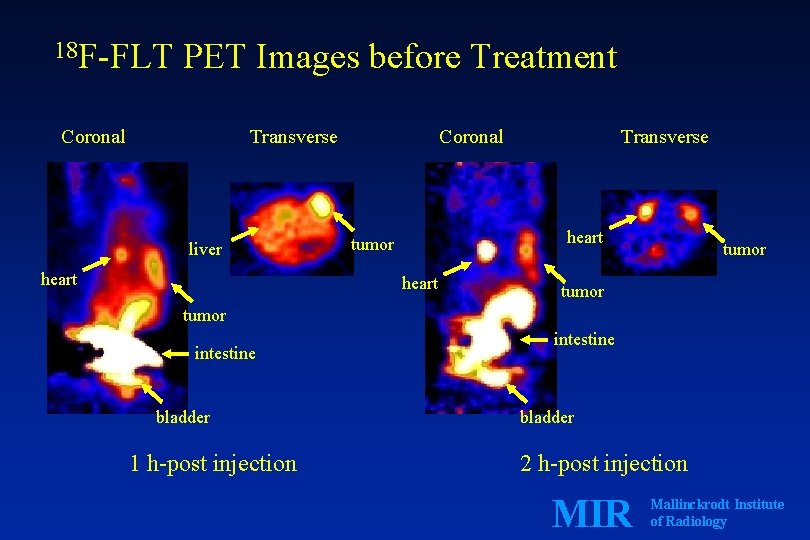
18 F-FLT PET Images before Treatment Coronal Transverse liver heart Coronal Transverse heart tumor intestine bladder 1 h-post injection intestine bladder 2 h-post injection MIR Mallinckrodt Institute of Radiology
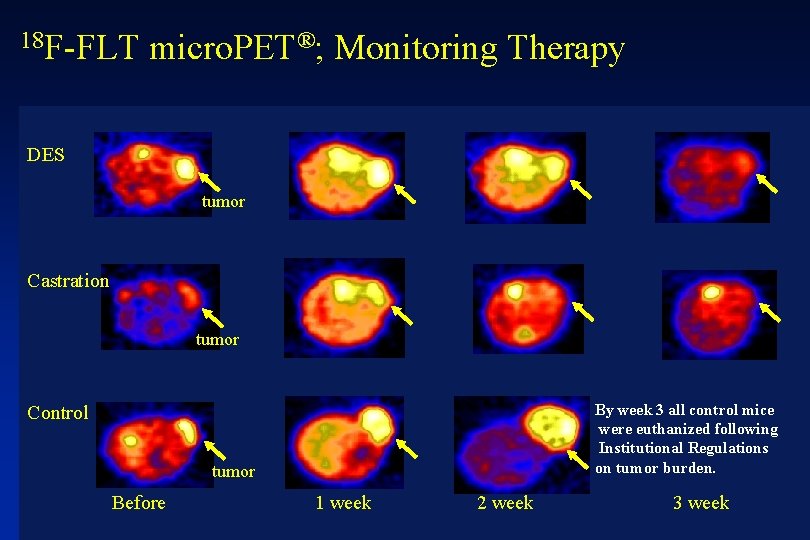
18 F-FLT micro. PET®; Monitoring Therapy DES tumor Castration tumor By week 3 all control mice were euthanized following Institutional Regulations on tumor burden. Control tumor Before 1 week 2 week MIR 3 week Institute Mallinckrodt of Radiology
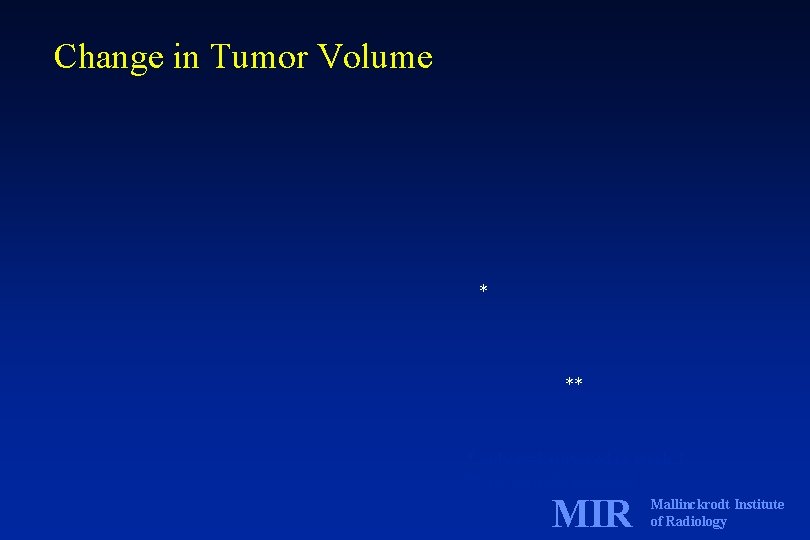
Change in Tumor Volume * ** * only n=2 survived to week 2 ** no animals survived MIR Mallinckrodt Institute of Radiology

Change of 18 F-FLT Uptake in Tumor * ** * only n=2 survived to week 2 ** no animals survived MIR Mallinckrodt Institute of Radiology

Rationale for use of FDG PET for response assessment • Earlier response assessment • Better predictor of clinical benefit than conventional imaging - biology rather than anatomy • ? Increased number of responders - more information on ‘stable disease’
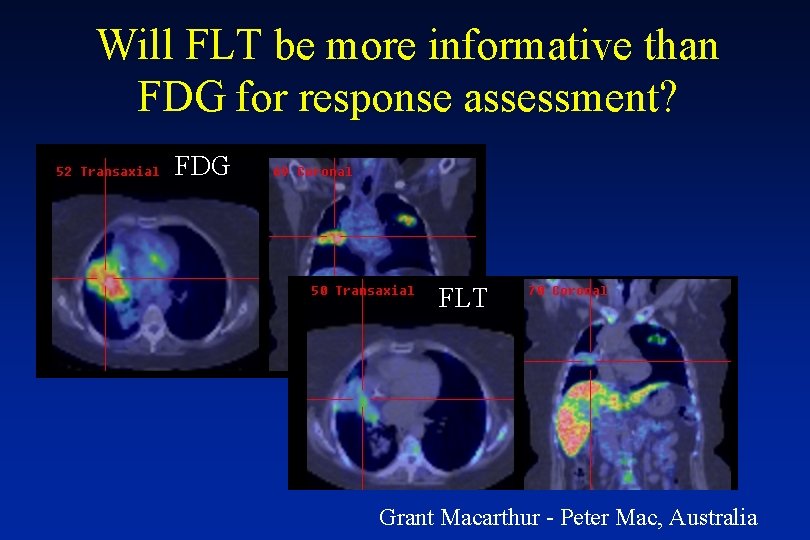
Will FLT be more informative than FDG for response assessment? FDG FLT Grant Macarthur - Peter Mac, Australia
Phase II/III - response prediction • Whole tumor imaging characteristics vs tissue biopsy • Implications of tumor heterogeneity • Serial non-invasive images vs serial biopsies
Potential utility of imaging. . • Imaging of receptor occupancy/ enzyme inhibition • Pre-clinical correlation with anti-tumor effect • Understanding of PK/PD relationship • Translatable technology from pre-clinic to clinic • Determination of reproducibility in clinic • High quality, multi-site imaging in trials • Early indication of efficacy • Response prediction

How do we get there? • Collaboration with academia - long term – limits of the technologies possible now – translational studies – what’s around the corner? • Work on QA/ imaging monitoring/analysis – delivery of high quality imaging in trials • Develop internal understanding of and expertise in imaging technology
- Slides: 34