Il Nobel per la medicina 2016 alle scoperte

Il Nobel per la medicina 2016 alle scoperte sulla "pulizia cellulare" Il Nobel per la medicina 2016 è stato assegnato a Yoshinori Ohsumi per le sue scoperte sui meccanismi dell'autofagia, un processo fondamentale di "pulizia cellulare", che ha un ruolo importante in molte patologie, da quelle neurodegenerative a malattie da accumulo lisosomiale a varie forme di tumore

The word autophagy is derived from Greek words “auto” meaning self and “phagy” meaning eating: ‘autophagy’, =‘eating of self’ The term was first coined by Christian de Duve over 40 years ago, and was largely based on the observed degradation of mitochondria and other intra-cellular structures within lysosomes of rat liver perfused with the pancreatic hormone, glucagon.

In recent years the scientific world has ‘rediscovered’ autophagy, with major contributions to our molecular understanding and appreciation of the physiological significance of this process coming from numerous laboratories. Autophagy is a normal physiological process in the body that deals with destruction of cells in the body. It maintains homeostasis or normal functioning by protein degradation and turnover of the destroyed cell organelles for new cell formation. During cellular stress the process of Autophagy is upscaled and increased. Cellular stress is caused when there is deprivation of nutrients and/or growth factors. Thus Autophagy may provide an alternate source of intracellular building blocks and substrates that may generate energy to enable continuous cell survival. Currently, 32 different autophagy-related genes (Atg) have been identified by genetic screening in yeast and, significantly, many of these genes are conserved in slime mould, plants, worms, flies and mammals, emphasizing the importance of the autophagic process in responses to starvation across phylogeny. Although the importance of autophagy is well recognized in mammalian systems, many of the mechanistic breakthroughs in delineating how autophagy is regulated and executed at the molecular level have been made in yeast (Saccharomyces cerevisiae).

Autophagy and cell death Autophagy also kills the cells under certain conditions. These are form of programmed cell death (PCD) and are called autophagic cell death. Programmed cell death is commonly termed apoptosis. Autophagy is termed a non-apoptotic programmed cell death with different pathways and mediators from apoptosis. Autophagy mainly maintains a balance between manufacture of cellular components and break down of damaged or unnecessary organelles and other cellular constituents.

Autophagy and stress Autophagy enables cells to survive stress from the external environment like nutrient deprivation and also allows them to withstand internal stresses like accumulation of damaged organelles and pathogen or infective organism invasion. Autophagy is seen in all eukaryotic systems including fungi, plants, slime mold, nematodes, fruit flies and insects, rodents (laboratory mice and rats), humans.

Types of autophagy There are several types of Autophagy. These are: • microautophagy – in this process the cytosolic components are directly taken up by the lysosome itself through the lysosomal membrane. • macroautophagy – this involves delivery of cytoplasmic cargo to the lysosome through the intermediary of a double membrane-bound vesicle. This is called an autophagosome that fuses with the lysosome to form an autolysosome. • Chaperone-mediated autophagy – in this process the targeted proteins are translocated across the lysosomal membrane in a complex with chaperone proteins (such as Hsc-70). • micro- and macropexophagy • piecemeal microautophagy of the nucleus • cytoplasm-to-vacuole targeting (Cvt) pathway
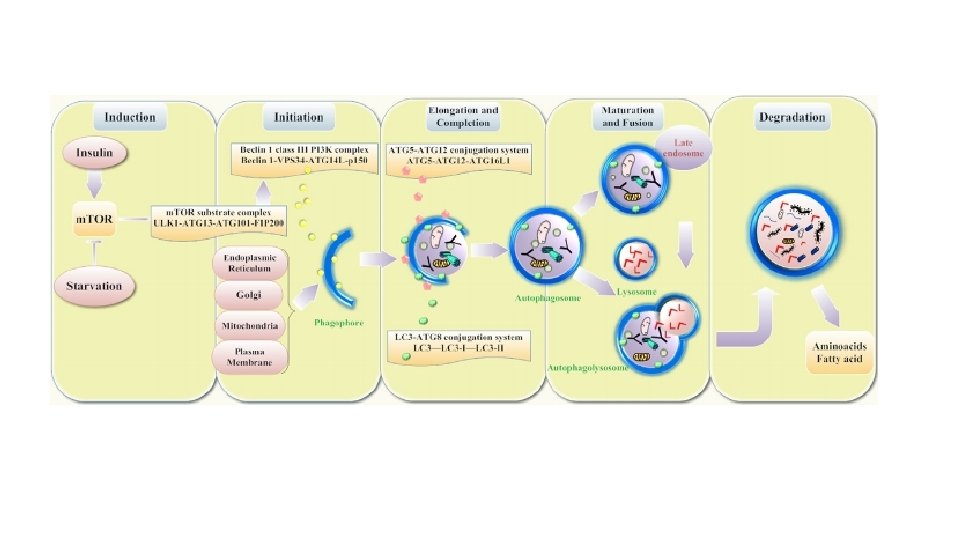

Figure 1 The molecular biology of autophagy Fougeray, S. & Pallet, N. (2014) Mechanisms and biological functions of autophagy in diseased and ageing kidneys Nat. Rev. Nephrol. doi: 10. 1038/nrneph. 2014. 201 Activation of AMPK in response to low cellular energy status inhibits m. TOR and activates ULK 1 via phosphorylation. Coordinated activation of the ULK 1–ATG 13–ATG 101–FIP 200 complex (which is inhibited by m. TORC 1) and the lipid kinase Vps 34–Beclin 1 complex (which is inactivated by Bcl-2), drives the formation of the isolation membrane. The transmembrane proteins ATG 9 and VMP 1 participate in the recruitment of lipids from endosomes and the Golgi to the isolation membrane. The ATG 5–ATG 12–ATG 16 L 1 complex and LC 3–PE system also have roles in cargo recruitment, membrane elongation and maturation. ATG 7 and ATG 10 catalyse the conjugation of ATG 12 to ATG 5, whereas ATG 7 and ATG 3 catalyse conjugation of PE to LC 3. The ATG 5– ATG 12–ATG 16 L 1 complex covalently links PE to LC 3, which becomes lipidated and associates with autophagosomes. SNARE proteins mediate fusion between autophagosomes and lysosomes and various lysosomal enzymes hydrolyze proteins, lipids and nucleic acids. Abbreviations: AMPK, AMPactivated protein kinase; ATG, autophagy-related protein; ATG 16 L 1, Autophagy-related protein 16 -1; DEPTOR, DEP domain-containing m. TOR-interacting protein; FIP 200, FAK family kinase-interacting protein of 200 k. Da; LC 3, microtubule-associated protein 1 light chain 3; m. LST 8, target of rapamycin complex subunit LST 8; m. TOR, mammalian target of rapamycin; m. TORC 1, m. TOR complex 1; PE, phosphatidyl ethanolamine; PRAS 40, proline-rich AKT 1 substrate 1; Raptor, regulatory-associated protein of m. TOR; SNARE, soluble NSF attachment protein receptor; ULK 1, serine/threonine-protein kinase ULK 1; VMP 1, vacuole membrane protein 1; Vps, vacuolar protein sorting.

Nature LETTER doi: 10. 1038/nature 14506 Receptor-mediated selective autophagy degrades the endoplasmic reticulum and the nucleus Keisuke Mochida 1, Yu Oikawa 2, Yayoi Kimura 3, Hiromi Kirisako 1, 4, Hisashi Hirano 3, Yoshinori Ohsumi 2 & Hitoshi Nakatogawa 1, 4
- Slides: 9