Illinois Institute of Technology PHYS 561 RADIATION BIOPHYSICS

Illinois Institute of Technology PHYS 561 RADIATION BIOPHYSICS: Lecture 3: Interaction of Photons with Matter; Radiation Chemistry; Introduction to Biology ANDREW HOWARD 10 June 2014 6/10/14 Rad Bio: interactions with matter p. 1 of 85

Radiation interacts with matter u u We’ll investigate the chemical changes that occur in matter, particularly soft tissue, when ionizing radiation impinges on it; But first we need to finish discussing the initial interactions themselves. 6/10/14 Rad Bio: interactions with matter p. 2 of 85

Outline of Session u. Left over from chapter 5: – – – – Pair production Bremsstrahlung Charged particles & matter Interaction of photons with matter Size scales and biological cells Energy deposition at different physical scales Neutrons 6/10/14 u. Chapter – – – u 6: Types of energy transfer from electrons Free Radicals Radiation Chemistry of water Fricke dosimeter Recombination, Restitution, Repair Biology 101 Rad Bio: interactions with matter p. 3 of 85

Relevant references u Some significant references on photoelectric effect and the interactions of photons with biological tissue: u 1. J. H. Hubbell (1977) Radiation Research 70: 58 -81. 2. J. H. Scofield (1973) Theoretical Photoionization Cross Sections from 1 to 1500 ke. V, Report UCRL-51326, University of California Lawrence Livermore National Laboratory, National Technical Information Center, Springfield, VA 3. A. M. Kellerer and H. H. Rossi (1971) Radiation Research 47: 1534 4. H. H. Rossi (1959) Radiation Research 10: 522 -531 u u u 6/10/14 Rad Bio: interactions with matter p. 4 of 85

Typos of the Day u u u Page 79, first paragraph under “IMPORTANCE OF THE COMPTON PROCESS”, 4 th line: “with attention the” “with attention to the” Page 86, List of possibilities, #3: “Either M or m 0 is at rest” “Both M and m 0 are at rest” Page 87, 2 nd paragraph, 1 st line: “The four principle” “The four principal” 6/10/14 Rad Bio: interactions with matter p. 5 of 85

Pair Production u u u e- + e+; see fig. 4. 7 Can happen if E > 1. 022 Me. V = 2 * m 0(e-)c 2 Rapidly increasing cross-section > 1. 022 Me. V This is the predominant mode of interaction over a range from a bit above that value up to ~5 Me. V Stopping power/atom varies as Z 2 Energy transferred is hn - 1. 022 Me. V 6/10/14 Rad Bio: interactions with matter p. 6 of 85

What pair production does u Scattering nucleus plays fairly passive role – – u not much momentum transferred to nucleus but it does soak up some momentum; otherwise we couldn’t get it to happen at all Generally the positron gets annihilated, giving off a pair of 0. 511 Me. V photons. These generally escape and are not part of the absorbed energy 6/10/14 Rad Bio: interactions with matter p. 7 of 85

Bremsstrahlung: Radiative Energy Loss u u u “Braking radiation”: A fast electron loses energy to its environment in a nonspecific way due to Coulombic interaction with neighboring charged particles. The static particles are much more massive than the electron, so they don’t get accelerated nearly as much as the electron does: but the electron does get accelerated. What happens when an electron is accelerated? It has to radiate! This type of Coulombically-motivated radiation is Bremsstrahlung 6/10/14 Rad Bio: interactions with matter p. 8 of 85

Dependence of Bremsstrahlung on Z u u High-Z elements have much higher cross sections for braking radiation for a given initial electron energy because the acceleration goes like Z Higher electron energies produce more Bremsstrahlung than lower electron energies 6/10/14 Rad Bio: interactions with matter p. 9 of 85

Significance of Bremsstrahlung u u Example in X-ray generators: 1. 5418Å (8 Ke. V) characteristic X-rays are produced in great quantity when we shoot fast electrons at a copper target: L-to-K shell transition emissions BUT: we also get a lot of radiative transfer of energy from the electrons as they move past the copper atoms. This gives rise to Bremsstrahlung, which has no characteristic energies. Thus the spectrum is like this: 6/10/14 Rad Bio: interactions with matter p. 10 of 85
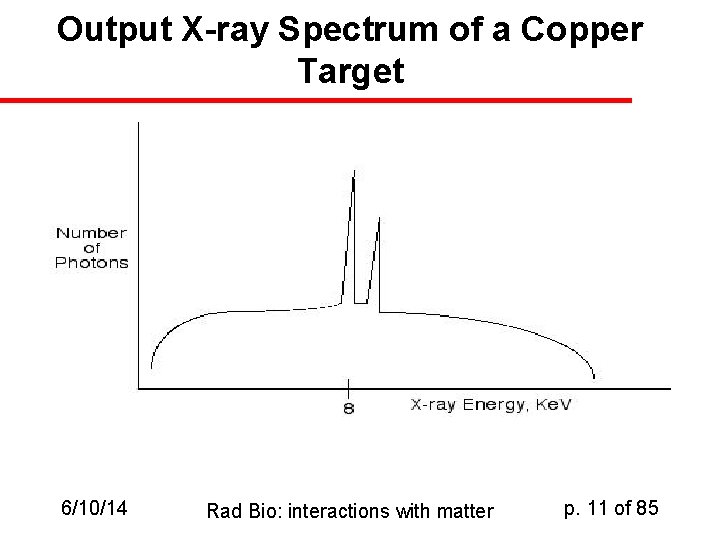
Output X-ray Spectrum of a Copper Target 6/10/14 Rad Bio: interactions with matter p. 11 of 85

Overview of photon-medium interactions u u u We’ve seen four mechanisms by which photons can transfer energy to a medium: Photoelectric effect (mostly below 1 Me. V) Coherent scattering (mostly below 20 ke. V) Compton scattering (peaks around 1 Me. V) Pair production (starts @ 1. 022 Me. V; dominates at high energy) We can write the overall event cross-section as µtot = PE + coh + Compton + pair 6/10/14 Rad Bio: interactions with matter p. 12 of 85

What predominates where? u u u Cf. fig. 5. 2(a), (b) For lead, Compton falls off from 100 ke. V upward and pair production takes over ~ 5 Me. V These figures make the distinction between absorption and transfer as well 6/10/14 Rad Bio: interactions with matter p. 13 of 85

How do mixtures absorb? u u 6/10/14 We’ll come back to this later: Mixtures, including polyatomic compounds, absorb according to their individual atomic attenuation properties, weighted by their mole fraction Rad Bio: interactions with matter p. 14 of 85

Attenuation Coefficients for Molecules (and mixtures) u Calculate mole fraction fmi for each atom type i in a molecule or mixture: – – u subject to Sifmi = 1 That’s why we call these mole fractions Recognize that, in a molecule, fmi is proportional to the product of the number of atoms of that type in the molecule, ni, and to the atomic weight of that atom, mi: fmi = Qni mi (Q a constant to be determined) 6/10/14 Rad Bio: interactions with matter p. 15 of 85

Attenuation coefficients, concl’d u u u Thus Sifmi = Si Qni mi = 1 so Q = (Si ni mi)-1 Then (s/r) for the compound will be (s/r)Tot = Sifmi (s/r)i = Si. Qni mi(s/r)i = (Si ni mi)-1 Sini mi(s/r)i This will, in fact, work even with mixtures of compounds, as long as you keep your mole fractions straight. 6/10/14 Rad Bio: interactions with matter p. 16 of 85

Calculating Mole Fractions and Attenuation Coefficients u Example 1: Water (in book): – – u H 2: n 1 = 2, m 1 = 1; O: n 2 = 1, m 2 = 16 Q = (Si ni mi)-1= (2*1 + 1 * 16)-1 = 1/18 Thus f. H 2 = 2/18, f. O = 16/18, (s/r)Tot = Sifmi(s/r)I = (2/18)*(0. 1129 cm 2 g-1) + (16/18)(0. 0570 cm 2 g -1)= 0. 0632 cm 2 g-1 Benzene (C 6 H 6): – – 6/10/14 C 6: n 1 = 6, m 1 = 12; H 6: n 2 = 6, m 2 = 1 Q = (6*12+6*1) = 1/78, f. C 6 = 72/78, f. H 6 = 6/78 Rad Bio: interactions with matter p. 17 of 85

Interaction of Charged Particles with Matter See pages 84 & 85 in the text. Provides solutions to the dynamical equations describing motion of a heavy charged particle past a stationary electron or (by relativity) motion of an electron past a stationary heavy particle: F = kze 2/r 2 along line MQ Q 6/10/14 Rad Bio: interactions with matter p. 18 of 85

Interaction of e- With Heavy Charged Particle u Momentum imparted to electron at distance of closest approach: classical coherent scattering by e-: non relativistic 6/10/14 Rad Bio: interactions with matter p. 19 of 85

Interaction of Charged Particles with Matter u u Recall diagram 5. 3, p. 84. The crucial equation is for DE(b), the energy imparted to the light particle: DE(b) = z 2 r 02 m 0 c 4 M/(b 2 E) where E is the (nonrelativistic) kinetic energy of the moving particle = (1/2)Mv 2. It increases with decreasing impact parameter b, which stands to reason Energy imparted is inversely proportional to the kinetic energy E of the incoming heavy particle! 6/10/14 Rad Bio: interactions with matter p. 20 of 85

Why does this formula matter? u u u Rate of energy loss is inversely proportional to the energy of the incoming particle So most of the energy is yielded up as the heavy particle approaches its resting state There are some details we’ve skipped, but they’re readily available in other textbooks. 6/10/14 Rad Bio: interactions with matter p. 21 of 85

Relativity tells us this is important! u u We usually think about the electron moving and the heavy nucleus being stationary, not the other way around. Why do we go through this derivation? (a) This could actually happen as described (b) Galilean (or Einsteinian!) relativity says this is equivalent to what happens if the electron is moving and the heavy particle is at rest. That’s the more typical situation: but the analysis still works. (c) If M=m 0, i. e. an electron-electron interaction, then the assumption of minimal interactivity fails, but we still find that the energy transfer goes like 1/v 2 or like 1/E. 6/10/14 Rad Bio: interactions with matter p. 22 of 85

Final steps in absorption u u Let’s think about a high-energy photon entering a biological medium It undergoes a modest number of scattering events that give rise to energetic (ke. V - Me. V) electrons These energetic electrons will themselves only affect the medium once they have transferred energy to other electrons in ~10 -50 e. V packets Four mechanisms for doing that: (a) Delta rays (c) Bremsstrahlung in motion (b) Photoelectric (d) Direct collision 6/10/14 Rad Bio: interactions with matter p. 23 of 85

Delta rays u u u We expect that each individual event transfers a small amount of momentum, so the incoming electron doesn’t change direction all that much There are exceptions: these are delta rays. The “delta ray” is the common name for a secondary electron that itself has enough energy to ionize other things The ionization events caused by the delta ray can produce electrons that have energies up to half of that of the primary electron So some delta rays will even produce tertiary electrons that are themselves delta rays! 6/10/14 Rad Bio: interactions with matter p. 24 of 85

Photoelectric processes u u If the primary photon is absorbed by an atom, a K or L-shell atomic electron is ejected This makes an outer-shell electron hop in to fill the inner-shell vacancy, giving rise to an output photon with an energy equal to the difference in shell energies The photon has to undergo ordinary scattering (probably Compton) in order to transfer energy This process is most likely in high-Z media, for which the capture cross section is measurable 6/10/14 Rad Bio: interactions with matter p. 25 of 85

Bremsstrahlung with movement u u Caused by Coulombic interaction of an emitted electron with nucleus Incoming electron decelerates a little due to these interactions This is not like the 1/E-dependent process we just described, because not much happens to the target in this instance Loss due to Bremsstrahlung is important for incoming electrons above 10 Me. V in lead or 100 Me. V in water: that corresponds to almost 200 times the rest energy of the electron 6/10/14 Rad Bio: interactions with matter p. 26 of 85
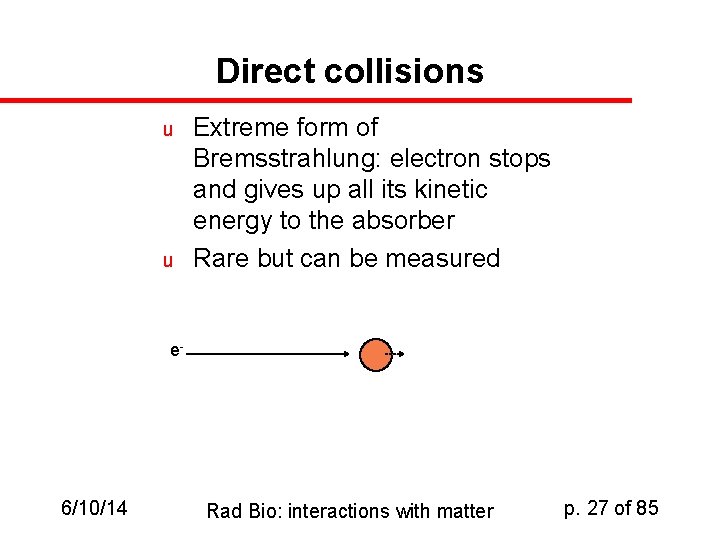
Direct collisions u u Extreme form of Bremsstrahlung: electron stops and gives up all its kinetic energy to the absorber Rare but can be measured e- 6/10/14 Rad Bio: interactions with matter p. 27 of 85

Dose: a definitional reminder u u Remember that dose is about deposition (or absorption) of energy per unit mass, while kerma is about transfer of energy per unit mass So energy that escapes the neighborhood of the initial event may not count in the dose in that region, but rather will count in the dose of some other region Kerma, by contrast happens at the moment of impact So kerma can be lower or higher than dose depending on circumstances 6/10/14 Rad Bio: interactions with matter p. 28 of 85
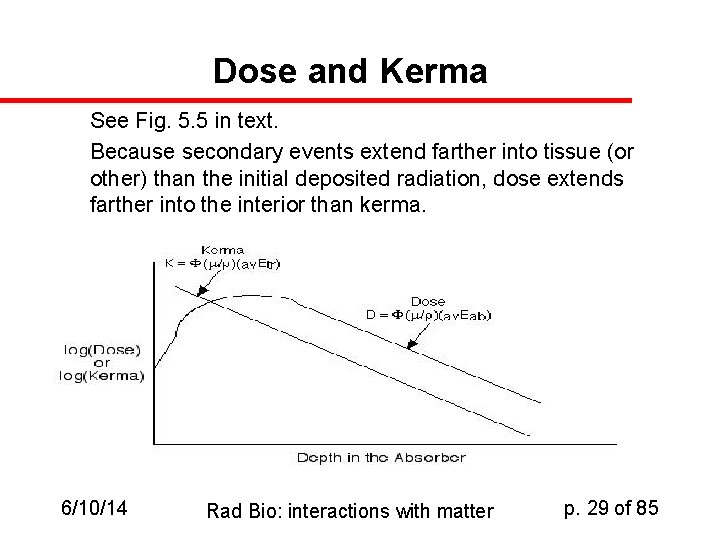
Dose and Kerma See Fig. 5. 5 in text. Because secondary events extend farther into tissue (or other) than the initial deposited radiation, dose extends farther into the interior than kerma. 6/10/14 Rad Bio: interactions with matter p. 29 of 85

Photons interacting with matter We mentioned at the beginning of the lecture that the interaction of a highenergy photon with a chunk of matter involves – – 6/10/14 Photoelectric effect Coherent scatter Compton scatter Pair production Rad Bio: interactions with matter p. 30 of 85

Compton Scattering, Revisited u u The most important of these processes for hn > 100 Ke. V is Compton scatter, especially if the matter is water or tissue See fig. 5. 2(B) in the text to see why: µab/r (Compton) predominates above 100 Ke. V 6/10/14 Rad Bio: interactions with matter p. 31 of 85

Compton Processes in Tissue Biological soft tissue is predominantly made up of H, C, N, O, and a little P and S. So attenuation of photons is dominated by those light elements (Z 16) 6/10/14 Rad Bio: interactions with matter p. 32 of 85

Dose Remember Dose = energy deposited per unit mass. What is the meaningful size scale for a mammalian cell? We’ll need to know this to estimate dose on a cell. size scales 5 mm r 1 g/cm 3 for water or soft tissue mass of (5 mm)3 r =(5 * 10 -4 cm)3 r =125 * 10 -12 cm 3 1 g/cm 3 =125 * 10 -12 g = 1. 25 * 10 -13 kg 6/10/14 Rad Bio: interactions with matter p. 33 of 85

Energy Absorbed in a Cell u u u Suppose N Joules of energy are deposited in a 70 kg human. Nominally the dose is N/70 Gy. How much energy is deposited in a single (5µm)3 cell? (N/70)Gy * 10 -13 kg = (N/70)*10 -13 J= (1. 3*10 -15)*N J = [(1. 3*10 -15)*N]/1. 609*10 -19 J = 8500*N e. V. So it’s a lot of energy! Is the Bethe-Blocke continuous slowing-down approximation applicable here? No! Too much energy is being stopped per cell for it to be applicable. But we try to use it anyway. 6/10/14 Rad Bio: interactions with matter p. 34 of 85

Track structure u u We draw different kinds of conclusions depending on the size range we think about The smaller the target, the bigger the fluctuations in dose that we have to recognize So deliver of 1 c. Gy to a large volume could mean we’re delivering anywhere between 0 and 103 Gy to a single chromatin fiber We can attempt to account for this in various ways; Rossi does it in terms of EY/d 6/10/14 Rad Bio: interactions with matter p. 35 of 85

Can LET tell us what we want? u u u Envision variations in dose in terms of the individual values of E/m for small m; these may differ from the bulk dose by many orders of magnitude Rossi defines E/m for these small masses as the local energy density, Z Remember that LET is d. EL/dl, where l is the length along the track of the ionization source. This might give us a handle on the effectiveness of a given bulk dose in causing damage, or it might not. Rossi uses LET in fudging his values, as we’ll now discuss. 6/10/14 Rad Bio: interactions with matter p. 36 of 85

Rossi’s alternative u u u He makes some assumptions: Particles and their secondaries deposit over spherical volumes of specific size Source and its secondaries can deposit an energy Ey within the sphere We call each deposition an event Event size Y is defined as Ey/d, where d is the sphere diameter The hope is that for idealized tracks, Y will be a constant independent of d 6/10/14 Rad Bio: interactions with matter p. 37 of 85

How this is used u u Rossi fudges the real spheres by looking at experimental systems involving spherical ionization chambers containing low-pressure gases; These become surrogates for tiny tissue-equivalent spheres Then he plots Y as the independent axis and the energy loss D(Y*), corrected for LET distribution See fig. 5. 6: for 1. 5 µm spheres the largest D(Y*) values occur around 70 ke. V/µm for protons 6/10/14 Rad Bio: interactions with matter p. 38 of 85

Local energy density u u u Tissues have densities close to 1 g cm-3 = 103 kg m-3 Alpen shows you that the amount of locally deposited energy Z ~ 30. 6 d-2 J kg-1 So for high LET and small d, this can be as high as 10. 9 Gy for reasonable values of Y For high LET radiation lots of events produce Z=0 and a few give us very big values For low LET is more boring: the distributions are Gaussian and centered on the absorbed dose General conclusion: high-LET radiation is harder to predict results at the single-cell level 6/10/14 Rad Bio: interactions with matter p. 39 of 85

Demonstration That Events Don’t Interact Much Spurs are 400 nm apart 1 nm = 10 -9 m 400 nm = 0. 4 mm Hydrogen radical diffusion (see below): 8 10 -5 cm 2 s-1 diffusion constant for H • Typical lifetime 10 -6 s Typical diffusion distance = 180 nm This is smaller than the distance between spurs! 6/10/14 Rad Bio: interactions with matter p. 40 of 85
![Mozumder & Magee [1 Me. V “typical” electron] u u u Spurs Blobs Tracks Mozumder & Magee [1 Me. V “typical” electron] u u u Spurs Blobs Tracks](http://slidetodoc.com/presentation_image_h2/6b81522752250c410fdc32dc8d1352e3/image-41.jpg)
Mozumder & Magee [1 Me. V “typical” electron] u u u Spurs Blobs Tracks 6/10/14 6 - 100 e. V 100 - 500 e. V 500 - 5000 e. V Portion of energy deposited 65% 15% 20% Rad Bio: interactions with matter p. 41 of 85
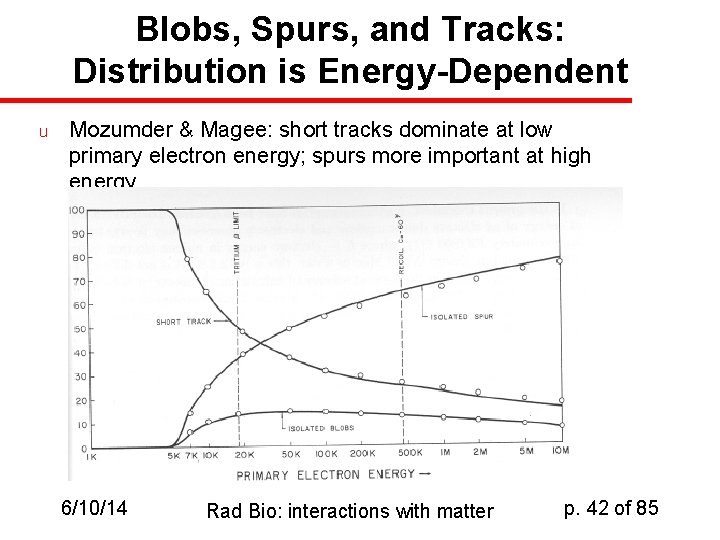
Blobs, Spurs, and Tracks: Distribution is Energy-Dependent u Mozumder & Magee: short tracks dominate at low primary electron energy; spurs more important at high energy 6/10/14 Rad Bio: interactions with matter p. 42 of 85

Neutrons u u 6/10/14 Neutrons are produced as byproducts of various reactions and are therefore moderately significant as portions of human or environmental exposure We can learn things from neutron studies that will help us in understanding the ways that ions and other heavy particles interact with tissue Rad Bio: interactions with matter p. 43 of 85
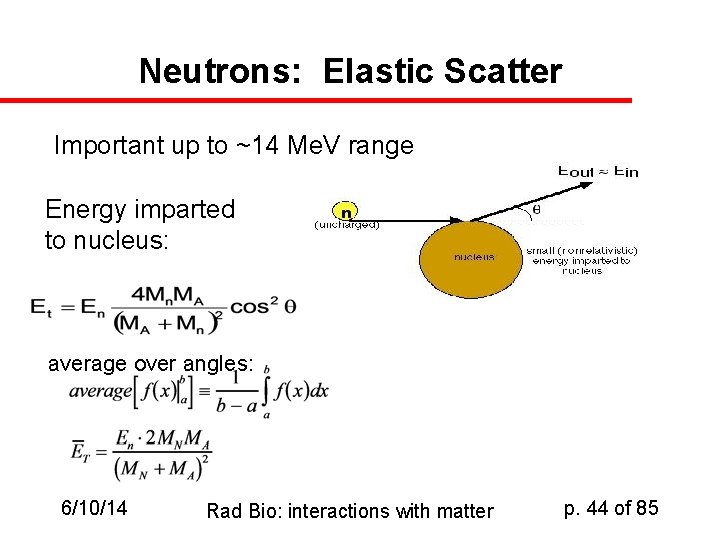
Neutrons: Elastic Scatter Important up to ~14 Me. V range Energy imparted to nucleus: average over angles: 6/10/14 Rad Bio: interactions with matter p. 44 of 85

How to average cos 2 q: u Addition formula for cosine: – – – u u cos(A+B) = cos. Acos. B - sin. Asin. B For A=B= , cos(2 ) = cos 2 - sin 2 Furthermore cos 2 + sin 2 = 1 so cos 2 = cos 2 - (1 - cos 2 ) = 2 cos 2 - 1 Therefore cos 2 = (1 + cos 2 ) / 2 This gives us the tools we need to integrate cos 2 over an interval. In general <f(x)> over an interval (a, b) is 6/10/14 Rad Bio: interactions with matter p. 45 of 85

<cos 2 q>, continued 6/10/14 Rad Bio: interactions with matter p. 46 of 85

Significance in Elastic Scatter u u u Recall we said that for any value of q, the energy transferred to the target nucleus, Et, is Et = En (4 Ma. Mn)cos 2 / (Ma + Mn)2 So the average energy imparted to the target nucleus is <Et> = {En (4 Ma. Mn) / (Ma + Mn)2} <cos 2 > We just spent three pages proving <cos 2 >=1/2 Thus <Et> = 2 En. Ma. Mn / (Ma + Mn)2 6/10/14 Rad Bio: interactions with matter p. 47 of 85
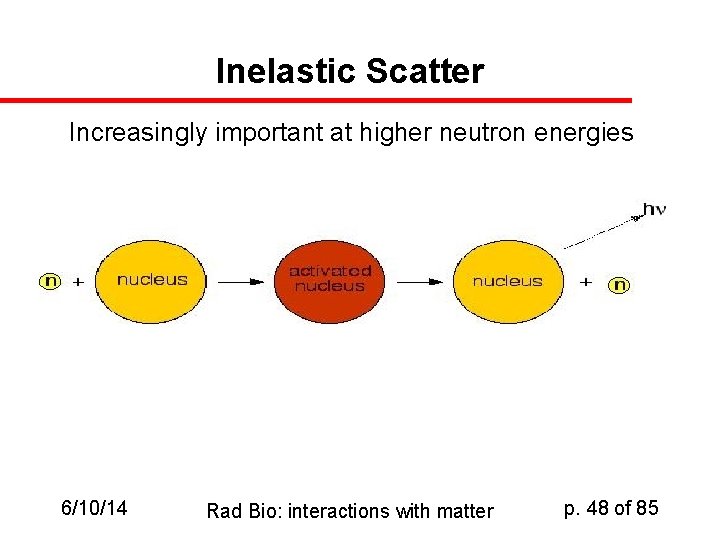
Inelastic Scatter Increasingly important at higher neutron energies 6/10/14 Rad Bio: interactions with matter p. 48 of 85

Neutrons: Other Mechanisms (III) Nonelastic (75 Me. V) + n 9 Be + a => KE ~1. 75 Me. V (IV) Neutron Capture 14 N + n 14 C + p 1 H + n 2 H + 2. 2 Me. V (V) Spallation: Nucleus fragments! Need very high-energy neutrons ( > 100 Me. V) 12 C 6/10/14 Rad Bio: interactions with matter p. 49 of 85

Free Radicals: Definitions and Illustrations u u A free radical is defined as a molecular species containing an unpaired electron. It may be charged or uncharged. Most biological free radicals, with the significant exception of superoxide (O 2 - • ), are uncharged. – – OH- Hydroxide ion (9 protons, 10 electrons) OH • Hydroxyl radical (9 protons, 9 electrons) Moses Gomberg: Characterized the triphenylmethyl radical in 1900 6/10/14 Rad Bio: interactions with matter p. 50 of 85

Reactivity of free radicals u u Free radicals are reactive because the unpaired electrons tend to seek out targets, either other unpaired electrons: H • + • H H 2 … or other acceptors of unpaired electrons. Reactivities vary considerably depending on presence or absence of stabilizing influences, such as channels through which the unpaired electron can be delocalized In the absence of those channels, free radicals tend to recombine in picoseconds With those channels, they can last seconds or longer, even at room temperature 6/10/14 Rad Bio: interactions with matter p. 51 of 85

Radical Stability u u This has nothing to do with political psychology : -) Highly unstable free radicals tend not to stay around long enough for ordinary spectroscopic methods to detect. Radicals where the unpaired electron can be highly delocalized last long enough to detect. Triphenylmethyl radical: H 6/10/14 C • C + H • Rad Bio: interactions with matter p. 52 of 85

Cartoons of Electron Distributions in ions, molecules, and radicals : : : O • H u u : : : : O u : : : O O: : : H : 6/10/14 : : O • O: : : u Hydroxyl radical (8 paired e-, 1 unpaired e-, 9 p+) Hydroxide ion (10 paired e-, 9 p+) Molecular oxygen (16 paired e-, 16 p+) Superoxide ionic radical (16 paired e-, 1 unpaired e-, 16 p+) Rad Bio: interactions with matter p. 53 of 85
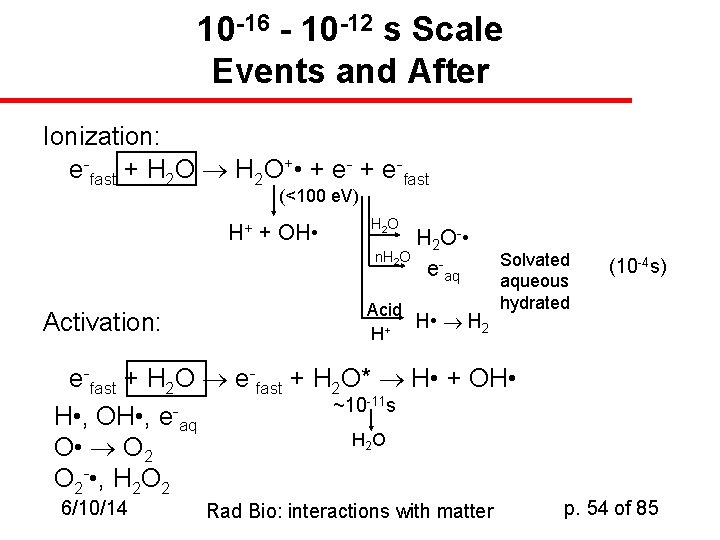
10 -16 - 10 -12 s Scale Events and After Ionization: e-fast + H 2 O+ • + e-fast (<100 e. V) H+ + OH • Activation: H 2 O H 2 O - • n. H 2 O e-aq Acid H • H 2 + H Solvated aqueous hydrated e-fast + H 2 O* H • + OH • ~10 -11 s H • , OH • , e aq H 2 O O • O 2 - • , H 2 O 2 6/10/14 Rad Bio: interactions with matter (10 -4 s) p. 54 of 85

Radiation Chemistry of Water u u u Since biological tissue is mostly water, we’re very interested in the products produced when water absorbs ionizing radiation The reactive species formed out of water are responsible for a large fraction of the biological activities of radiation Ordinary ions (H+, OH-, H 3 O+) are among these species, as is hydrogen peroxide (H 2 O 2); So are free radicals: H • , O 2 - • , HO 2 • We often discuss the “solvated electron”, eaq-. 6/10/14 Rad Bio: interactions with matter p. 55 of 85

Fricke Dosimeter u u u Bookkeeping tool for aqueous radical chemistry, based on Fe 2+ Fe 3++ eferrous ferric Sequence of reactions: H • + O 2 HO 2 • (i. e. H-O=O • ) HO 2 • + Fe 2+ HO 2 - + Fe 3+ HO 2 - + H+ H 2 O 2 OH • + Fe 2+ Fe 3+ + OHH 2 O 2 + Fe 2+ Fe 3+ + OH- + OH • In absence of O 2: H • + H 2 O OH • + H 2 6/10/14 Rad Bio: interactions with matter p. 56 of 85

Fricke Dosimeter: bookkeeping u u Each hydrogen radical H • causes the oxidation of three molecules of ferrous ion H 2 O 2 produced by radiolysis will oxidize two ferrous ions: one directly, one indirectly. A radiolytically-produced OH • radical gives rise to one more oxidation Therefore at acidic p. H in the presence of oxygen, G(Fe 3+) = 2 G(H 2 O 2) + 3 G(H • ) + G(OH • ) 6/10/14 Rad Bio: interactions with matter p. 57 of 85

Definition of Yield G = Yield Number of events produced per 100 e. V energy deposition We’re often interested in d. G(E)/d. E Yield is either dimensionless or has dimensions of (energy)-1 depending on your perspective Fricke dosimeter provides a way of measuring yield 6/10/14 Rad Bio: interactions with matter p. 58 of 85

Fricke bookkeeping u u Results on p. 112 for 60 Co photons: G(H • ) = 3. 65 G(H 2 O 2) = 0. 75 G(OH • ) = 3. 15 We then apply formula 6. 8 to determine G(Fe 3+) Recall that under appropriate conditions G(Fe 3+) = 3 * G(H • ) + 2 * G(H 2 O 2) + G(OH • ) = 3 * 3. 65 + 2* 0. 75 + 3. 15 = 15. 6 Under anaerobic conditions: eqn. 6. 9 applies: G(Fe 3+) = G(H • ) + G(OH • ) + 2 G(H 2 O 2) = 8. 3 6/10/14 Rad Bio: interactions with matter p. 59 of 85
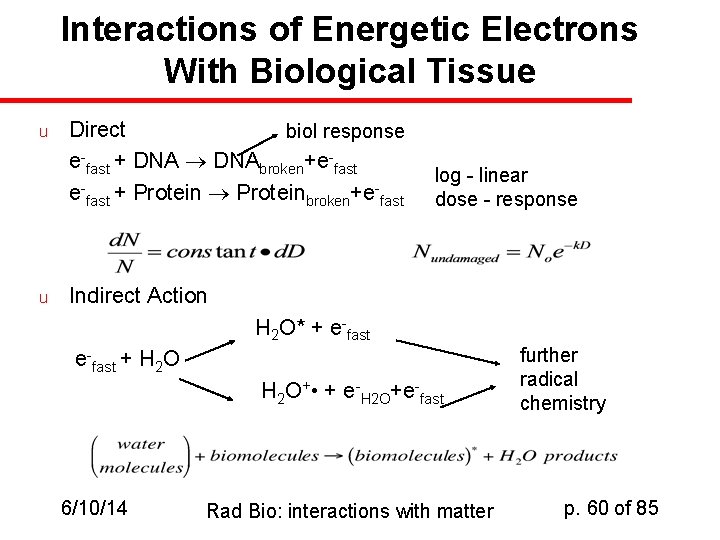
Interactions of Energetic Electrons With Biological Tissue u u Direct biol response e-fast + DNAbroken+e-fast + Proteinbroken+e-fast log - linear dose - response Indirect Action H 2 O* + e-fast + H 2 O+ • + e-H 2 O+e-fast 6/10/14 Rad Bio: interactions with matter further radical chemistry p. 60 of 85

Direct Action: the model u Direct action of radiation on a species says that a single hit of radiation onto a molecule damages it. Then if N is the number of undamaged molecules after irradiation with dose D, we expect the change in N, with a small increase D in dose is proportional to N and to D. Radiation in ******************* N 0 Total molecules N Undamaged molecules 6/10/14 Rad Bio: interactions with matter p. 61 of 85

Physical model and mathematics u u u Let N = number of undamaged molecules after irradiation with dose D. Then d. N N d. D. More radiation dose implies more response More undamaged molecules implies more damage. f A A+ + e or other chemistry 6/10/14 Rad Bio: interactions with matter p. 62 of 85

Why should the damage be loglinear? u u u The relationship d. N N d. D can be rewritten d. N = -k. N d. D, where k = inactivation constant. Then d. N / N = -kd. D. Integrating both sides, ln N = -k. D + C. Raising e to a power on both sides, eln. N = e(-k. D+ C) = e-k. D * e. C. Defining e. C = N 0, N = N 0 e-k. D Thus the physical meaning (boundary condition) of N 0 is that it is the number of entities present in the case where the dose is 0. 6/10/14 Rad Bio: interactions with matter p. 63 of 85

Significance of the inactivation constant u u u Inactivation constant, k, is in dimensions of inverse dose (e. g. units of Gy-1) and is the reciprocal of the dose required to reduce the number of undamaged molecules down to 1/e times the original count. N = N 0 e-k. D; if Di = 1/k, then N(Di)= N 0 e-k. Di = N 0 e-k/k = N 0 e-1 = N 0/e We could define a half-inactivation dose D 1/2, analogous to the half-life of an emitter: For D =D 1/2, N=N 0/2 = N 0 e-k. D 1/2, ln 1/2 = -k. D 1/2 Thus -ln 2 = -k. D 1/2, so D 1/2 = (ln 2)/ k = 0. 693 / k 6/10/14 Rad Bio: interactions with matter p. 64 of 85

Indirect action of radiation u u u Initial absorption of radiative energy gives rise to secondary chemical events Specifically, in biological tissue R + H 2 O* (R = radiation) H 2 O* + biological macromolecules damaged biological macromolecules The species “H 2 O*” may be a free radical or an ion, but it’s certainly an activated species derived from water. Effects are usually temperature-dependent, because they depend on diffusion of the reactive species to the biological macromolecule. 6/10/14 Rad Bio: interactions with matter p. 65 of 85
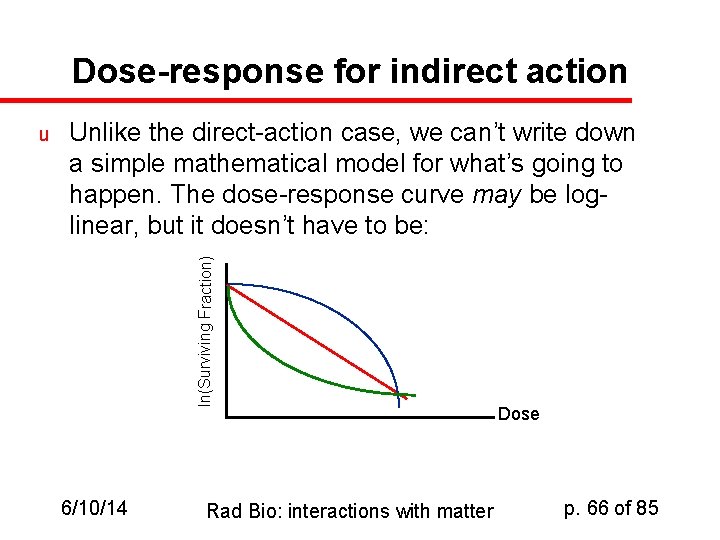
Dose-response for indirect action Unlike the direct-action case, we can’t write down a simple mathematical model for what’s going to happen. The dose-response curve may be loglinear, but it doesn’t have to be: ln(Surviving Fraction) u 6/10/14 Rad Bio: interactions with matter Dose p. 66 of 85
Interaction of energetic electrons with biological tissue u u Direct action: e-fast + DNAbroken + e-fast (log-linear) e-fast + proteinbroken + e-fast (log-linear) d. N/d. D = -k*D; Nundamaged = N 0 e-k. D Indirect action: H 2 O* + e-fast + H 2 O further radical chemistry H 2 O+. + e-H 2 O + e-fast (water molecules) + (biomolecules)* + radical water products 6/10/14 Rad Bio: interactions with matter p. 67 of 85

Radical Fates/ Damaged Biomolecule Fates u u Recombination A • + B • A - B (timescale 10 -11 s) Generally A = B i. e. A • + • A A - A Restitution: Non catalyzed regeneration of non-radical species (microsecond timescale) A • + X A + X • biol diffuseable molecule u out Repair: Catalyzed regeneration of undamaged species A • + E + R Amod + E + R • where E is enzyme (ms-sec) biol 6/10/14 biol Rad Bio: interactions with matter p. 68 of 85

Fundamentals of Biology for the Radiation Biophysicist u u u It would be presumptuous of me to try to summarize all of biology in half of one lecture I’ll therefore content myself with pointing out a few fundamentals that will be relevant to our studies of the interactions between ionizing radiation and biological tissue As an endpoint, we’re primarily concerned with the effects of radiation on vertebrate, especially human, tissues; but we do need to have some feel for how radiation affects bacteria and yeast as well 6/10/14 Rad Bio: interactions with matter p. 69 of 85

What is life? u u u That was the title of a series of lectures given by Erwin Schrödinger in Dublin in 1943 His focus was on articulating how quantum physics could help to explain the organizational principles found in organisms But we can kidnap that name to focus on what we actually do want to do, which is differentiate living organisms from non-living entities 6/10/14 Rad Bio: interactions with matter p. 70 of 85

What are living organisms? u Entities capable of: – – Reproduction Energy processing Adaptation to changes in environment Capable of local decreases in entropy 6/10/14 Rad Bio: interactions with matter p. 71 of 85

Based on that definition, what is alive? u u On this basis, conventional life from archaea and eubacteria up through elephants and redwood trees are living Viruses are a borderline case; prions highly questionable 6/10/14 Rad Bio: interactions with matter p. 72 of 85

Organisms are made up of cells u u Cells are somewhat self-contained objects within which chemical reactions and reproduction can occur Every cell interacts with its environment Cells are surrounded by a differentially permeable boundary called a cell membrane Sizes of cells vary but they’re generally between 1 and 10 micrometers on a side 6/10/14 Rad Bio: interactions with matter p. 73 of 85

Highest-level taxonomic distinctions u Most fundamental distinction is made on the basis of whether the organism’s cells have nuclei – – u u Eukaryotic cells have nuclei Prokaryotic (archaeal and eubacterial) cells don’t Eubacteria are simple, generally unicellular, organisms lacking nuclei Archaea are like that too, but they appear to derive from a separate evolutionary history; many are extremophiles 6/10/14 Rad Bio: interactions with matter p. 74 of 85

Eukaryotes u u These organisms have nuclei and generally have other definable organelles as well: mitochondria, endoplasmic reticulum, Golgi apparatus, vacuoles, chloroplasts, … Often but not always multicellular Yeast, e. g. Saccharomyces, is a unicellular eukaryote Some organisms are complex but unicellular 6/10/14 Rad Bio: interactions with matter p. 75 of 85

Reproduction u u u We included this in our definition of life Unicellular organisms generally reproduce by fission, but not inevitably; there are cooperative (sexual) events that occur even among bacteria Multicellular organisms undergo mitosis at the cellular level but they also have a level of organization wherein an entire organism can be engendered via combinations of meiosis, mitosis, and growth 6/10/14 Rad Bio: interactions with matter p. 76 of 85

Eukaryotic organelles u u u u Nucleus: site of replication and transcription contains DNA in various levels of organization Mitochondrion: site of most catabolic (energyproducing) reactions, which typically yield ATP Endoplasmic reticulum: vehicle for lipid synthesis and protein processing Golgi apparatus: vehicle for trafficking Cytoskeleton: stiff proteinaceous organizers Vacuoles: sacs containing fluids Chloroplasts: sites of photosynthesis 6/10/14 Rad Bio: interactions with matter p. 77 of 85

How do we study biological systems? u u u u Observations of ecosystems Direct observations of whole organisms Recognition of tissue-tissue interactions Delineation of tissue types Recognition of cell-cell interactions Delineation of cell types Characterization of molecular events in a cell and in the extracellular matrix Understanding of the underlying physics that enables those molecular events to occur 6/10/14 Rad Bio: interactions with matter p. 78 of 85

Biological activity depends on chemistry u u u That’s a commonplace now, but it wasn’t fully recognized until sometime in the nineteenth century It applies to chemical transformations within cells, but it also applies to extracellular systems and macroscopic movements, such as muscle contraction Understanding the energetics (does the activity require or release energy) and the kinetics (how does one overcome an activation barrier) is critical to understanding biochemical processes 6/10/14 Rad Bio: interactions with matter p. 79 of 85

Biological reactions are catalyzed u u A catalyst is an entity that participates in a reaction but is ultimately returned to its original state after the reaction completes Therefore a catalyst influences kinetics but not equilibrium Biological catalysts are called enzymes Enzymes have three fundamental properties: – – – They are catalytic They are specific They can be regulated 6/10/14 Rad Bio: interactions with matter p. 80 of 85

Most, but not all, enzymes are proteins u u Note that many proteins aren’t enzymes The recognition that enzymes are proteins arose in the 1920’s and 1930’s By the 1970’s it became clear that certain RNA molecules are catalytic Some RNA-catalyzed reactions, notably the creation of new protein molecules, are central to biological function 6/10/14 Rad Bio: interactions with matter p. 81 of 85

DNA is the vehicle for heritance u u Deoxyribonucleic acid is a polymer found in all organisms and many viruses Building blocks: phosphodeoxyribose backbone, N-containing side-groups called nucleic acid bases DNA contains information enabling parent cell or organism to produce nearly-identical offspring Differences arise from: – – Mutations Recombination Sexual segregation of chromosomes Epigenetic effects 6/10/14 Rad Bio: interactions with matter p. 82 of 85

Central dogma, modern form u DNA becomes replicated prior to each cell division – – u DNA is transcribed into various forms of RNA – – u Double-stranded DNA uncoils Fresh copy of each strand created and coiled up Gene transcribed when RNA is called for One strand of DNA provides template Messenger RNA is translated at ribosome: – – m. RNA sequence defines amino acid sequence of resulting protein Protein then folds, either spontaneously or not 6/10/14 Rad Bio: interactions with matter p. 83 of 85

DNA replication is protected against error u u u Inherent error rate might be one base substitution in 104 or 105 replicated bases Error correction within DNA polymerase drops that to about 1 in 107 Further error correction (“repair”) by external enzymes drops it to about 1 in 109 6/10/14 Rad Bio: interactions with matter p. 84 of 85

Why is that relevant? u u In the human genome, 1 in 109 is still several surviving base substitutions in each replication! Most substitutions are harmful; most aren’t fatal Exposure to ionizing radiation and certain chemical mutagens, particularly at certain stages in the cell cycle, can significantly increase the error rate Human genetic conditions that hinder DNA repair substantially increase radiation sensitivity 6/10/14 Rad Bio: interactions with matter p. 85 of 85
- Slides: 85