Illinois Institute of Technology Physics 561 Radiation Biophysics

Illinois Institute of Technology Physics 561 Radiation Biophysics Lecture 6: Modifiers of the Response to Radiation; Response of Tumors to Radiation Andrew Howard 19 June 2014 11/2/2020 Response Modifiers; Tumors p. 1 of 75

Class Plan u u u Chemical modifiers of Radiation Response Cell types and cell cycles Tumors – – – Definitions Multistage model of carcinogenesis Radiosensitivity 11/2/2020 u Tumors, concluded – – u u Tools for studying response of tumors Role of oxygenation in tumor response Homework hints Radiosensitivity of normal tissues: Experimental systems Response Modifiers; Tumors p. 2 of 75
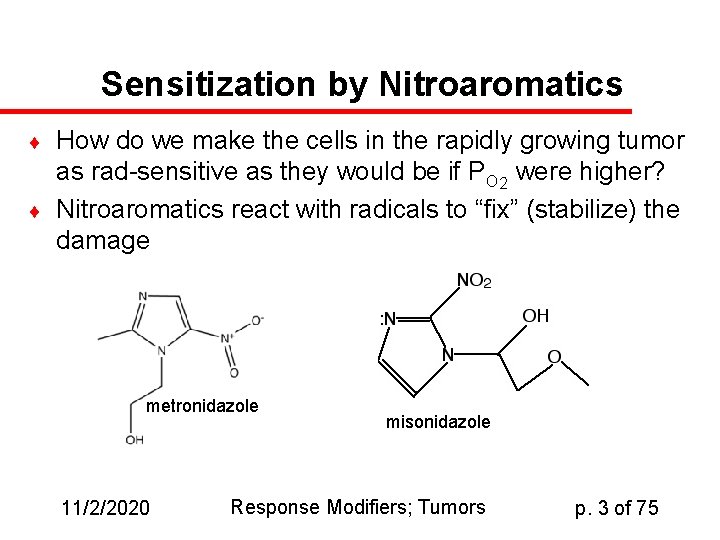
Sensitization by Nitroaromatics How do we make the cells in the rapidly growing tumor as rad-sensitive as they would be if PO 2 were higher? Nitroaromatics react with radicals to “fix” (stabilize) the damage metronidazole 11/2/2020 misonidazole Response Modifiers; Tumors p. 3 of 75

Nitroaromatic action u u u Like oxygen, nitroaromatics react with short-lived radicals to produce longer-lived and therefore more reactive radicals Concentrations required to enhance radiosensitivity are much higher than with O 2 These compounds have other applications, but they can be used therapeutically as potentiators of radiation damage 11/2/2020 Response Modifiers; Tumors p. 4 of 75
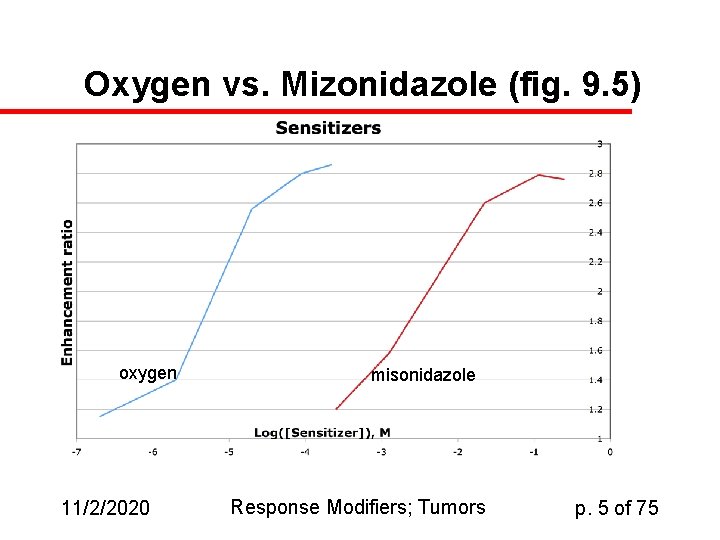
Oxygen vs. Mizonidazole (fig. 9. 5) oxygen 11/2/2020 misonidazole Response Modifiers; Tumors p. 5 of 75

5 -Halogen-substituted pyrimidines u u These are molecules that resemble thymine The halogen at the 5 - position looks like the methyl group in thymine and can be incorporated in place of thymine in DNA Most common: 5 -bromodeoxyuridine Sensitization produced by ready reaction with the aqueous electron 11/2/2020 Response Modifiers; Tumors p. 6 of 75
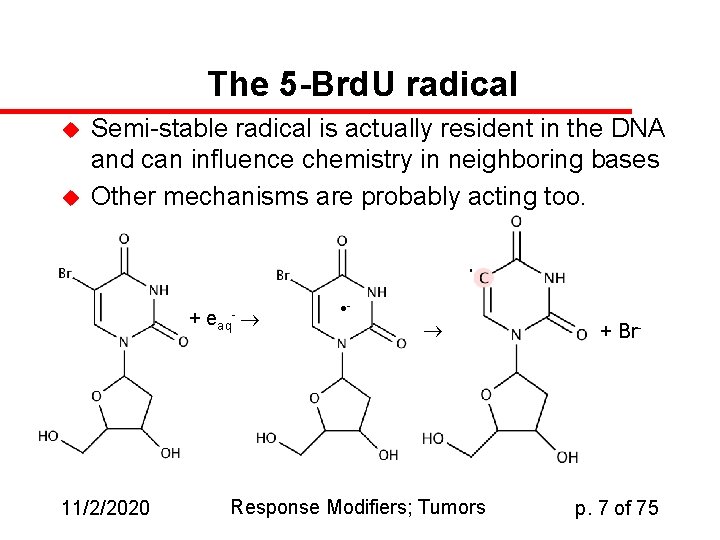
The 5 -Brd. U radical u u Semi-stable radical is actually resident in the DNA and can influence chemistry in neighboring bases Other mechanisms are probably acting too. + eaq 11/2/2020 - - Response Modifiers; Tumors + Br- p. 7 of 75

Cell Death u u Clonogenic cell death: inability to produce several generations’ worth of progeny Acute pathological cell death: necrosis – – u Cells typically swell, then lyse Accompanied by inflammation Apoptosis – – – Programmed cell death Shrinkage, fragmentation, phagocytosis p 53 is activator of genes that regulate it 11/2/2020 Response Modifiers; Tumors p. 8 of 75
Gilbert & Lajtha’s cell types See next slide for explanation! A D 11/2/2020 B E Response Modifiers; Tumors C F p. 9 of 75
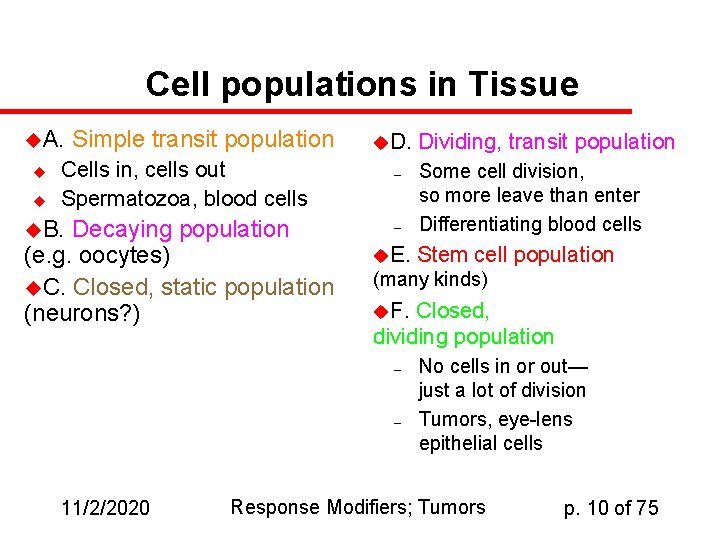
Cell populations in Tissue u. A. u u Simple transit population Cells in, cells out Spermatozoa, blood cells u. B. Decaying population (e. g. oocytes) u. C. Closed, static population (neurons? ) u. D. – – u. E. Some cell division, so more leave than enter Differentiating blood cells Stem cell population (many kinds) u. F. Closed, dividing population – – 11/2/2020 Dividing, transit population No cells in or out— just a lot of division Tumors, eye-lens epithelial cells Response Modifiers; Tumors p. 10 of 75
Cell population kinetics u u u Cell types that divide are the most sensitive. Cells are most sensitive during G 2 and M, so cells that spend a lot of time in G 2 and M are more sensitive If a cell population is exposed to radiation, the outcome depends on there being an adequate number of (clonogenically) surviving cells. 11/2/2020 Response Modifiers; Tumors p. 11 of 75

Growth Fraction u u Lajtha (1963): described “G 0” phase in cell cycle: cell is not engaged in proliferation but could later re-enter proliferative stage Growth fraction is defined as fraction of total cellular population that is clonogenically competent and actually in the process of DNA replication and cell division. Measurement: uses 3 H-thymidine uptake Significance: cells in G 0 have time to repair DNA damage – – Works even if [repair enzymes] is low during G 0 This is suspected but not proven 11/2/2020 Response Modifiers; Tumors p. 12 of 75
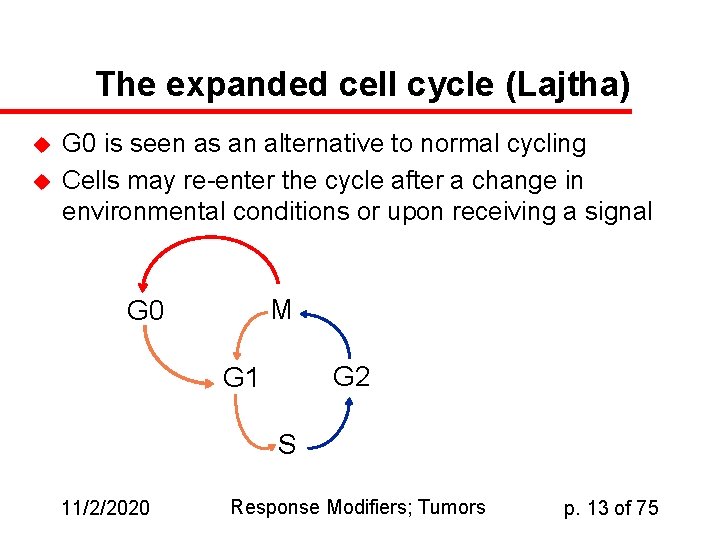
The expanded cell cycle (Lajtha) u u G 0 is seen as an alternative to normal cycling Cells may re-enter the cycle after a change in environmental conditions or upon receiving a signal M G 0 G 2 G 1 S 11/2/2020 Response Modifiers; Tumors p. 13 of 75

Why study tumors in this class u This is a relevant area of research for at least two reasons: – – u Ionizing radiation cause or contribute to tumorigenesis Ionizing radiation can kill tumor cells, sometimes preferentially relative to healthy cells; so it’s used in cancer therapy So we want to understand the interaction between ionizing radiation and tumors for both reasons 11/2/2020 Response Modifiers; Tumors p. 14 of 75

What is a Tumor? u u A tumor is an mass of undifferentiated or minimally differentiated tissue growing amidst differentiated tissue. A tumor may be malignant, i. e. growing uncontrollably and with a propensity for spreading to other tissues. Or it may be benign, i. e. growing slowly or not at all and without a propensity for spreading The phenomenon of spreading is called metastasis. 11/2/2020 Response Modifiers; Tumors p. 15 of 75

Cancer is the growth of one or more malignant tumors. The process by which cancer develops is called carcinogenesis. The causes, rapidity of onset, course of disease, treatment possibilities, and likely outcomes of cancer depend enormously on what tissue is being attacked, i. e. on the kind of cells from which the tumor grew. 11/2/2020 Response Modifiers; Tumors p. 16 of 75
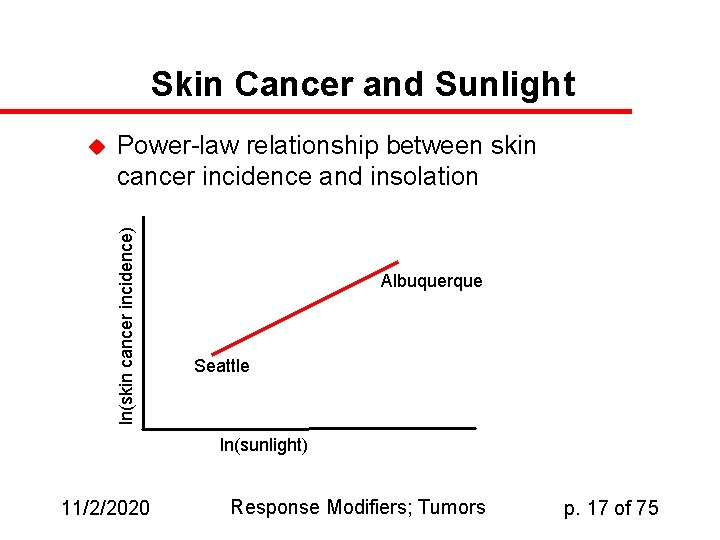
Skin Cancer and Sunlight Power-law relationship between skin cancer incidence and insolation ln(skin cancer incidence) u Albuquerque Seattle ln(sunlight) 11/2/2020 Response Modifiers; Tumors p. 17 of 75
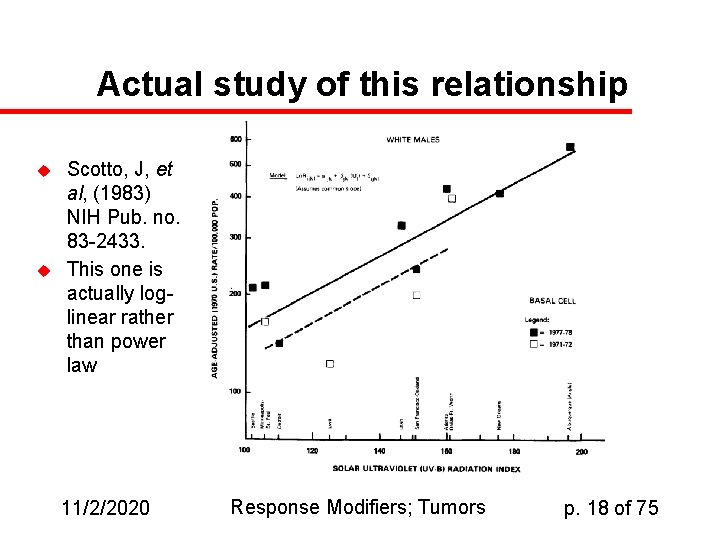
Actual study of this relationship u u Scotto, J, et al, (1983) NIH Pub. no. 83 -2433. This one is actually loglinear rather than power law 11/2/2020 Response Modifiers; Tumors p. 18 of 75
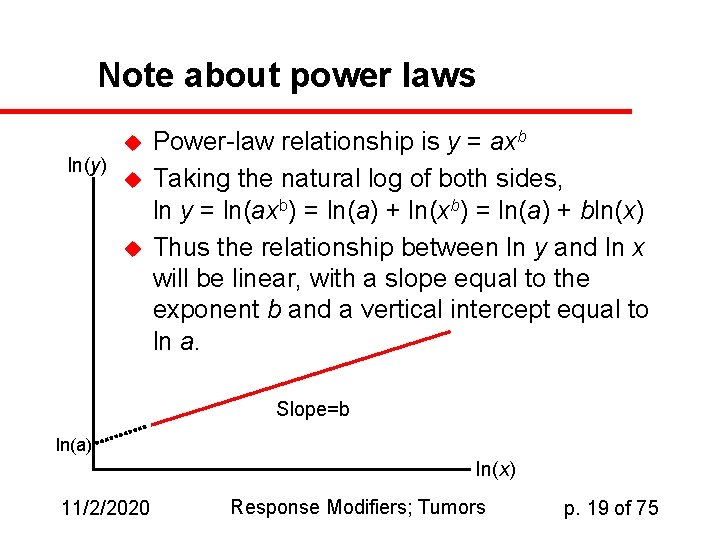
Note about power laws u ln(y) u u Power-law relationship is y = axb Taking the natural log of both sides, ln y = ln(axb) = ln(a) + ln(xb) = ln(a) + bln(x) Thus the relationship between ln y and ln x will be linear, with a slope equal to the exponent b and a vertical intercept equal to ln a. Slope=b ln(a) ln(x) 11/2/2020 Response Modifiers; Tumors p. 19 of 75

Characteristics of Cancer Cells u u Cancer cells lack differentiation Cancer cells have abnormal nuclei – – u u May have an abnormal number of chromosomes Gene amplification (abnormal # of copies of specific genes) is common Not subject to apoptotic controls Sometimes they have enhanced mitotic rates Cancer cells form tumors Cancer cells metastasize 11/2/2020 Response Modifiers; Tumors p. 20 of 75
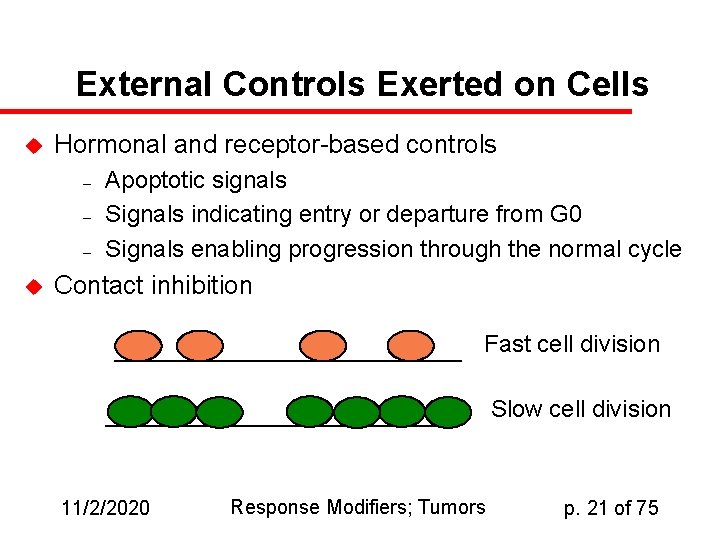
External Controls Exerted on Cells u Hormonal and receptor-based controls – – – u Apoptotic signals Signals indicating entry or departure from G 0 Signals enabling progression through the normal cycle Contact inhibition Fast cell division Slow cell division 11/2/2020 Response Modifiers; Tumors p. 21 of 75

Cell types and cancer u u Epithelial cells (skin, digestive tract, tracheal lining, glands, . . . ) give rise to carcinomas. Connective tissue cells (bone, cartilage) give rise to sarcomas. Blood cells give rise to leukemia. Lymphatic tissue gives rise to lymphomas. 11/2/2020 Response Modifiers; Tumors p. 22 of 75

Multistage model of carcinogenesis u u u Fundamental notion is that the formation of malignancies depends on a sequence of events, some of which are mutational and some of which are not. Specifically: Initiation is a set of mutational events (~5 in a cell? ) through which the DNA of a cell is transformed without its being unable to replicate Promotion is the process by which morphological and functional characteristics of the mutagenized cell change * * * clonal and (possibly) uncontrolled growth begins here * * * Progression involves invasion of the tumor into tissues separate from that in which the initial event occurs 11/2/2020 Response Modifiers; Tumors p. 23 of 75

Time-Course of Cancer u Steps in causation: – – – u Initiation Promotion Progression S. D. Hursting Steps in clinical outcome: – – Exposure Latency Onset Disease Hursting, et al, (1999) JNCI 91: 215 11/2/2020 Response Modifiers; Tumors p. 24 of 75

Initiation is typically a series of mutational events, often single-base changes in DNA in a single cell. u The clonal hypothesis states that cancer typically arises from clonal growth out of a single damaged cell. u In most cases it does appear that the number of mutations that have to occur in order for a tumor to grow out of a single cell is more than one. u Initiation events can arise over a short time span if the exposure to the mutagen is intense and short (or if only one event is required). u 11/2/2020 Response Modifiers; Tumors p. 25 of 75
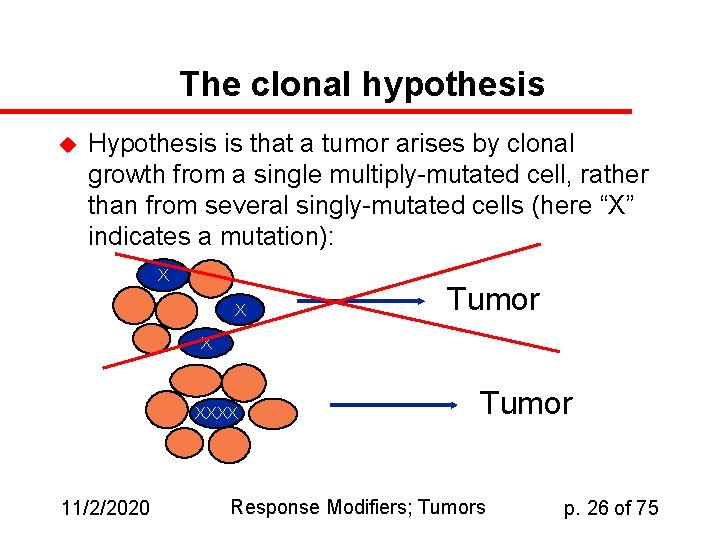
The clonal hypothesis u Hypothesis is that a tumor arises by clonal growth from a single multiply-mutated cell, rather than from several singly-mutated cells (here “X” indicates a mutation): X X Tumor X XXXX 11/2/2020 Tumor Response Modifiers; Tumors p. 26 of 75

Promotion u u u Promotion is a process in which metabolic and then morphological changes in the mutated cell occur. It does not typically involve mutations in the affected cell, but rather interference with some of the surveillance mechanisms by which these metabolic and morphological changes are controlled. Among the systems involved are the arachidonic acid cascade, by which the cell’s differentiation capacity is regulated; and apoptosis factors like p 53. 11/2/2020 Response Modifiers; Tumors p. 27 of 75
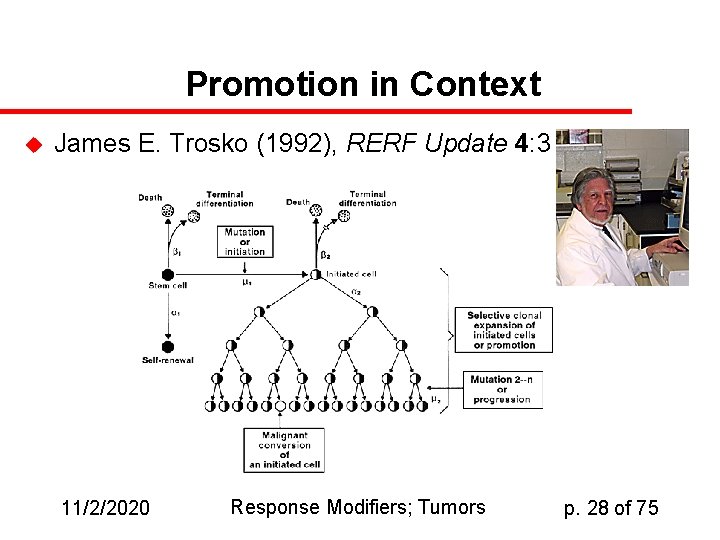
Promotion in Context u James E. Trosko (1992), RERF Update 4: 3 11/2/2020 Response Modifiers; Tumors p. 28 of 75
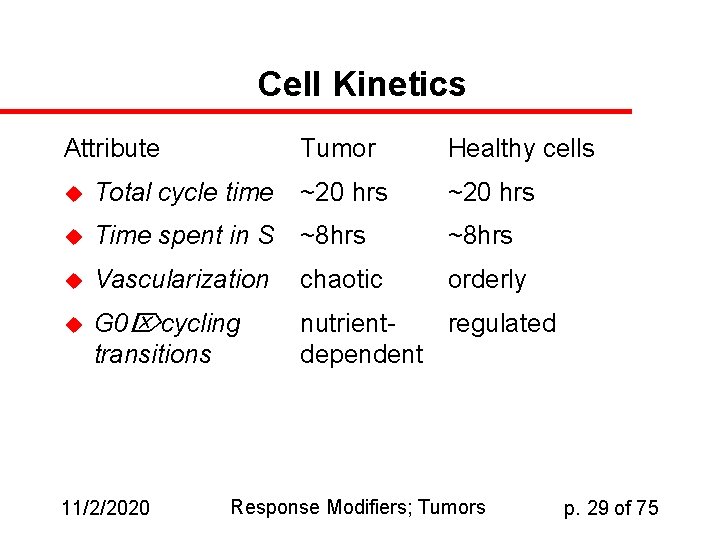
Cell Kinetics Attribute Tumor Healthy cells u Total cycle time ~20 hrs u Time spent in S ~8 hrs u Vascularization chaotic orderly u G 0 cycling transitions nutrientregulated dependent 11/2/2020 Response Modifiers; Tumors p. 29 of 75

Modeling Sensitivity in Tumors u Why do we bother? Because it enables us to optimize treatment regimens when exposing patients to radiation – – u Maximize cell killing in the tumor Minimize damage to normal tissues Also provides test-bed for understanding interactions between tissues and radiation in general 11/2/2020 Response Modifiers; Tumors p. 30 of 75

Hewitt Dilution Assay u u Tumor cells grown in peritoneal (gut) cavity of mouse--“ascites” tumor Tumor cells can be harvested and injected into recipient mice Inject varying number of tumor cells and plot fraction killed against number of cells injected Result: if you pre-irradiate the tumor cells, they don’t kill as many hosts. 11/2/2020 Response Modifiers; Tumors p. 31 of 75
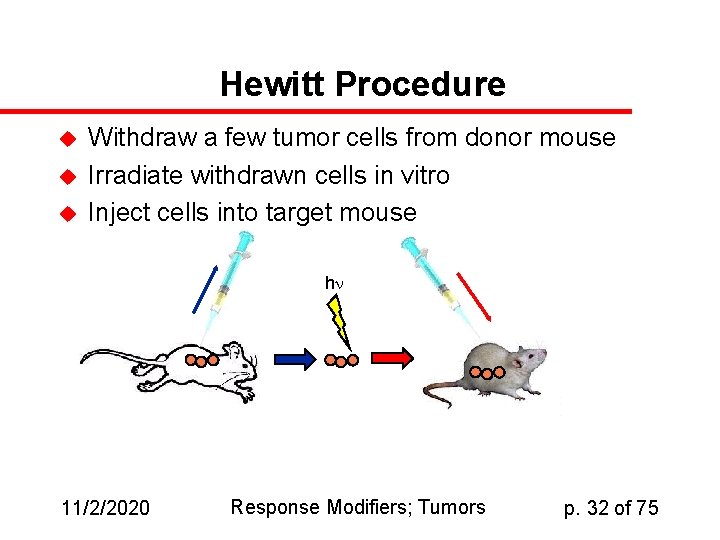
Hewitt Procedure u u u Withdraw a few tumor cells from donor mouse Irradiate withdrawn cells in vitro Inject cells into target mouse h 11/2/2020 Response Modifiers; Tumors p. 32 of 75
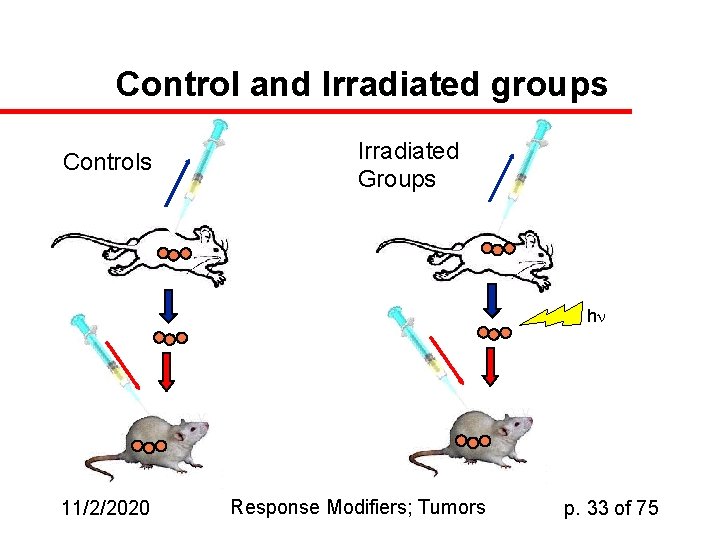
Control and Irradiated groups Controls Irradiated Groups h 11/2/2020 Response Modifiers; Tumors p. 33 of 75
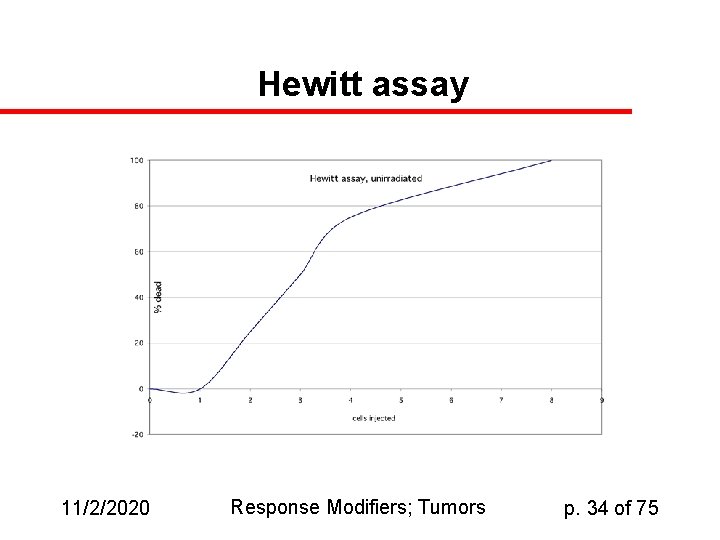
Hewitt assay 11/2/2020 Response Modifiers; Tumors p. 34 of 75
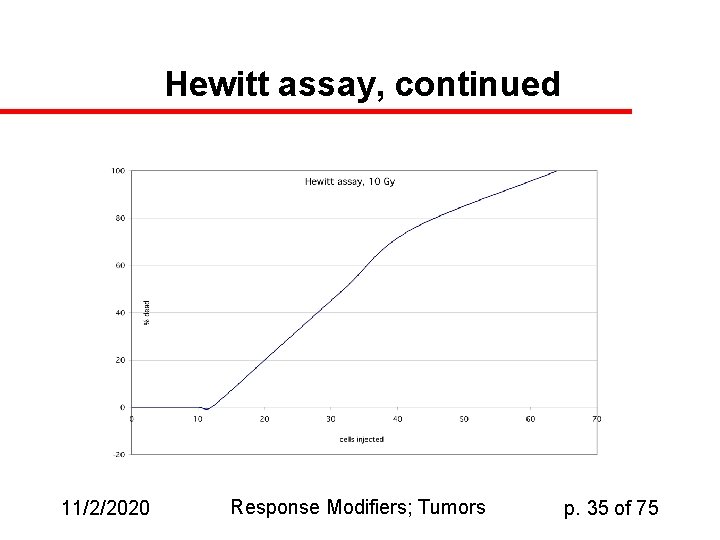
Hewitt assay, continued 11/2/2020 Response Modifiers; Tumors p. 35 of 75

Hewitt assay: analysis u u LD 50 is used to construct survival curve In our example, 3 cells are enough to kill half the host animals if no radiation is used; 32 are required if 10 Gy are used Evidently S/S 0 = 3/32 = 0. 094 of the initial cells were functional enough to kill the host. We can calculate similar numbers for each dose level and calculate a dose-response curve (dose vs. log(S/S 0), perhaps under multiple conditions. 11/2/2020 Response Modifiers; Tumors p. 36 of 75
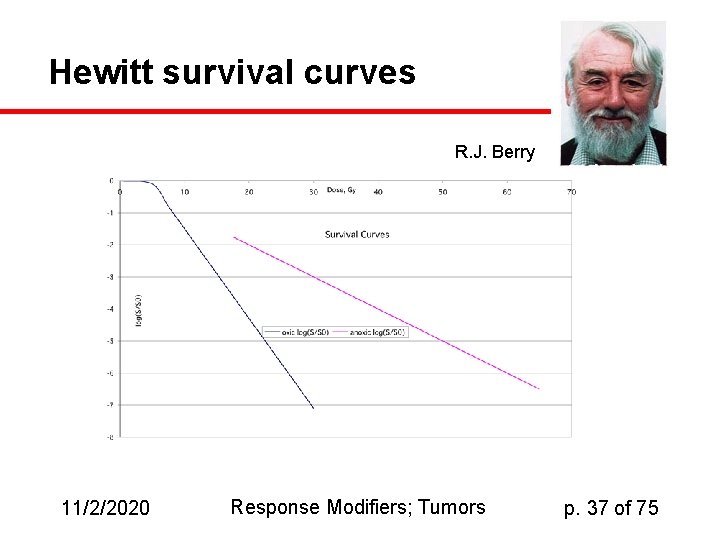
Hewitt survival curves R. J. Berry 11/2/2020 Response Modifiers; Tumors p. 37 of 75

OER in Hewitt Assay u Remember that OER is defined as ratio of dose required to get a given effect in the absence of oxygen to the dose required in the presence of oxygen. Since you need more dose to get the same effect if oxygen is absent, the OER is greater than one. For the data in fig. 10. 3, the OER is about 2. 2, since we need about 40 Gy of dose to damage anoxic cells the same amount as would require only 18 Gy with oxic cells. (40/18=2. 2) 11/2/2020 Response Modifiers; Tumors p. 38 of 75

Lung Colony Assay u u u Tumor is injected into a recipient mouse’s lung Number of tumor colonies in lung is counted Tumor may be irradiated: – – u u u In vivo in the donor mouse After dissection and cell dissociation but before injection Linear relationship between number of cells injected and number of colonies counted 10 -50 X enhancement in number of colonies if heavily irradiated, nonclonogenic cells are injected Irradiation increases the # of cells required to produce a given number of colonies 11/2/2020 Response Modifiers; Tumors p. 39 of 75
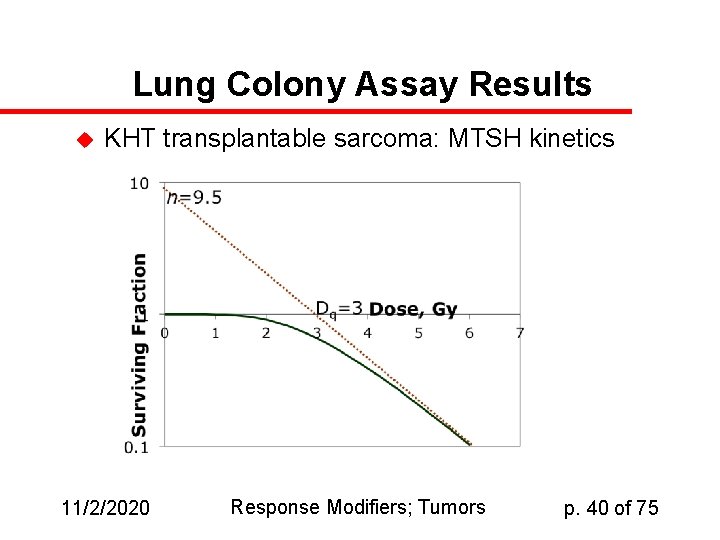
Lung Colony Assay Results u KHT transplantable sarcoma: MTSH kinetics 11/2/2020 Response Modifiers; Tumors p. 40 of 75
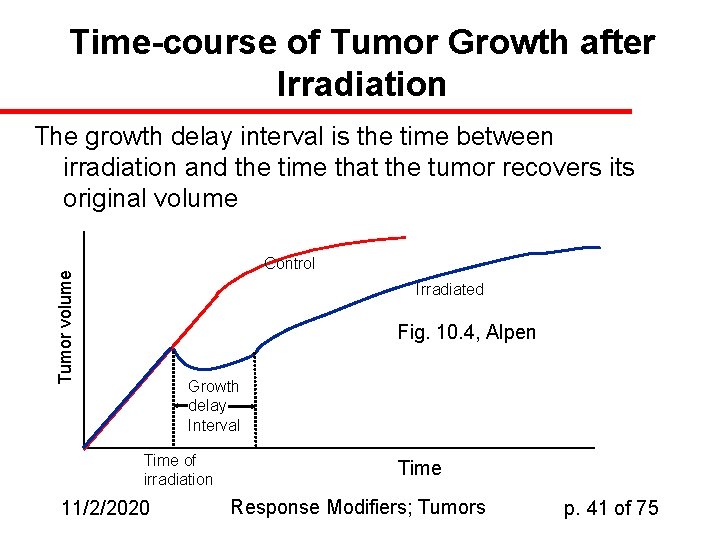
Time-course of Tumor Growth after Irradiation The growth delay interval is the time between irradiation and the time that the tumor recovers its original volume Tumor volume Control Irradiated Fig. 10. 4, Alpen Growth delay Interval Time of irradiation 11/2/2020 Time Response Modifiers; Tumors p. 41 of 75
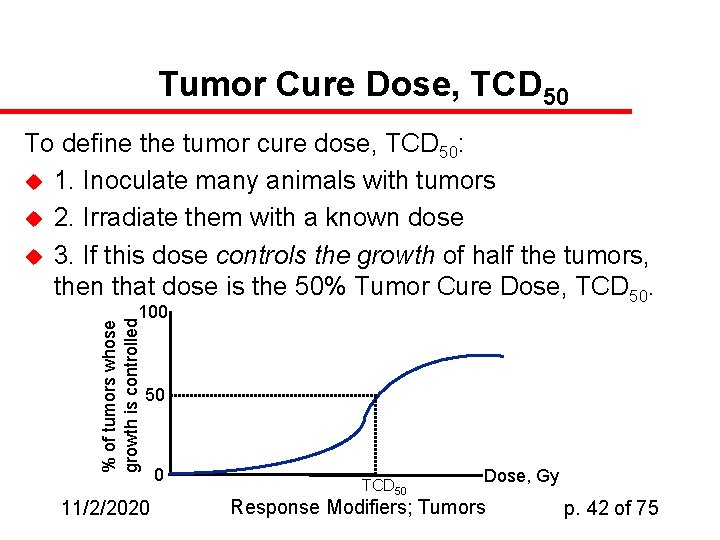
Tumor Cure Dose, TCD 50 To define the tumor cure dose, TCD 50: u 1. Inoculate many animals with tumors u 2. Irradiate them with a known dose u 3. If this dose controls the growth of half the tumors, then that dose is the 50% Tumor Cure Dose, TCD 50. % of tumors whose growth is controlled 100 50 11/2/2020 0 TCD 50 Dose, Gy Response Modifiers; Tumors p. 42 of 75

How cancer cell lines respond to radiation u Transformed (immortalized) cell lines fairly similar – – – u Reasonable agreement with MTSH kinetics D 0 values range over a factor of 2 -3 Gy n values between 5 and 20 Exceptions: DNA-repair-deficient cells – – – Xeroderma pigmentosum Ataxia telangectasia Results here: n~ 1, D 0 smaller than otherwise 11/2/2020 Response Modifiers; Tumors p. 43 of 75

How fresh tumors respond to radiation u u Results from fresh tumors are somewhat different: Fit LQ models somewhat better than MTSH 3 categories: high, medium, and low sensitivity Correlation with responsiveness to radiotherapy 11/2/2020 Response Modifiers; Tumors p. 44 of 75
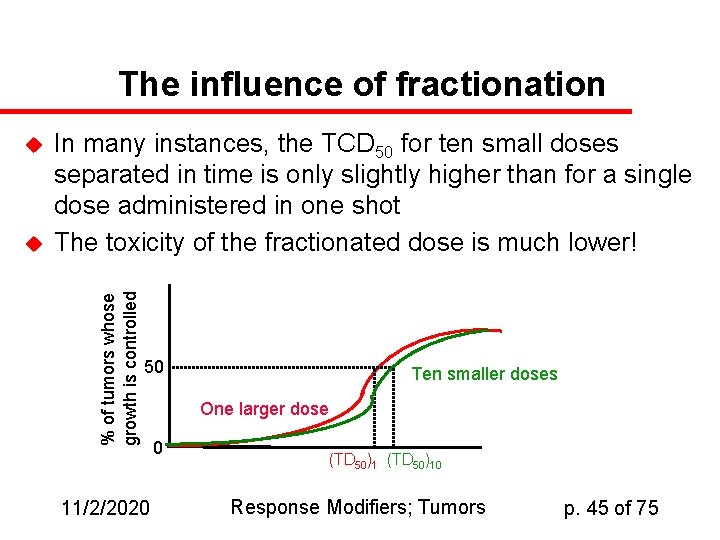
The influence of fractionation u In many instances, the TCD 50 for ten small doses separated in time is only slightly higher than for a single dose administered in one shot The toxicity of the fractionated dose is much lower! % of tumors whose growth is controlled u 50 11/2/2020 Ten smaller doses One larger dose 0 (TD 50)10 Response Modifiers; Tumors p. 45 of 75
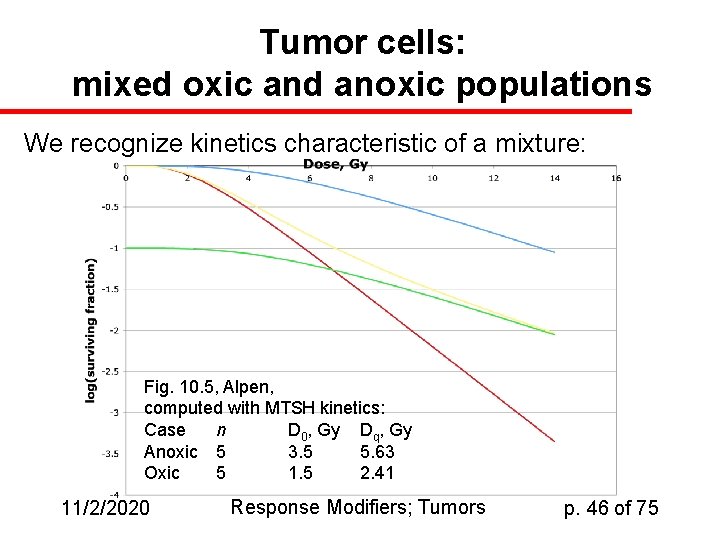
Tumor cells: mixed oxic and anoxic populations We recognize kinetics characteristic of a mixture: Fig. 10. 5, Alpen, computed with MTSH kinetics: Case n D 0, Gy Dq, Gy Anoxic 5 3. 5 5. 63 Oxic 5 1. 5 2. 41 11/2/2020 Response Modifiers; Tumors p. 46 of 75
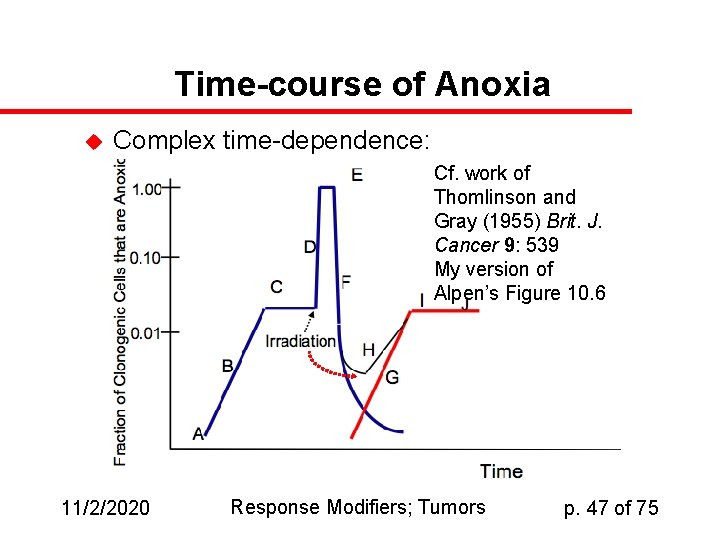
Time-course of Anoxia u Complex time-dependence: Cf. work of Thomlinson and Gray (1955) Brit. J. Cancer 9: 539 My version of Alpen’s Figure 10. 6 11/2/2020 Response Modifiers; Tumors p. 47 of 75

Other influences on tumor response u COX-2 inhibitors potentiate radiation’s effects on tumors: – – – u Cyclooxygenase-II is an enzyme that primarily influences inflammatory states and cancer Overexpression of this enzyme tends to make tumors grow and metabolize more aggressively So: inhibiting this enzyme makes the radiation work better E. Nakata et al. (2004) Int. J. Radiat. Oncol. Biol. Phys. 58: 369 11/2/2020 Response Modifiers; Tumors p. 48 of 75

… still others u The chemical modifiers of tumor response discussed in the previous chapter are pertinent – – u u Halogenated pyrimidines Nitroaromatics Hypoxia inducers (e. g. tirapazamine) Erythropoietin (increases production of blood cells to offset anemia caused by radiation therapy) 11/2/2020 Response Modifiers; Tumors p. 49 of 75

An upcoming assignment u u Alpen, chapter 10, problem 1. I’m not going to write this one out: it’s a lot of text. Assume that ionizing radiation exerts its tumorigenic effects primarily through mutational events. Assume further that cigarette tar contains large numbers of cancer promoters. Which scenario would you expect would cause a higher incidence of cancer, and why? : – – Irradiation followed by ten years of smoking Ten years of smoking followed by irradiation 11/2/2020 Response Modifiers; Tumors p. 50 of 75

Homework problem 9. 1 u u See next few slides for specifics General point: you don’t actually need the MTSH n and D 0 values to solve the problem—just the OER values and the recognition that in this situation the OER at high [O 2 ] values is the m value (see eqn. 9. 5) Then if you plot OER against [O 2], find the point where S/SN = (m+1)/2 = 1. 905. That point is at [O 2] = 0. 61%. Ratio of type 1 to type 2 damage = n 1/n 2 = m-1. 11/2/2020 tumor and normal-tissue responses p. 51 of 75

Alpen, Chapter 9, Problem 1 u A mammalian cell line has a D 0 of 120 c. Gy and an extrapolation number of 12. The OER is found to be of a dose modifying character with a value of 2. 81 at high oxygen concentration. What is the value of m in the Alper-Howard-Flanders equation? The following data have been found for this cell line for OER as a function of oxygen partial pressure: 11/2/2020 tumor and normal-tissue responses p. 52 of 75
![Problem 9. 1, continued [O 2], % 0. 1353 0. 223 0. 2865 0. Problem 9. 1, continued [O 2], % 0. 1353 0. 223 0. 2865 0.](http://slidetodoc.com/presentation_image/3dc4fb5d43e1e08c25a4f0b2fec0290f/image-53.jpg)
Problem 9. 1, continued [O 2], % 0. 1353 0. 223 0. 2865 0. 3679 0. 6065 OER 1. 10 1. 20 1. 33 1. 64 1. 88 [O 2], % 0. 7788 1. 00 1. 75 2. 50 OER 2. 19 2. 59 2. 77 2. 81 • What is the value of K in the same expression? • What is the ratio of type 1 to type 2 damage in this cell line? 11/2/2020 tumor and normal-tissue responses p. 53 of 75
![Remember how this works: u u u Alpen, p. 205: When [O 2] is Remember how this works: u u u Alpen, p. 205: When [O 2] is](http://slidetodoc.com/presentation_image/3dc4fb5d43e1e08c25a4f0b2fec0290f/image-54.jpg)
Remember how this works: u u u Alpen, p. 205: When [O 2] is very much larger than K, S/SN = (m[O 2] + K) / ([O 2] + K) = (m[O 2]) / ([O 2]) = m Therefore we can compute m by finding the asymptotic value of S/SN (for [O 2] very large) Then we recognize that if K = [O 2], then our general formula becomes S/SN = (m[O 2] + [O 2]) / ([O 2] + [O 2]) = (m+1) / 2 So we find the value of [O 2] for which S/SN = (m+1)/2 and that value of [O 2] will be K. Shazam. 11/2/2020 tumor and normal-tissue responses p. 54 of 75

Computing n 1/n 2 u u Remember that the ratio of type-1 to type-2 lesions is n 1/n 2 = (m - 1) and K = krep / kfix So once we know m we can compute n 1/n 2. 11/2/2020 tumor and normal-tissue responses p. 55 of 75

Homework for early July u Alpen, chapter 10, problem 1: Hewitt Dilution Assay with mortality rates listed for doses of 0, 1, 2, 4, 5, 6, and 8 Gy. Construct the curves for animal lethality versus dose of injected cells for each of the irradiation doses given. Construct the derived survival versus dose curve for the irradiation of the line of lymphoma cells. Estimate D 0 and n for an MTSH model. Then plot the survival curve in LQ format using the linearized form of the linear-quadratic expression. Which fits better? 11/2/2020 tumor and normal-tissue responses p. 56 of 75
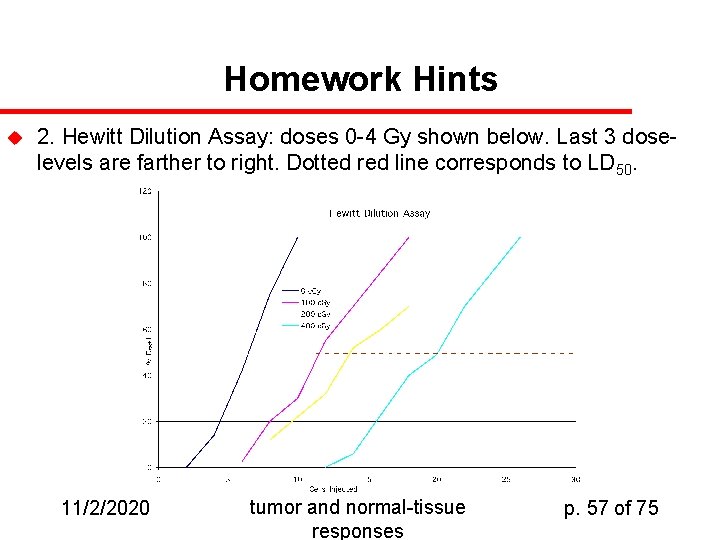
Homework Hints u 2. Hewitt Dilution Assay: doses 0 -4 Gy shown below. Last 3 doselevels are farther to right. Dotted red line corresponds to LD 50. 11/2/2020 tumor and normal-tissue responses p. 57 of 75

Homework Hints, Continued 2. Hewitt dilution assay: from curves shown, we can estimate the LD 50 for each radiation dose: Dose, Gy LD 50 0 6. 5 1 11. 6 2 13. 8 4 20. 1 5 65 6 159 8 598 So S/S 0 = (LD 50 at a given dose)/ (LD 50 at Dose=0). 11/2/2020 tumor and normal-tissue responses p. 58 of 75
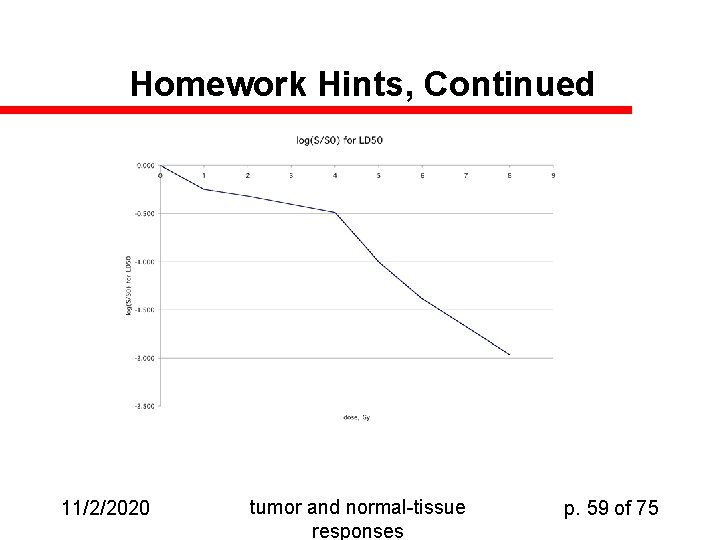
Homework Hints, Continued 11/2/2020 tumor and normal-tissue responses p. 59 of 75

Linearized Linear-Quadratic Model u u u Recall that in the LQ model S = exp(-p( D + D 2)) Note that this form is the one articulated in the “molecular model” section of the text (p. 146 -151) The fudge-parameter p is the biological effectiveness factor for double-strand breaks (is that useful? ) So ln S = -p( D + D 2) = (-p( + D)) D Therefore ln S / D = -p -p D Therefore a plot of ln S / D against D should have a slope of -p and a Y intercept of -p. 11/2/2020 tumor and normal-tissue responses p. 60 of 75
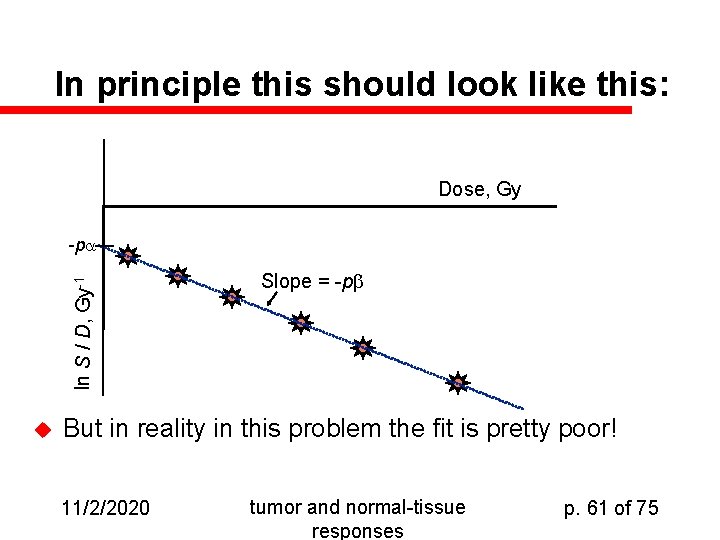
In principle this should look like this: Dose, Gy ln S / D, Gy-1 -p u Slope = -p But in reality in this problem the fit is pretty poor! 11/2/2020 tumor and normal-tissue responses p. 61 of 75

Non-Alpen problem Assume that ionizing radiation exerts its tumorigenic effects primarily through mutational events. Assume further the cigarette tar contains large numbers of cancer promoters. Which scenario would you expect would cause a higher incidence of cancer, and why? – – Irradiation followed by ten years of smoking Ten years of smoking followed by irradiation 11/2/2020 tumor and normal-tissue responses p. 62 of 75

So let’s get back to chapter 10 u u We’ve been discussing models for radiosensitivity of tumor cells This is important to oncologists and to general radiation biologists, but (perhaps) for different reasons: – – u The oncologist wants to know how to exploit differential radiosensitivity of tumor cells The radiation biologist hopes that results derived from studying tumor cells (which are relatively easy to culture) will help us to understand normal cells So: now we move on to those normal cells. 11/2/2020 tumor and normal-tissue responses p. 63 of 75

Approaches to studying normal cells u u We pick some systems because they’re easy to study We pick others because they’re inherently important The built-in problem with normal cell systems is that they are subject to apoptotic and telomeric controls, which means that they’re often a bit harder to handle than cancer cell lines or injectible tumors Unlike tumors, normal cells pay attention to their environment, and that can complicate data analysis 11/2/2020 tumor and normal-tissue responses p. 64 of 75
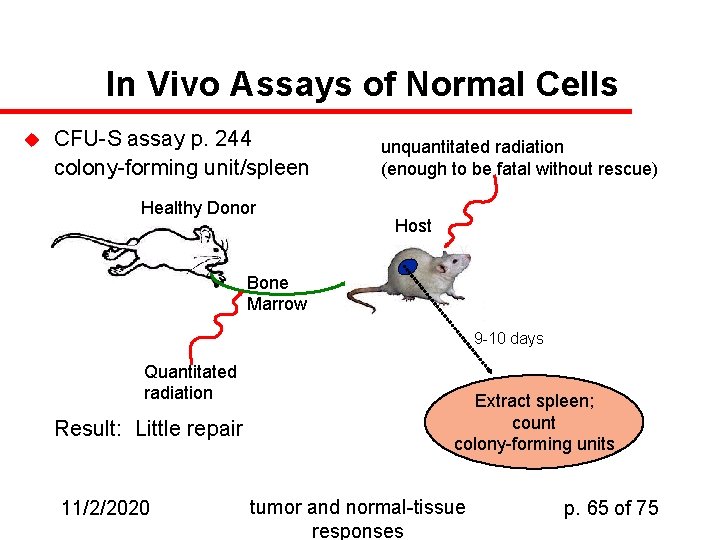
In Vivo Assays of Normal Cells u CFU-S assay p. 244 colony-forming unit/spleen Healthy Donor unquantitated radiation (enough to be fatal without rescue) Host Bone Marrow 9 -10 days Quantitated radiation Result: Little repair 11/2/2020 Extract spleen; count colony-forming units tumor and normal-tissue responses p. 65 of 75
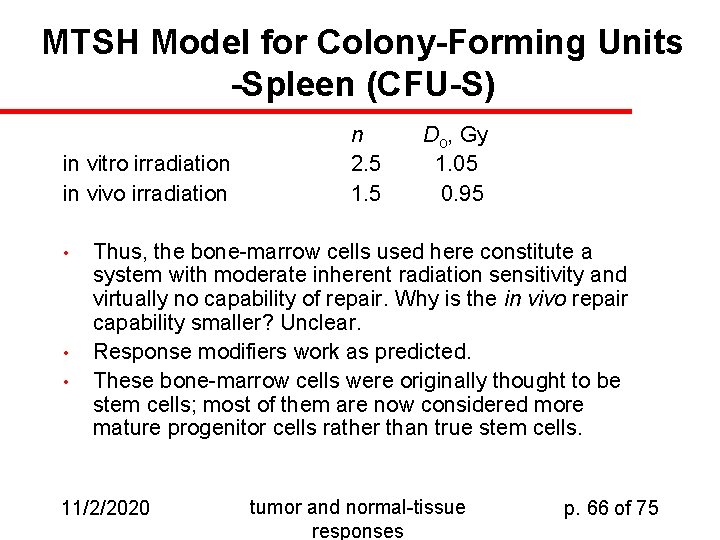
MTSH Model for Colony-Forming Units -Spleen (CFU-S) in vitro irradiation in vivo irradiation • • • n 2. 5 1. 5 Do, Gy 1. 05 0. 95 Thus, the bone-marrow cells used here constitute a system with moderate inherent radiation sensitivity and virtually no capability of repair. Why is the in vivo repair capability smaller? Unclear. Response modifiers work as predicted. These bone-marrow cells were originally thought to be stem cells; most of them are now considered more mature progenitor cells rather than true stem cells. 11/2/2020 tumor and normal-tissue responses p. 66 of 75
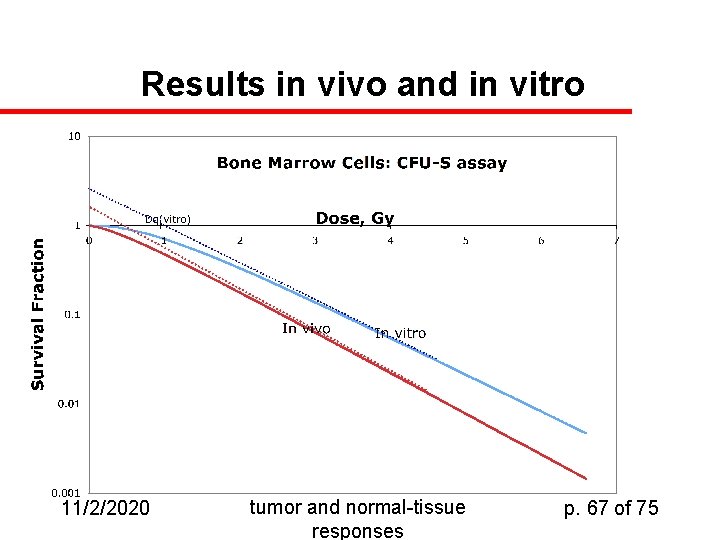
Results in vivo and in vitro 11/2/2020 tumor and normal-tissue responses p. 67 of 75
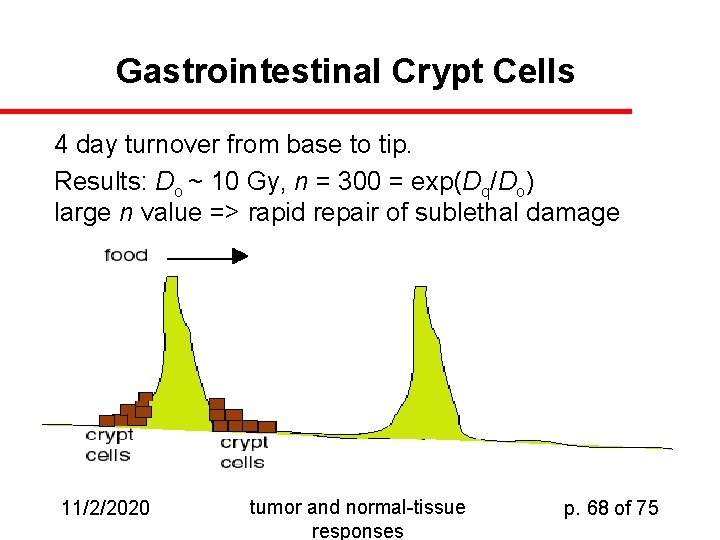
Gastrointestinal Crypt Cells 4 day turnover from base to tip. Results: Do ~ 10 Gy, n = 300 = exp(Dq/Do) large n value => rapid repair of sublethal damage 11/2/2020 tumor and normal-tissue responses p. 68 of 75
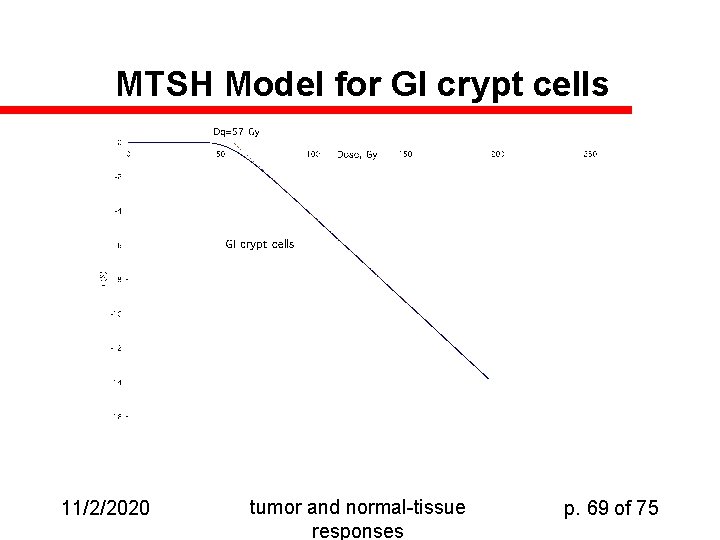
MTSH Model for GI crypt cells 11/2/2020 tumor and normal-tissue responses p. 69 of 75
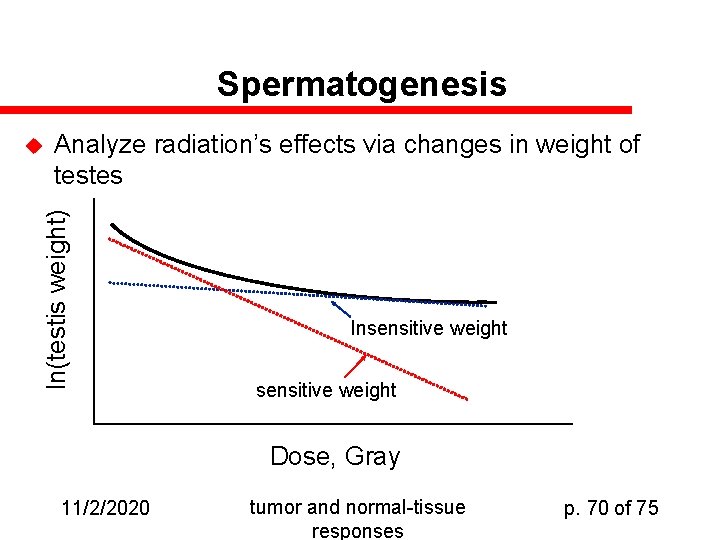
Spermatogenesis Analyze radiation’s effects via changes in weight of testes ln(testis weight) u Insensitive weight Dose, Gray 11/2/2020 tumor and normal-tissue responses p. 70 of 75

Developmental Stages in Spermatogenesis in the Mouse Cell type Type A spermatogonia (type AS, A 1 -A 4) Intermediate spermatogonia Type B spermatogonia 11/2/2020 Days to mature LD 50 spermatozoon (Gy) 35 -45 >2 32 -35 0. 2 30 -35 1. 0 tumor and normal-tissue responses p. 71 of 75

Spermatogenesis, concluded Cell type Primary spermatocytes* Days to mature spermatozoon 20 -35 Resting (preleptotene) Leptotene, Zygotene Pachytene Diplotene Diakinesis 2 5 Unknown 8 9 Secondary spermatocytes Spermatids Spermatozoa 11/2/2020 LD 50 (Gy) 20 -22 7 -20 0 -7 tumor and normal-tissue responses 10 15 500. p. 72 of 75

Testis Weight-Loss Assay u u Very early studies showed that a single exponential didn’t describe the radiosensitivity of testes Endpoint studied was the change in weight of the testis as a function of radiation exposure Result: WD = Wsexp(-ks. D) + Wiexp(-ki. D), where Ws + Wi = total pre-irradiation mass Notion is that testis contains (at least) two kinds of tissues, one of which is more radiosensitive than the other. 11/2/2020 tumor and normal-tissue responses p. 73 of 75
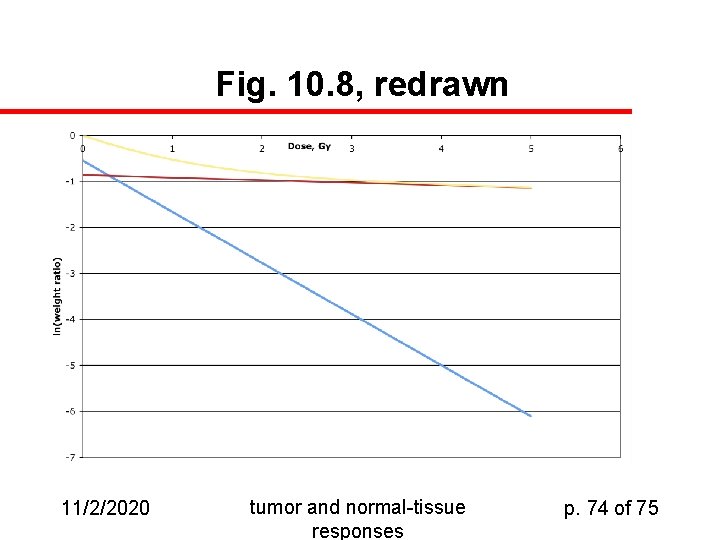
Fig. 10. 8, redrawn 11/2/2020 tumor and normal-tissue responses p. 74 of 75
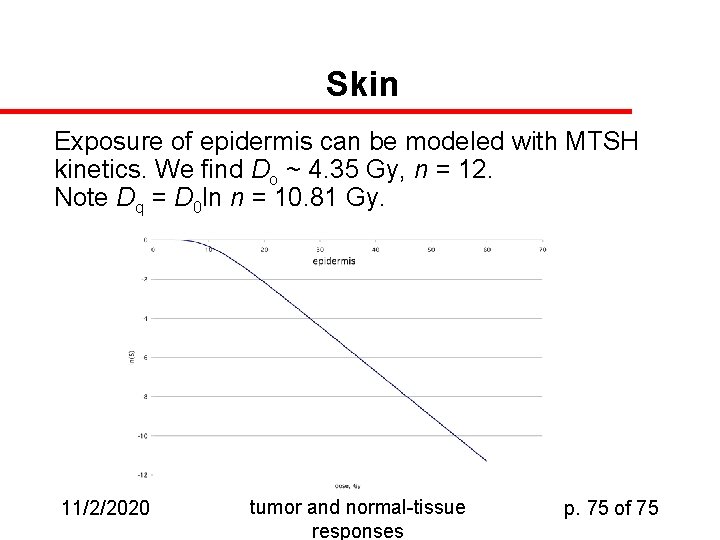
Skin Exposure of epidermis can be modeled with MTSH kinetics. We find Do ~ 4. 35 Gy, n = 12. Note Dq = D 0 ln n = 10. 81 Gy. 11/2/2020 tumor and normal-tissue responses p. 75 of 75
- Slides: 75