Imagine you are baking chocolate chip cookies What

Imagine you are baking chocolate chip cookies What materials do you need?

If you have all the flour, eggs, sugar, etc you need but only 20 chips, how many cookies will you make?

In dealing with manufactured goods (paint, soaps, food), the main purpose is to make a profit; any waste will cut profits

How do the manufacturers know how much to mix & how much they will get?

By knowing how to write: Balanced Equations They are the “recipes” that tell the amounts to mix & what they will get It allows one to calculate quantities of reactants & products

Stoichiometry – the calculation of quantities in chemical equations

STOICHIOMETRIC CALCULATIONS

Interpreting Equations – Coefficients in a balanced equation shows a relationship between moles of reactants vs. moles of products – Moles can then be converted into grams, r. p. , or Liters

ex. N 2(g) + 3 H 2(g) -> 2 NH 3(g) – Molecules: 1 + 3 -> 2 – Moles: 1 + 3 -> 2 – Grams: 1(28) + 3(2) -> 2(17) – Volume: 1(22. 4) + 3(22. 4) -> 2(22. 4) (at STP) – R. P. ’s: 1(6. 02 ) + 3(6. 02) -> 2(6. 02)

Mole -> Mole –Coefficients of balanced equations tells the “conversion factor” or mole ratio”

How many moles of NH 3 are produced when 0. 60 mol of N 2 reacts with H 2? 1. Write a balanced equation: 2. Determine X & Y: 3. Factor/Calculate: (Start -> Finish)

Ex. Calculate the # of moles of reactants needed to make 7. 24 mol of NH 3 – Answers: 3. 62 mol N 2 10. 9 mol H 2 •

Mass -> Mass or Teoretical Yeild – *since we can’t measure moles in a lab, we must be able to measure the reactants or products by their mass from the moles or mass of the other

Calculate: # of grams of NH 3 produced by the reaction of 5. 40 g of H 2 with N 2 1. Write a balanced equation: 2. Determine X & Y: 3. Factor/Calculate: = 30. 4 g (Start -> Finish)

Ex. Calculate the # of grams of reactants needed to produce 30. 6 g NH 3 Answer: 25. 2 g N 2 • 5. 44 g H 2

The quantities calculated in mass-mass problems are theoretical amounts; only the maximum possible yield is calculated. In reality, actual amounts of products are less.

A comparison of the actual vs. theoretical is expressed in percent and is called: –PERCENT YIELD

PERCENT YIELD is calculated as follows: actual amount theoretical amount (MASS - MASS) x 100

ex. Oil of wintergreen is made by heating salicylic acid, C 7 H 6 O 3, with methanol, CH 3 OH, by the following equation: C 7 H 6 O 3 +CH 3 OH---> C 8 H 8 O 3 +H 2 O If you start with 1. 75 g of salicylic acid, what is theoretical yield of wintergreen oil? Theoretical Yield = 1. 93 g

If the actual is 1. 42 g, what is the percent yield? Percent Yield: 73. 6%

LIMITING REAGENT When following a recipe, sufficient quantities of all ingredients must be present to react.

Ex. No matter how much P & J you have with 8 slices of bread, you can only make 4 sandwiches.

A limiting reagent “limits” or determines the amount of product that can be produced in a reaction. In comparison, the excess reagent is more than enough to react with a limiting reagent.

How to determine the limiting reagent:

What will occur if 6. 70 mol Na reacts with 3. 20 mol of Cl 2? 1. Write a balanced equation 2. Determine X & Y 3. Calculate needed amount of one reactant from the other (Start -> Finish) 4. Compare the needed with the given 5. LR = Cl 2

What is the amount of product formed? Calculate: (start -> Finish) (Be sure to use the amount of LR you started with)
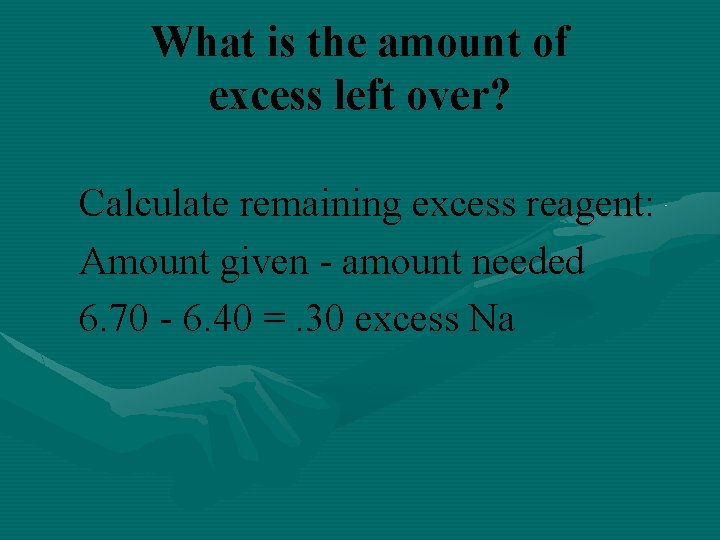
What is the amount of excess left over? Calculate remaining excess reagent: Amount given - amount needed 6. 70 - 6. 40 =. 30 excess Na

Copper reacts with sulfur to form copper (I) sulfide. What is the maximum number of grams of copper (I) sulfide that can be formed when 80. 0 g of copper reacts with 25. 0 g of sulfur?
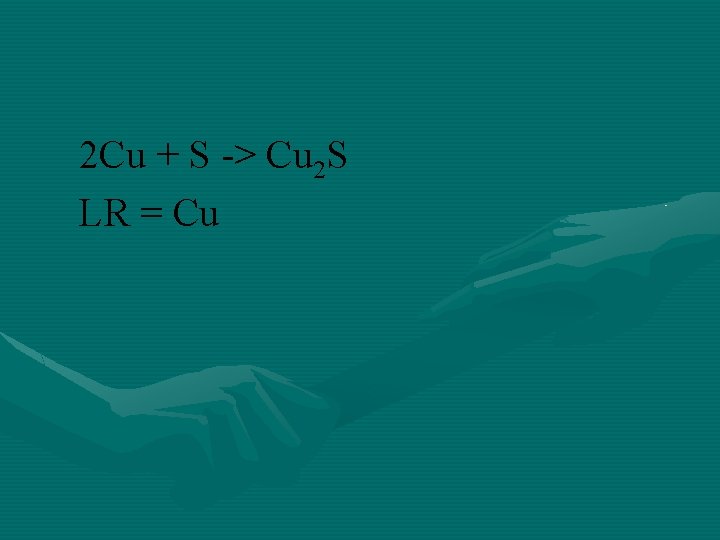
2 Cu + S -> Cu 2 S LR = Cu

Calculate Maximum Product: Amount LR x Ratio: = 1. 00 x 102 g Cu 2 S

OTHER FORMULAS Because most reactions occur in solutions, scientists must express the relationship between the solute and the solvent. This expression is called: MOLARITY

Molarity – the ratio between moles of solute to the volume of solution in dm 3 M = mol solute dm 3 of solution

Molarity Solving Process: – Convert volume from cm 3 -> dm 3 – Put givens into equation:

ex. Calculate the molarity of 2 3 2. 50 x 10 cm of solution containing 9. 46 g of Cs. Br. = 0. 178 M Cs. Br

What if you wanted to make a solution? – use the molarity equation and solve for grams. ex. How would you make 5. 00 x 102 cm 3 of a 0. 133 M solution of Mn. Se. O 4? = 13. 2 g of Mn. Se. O 4

= 13. 2 g of Mn. Se. O 4
- Slides: 36