Im Bond Chemical Bond Atom the smallest unit

I’m Bond, Chemical Bond
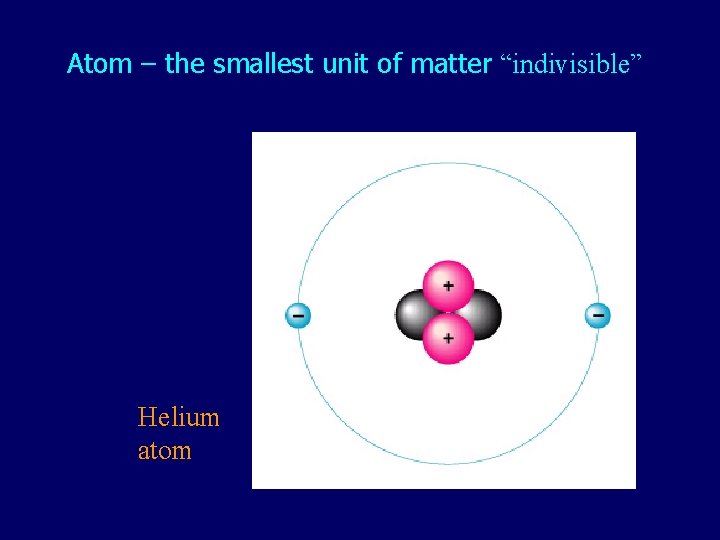
Atom – the smallest unit of matter “indivisible” Helium atom

electron shells a) Atomic number = number of Electrons b) Electrons vary in the amount of energy they possess, and they occur at certain energy levels or electron shells.

Electrons are placed in shells according to rules: 1) The 1 st shell can hold up to two electrons, and each shell thereafter can hold up to 8 electrons.
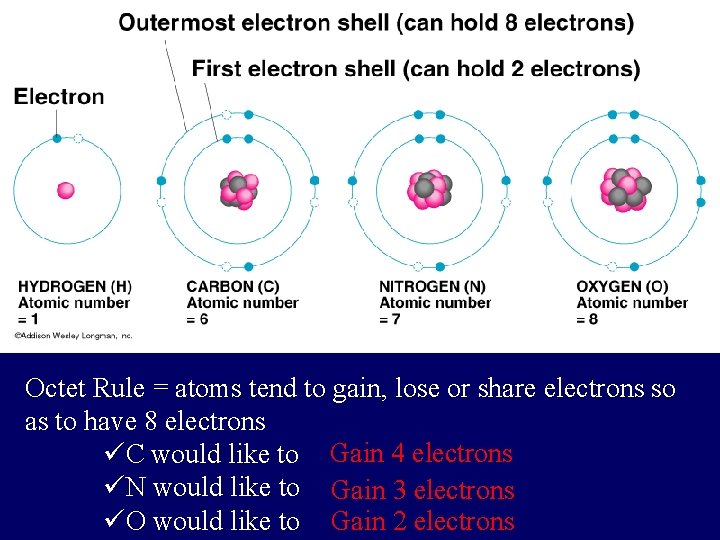
Octet Rule = atoms tend to gain, lose or share electrons so as to have 8 electrons üC would like to Gain 4 electrons üN would like to Gain 3 electrons üO would like to Gain 2 electrons

Why are electrons important? 1) Elements have different electron configurations § different electron configurations mean different ways of bonding – and different products from their reactions
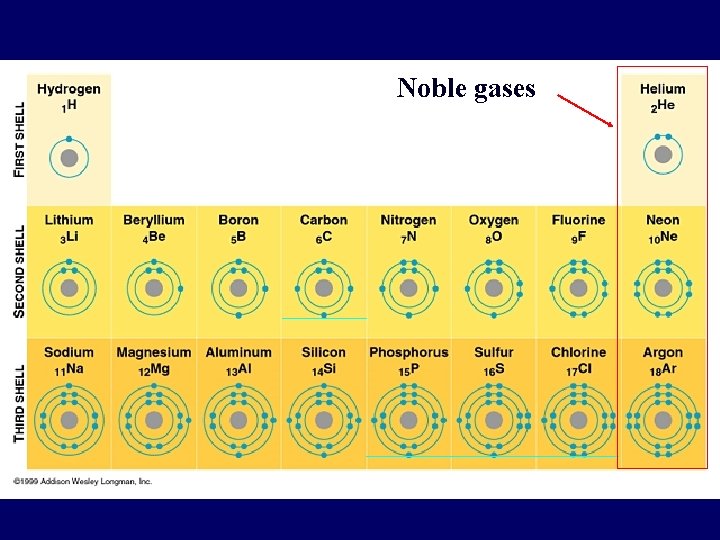
Noble gases

Electron Dot Structures Symbols of atoms with dots to represent the outermost shell electrons 1 2 3 4 5 6 7 H 2/8 He: Li Be B Na Mg Al C N O Si P S : F : Ne : : Cl : Ar :

Chemical bonds: an attempt to fill electron shells 1. Ionic bonds – 2. Covalent bonds – 3. Metallic bonds
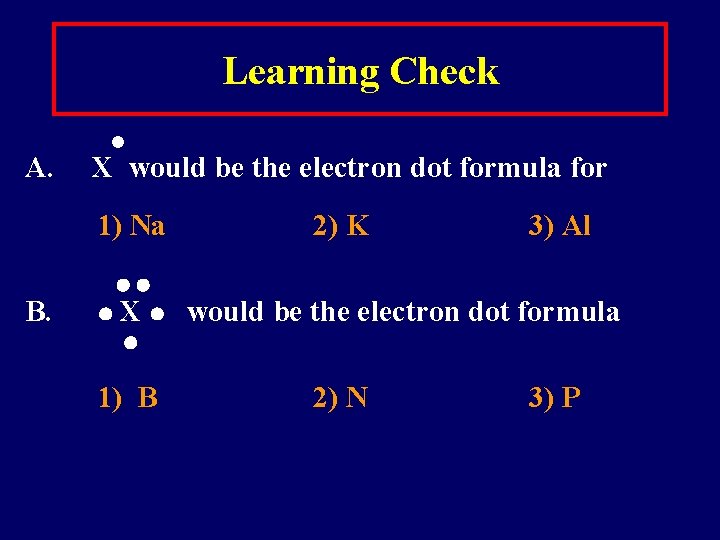
Learning Check A. X would be the electron dot formula for 1) Na B. X 1) B 2) K 3) Al would be the electron dot formula 2) N 3) P

IONIC BOND bond formed between two ions by the transfer of electrons

Formation of Ions from Metals Ionic compounds result when metals react with nonmetals Metals lose electrons to match the number of outermost shell electrons of their nearest noble gas Positive ions form when the number of electrons are less than the number of protons Group 1 metals • ion 1+ Group 2 metals ion 2+ Group 13 metals ion 3+
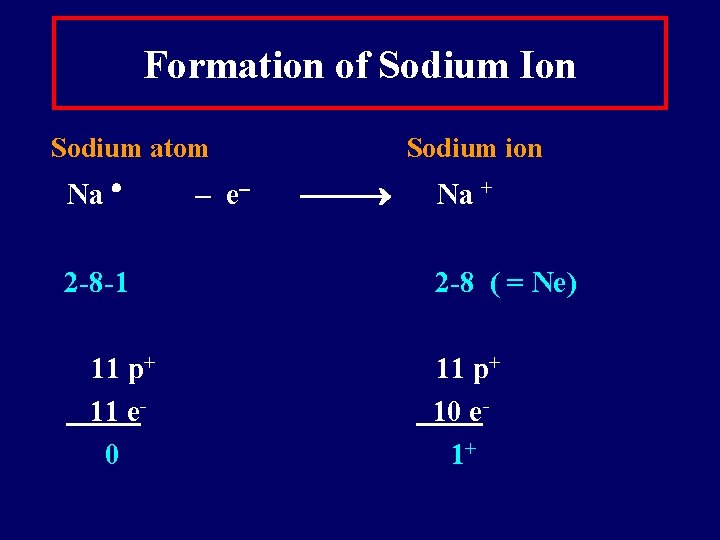
Formation of Sodium Ion Sodium atom Na 2 -8 -1 11 p+ 11 e 0 – e Sodium ion Na + 2 -8 ( = Ne) 11 p+ 10 e 1+

Formation of Magnesium Ion Magnesium atom Mg 2 -8 -2 12 p+ 12 e 0 – 2 e Magnesium ion Mg 2+ 2 -8 (=Ne) 12 p+ 10 e 2+

Some Typical Ions with Positive Charges (Cations) Group 1 Group 2 Group 13 H+ Mg 2+ Al 3+ Li+ Ca 2+ Na+ K+

Learning Check A. Number of outermost shell electrons in aluminium 1) 1 e 2) 2 e 3) 3 e. B. Change in electrons for octet 1) lose 3 e 2) gain 3 e. Ionic charge of aluminium 1) 32) 5 - 3) gain 5 e- 3) 3+

Solution A. Number of outermost shell electrons in aluminium 3) 3 e. B. Change in electrons for octet 1) lose 3 e- C. Ionic charge of aluminium 3) 3+

Learning Check Give the ionic charge for each of the following: A. 12 p+ and 10 e 1) 0 2) 2+ 3) 2 B. 50 p+ and 46 e 1) 2+ 2) 4+ 3) 4 - C. 15 p+ and 18 e 2) 3+ 2) 3 - 3) 5 -

Ions from Nonmetal Ions n. In ionic compounds, nonmetals gain electrons from metals n. Nonmetal add electrons to achieve the octet arrangement n. Nonmetal ionic charge: 3 -, 2 -, or 1 -
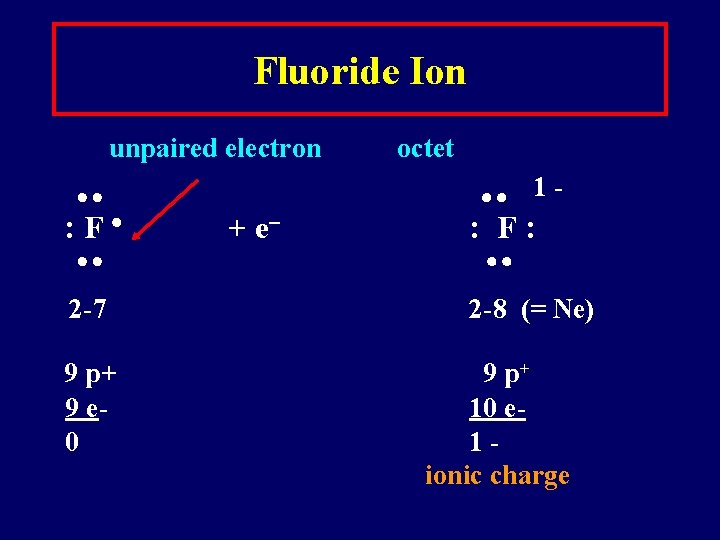
Fluoride Ion unpaired electron : F 2 -7 9 p+ 9 e 0 + e octet 1 - : F: 2 -8 (= Ne) 9 p+ 10 e 1 ionic charge

Ionic Bond • Between atoms of metals and nonmetals • Bond formed by transfer of electrons • Produce charged ions. Conductors and have high melting point. • Examples; Na. Cl, Ca. Cl 2, K 2 O
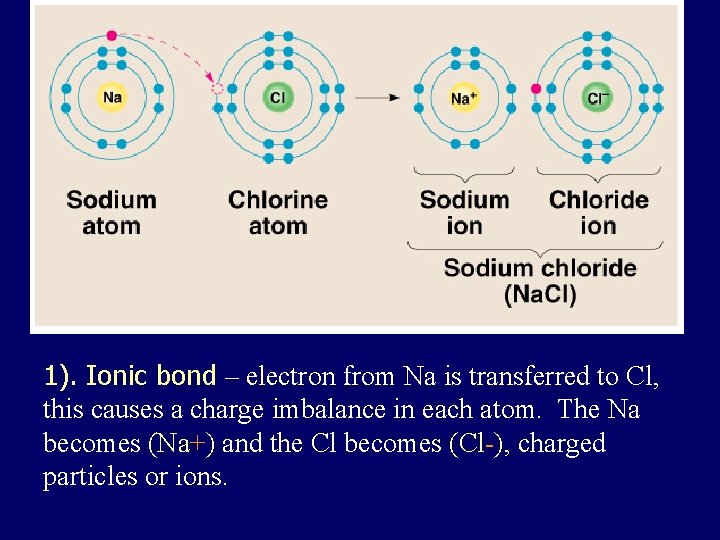
1). Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions.
http: //www. youtub e. com/watch? v=Ft w 7 a 5 ccubs

COVALENT BOND bond formed by the sharing of electrons

Covalent Bond • Between nonmetallic elements • Formed by sharing electron pairs • Stable non-charged particles, they are not conductors at any state • Examples; O 2, C 2 H 6, H 2 O, Si. C

Bonds in all the polyatomic ions and diatomics are all covalent bonds

COVALENT BOND in elements when electrons are shared equally H 2 or Cl 2
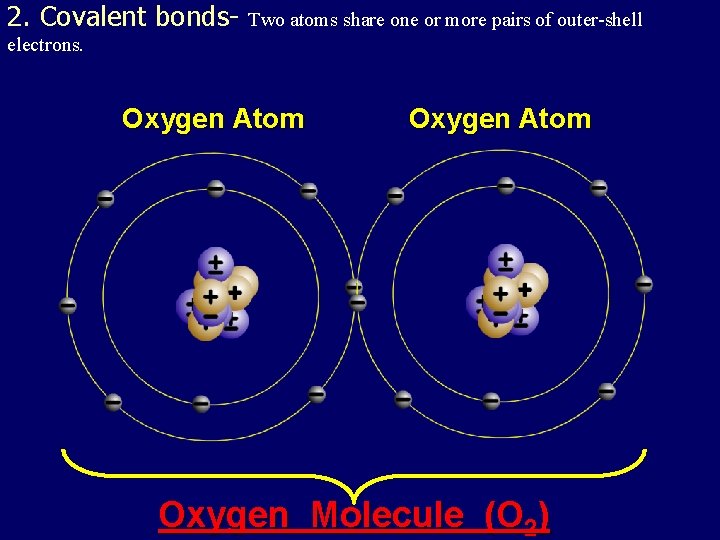
2. Covalent bonds- Two atoms share one or more pairs of outer-shell electrons. Oxygen Atom Oxygen Molecule (O 2)

COVALENT BONDS in compounds when electrons are shared but shared unequally H 2 O
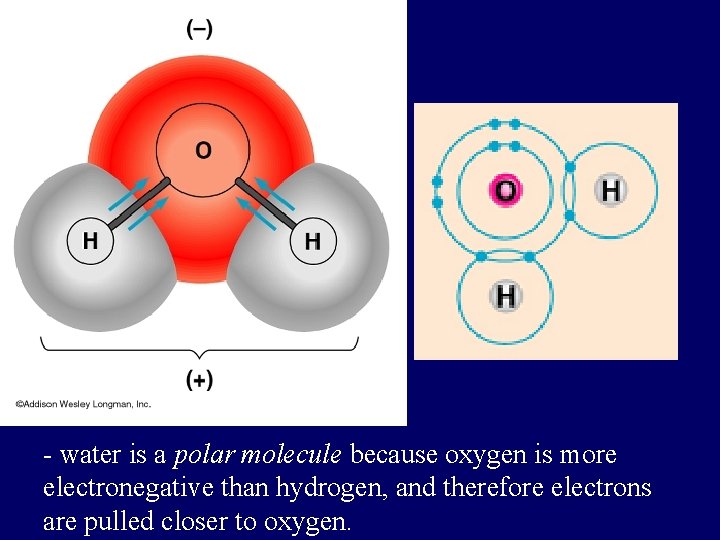
- water is a polar molecule because oxygen is more electronegative than hydrogen, and therefore electrons are pulled closer to oxygen.

METALLIC BOND bond found in metals; holds metal atoms together very strongly

Metallic Bond • Formed between atoms of metallic elements • Electron cloud around atoms • Good conductors at all states, lustrous, very high melting points • Examples; Na, Fe, Al, Au, Co
Ionic Bond, A Sea of Electrons

Metals Form Alloys Metals do not combine with metals. They form Alloys which is a solution of a metal in a metal. Examples are steel, brass, bronze and pewter.


Compounds and Their Bonds Ionic Compounds Naming Ionic Formulas

Ionic Compounds Attraction between + ions and - ions Electrons go from metals to nonmetals electron transfer metal nonmetal ion+ Electrons lost = Electrons gain ion–

Formulas of Ionic Compounds Formulas of ionic compounds are determined from the charges on the ions atoms Na + F : sodium fluorine Charge balance: ions – Na+ : F : Na. F sodium fluoride 1+ 1 - formula = 0

Writing a Formula Write the formula for the ionic compound that will form between Ba 2+ and Cl. Solution: 1. Balance charge with + and – ions 2. Write the positive ion of metal first, and the negative ion Ba 2+ Cl Cl 3. Write the number of ions needed as subscripts Ba. Cl 2

Learning Check Write the correct formula for the compounds containing the following ions: A. Na+, S 21) Na. S 2) Na 2 S 3) Na. S 2 B. Al 3+, Cl 1) Al. Cl 3 2) Al. Cl 3) Al 3 Cl C. Mg 2+, N 31) Mg. N 2) Mg 2 N 3 3) Mg 3 N 2

Solution A. Na+, S 22) Na 2 S B. Al 3+, Cl 1) Al. Cl 3 C. Mg 2+, N 33) Mg 3 N 2

Naming Binary Ionic Compounds Contain 2 different elements Name the metal first, then the nonmetal as -ide. Use name of a metal with a fixed charge Groups 1 A, 2 A, 3 A and Ag, Zn, and Cd Examples: Na. Cl sodium chloride Zn. I 2 zinc iodide Al 2 O 3 aluminum oxide

Learning Check Complete the names of the following binary compounds: Na 3 N sodium ________ KBr potassium ________ Al 2 O 3 aluminum ________ Mg. S _____________

Solution Complete the names of the following binary compounds: Na 3 N sodium nitride KBr potassium bromide Al 2 O 3 aluminum oxide Mg. S magnesium sulfide
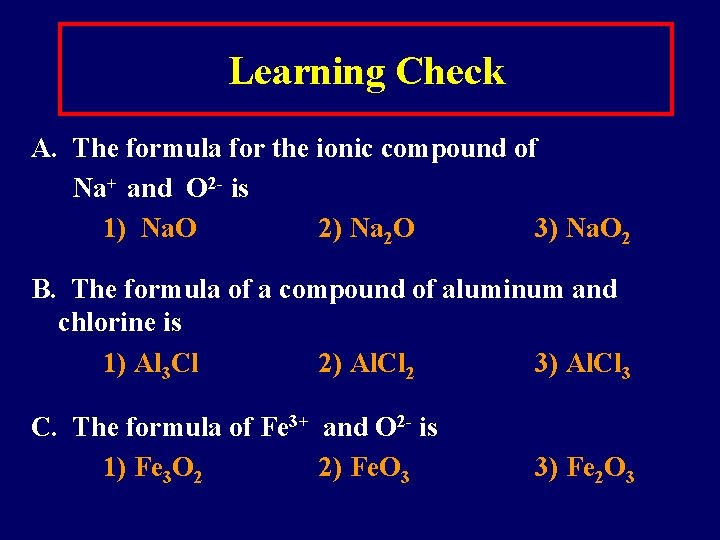
Learning Check A. The formula for the ionic compound of Na+ and O 2 - is 1) Na. O 2) Na 2 O 3) Na. O 2 B. The formula of a compound of aluminum and chlorine is 1) Al 3 Cl 2) Al. Cl 2 3) Al. Cl 3 C. The formula of Fe 3+ and O 2 - is 1) Fe 3 O 2 2) Fe. O 3 3) Fe 2 O 3

Solution A. The formula for the ionic compound of Na+ and O 2 - is 2) Na 2 O B. The formula of a compound of aluminum and chlorine is 3) Al. Cl 3 C. The formula of Fe 3+ and O 2 - is 3) Fe 2 O 3

Learning Check Name the following compounds: A. Ca. O 1) calcium oxide 2) calcium(I) oxide 3) calcium (II) oxide B. Sn. Cl 4 1) tin tetrachloride 3) tin(IV) chloride C. Co 2 O 3 1) cobalt oxide 3) cobalt trioxide 2) tin(II) chloride 2) cobalt (III) oxide

Solution Name the following compounds: A. Ca. O 1) calcium oxide B. Sn. Cl 4 3) tin(IV) chloride C. Co 2 O 3 2) cobalt (III) oxide

Q 1. Name a metal in group 1 that is in the same period as Magnesium. Q 2. Name a metal in group 2 that in the same period as Lithium. Q 3. Name a non-metal in the same group as Nitrogen. Q 4. Name a noble gas in the same period as Oxygen. Q 5. Name a gas in group 7 that is in the same period as Aluminium.

Objective: 1. Learn the scientific method for naming compounds. 2. There are three rules for naming compounds: the “ide” rule, the “ate” rule and the “same” rule.

Rule 1: When two elements combine the ending is usually ” ……………ide”. metal goes first Sodium Chloride Magnesium Oxide Iron Sulphide Cl Na Mg Fe O S
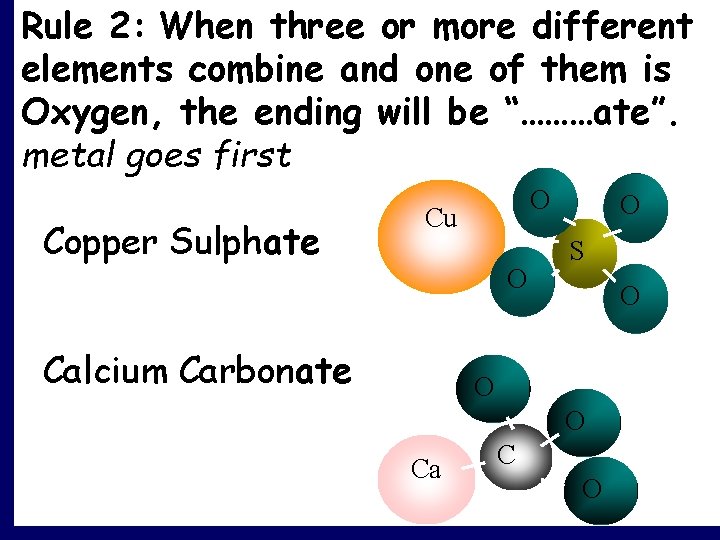
Rule 2: When three or more different elements combine and one of them is Oxygen, the ending will be “………ate”. metal goes first Copper Sulphate O Cu O Calcium Carbonate O S O O O Ca C O
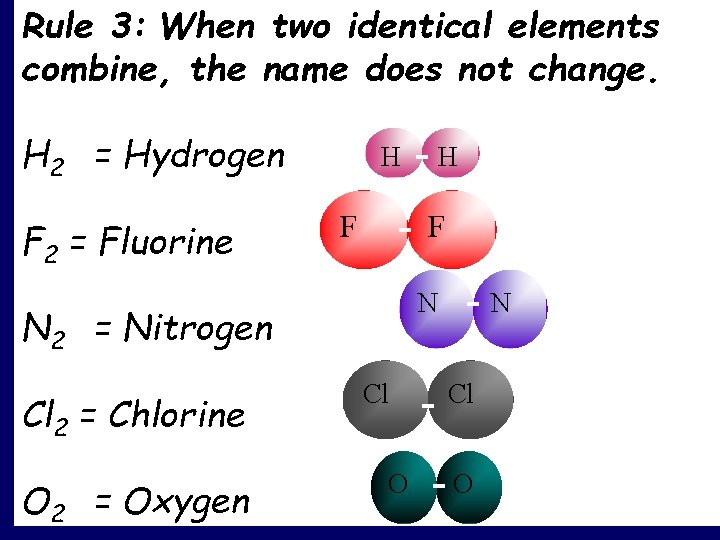
Rule 3: When two identical elements combine, the name does not change. H 2 = Hydrogen F 2 = Fluorine H F F N N 2 = Nitrogen Cl 2 = Chlorine O 2 = Oxygen H Cl O N Cl O

Name the following compound Na 2 O

Name the following compound Cu. SO 4

Name the following compound Mg. SO 4

Name the following compound Zn. O

Name the following compound Mg. Cl 2

Name the following molecule H 2

Name the following compound KI

Name the following compound Ag. NO 3
- Slides: 63