III METABOLIC BIOCHEMISTRY 3 1 Glucose Catabolism 3

III. METABOLIC BIOCHEMISTRY § 3. 1 Glucose Catabolism § 3. 1 a Metabolic Overview § 3. 1 b Glycolysis Pathway § 3. 1 c Glycolysis Regulation § 3. 1 d Anaerobic Glycolysis
§ 3. 1 a Metabolic Overview

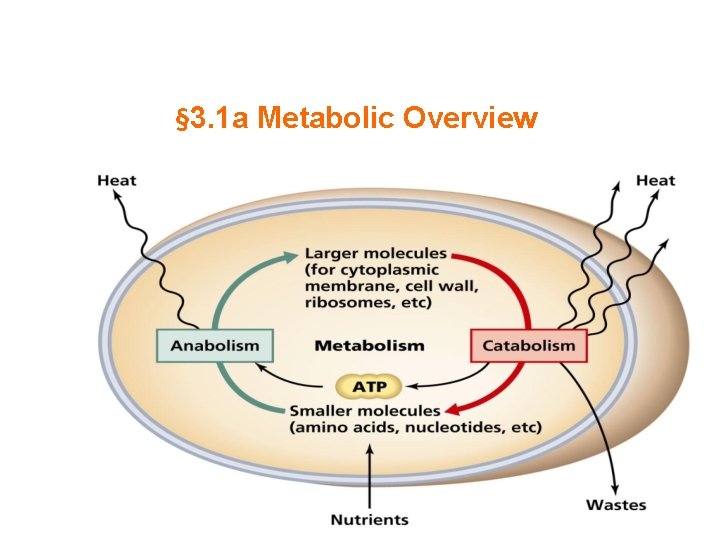
Synopsis 3. 1 a - Metabolism is the process through which living organisms break down nutrients (or synthesize biomolecules) to generate energy needed to drive biochemical processes needed for the maintenance of life—ie the sum of all chemical reactions in the body! - In metabolic processes, nutrients can be broken down to generate energy (catabolism), or biomolecules can be synthesized from simpler chemicals (anabolism) - Metabolic processes usually occur via a series of enzyme-catalyzed reactions that are collectively referred to as a “metabolic pathway” - A typical metabolic pathway involves the breakdown of a substrate into a product (or vice versa) via a series of intermediates because such metabolites usually “feed” into other pathways and serve as “entry points” for other pathways to “offload” their excess products—simply put, such intermediates allow metabolic pathways to “communicate” or “cross-talk” with each other so as to work together and cooperate in order to maintain cellular homeostasis - The term “metabolite” refers to small molecules involved in metabolism (eg substrates, products, intermediates, vitamins, cofactors, and so forth)—nearly almost all chemicals (excluding macromolecules) in the body are essentially metabolites - The metabolic breakdown of nutrients (digestion) largely occurs in the gut—technically referred to as the gastrointestinal (GI) tract, digestive tract, or alimentary canal
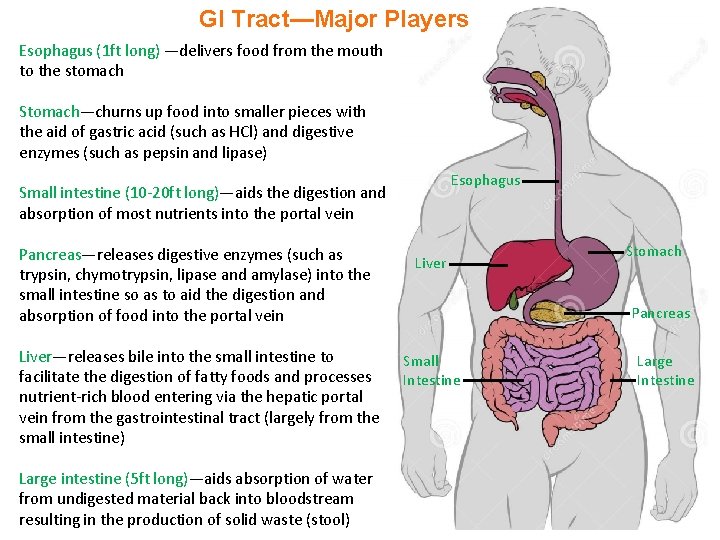
GI Tract—Major Players Esophagus (1 ft long) —delivers food from the mouth to the stomach Stomach—churns up food into smaller pieces with the aid of gastric acid (such as HCl) and digestive enzymes (such as pepsin and lipase) Esophagus Small intestine (10 -20 ft long)—aids the digestion and absorption of most nutrients into the portal vein Pancreas—releases digestive enzymes (such as trypsin, chymotrypsin, lipase and amylase) into the small intestine so as to aid the digestion and absorption of food into the portal vein Liver—releases bile into the small intestine to facilitate the digestion of fatty foods and processes nutrient-rich blood entering via the hepatic portal vein from the gastrointestinal tract (largely from the small intestine) Large intestine (5 ft long)—aids absorption of water from undigested material back into bloodstream resulting in the production of solid waste (stool) Liver Stomach Pancreas Small Intestine Large Intestine
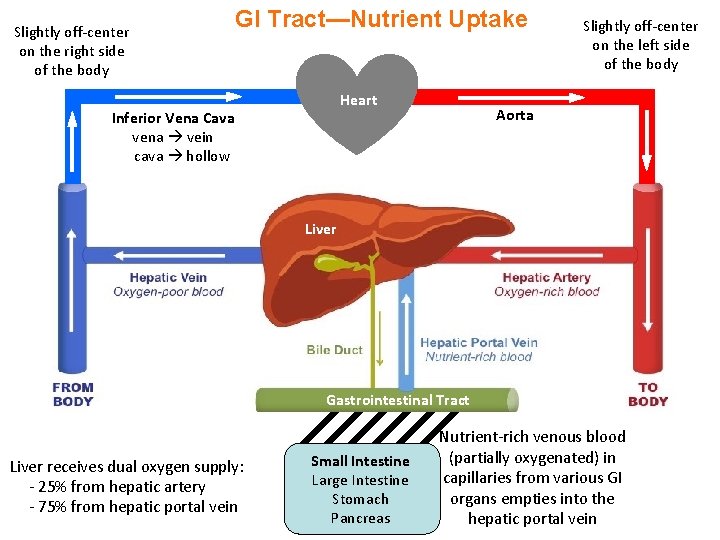
Slightly off-center on the right side of the body GI Tract—Nutrient Uptake Heart Inferior Vena Cava vena vein cava hollow Slightly off-center on the left side of the body Aorta Liver Gastrointestinal Tract Liver receives dual oxygen supply: - 25% from hepatic artery - 75% from hepatic portal vein Small Intestine Large Intestine Stomach Pancreas Nutrient-rich venous blood (partially oxygenated) in capillaries from various GI organs empties into the hepatic portal vein
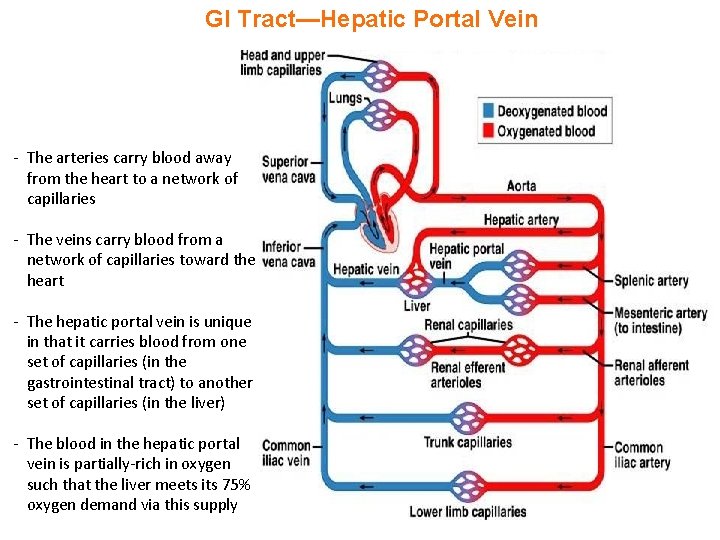
GI Tract—Hepatic Portal Vein - The arteries carry blood away from the heart to a network of capillaries - The veins carry blood from a network of capillaries toward the heart - The hepatic portal vein is unique in that it carries blood from one set of capillaries (in the gastrointestinal tract) to another set of capillaries (in the liver) - The blood in the hepatic portal vein is partially-rich in oxygen such that the liver meets its 75% oxygen demand via this supply
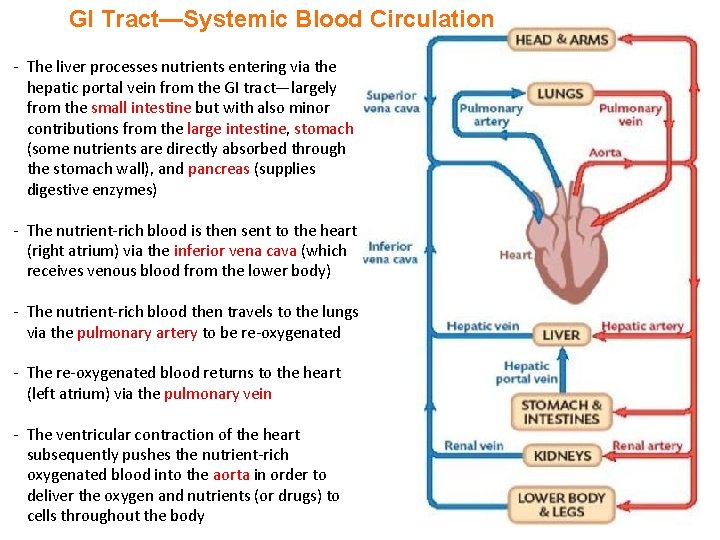
GI Tract—Systemic Blood Circulation - The liver processes nutrients entering via the hepatic portal vein from the GI tract—largely from the small intestine but with also minor contributions from the large intestine, stomach (some nutrients are directly absorbed through the stomach wall), and pancreas (supplies digestive enzymes) - The nutrient-rich blood is then sent to the heart (right atrium) via the inferior vena cava (which receives venous blood from the lower body) - The nutrient-rich blood then travels to the lungs via the pulmonary artery to be re-oxygenated - The re-oxygenated blood returns to the heart (left atrium) via the pulmonary vein - The ventricular contraction of the heart subsequently pushes the nutrient-rich oxygenated blood into the aorta in order to deliver the oxygen and nutrients (or drugs) to cells throughout the body
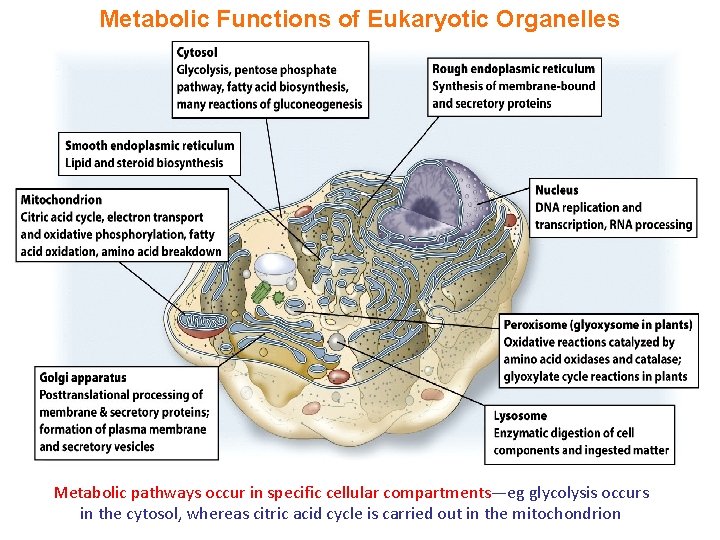
Metabolic Functions of Eukaryotic Organelles Metabolic pathways occur in specific cellular compartments—eg glycolysis occurs in the cytosol, whereas citric acid cycle is carried out in the mitochondrion
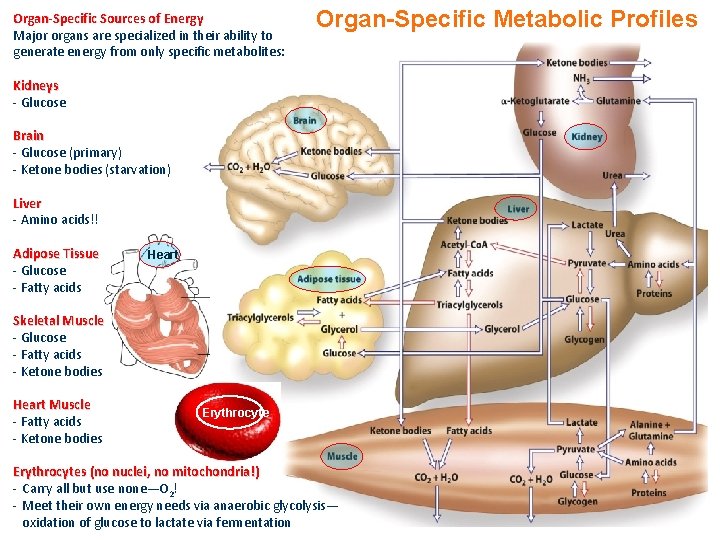
Organ-Specific Sources of Energy Major organs are specialized in their ability to generate energy from only specific metabolites: Organ-Specific Metabolic Profiles Kidneys - Glucose Brain - Glucose (primary) - Ketone bodies (starvation) Liver - Amino acids!! Adipose Tissue - Glucose - Fatty acids Heart Skeletal Muscle - Glucose - Fatty acids - Ketone bodies Heart Muscle - Fatty acids - Ketone bodies Erythrocytes (no nuclei, no mitochondria!) - Carry all but use none—O 2! - Meet their own energy needs via anaerobic glycolysis— oxidation of glucose to lactate via fermentation

Types of Metabolic Nutrients Nutrition (dietary intake) can be divided into two major categories: 1) Macronutrients—required by the body in macro quantities (typically g)! These generally serve as body’s fuel (source of energy) and also provide the body with raw materials for the synthesis of more complex molecules. Examples include proteins, carbohydrates, lipids, and water (non-caloric). 2) Micronutrients—required by the body in micro quantities (typically g)! These have no intrinsic caloric value but they are essential for the body to carry out metabolic reactions involved in supplying the body with its energy needs as well as in the synthesis of complex molecules. Examples include vitamins and minerals.
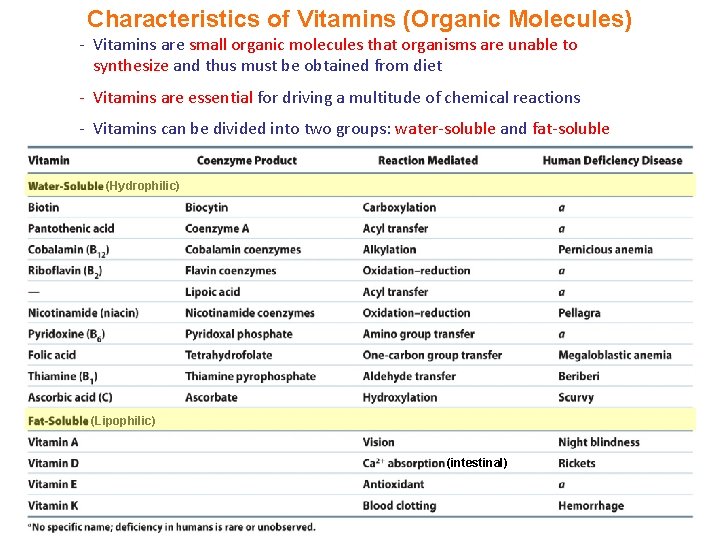
Characteristics of Vitamins (Organic Molecules) - Vitamins are small organic molecules that organisms are unable to synthesize and thus must be obtained from diet - Vitamins are essential for driving a multitude of chemical reactions - Vitamins can be divided into two groups: water-soluble and fat-soluble (Hydrophilic) (Lipophilic) (intestinal)
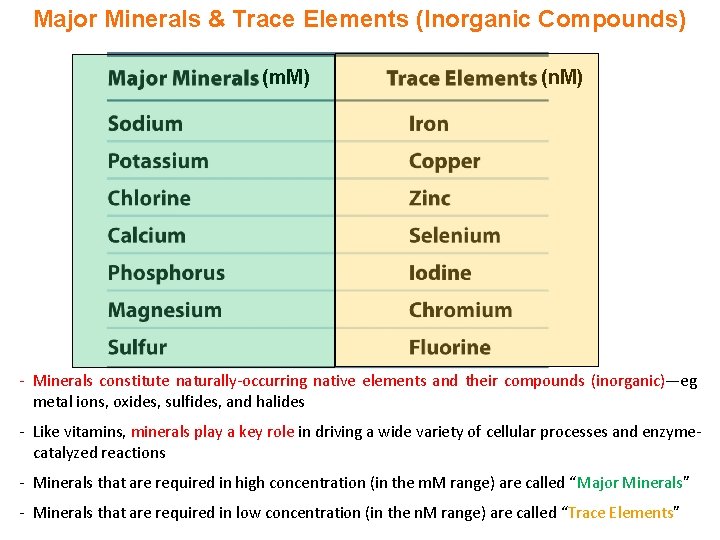
Major Minerals & Trace Elements (Inorganic Compounds) (m. M) (n. M) - Minerals constitute naturally-occurring native elements and their compounds (inorganic)—eg metal ions, oxides, sulfides, and halides - Like vitamins, minerals play a key role in driving a wide variety of cellular processes and enzymecatalyzed reactions - Minerals that are required in high concentration (in the m. M range) are called “Major Minerals” - Minerals that are required in low concentration (in the n. M range) are called “Trace Elements”
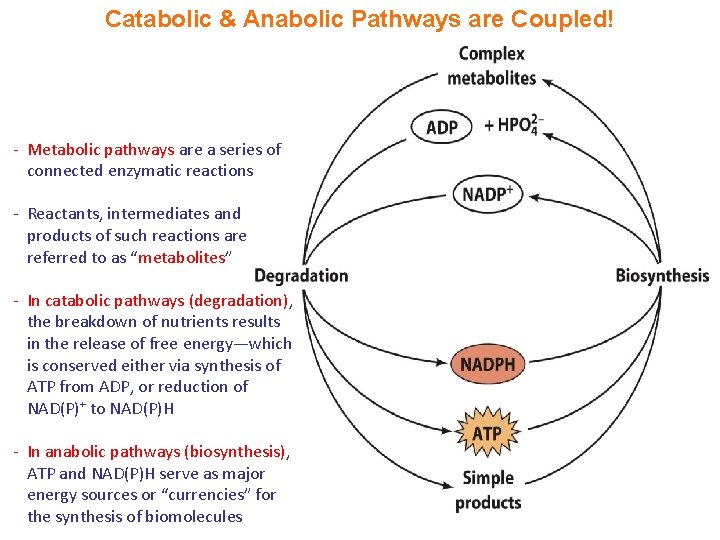
Catabolic & Anabolic Pathways are Coupled! - Metabolic pathways are a series of connected enzymatic reactions - Reactants, intermediates and products of such reactions are referred to as “metabolites” - In catabolic pathways (degradation), the breakdown of nutrients results in the release of free energy—which is conserved either via synthesis of ATP from ADP, or reduction of NAD(P)+ to NAD(P)H - In anabolic pathways (biosynthesis), ATP and NAD(P)H serve as major energy sources or “currencies” for the synthesis of biomolecules
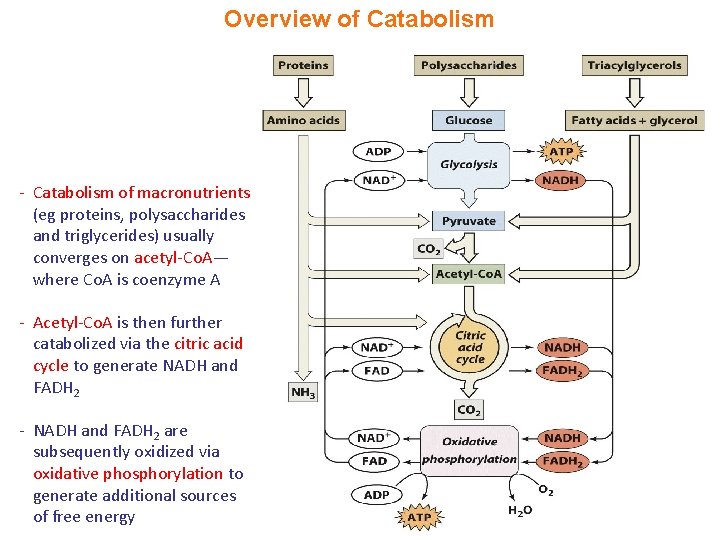
Overview of Catabolism - Catabolism of macronutrients (eg proteins, polysaccharides and triglycerides) usually converges on acetyl-Co. A— where Co. A is coenzyme A - Acetyl-Co. A is then further catabolized via the citric acid cycle to generate NADH and FADH 2 - NADH and FADH 2 are subsequently oxidized via oxidative phosphorylation to generate additional sources of free energy

Major Characteristics of Metabolic Pathways (1) Metabolic pathways are irreversible - While most enzymatically-driven steps in a metabolic pathway operate near equilibrium ( G 0), metabolic pathways usually harbor one or more exergonic steps that operate far from equilibrium ( G << 0) - Such thermodynamic behavior imparts directionality on metabolic pathways (2) Metabolic pathways harbor a committed step - Metabolic pathways are usually characterized by an exergonic step ( G << 0) early in the pathway that “commits” the pathway to continue downstream (3) Catabolic and anabolic pathways differ - Given that the conversion of metabolite 1 to metabolite 2 via intermediate A is exergonic ( G << 0), the same route cannot be used for the synthesis of metabolite 1 from metabolite 2 due to unfavorable thermodynamics - Rather, a separate pathway must be employed to convert metabolite 2 back to metabolite 1 via intermediates X and Y - Existence of such independent pathways is an important property of metabolic pathways as it allows independent control of degradation versus biosynthesis

Metabolic Flux - Living organisms are thermodynamically open systems that tend to maintain a steady-state rather than reach equilibrium—doing so would equate to death! - “Steady-state” implies that the rates of synthesis and degradation of metabolic intermediates within a cell are more or less equal such that their concentrations change little over time—eg the blood glucose level is maintained @ 1 mg/ml! - The rate of flow of metabolites through a metabolic pathway is referred to as “metabolic flux”—it is determined by the slowest or the rate-determining step in a metabolic pathway - The metabolic flux of a pathway can be altered or modulated depending on the needs of the cell and/or organism - This is usually achieved by altering the rate of the rate-determining step in a metabolic path via one of the following mechanisms: (1) Allosteric Control (2) Feedback Inhibition (3) Feedforth Activation (4) Post-Translational Modification (5) Substrate Cycling (6) Gene Expression
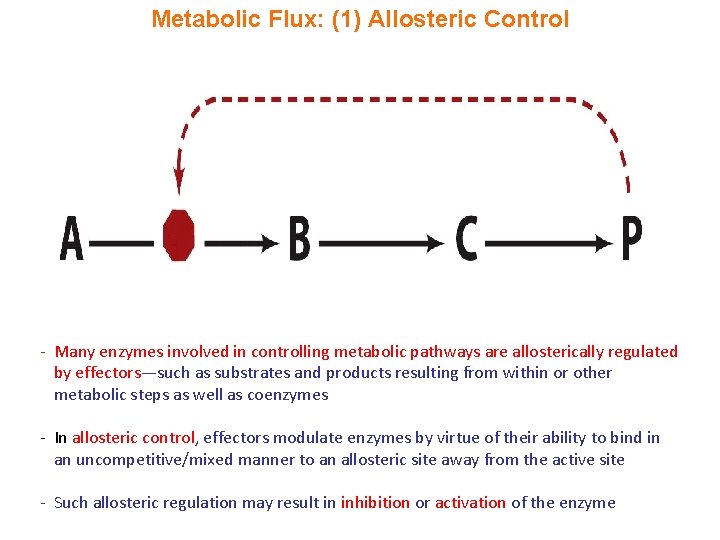
Metabolic Flux: (1) Allosteric Control - Many enzymes involved in controlling metabolic pathways are allosterically regulated by effectors—such as substrates and products resulting from within or other metabolic steps as well as coenzymes - In allosteric control, effectors modulate enzymes by virtue of their ability to bind in an uncompetitive/mixed manner to an allosteric site away from the active site - Such allosteric regulation may result in inhibition or activation of the enzyme
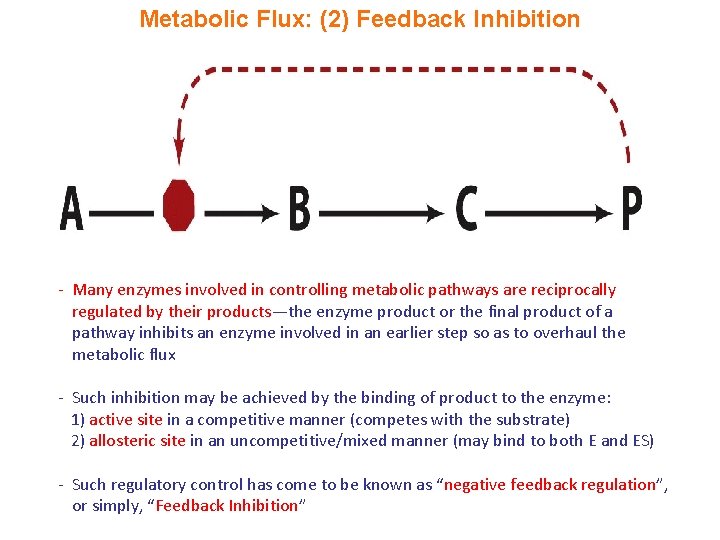
Metabolic Flux: (2) Feedback Inhibition - Many enzymes involved in controlling metabolic pathways are reciprocally regulated by their products—the enzyme product or the final product of a pathway inhibits an enzyme involved in an earlier step so as to overhaul the metabolic flux - Such inhibition may be achieved by the binding of product to the enzyme: 1) active site in a competitive manner (competes with the substrate) 2) allosteric site in an uncompetitive/mixed manner (may bind to both E and ES) - Such regulatory control has come to be known as “negative feedback regulation”, or simply, “Feedback Inhibition”

Metabolic Flux: (3) Feedforth Activation E 2 E 1 S P - Enzymes may be activated by their own substrates—the enzyme E 1 substrate may augment E 1 activity, usually by inhibiting enzyme E 2 involved in inhibiting E 1 through mechanisms such as post-translational modification (PTM) - Such inhibition is usually achieved by the binding of enzyme E 1 substrate to an allosteric site in enzyme E 2 in an uncompetitive/mixed manner (may bind to both E and ES) - Such regulatory control is called “Feedforth activation”
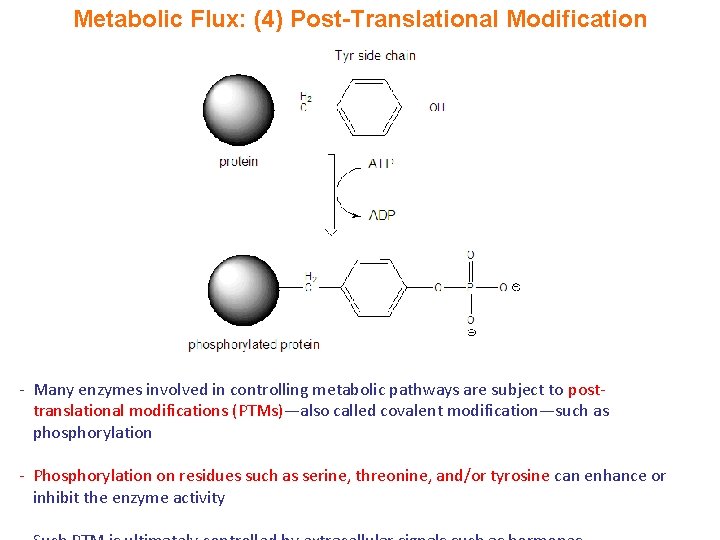
Metabolic Flux: (4) Post-Translational Modification - Many enzymes involved in controlling metabolic pathways are subject to posttranslational modifications (PTMs)—also called covalent modification—such as phosphorylation - Phosphorylation on residues such as serine, threonine, and/or tyrosine can enhance or inhibit the enzyme activity
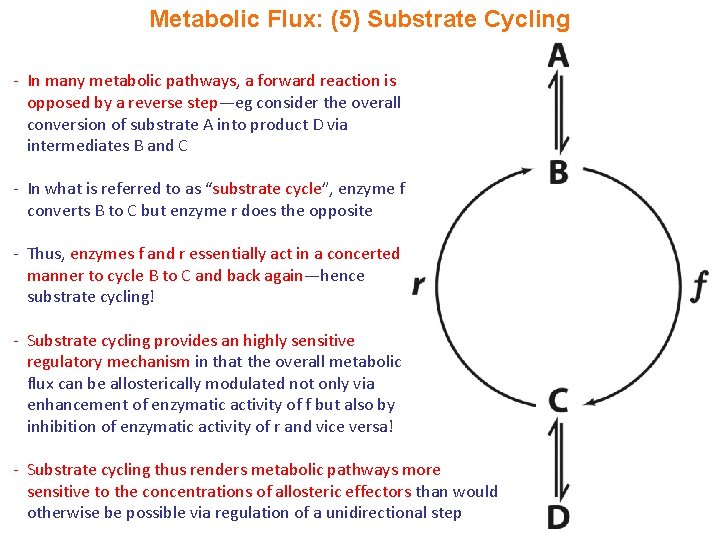
Metabolic Flux: (5) Substrate Cycling - In many metabolic pathways, a forward reaction is opposed by a reverse step—eg consider the overall conversion of substrate A into product D via intermediates B and C - In what is referred to as “substrate cycle”, enzyme f converts B to C but enzyme r does the opposite - Thus, enzymes f and r essentially act in a concerted manner to cycle B to C and back again—hence substrate cycling! - Substrate cycling provides an highly sensitive regulatory mechanism in that the overall metabolic flux can be allosterically modulated not only via enhancement of enzymatic activity of f but also by inhibition of enzymatic activity of r and vice versa! - Substrate cycling thus renders metabolic pathways more sensitive to the concentrations of allosteric effectors than would otherwise be possible via regulation of a unidirectional step
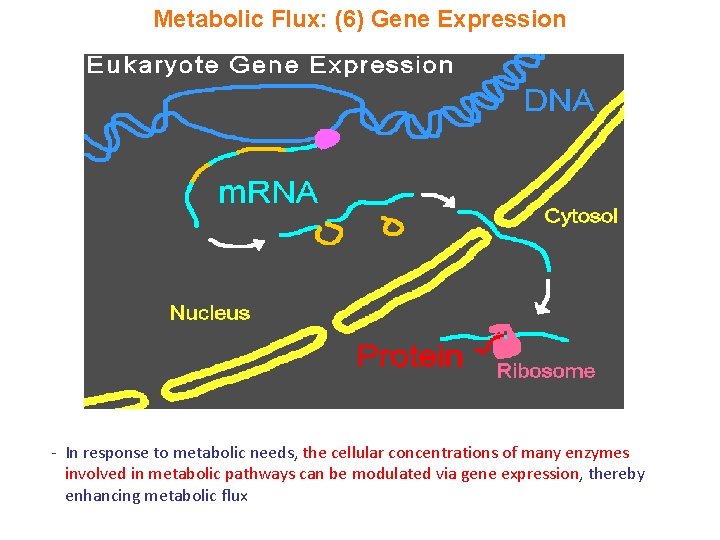
Metabolic Flux: (6) Gene Expression - In response to metabolic needs, the cellular concentrations of many enzymes involved in metabolic pathways can be modulated via gene expression, thereby enhancing metabolic flux

Exercise 3. 1 a - List the categories of macronutrients and micronutrients required for mammalian metabolism and provide examples of each - Explain the roles of ATP and NADPH in catabolic and anabolic reactions - Give some reasons why enzymes are essential for the operation of metabolic pathways - Explain the metabolic significance of reactions that function near equilibrium and reactions that function far from equilibrium - Discuss the mechanisms by which the flux through a metabolic pathway can be controlled. Which mechanisms can rapidly alter flux?
§ 3. 1 b Glycolysis Pathway

Synopsis 3. 1 b - Dietary starch (eg bread, rice and potatoes) is hydrolyzed into glucose by the combined action of enzymes such as amylase (saliva) and maltase (small intestine) - Glycolysis involves the breakdown/oxidation of glucose into pyruvate using a wide array of enzymes—and the free energy released in the process is either used to synthesize ATP or captured in the form of NADH - In terms of chemical reactions, the glycolytic enzymes catalyze phosphorylation (transferase), isomerization (isomerase), bond cleavage (lyase), dehydrogenation (oxidoreductase), and hydrolysis (hydrolase) - Of the six major classes/families of enzymes (see § 2. 5), all but ligase are involved in mediating glycolysis! - The 10 -reaction sequence of glycolysis is divided into two stages: Stage I Energy investment/expenditure Stage II Energy recovery/payoff - Overall glycolytic reaction: Glucose + 2 NAD+ + 2 ADP + 2 Pi <=> 2 Pyruvate + 2 NADH + 2 ATP + 2 H 2 O + 2 H+
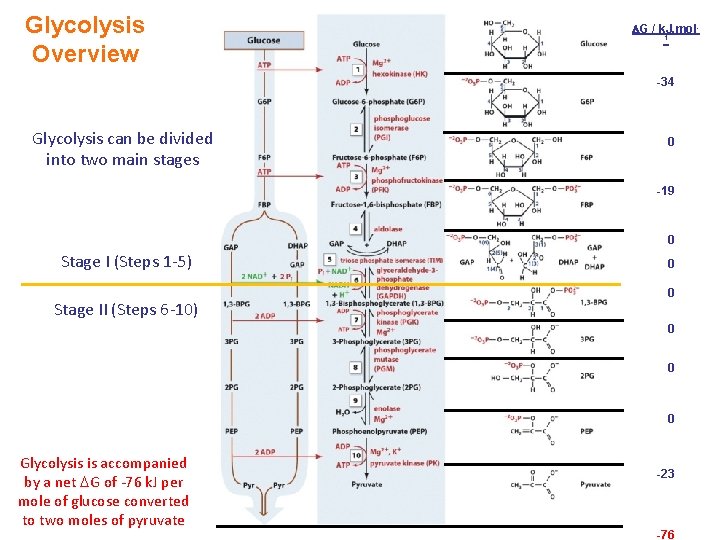
Glycolysis Overview G / k. J. mol 1 -34 Glycolysis can be divided into two main stages 0 -19 0 Stage I (Steps 1 -5) Stage II (Steps 6 -10) 0 0 0 Glycolysis is accompanied by a net G of -76 k. J per mole of glucose converted to two moles of pyruvate -23 -76

Glycolysis Stage I (Investment): Glucose GAP 1 2 3 In Stage I (Steps 1 -5): - 1 molecule of glucose is converted to 2 molecules of glyceraldehyde-3 phosphate (GAP) 4 - 2 molecules of ATP are utilized (energy investment) 5
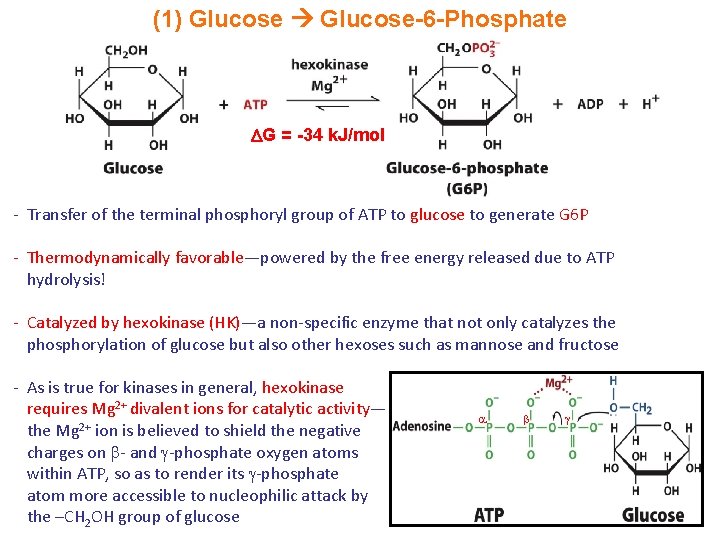
(1) Glucose-6 -Phosphate G = -34 k. J/mol - Transfer of the terminal phosphoryl group of ATP to glucose to generate G 6 P - Thermodynamically favorable—powered by the free energy released due to ATP hydrolysis! - Catalyzed by hexokinase (HK)—a non-specific enzyme that not only catalyzes the phosphorylation of glucose but also other hexoses such as mannose and fructose - As is true for kinases in general, hexokinase requires Mg 2+ divalent ions for catalytic activity— the Mg 2+ ion is believed to shield the negative charges on - and -phosphate oxygen atoms within ATP, so as to render its -phosphate atom more accessible to nucleophilic attack by the –CH 2 OH group of glucose
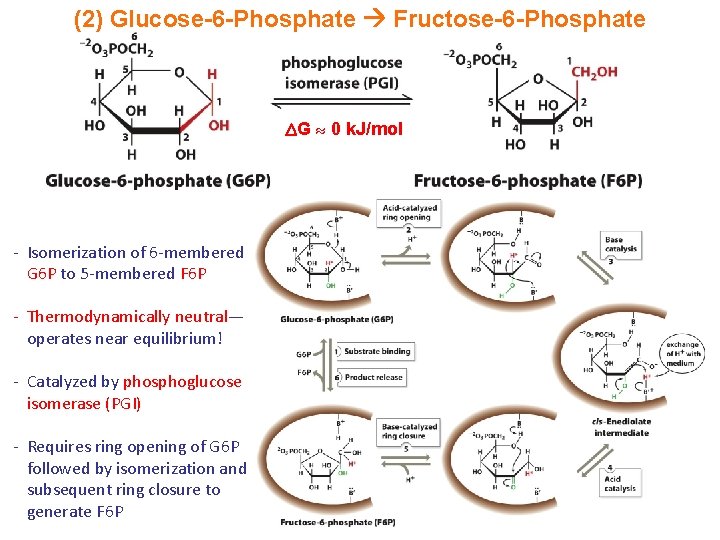
(2) Glucose-6 -Phosphate Fructose-6 -Phosphate G 0 k. J/mol - Isomerization of 6 -membered G 6 P to 5 -membered F 6 P - Thermodynamically neutral— operates near equilibrium! - Catalyzed by phosphoglucose isomerase (PGI) - Requires ring opening of G 6 P followed by isomerization and subsequent ring closure to generate F 6 P
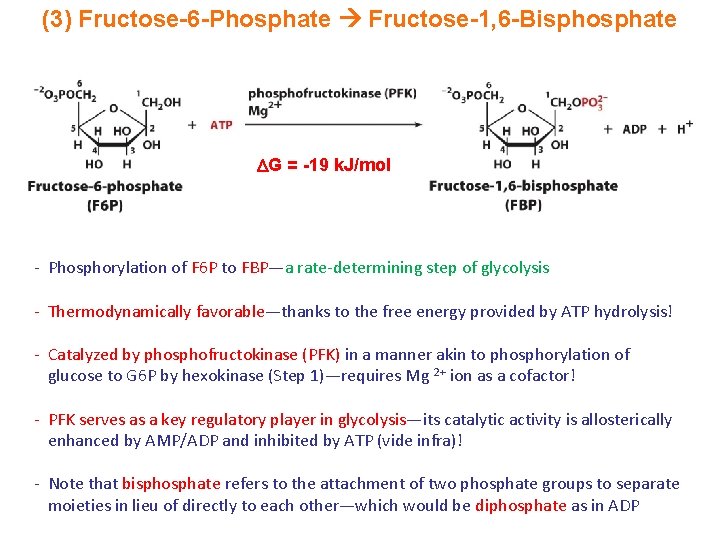
(3) Fructose-6 -Phosphate Fructose-1, 6 -Bisphosphate G = -19 k. J/mol - Phosphorylation of F 6 P to FBP—a rate-determining step of glycolysis - Thermodynamically favorable—thanks to the free energy provided by ATP hydrolysis! - Catalyzed by phosphofructokinase (PFK) in a manner akin to phosphorylation of glucose to G 6 P by hexokinase (Step 1)—requires Mg 2+ ion as a cofactor! - PFK serves as a key regulatory player in glycolysis—its catalytic activity is allosterically enhanced by AMP/ADP and inhibited by ATP (vide infra)! - Note that bisphosphate refers to the attachment of two phosphate groups to separate moieties in lieu of directly to each other—which would be diphosphate as in ADP
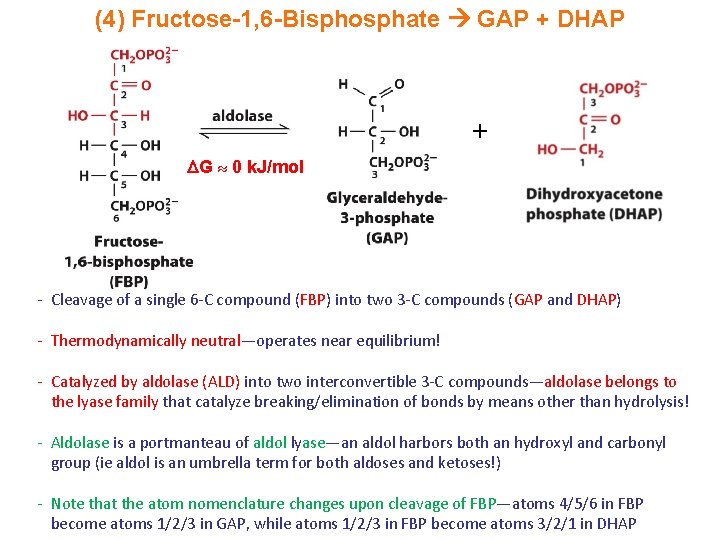
(4) Fructose-1, 6 -Bisphosphate GAP + DHAP + G 0 k. J/mol - Cleavage of a single 6 -C compound (FBP) into two 3 -C compounds (GAP and DHAP) - Thermodynamically neutral—operates near equilibrium! - Catalyzed by aldolase (ALD) into two interconvertible 3 -C compounds—aldolase belongs to the lyase family that catalyze breaking/elimination of bonds by means other than hydrolysis! - Aldolase is a portmanteau of aldol lyase—an aldol harbors both an hydroxyl and carbonyl group (ie aldol is an umbrella term for both aldoses and ketoses!) - Note that the atom nomenclature changes upon cleavage of FBP—atoms 4/5/6 in FBP become atoms 1/2/3 in GAP, while atoms 1/2/3 in FBP become atoms 3/2/1 in DHAP
![(5) Dihydroxyacetone Phosphate Glyceraldehyde-3 Phosphate triose phosphate isomerase [DHAP] G 0 k. J/mol [GAP] (5) Dihydroxyacetone Phosphate Glyceraldehyde-3 Phosphate triose phosphate isomerase [DHAP] G 0 k. J/mol [GAP]](http://slidetodoc.com/presentation_image/184c1c0c64c2cd61420d975891d67e1e/image-32.jpg)
(5) Dihydroxyacetone Phosphate Glyceraldehyde-3 Phosphate triose phosphate isomerase [DHAP] G 0 k. J/mol [GAP] - Interconversion of DHAP to GAP via an enediol intermediate - Thermodynamically neutral—operates near equilibrium! - Catalyzed by triose phosphate isomerase (TIM)—a “perfect” enzyme in that it operates near the diffusion-controlled limit with kcat/KM 109 M -1 s-1 (see § 2. 6) - DHAP and GAP are ketose-aldose isomers - Only GAP continues along the glycolytic pathway—as GAP is siphoned off, more DHAP is converted to GAP due to “equilibrium shift”!
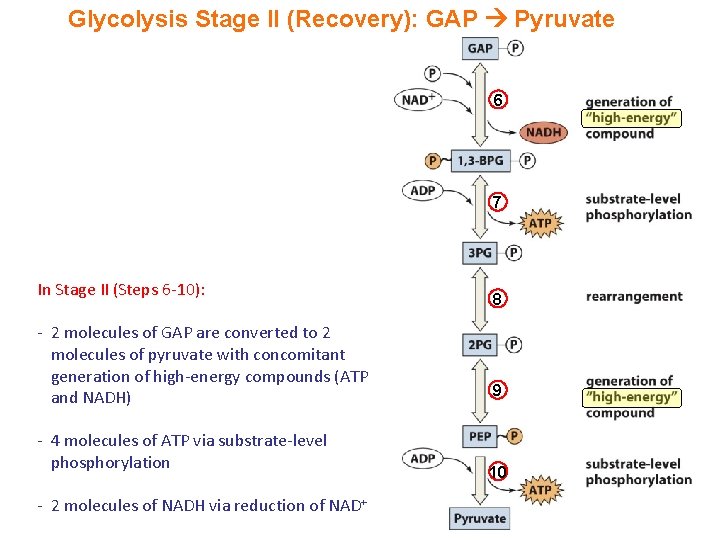
Glycolysis Stage II (Recovery): GAP Pyruvate 6 7 In Stage II (Steps 6 -10): - 2 molecules of GAP are converted to 2 molecules of pyruvate with concomitant generation of high-energy compounds (ATP and NADH) - 4 molecules of ATP via substrate-level phosphorylation - 2 molecules of NADH via reduction of NAD+ 8 9 10
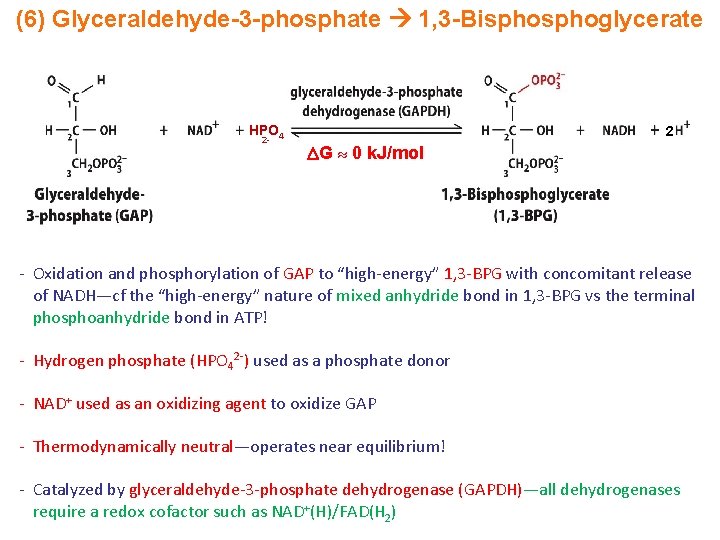
(6) Glyceraldehyde-3 -phosphate 1, 3 -Bisphoglycerate HPO 4 2 - 2 G 0 k. J/mol - Oxidation and phosphorylation of GAP to “high-energy” 1, 3 -BPG with concomitant release of NADH—cf the “high-energy” nature of mixed anhydride bond in 1, 3 -BPG vs the terminal phosphoanhydride bond in ATP! - Hydrogen phosphate (HPO 42 -) used as a phosphate donor - NAD+ used as an oxidizing agent to oxidize GAP - Thermodynamically neutral—operates near equilibrium! - Catalyzed by glyceraldehyde-3 -phosphate dehydrogenase (GAPDH)—all dehydrogenases require a redox cofactor such as NAD+(H)/FAD(H 2)
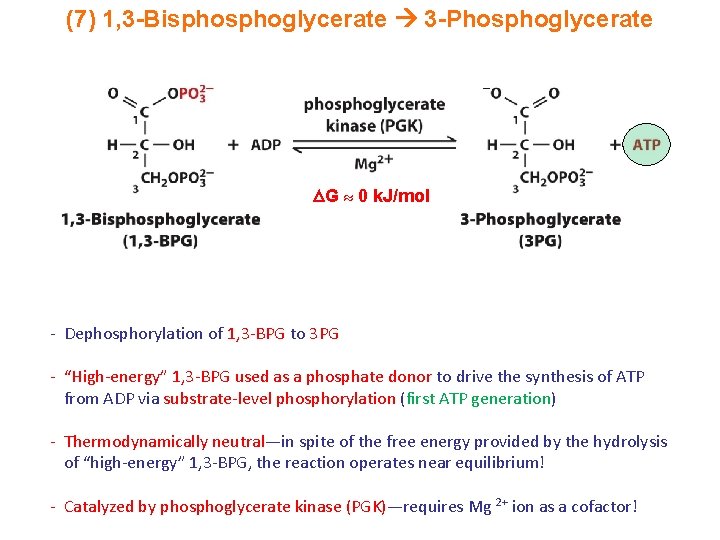
(7) 1, 3 -Bisphoglycerate 3 -Phosphoglycerate G 0 k. J/mol - Dephosphorylation of 1, 3 -BPG to 3 PG - “High-energy” 1, 3 -BPG used as a phosphate donor to drive the synthesis of ATP from ADP via substrate-level phosphorylation (first ATP generation) - Thermodynamically neutral—in spite of the free energy provided by the hydrolysis of “high-energy” 1, 3 -BPG, the reaction operates near equilibrium! - Catalyzed by phosphoglycerate kinase (PGK)—requires Mg 2+ ion as a cofactor!
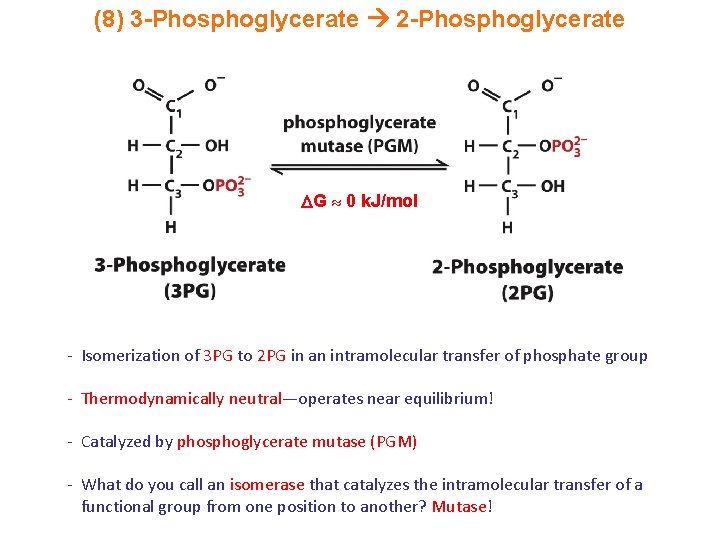
(8) 3 -Phosphoglycerate 2 -Phosphoglycerate G 0 k. J/mol - Isomerization of 3 PG to 2 PG in an intramolecular transfer of phosphate group - Thermodynamically neutral—operates near equilibrium! - Catalyzed by phosphoglycerate mutase (PGM) - What do you call an isomerase that catalyzes the intramolecular transfer of a functional group from one position to another? Mutase!
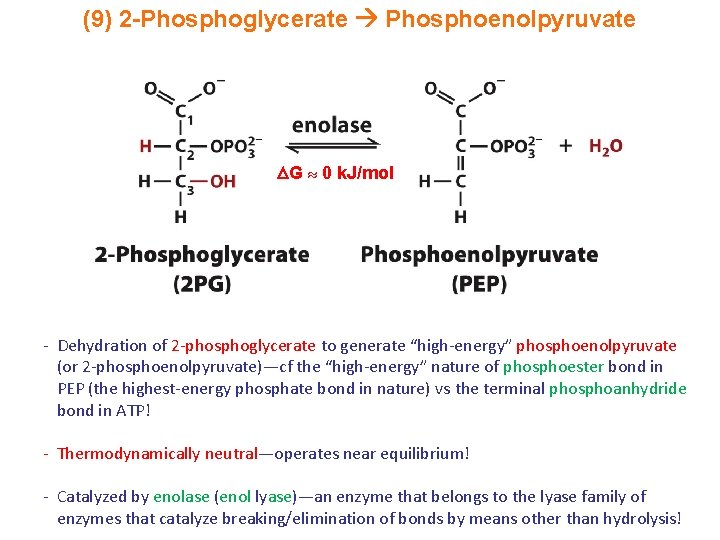
(9) 2 -Phosphoglycerate Phosphoenolpyruvate G 0 k. J/mol - Dehydration of 2 -phosphoglycerate to generate “high-energy” phosphoenolpyruvate (or 2 -phosphoenolpyruvate)—cf the “high-energy” nature of phosphoester bond in PEP (the highest-energy phosphate bond in nature) vs the terminal phosphoanhydride bond in ATP! - Thermodynamically neutral—operates near equilibrium! - Catalyzed by enolase (enol lyase)—an enzyme that belongs to the lyase family of enzymes that catalyze breaking/elimination of bonds by means other than hydrolysis!
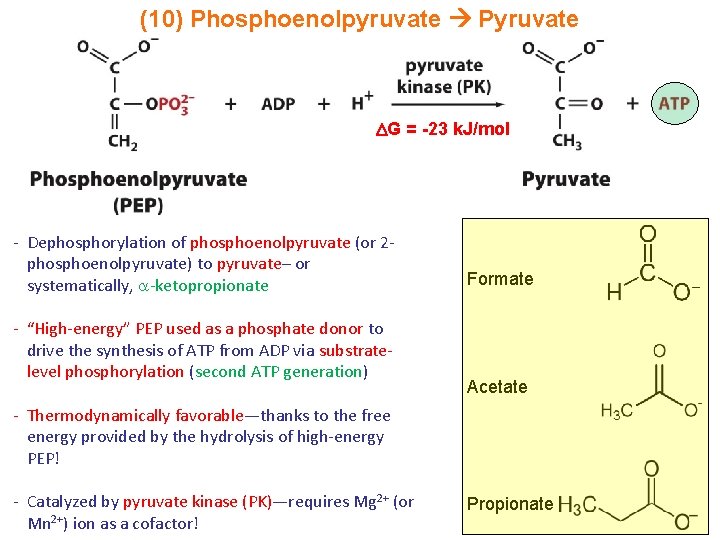
(10) Phosphoenolpyruvate Pyruvate G = -23 k. J/mol - Dephosphorylation of phosphoenolpyruvate (or 2 phosphoenolpyruvate) to pyruvate– or systematically, -ketopropionate - “High-energy” PEP used as a phosphate donor to drive the synthesis of ATP from ADP via substratelevel phosphorylation (second ATP generation) Formate Acetate - Thermodynamically favorable—thanks to the free energy provided by the hydrolysis of high-energy PEP! - Catalyzed by pyruvate kinase (PK)—requires Mg 2+ (or Mn 2+) ion as a cofactor! Propionate

Exercise 3. 1 b - What happens during the two phases of glycolysis? - How many ATP molecules are invested and how many are recovered from each molecule of glucose that follows the glycolytic pathway? - Write the reactions of glycolysis, showing the structural formulas of the intermediates and the names of the enzymes that catalyze the reactions. Distinguish between thermodynamically neutral and favorable steps. - Summarize the types of catalytic mechanisms involved. Do any glycolytic enzymes require cofactors? - What “high-energy” compounds are synthesized during glycolysis?
§ 3. 1 c Glycolysis Regulation

Synopsis 3. 1 c - Enzymes that function with a large negative G are candidates for flux-control points - Phosphofructokinase (PFK), the major regulatory point for glycolysis in muscle, is allosterically inhibited by ATP and activated by AMP/ADP - Substrate cycling allows the rate of glycolysis to respond rapidly to changing needs
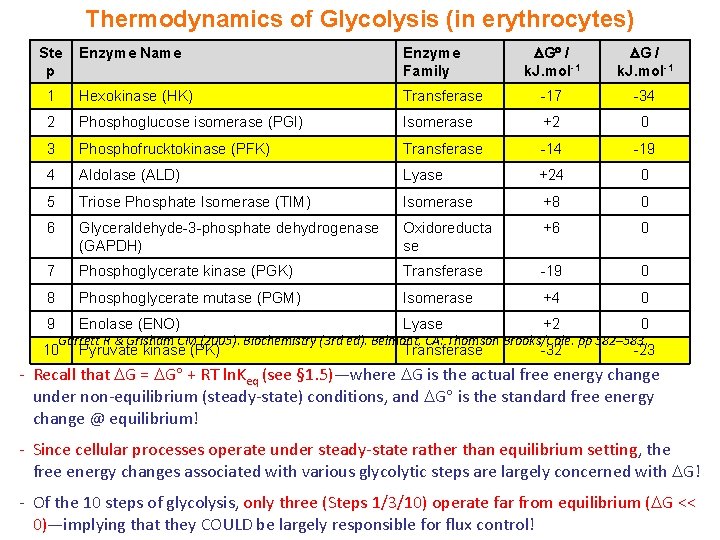
Thermodynamics of Glycolysis (in erythrocytes) G / k. J. mol-1 Transferase -17 -34 Phosphoglucose isomerase (PGI) Isomerase +2 0 3 Phosphofrucktokinase (PFK) Transferase -14 -19 4 Aldolase (ALD) Lyase +24 0 5 Triose Phosphate Isomerase (TIM) Isomerase +8 0 6 Glyceraldehyde-3 -phosphate dehydrogenase (GAPDH) Oxidoreducta se +6 0 7 Phosphoglycerate kinase (PGK) Transferase -19 0 8 Phosphoglycerate mutase (PGM) Isomerase +4 0 9 Enolase (ENO) Lyase +2 0 Pyruvate kinase (PK) Transferase -32 -23 Ste p Enzyme Name Enzyme Family 1 Hexokinase (HK) 2 Garrett R & Grisham CM (2005). Biochemistry (3 rd ed). Belmont, CA: Thomson Brooks/Cole. pp 582– 583. 10 - Recall that G = G + RT ln. Keq (see § 1. 5)—where G is the actual free energy change under non-equilibrium (steady-state) conditions, and G is the standard free energy change @ equilibrium! - Since cellular processes operate under steady-state rather than equilibrium setting, the free energy changes associated with various glycolytic steps are largely concerned with G! - Of the 10 steps of glycolysis, only three (Steps 1/3/10) operate far from equilibrium ( G << 0)—implying that they COULD be largely responsible for flux control!
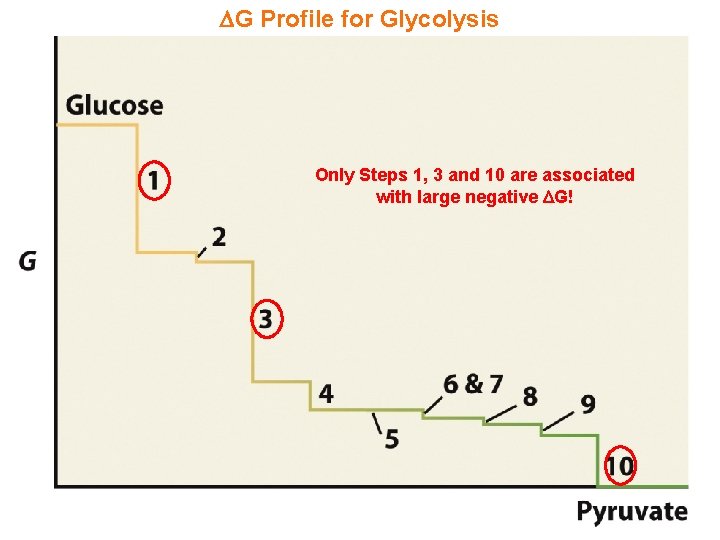
G Profile for Glycolysis Only Steps 1, 3 and 10 are associated with large negative G!

PFK Is the Major Flux-Controlling Enzyme - Actual free energy changes ( G) associated with various glycolytic steps suggest that the major candidates for flux control are: - Hexokinase (HK) => Step 1 - Phosphofructokinase (PFK) => Step 3 - Pyruvate kinase (PK) => Step 10 - Of these three enzymes, only PFK plays a central role in controlling the rate of flow of metabolites (or flux) through glycolysis—why? ! - HK is not absolutely critical for glycolysis (much less serve as a regulatory point!)—since glycolysis in skeletal muscle often does not require HK (due to the breakdown of glycogen into glucose-6 -phosphate via glycogenolysis)—see § 3. 2 - On the other hand, PK catalyzes the final step of glycolysis—thus its ability to control flux through glycolysis becomes somewhat moot - Simply put, the conversion of F 6 P to FBP in Step 3 by PFK is the major ratedetermining or rate-limiting (or the slowest) step of glycolysis - How is PFK regulated? In PFK, ATP not only binds to the active site (to serve as a substrate) but also to an allosteric site (to serve as an allosteric inhibitor)
![Allosteric Regulation of PFK In respiring muscle: PFK Activity [ATP]/[ADP] ~ 10: 1 F Allosteric Regulation of PFK In respiring muscle: PFK Activity [ATP]/[ADP] ~ 10: 1 F](http://slidetodoc.com/presentation_image/184c1c0c64c2cd61420d975891d67e1e/image-45.jpg)
Allosteric Regulation of PFK In respiring muscle: PFK Activity [ATP]/[ADP] ~ 10: 1 F 6 P / m. M - PFK is allsoterically inhibited by ATP—a built-in control to ensure that when ATP is in excess (there is little demand for energy production), glycolysis is shut off! - On the other hand, AMP/ADP serve as allosteric activators of PFK—rising cellular concentrations of AMP/ADP are indicative of energy shortage and thus serve as signals for the production of ATP - Acting in concert, AMP/ADP and ATP allsoterically modulate the enzymatic activity of PFK—thereby enabling PFK to play a key role in the control of flux through glycolysis
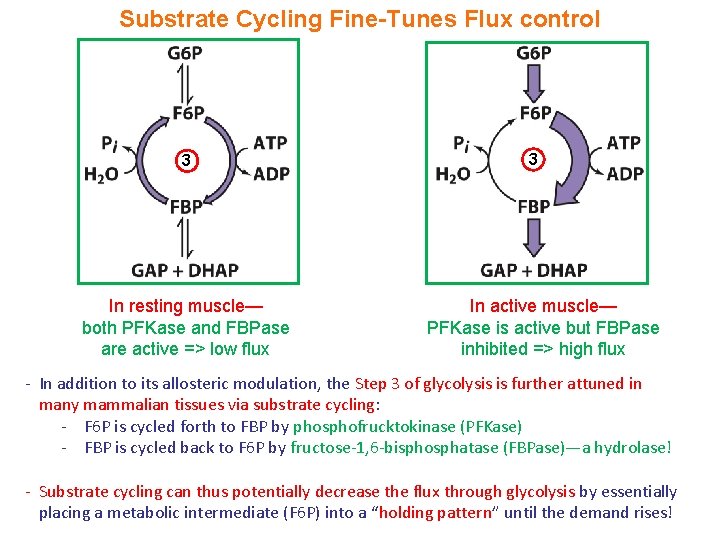
Substrate Cycling Fine-Tunes Flux control 3 In resting muscle— both PFKase and FBPase are active => low flux 3 In active muscle— PFKase is active but FBPase inhibited => high flux - In addition to its allosteric modulation, the Step 3 of glycolysis is further attuned in many mammalian tissues via substrate cycling: - F 6 P is cycled forth to FBP by phosphofrucktokinase (PFKase) - FBP is cycled back to F 6 P by fructose-1, 6 -bisphosphatase (FBPase)—a hydrolase! - Substrate cycling can thus potentially decrease the flux through glycolysis by essentially placing a metabolic intermediate (F 6 P) into a “holding pattern” until the demand rises!

Exercise 3. 1 c - Which glycolytic enzymes are potential control points? - Describe the mechanisms that control phosphofructokinase activity - What is the metabolic advantage of a substrate cycle?

§ 3. 1 d Anaerobic Glycolysis (Fermentation)

Synopsis 3. 1 d - Under aerobic conditions, the glycolytic end-product pyruvate is completely oxidized to CO 2 and H 2 O via the Krebs cycle (see § 3. 5) - Under anaerobic conditions, the glycolytic end-product pyruvate is converted (or reduced) to either lactate or ethanol in a metabolic process referred to as “fermentation” - Fermentation can be classified into two major groups: (1) Homolactic (or lactic acid) fermentation: —largely occurs in the muscle cells of animals but also in some bacteria (2) Alcoholic (or ethanol) fermentation: —largely occurs in yeast but also in some species of fish and plants - In each case, the physiological significance of fermentation is to reoxidize/recycle NADH back to NAD+ so as to to ensure glycolytic continuity—thus fermentation plays a key role in the regeneration of energy under anaerobic conditions! - In anaerobic glycolysis, glycolysis and fermentation are essentially coupled in that glucose is oxidized to either lactate or ethanol in a seamless fashion—erythrocytes obtain their energy exclusively through anaerobic glycolysis
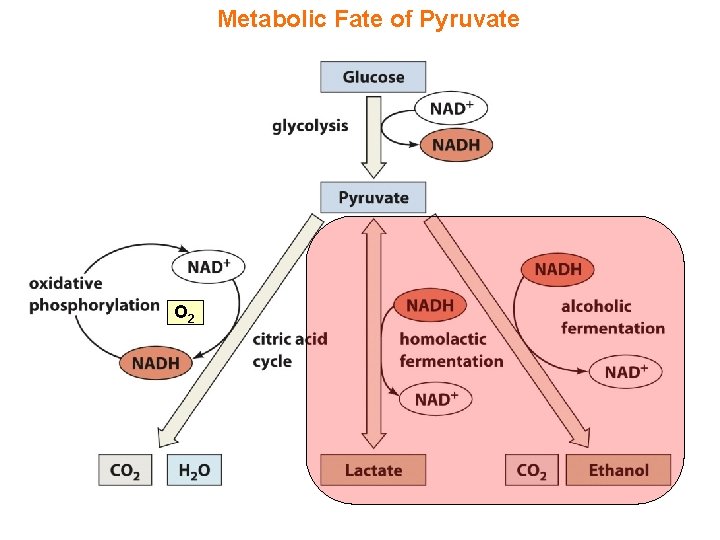
Metabolic Fate of Pyruvate O 2

Homolactic Fermentation (in muscle) - During strenuous activity when oxygen is in short supply, ATP is largely synthesized via anaerobic glycolysis in muscles - The pyruvate end-product of glycolysis is reduced to lactate in order to regenerate NAD+ (from NADH)—which is required for the continuity of anaerobic glycolysis—in a process known as “homolactic fermentation” - Homolactic fermentation is catalyzed by lactate dehydrogenase (LDH) - Under resting conditions when oxygen is no longer limiting, lactate is converted back to pyruvate via “equilibrium shift” to allow its complete oxidation via the citric acid cycle - When transported to the liver via the bloodstream, lactate can also be used to synthesize glucose via gluconeogenesis—generation of glucose from noncarbohydrate sources (including amino acids)
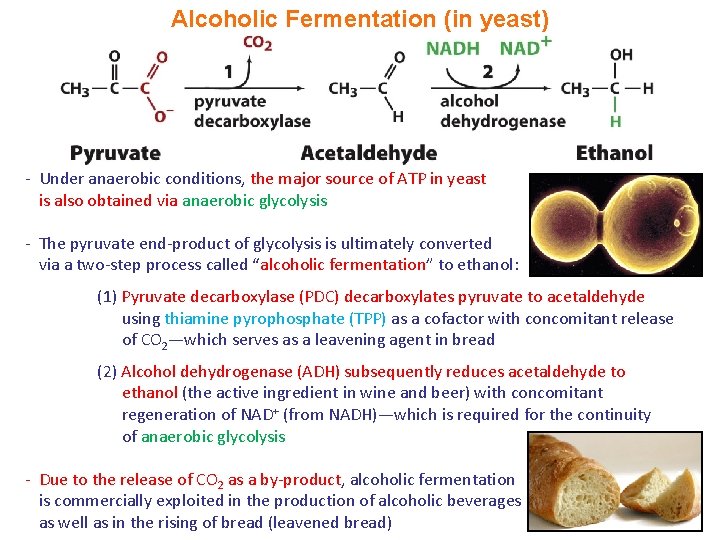
Alcoholic Fermentation (in yeast) - Under anaerobic conditions, the major source of ATP in yeast is also obtained via anaerobic glycolysis - The pyruvate end-product of glycolysis is ultimately converted via a two-step process called “alcoholic fermentation” to ethanol: (1) Pyruvate decarboxylase (PDC) decarboxylates pyruvate to acetaldehyde using thiamine pyrophosphate (TPP) as a cofactor with concomitant release of CO 2—which serves as a leavening agent in bread (2) Alcohol dehydrogenase (ADH) subsequently reduces acetaldehyde to ethanol (the active ingredient in wine and beer) with concomitant regeneration of NAD+ (from NADH)—which is required for the continuity of anaerobic glycolysis - Due to the release of CO 2 as a by-product, alcoholic fermentation is commercially exploited in the production of alcoholic beverages as well as in the rising of bread (leavened bread)

Exercise 3. 1 d - Describe three possible fates of pyruvate - Compare homolactic and alcoholic fermentation in terms of the products and the cofactors required
- Slides: 53