IHC Troubleshooting Jason A Ramos Ph D Director

IHC Troubleshooting Jason A. Ramos, Ph. D. Director of Education and Technical Support Office: 925. 603. 8098 Mobile: 925. 768. 2792 Email: jramos@biocare. net 1

Something doesn’t look right……. } When things go wrong, where do you start?

IHC Refresher
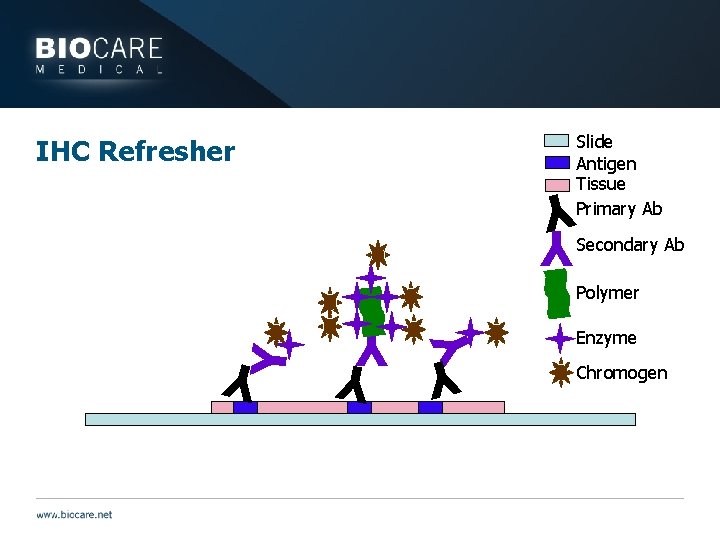
IHC Refresher Slide Antigen Tissue Primary Ab Secondary Ab Y Y Y Y Polymer Enzyme Chromogen Y

The Best Defense is a Good Offense } More than 95% of the time IHC works } Avoid the need for troubleshooting by following established protocols } New staff training and evaluation } See one, do one, teach one

You can never have too many controls! } Positive Control } Lets you know all reagents are working properly } Example: Normal tissue that expresses antigen of interest } CD 8 – Tonsil } Example: appropriate typical cancer tissue or TMA

You can never have too many controls! } Negative Control(s) } Lets you know reagents are working specifically } Tissue Controls: don’t express the antigen of interest } Example: Brain tissue for Prostate Specific Antigen } Reagent Controls: } Isotype control for antibody } If your Positive and Negative controls stained appropriately } Tissue sample is the problem if controls used are appropriate for the tissue in question

Take a step back } Detach emotionally from the issue } Use an analytical and practical approach to finding your problem } Get a complete and accurate description of the problem } Just the Facts } Haste Makes Waste! } When in doubt: CHECK THE DATA SHEET!

Investigative Reporting: The Five W’s } When? } What? } Where? } Why? } How? } Who?

Evaluation of problem } Is the problem on the controls, the patient samples or both? } Does something need to be replaced, remade or outsourced? } Once you think the problem has been identified, can it be reproduced or confirmed? } Can the root cause be mitigated to avoid repeat incidences?

Check your protocol } Did anything in the protocol deviate from the last time it was successfully performed? } New batch of reagent? } Can you repeat with old reagent? } New technician? } Even if you cannot determine a change in a parameter, there may have been an unintentional change } Mislabeled reagent } Misused reagent } Improperly prepared reagent } Contamination

Begin at the Beginning

The Biggest Problem Areas } More than 50% or problems can be traced to: } Improper fixation/processing } Contaminated or expired fixative } Too long or too short fixation/processing } Improper temperatures } Improper pretreatment } Wrong pretreatment method or solution } Poorly executed pretreatment } Time or temperature incorrect } Start with these } Work your way systematically through your protocol

IHC Protocol 1. Tissue Fixation and Processing
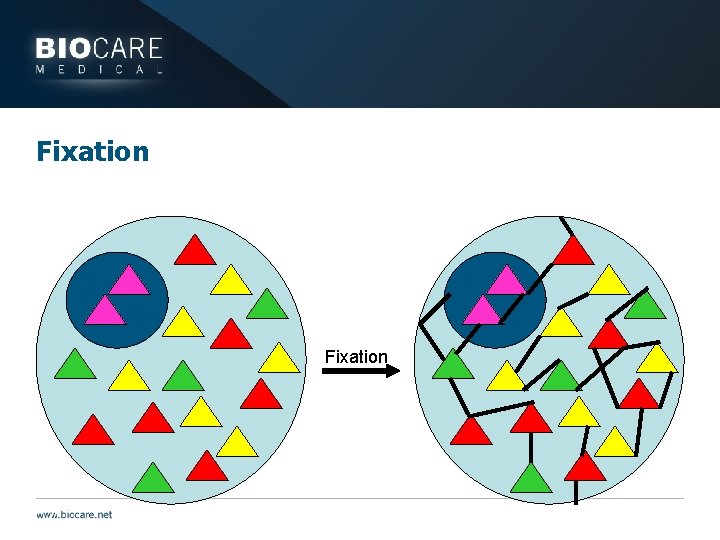
Fixation
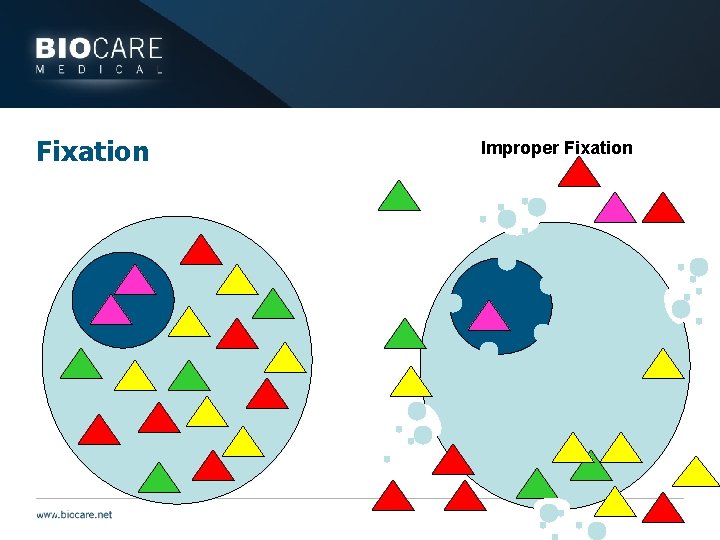
Fixation Improper Fixation

Fixation Problems: H&E Good vs. Bad

Problems with the Antigen } Fixation ensures that proteins remain unchanged in tissue } Holds antigen in place } Prevents degradation of antigen } Once an antigen is lost it cannot be retrieved } Rare problem
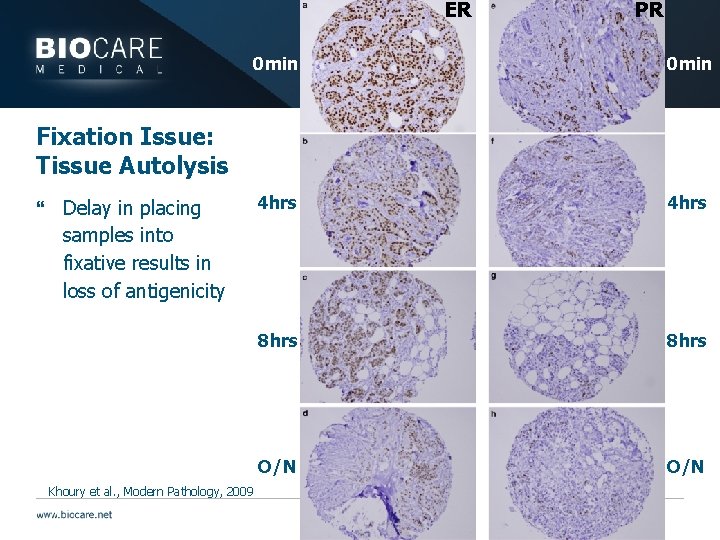
ER PR 0 min 4 hrs 8 hrs O/N Fixation Issue: Tissue Autolysis } Delay in placing samples into fixative results in loss of antigenicity Khoury et al. , Modern Pathology, 2009
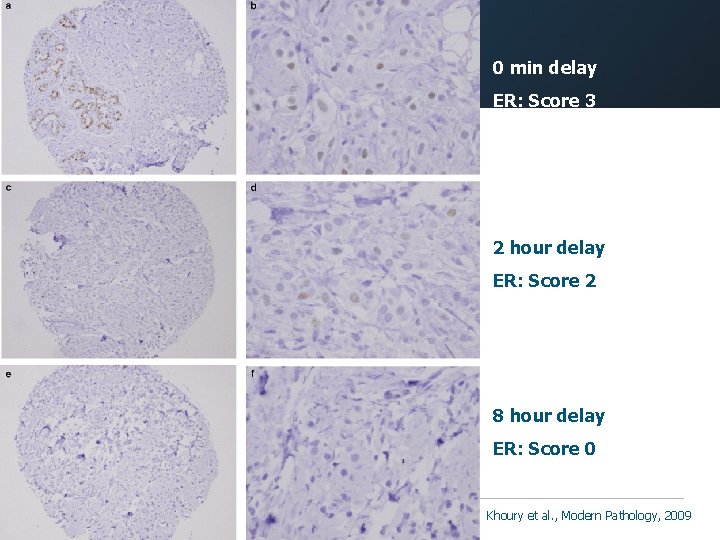
0 min delay ER: Score 3 2 hour delay ER: Score 2 8 hour delay ER: Score 0 Khoury et al. , Modern Pathology, 2009
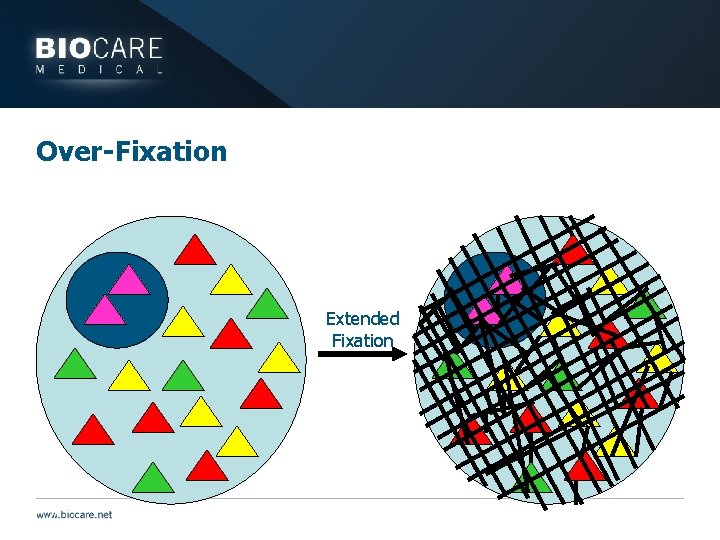
Over-Fixation Extended Fixation

Problems with the Antigen } Over-fixation may result in weak staining or false negatives } Inaccessibility of the antigen may be remedied } Antigen retrieval } HIER or Enzymes
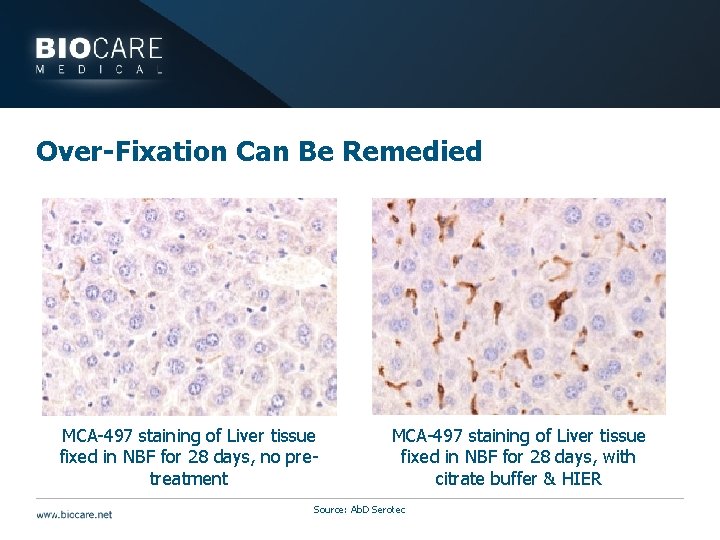
Over-Fixation Can Be Remedied MCA-497 staining of Liver tissue fixed in NBF for 28 days, no pretreatment MCA-497 staining of Liver tissue fixed in NBF for 28 days, with citrate buffer & HIER Source: Ab. D Serotec

To Ensure Antigen Preservation } Set protocols for fixation and processing } Avoid variation in parameters } FFPE optimal at 6 to 48 hours total in formalin } Some tissues require special fixation for optimal results } Bone: formic acid or Ion Exchange Decalcification (Biocare) } Closely Monitor temperature } Paraffin bath, minimize tissue time on hot plates } Avoid drying slides at high temperatures (no higher than 60°C) } Recommendation: 37°C for 30 min, 60°C for 30 min

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting

Slide Age } Most relevant for control tissues and slides } Antigenicity decreases over time even with fixation } Controls with false negatives can cause diagnostic inaccuracies
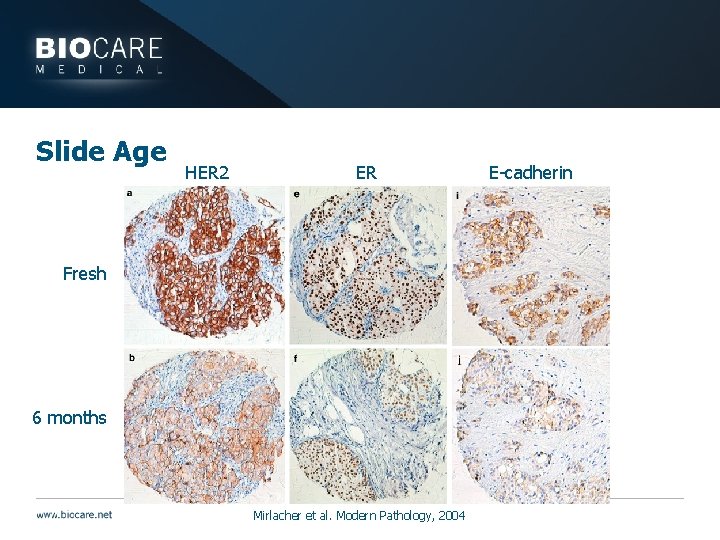
Slide Age HER 2 ER Fresh 6 months Mirlacher et al. Modern Pathology, 2004 E-cadherin
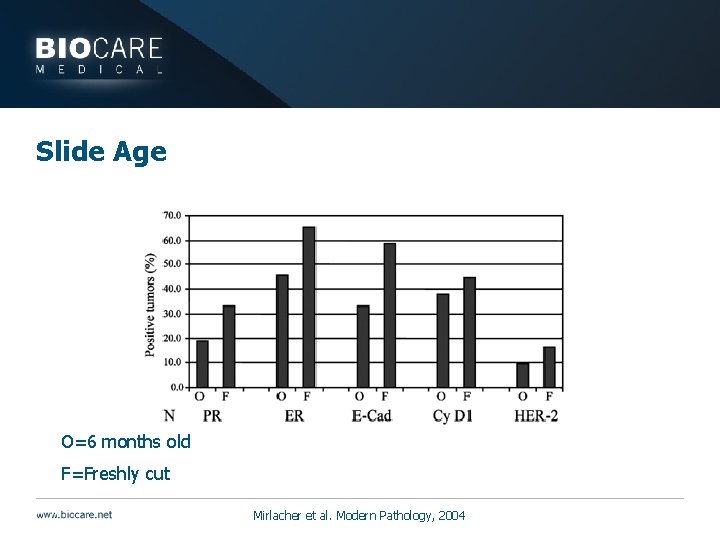
Slide Age O=6 months old F=Freshly cut Mirlacher et al. Modern Pathology, 2004

Slide Age } Antigenicity may be compromised by: } Time } Problem for controls that need to be stored } Control slides should be prepared and used quickly } Treatment with 10% sucrose may help restore antigenicity (1 hr to overnight at room temp to 37°C) } Keep careful records for control slides including date cut and staining success over time } Check first and last slide cut from block to ensure antigen is present throughout } If pre-cut controls look weak, go back to control block } Oven drying prior to storage may contribute to antigen loss

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment

Pretreatment } Closely monitor equipment } Are timers working accurately? } Thermometers in ovens, calibrated equipment } Use fresh reagents } Use the recommended pretreatment method } Use appropriate amounts of reagents } Decloaking Chamber™ Pro and Decloaking Chamber™ Plus are calibrated for specific volumes of water and retrieval solution } Use no less that one Tissue-Tek and one Coplin jar even if you must fill one with water } Volume of liquid in chamber affects pressure and temperature

Pretreatment } Enzymatic Digestion } May help eliminate background staining by breaking down sticky proteins } Example: Mucous } May be used as antigen retrieval } Weak staining may result if enzyme digestion is not performed or inadequate } Weak staining may occur if tissue is digested for too long leading to antigen degradation } Background staining may result if enzyme digestion is excessive } Tissue may fall off slides if digestion is excessive
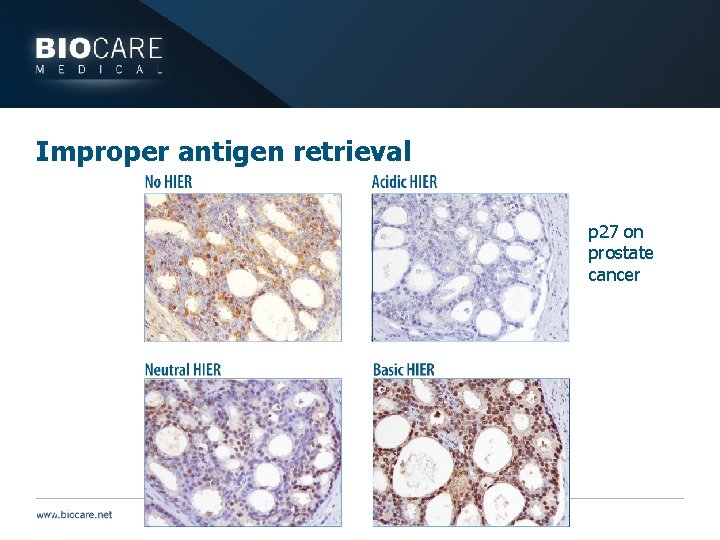
Improper antigen retrieval p 27 on prostate cancer
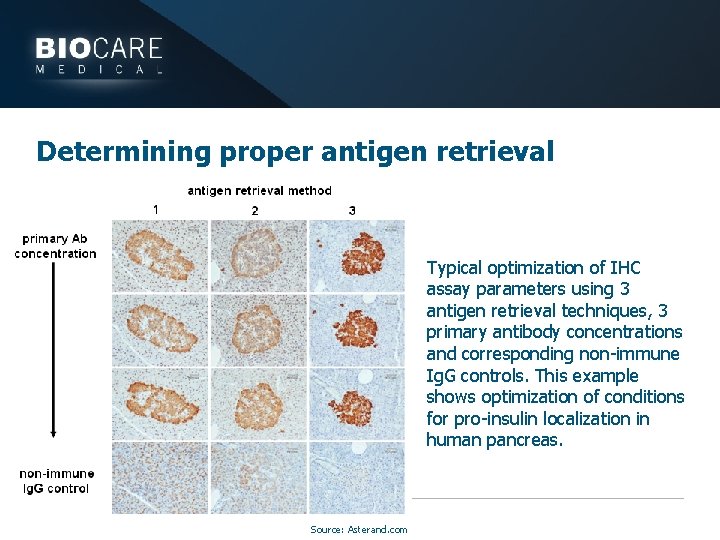
Determining proper antigen retrieval Typical optimization of IHC assay parameters using 3 antigen retrieval techniques, 3 primary antibody concentrations and corresponding non-immune Ig. G controls. This example shows optimization of conditions for pro-insulin localization in human pancreas. Source: Asterand. com

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment 4. Blocking

Blocking } High background staining may be caused by: } Endogenous molecules } Peroxidase } Peroxidazed 1/Peroxide solutions do break down over time } Replace in a timely fashion } Phosphatase } Endogenous biotin if using avidin-biotin systems } Biotin is a vitamin found in most cells at some level } Use avidin-biotin block } Use polymer based detection systems
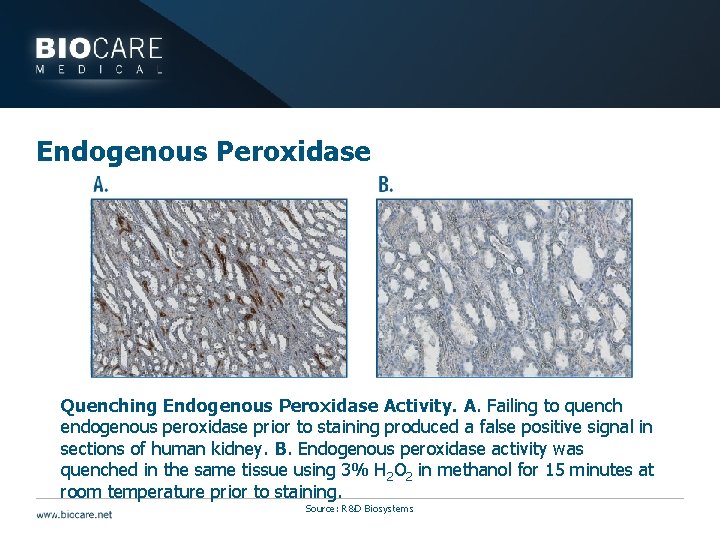
Endogenous Peroxidase Quenching Endogenous Peroxidase Activity. A. Failing to quench endogenous peroxidase prior to staining produced a false positive signal in sections of human kidney. B. Endogenous peroxidase activity was quenched in the same tissue using 3% H 2 O 2 in methanol for 15 minutes at room temperature prior to staining. Source: R&D Biosystems
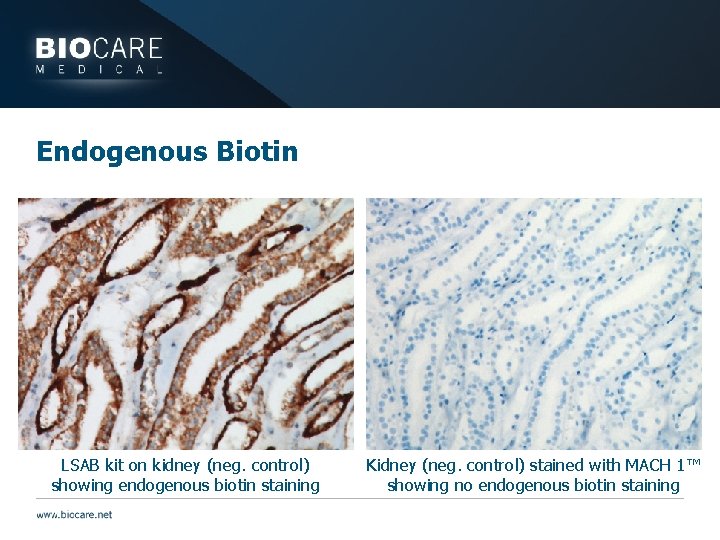
Endogenous Biotin LSAB kit on kidney (neg. control) showing endogenous biotin staining Kidney (neg. control) stained with MACH 1™ showing no endogenous biotin staining
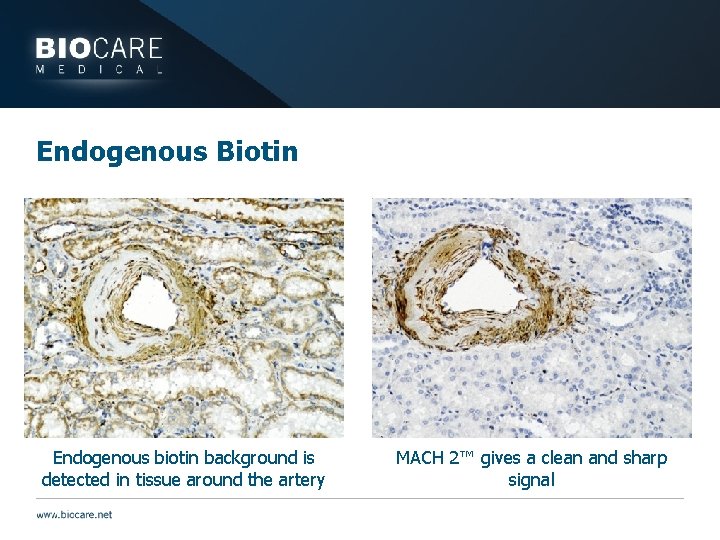
Endogenous Biotin Endogenous biotin background is detected in tissue around the artery MACH 2™ gives a clean and sharp signal

Blocking } High background staining may be caused by: } Cells with Fc. R bind antibodies } Use an Fc. R block agent } Adequate competitive blocking } Use F(ab) or F(ab)’ 2 antibodies instead of intact antibodies } Inadequate blocking } Casein based blocking solutions are excellent blockers (Background Sniper and Background Punisher) } Serum based blocking should sequester Fc binding regions and other sticky endogenous proteins (Background Eraser and Background Terminator): should be from secondary antibody host
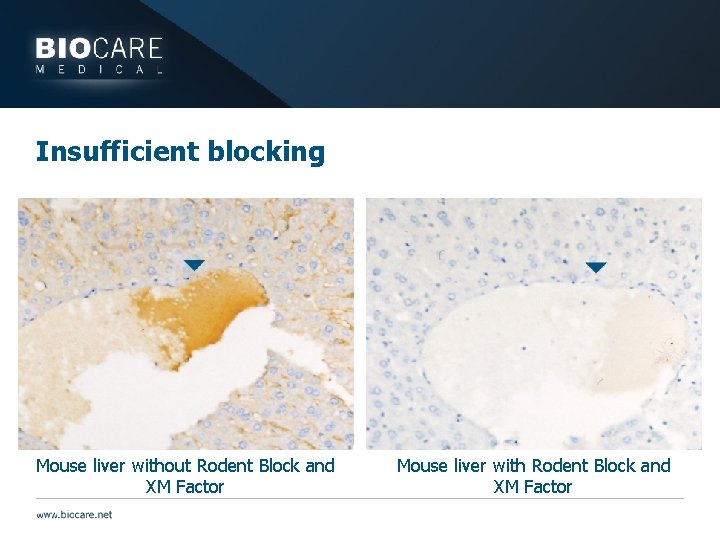
Insufficient blocking Mouse liver without Rodent Block and XM Factor Mouse liver with Rodent Block and XM Factor

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment 4. Blocking 5. Antibody Incubation

Antibody Problems } Interactions between antibodies and antigens are robust } Subject to interference by ions and detergents } Proper diluent choice is critical } Proper concentration of ions ensures optimal interactions } Check manufacturers instructions (Biocare Data Sheet diluent recommendation) } Test several different diluents

Antibody Problems } Antibody concentration must be optimized to account for } Different affinities of antibodies } Unknowable: empirically determined } Different expression levels of antigen } Vary between tissues, cell types, cancer types } Performing a titration of new antibody is key to finding optimal dilution that works for a specific tissue } Modify incubation time and/or temperature (offline) } Shorter if nonspecific staining } Longer if weak staining

Antibody Problems } Antibody Selection } Choose antibodies that have been verified for specific application } Example: FFPE vs. Frozen } Drug treatments may affect antigen availability to antibody } Tamoxifen and Herceptin treatment can mask ER and HER 2 expression } Immuno-based therapies: antibodies in treatment may block epitopes

Antibody type issues } Monoclonals } Recognize only one epitope } Immunizing antigen must be carefully designed to minimize cross reactivity } Prone to false negatives } A negative result cannot be interpreted to definitively mean the antigen is not present, only the epitope was not accessible
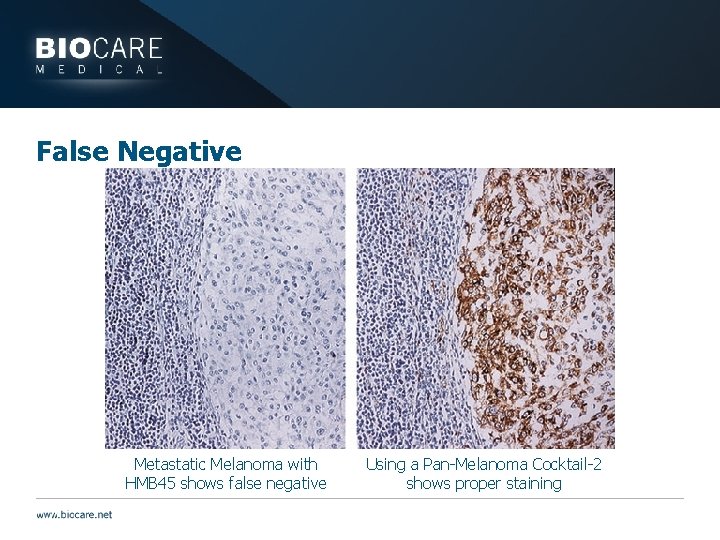
False Negative Metastatic Melanoma with HMB 45 shows false negative Using a Pan-Melanoma Cocktail-2 shows proper staining

Antibody type issues } Polyclonals } Bind more than one epitope } Increased sensitivity } Increased potential for cross-reaction and nonspecific binding } Significant lot-to-lot variability } Each lot is the serum from a new bleed or of a new animal } Each animal’s immune response to an antigen varies significantly

Antibody type issues } Affinity Purification: } Antibodies are incubated with immobilized desired epitope } Antibodies that do not bind are washed away } High affinity antibodies are eluted and used
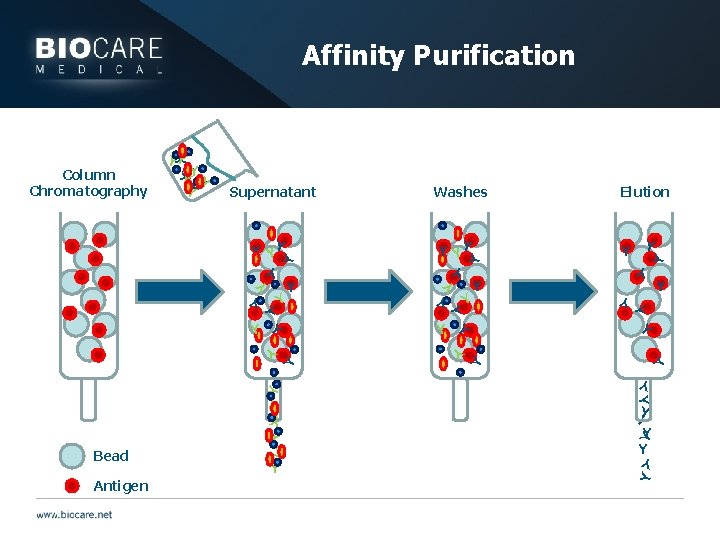
Affinity Purification Y Y Y Y Y Y Y Y Y Y Y YY Y Antigen Elution Y YY Y Bead Washes Y Y Y Supernatant Y Column Chromatography YY Y

Antibody type issues } Pre-adsorption: Pertains mostly to secondary antibodies } Can eliminate non-specific staining by pre-adsorbing antibodies against the tissues you wish to use } Removes any antibodies that bind non-specifically

Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Binds unrelated epitope Bind Charged Molecules Bind Mouse Igs Serum: Secondary anti-Mouse Y Y Y Y Y Pre-Adsorption
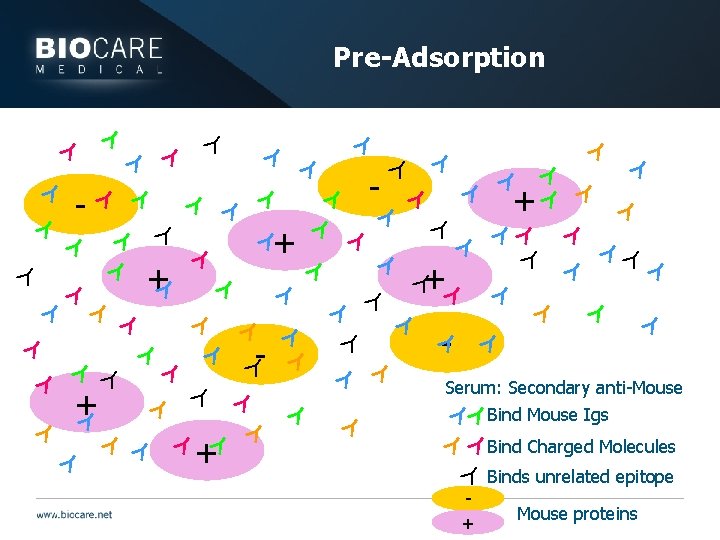
Pre-Adsorption Y Y Y Y Y YY Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y Y + + - + Y - - + - Serum: Secondary anti-Mouse + Bind Mouse Igs + Bind Charged Molecules Binds unrelated epitope + Mouse proteins
Pre-Adsorption Y Y Y Y YY Y Y Y Y Y Y Y Y + Y Y Y Y Y Y Y Y Y Y + + Y Y Y - Y Y Y + Y - - + - Serum: Secondary anti-Mouse Bind Mouse Igs Bind Charged Molecules Binds unrelated epitope + Mouse proteins Y Y Y

Pre-Adsorption Y Y Y Y Y Y Serum: Secondary anti-Mouse Bind Mouse Igs Y YY Bind Charged Molecules Binds unrelated epitope Y Y Y Y Y Y Y Y Y Y Y
Y YY Binds unrelated epitope Serum: Secondary anti-Mouse Bind Mouse Igs Y Y Y Y Y YYY YYYY YYY Y Y Y Y Y Y Y Y Y Y Pre-Adsorption
Y YY Binds unrelated epitope Serum: Secondary anti-Mouse Bind Mouse Igs Y Y YYY Y Y Y YY Y Y Y YYYYY YY YYYYYYY Y Y Y Y Y Pre-Adsorption

Pre-Adsorption Y Y YYYY Serum: Secondary anti-Mouse Bind Mouse Igs Y YY Binds unrelated epitope Y Y YYYYY YY YYYYYYY YYYYY

Antibody Format } Prediluted Antibodies } Designed and optimized for a given manufacturer’s detection system } Concentrated Antibodies } Choose these and do your own titrations especially if you are using reagents from multiple manufacturers

Titration } Titration is the process of adding sequentially smaller amounts of antibody to diluent } Titration series: several different dilutions of the same antibody tested in the same way to achieve optimal staining
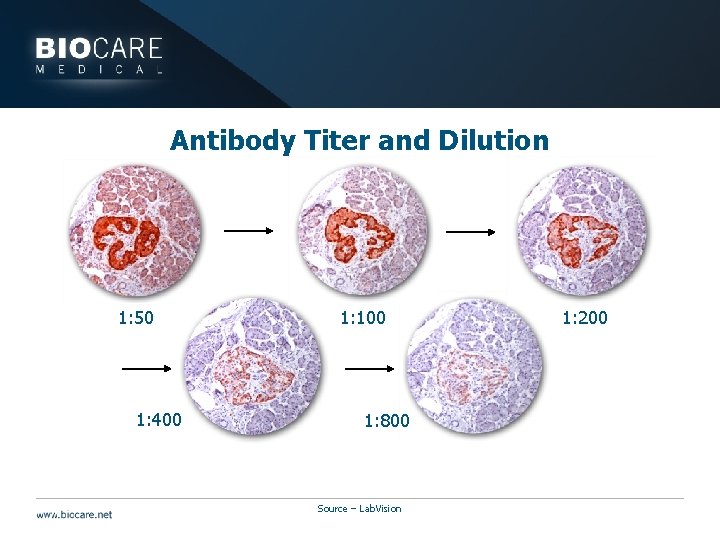
Antibody Titer and Dilution 1: 50 1: 400 1: 100 1: 800 Source – Lab. Vision 1: 200

Calculating Titrations } 1 mg/m. L monoclonal antibody stock solution } Recommended working solution protein concentration: 1 microgram/m. L } 1000 micrograms in 1 mg } Dilution factor: 1 in 1000 } 1 microliter concentrate: 999 microliters diluent } 5 microliters concentrate: 4995 microliters diluent

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment 4. Blocking 5. Antibody Incubation 6. Detection

Detection Problems } Problem would be evident with multiple antibodies that use the same detection } Weak staining } Component may have gone bad or become contaminated } Be careful of stability issues (Check Data Sheet) } Insufficient incubation times } Insufficient amount of reagents } High Background } Excessive incubation time } Detection system too sensitive } May dilute } No staining } Components applied in wrong order
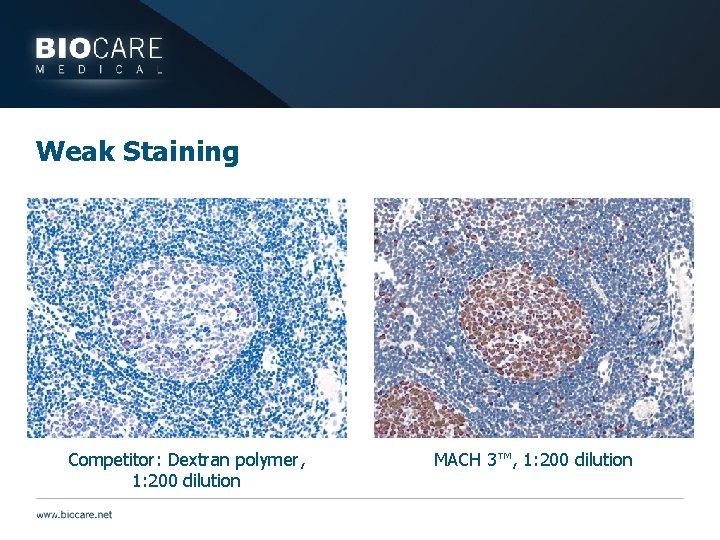
Weak Staining Competitor: Dextran polymer, 1: 200 dilution MACH 3™, 1: 200 dilution

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment 4. Blocking 5. Antibody Incubation 6. Detection 7. Wash Steps

Wash Problems } High background may be the result of insufficient washing } } Weak staining may be the result of excessive washing or excessive strength } } } Omit detergent from wash buffers Use low salt wash buffers Contamination of wash buffers may cause weak or spurious staining } } Increase number of washes Adequate wash incubation time Increase strength of washes (more detergent or salt) Clean tissue baths daily and disinfect regularly Improper preparation of wash buffers may cause weak or spurious staining } } Use good quality water and reagents Check p. H

IHC Protocol 1. Tissue Fixation and Processing 2. Slide Cutting 3. Pretreatment 4. Blocking 5. Antibody Incubation 6. Detection 7. Wash Steps 8. Chromogen

Chromogen Problems } Alkaline Phosphatase rapidly oxidizes in buffer } Chromogenic enzymes are p. H sensitive } Use indicated amounts of component solutions } Chromogen products maybe sensitive to alcohols and xylenes } AP systems not as robust as HRP } Do not leave slides in alcohols or xylenes for more than a couple minutes, better yet do 20 dips } Air dry slides (no alcohols) place directly into xylene (AP particularly) } Chromogens may form crystals when reacting with mounting media
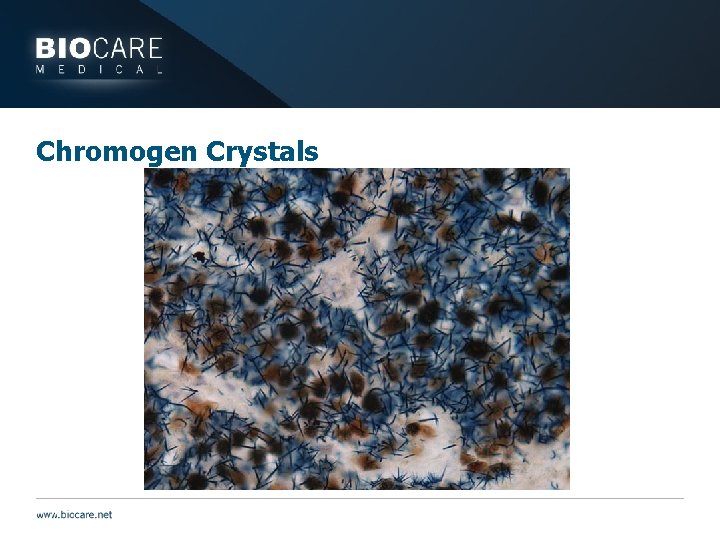
Chromogen Crystals

If using a kit } Follow manufacturer’s directions } Apply reagents in proper order } Prepare your “mise en place” } Everything is prepared and arranged in order of application } Ensures you have adequate amounts of all reagents } Prevents starting a protocol only to discover you’re out of a vital reagent

Other Problems } Reagents } Equipment } Tissues } Slides } Poor quality sectioning } Impatience } Improper handling

Tissue Issues } Pigmentation } Can mask chromogen } Example: Melanin in skin } Use a red chromogen } Fast Red or Warp Red using AP system } Romulin AEC or Bajoran Purple if using HRP system } Use Azure blue counter stain

Tissue Issues } Slides are critical } Positively charged } Overcharged slides may be hydrophobic, blocking solutions may help } Environment alters slides } Humidity affects charge } Keep slide boxes closed, use up box before opening new } High quality } Inexpensive slides may have an uneven surface and trap reagents under tissue } Dry slides appropriately for tissue adherence } Water trapped behind tissue will hinder binding } Blistering on slide } Sequential adherence of tissues not recommended } If staining control and patient on same slide, cut and put onto slide same day } Turn slide upside down so as not to wet end where second section will go

Blistering and Rolling
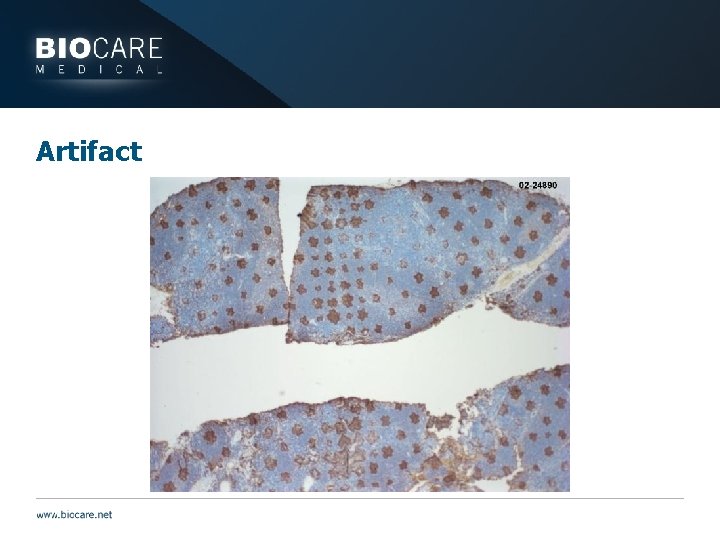
Artifact

Tissue Issue: Formalin fixed, then frozen
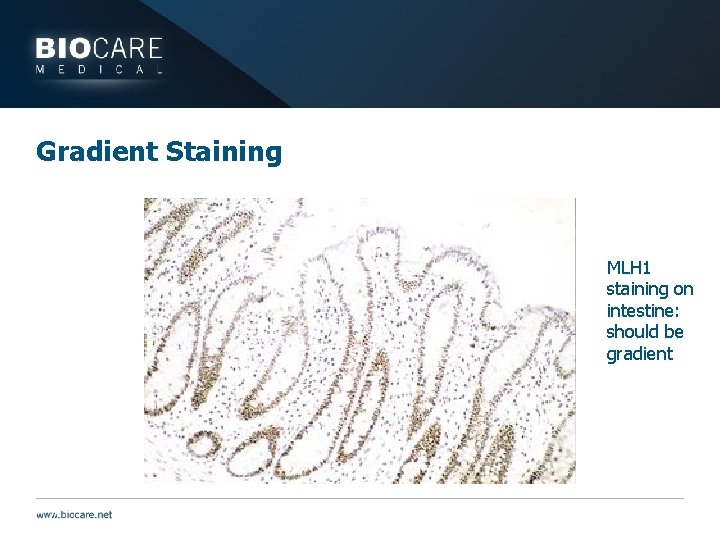
Gradient Staining MLH 1 staining on intestine: should be gradient
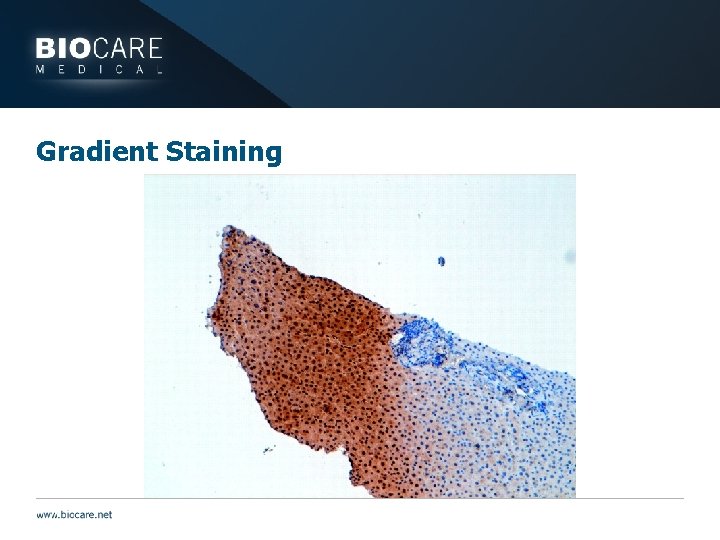
Gradient Staining
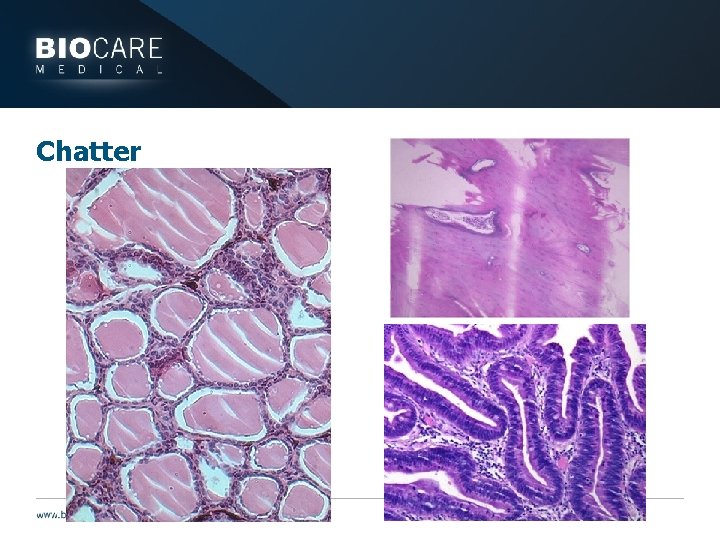
Chatter

More Sections…. More Problems Incomplete decalcification Sectioning Artifact (mouse spleen)
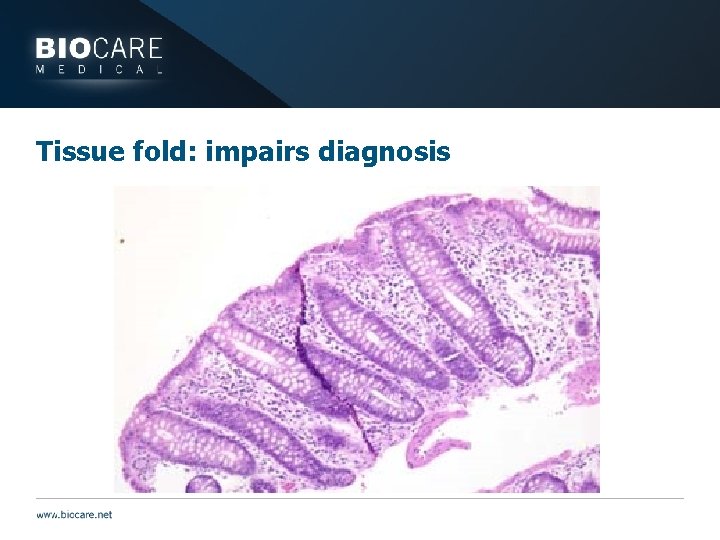
Tissue fold: impairs diagnosis
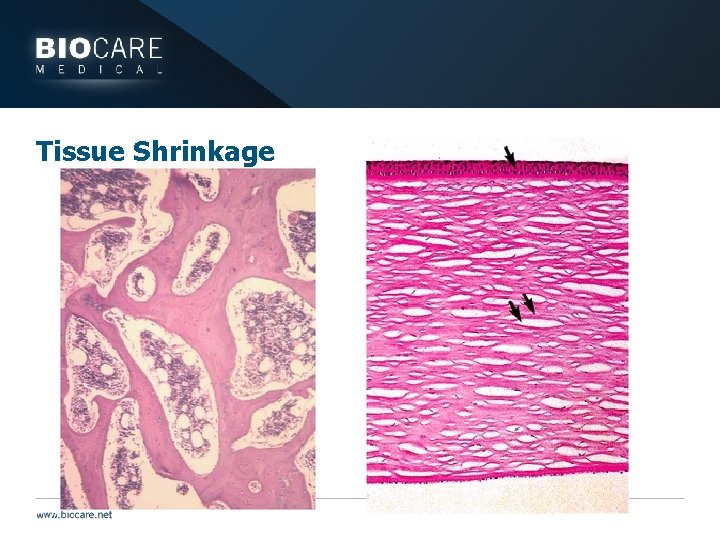
Tissue Shrinkage
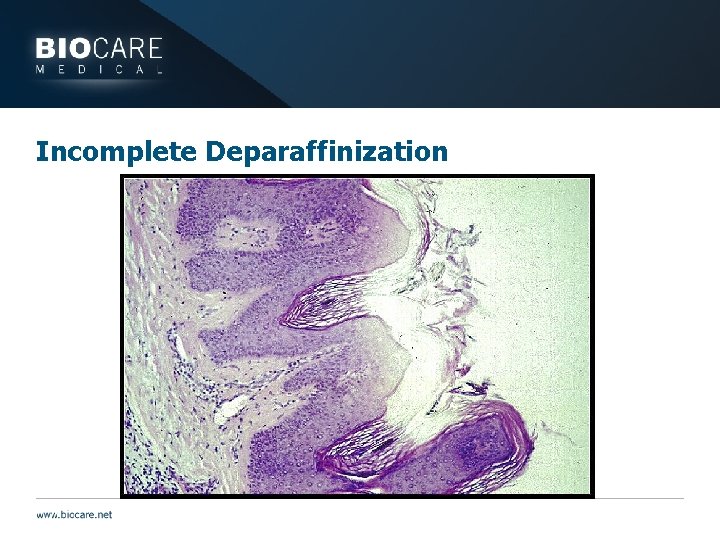
Incomplete Deparaffinization
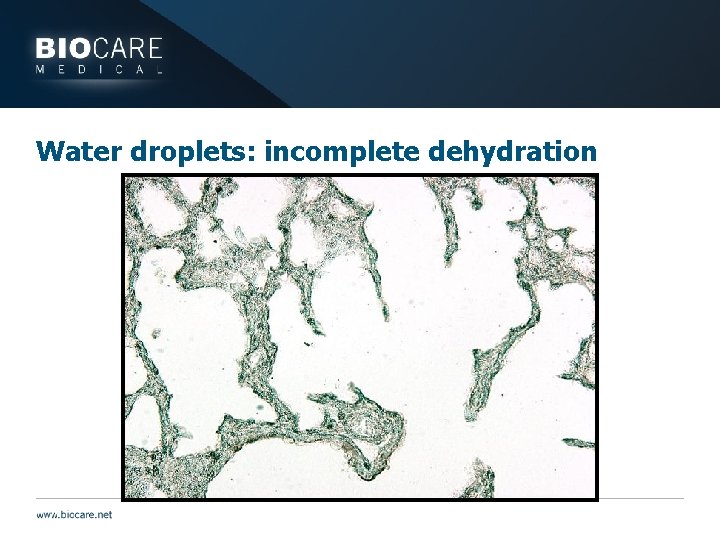
Water droplets: incomplete dehydration
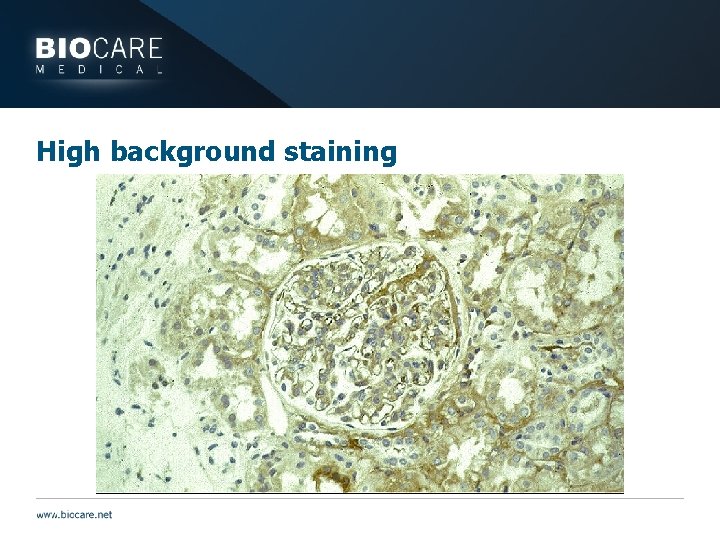
High background staining
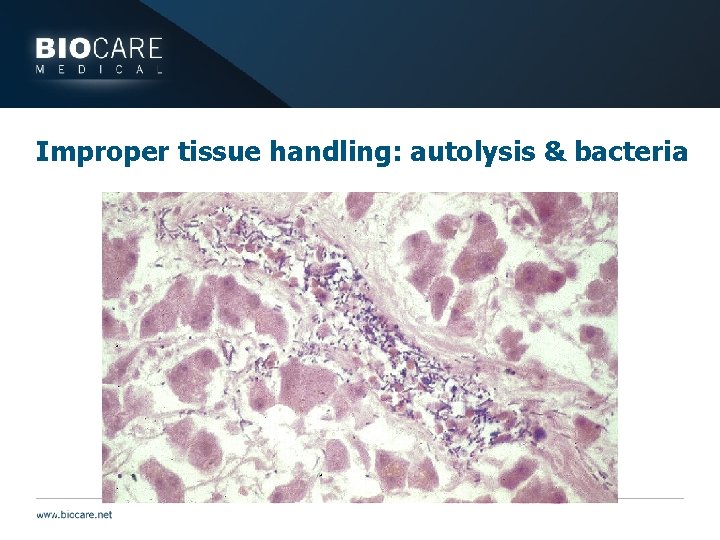
Improper tissue handling: autolysis & bacteria

General considerations } Never allow slides to dry during staining } } Use surfactant in all wash steps to ensure proper spreading of reagent Maintain moisture in staining environment during extended incubations Repeat applications for extended incubations Precision is absolutely essential } Always measure reagents as accurately as possible } Graduated cylinders and pipettes } Maintain Equipment } } Verify Equipment function } Steam strips } Test runs Consistency in reagents } } Clean regularly Replace gaskets or parts Verify p. H of solutions Don’t change multiple parameters simultaneously when troubleshooting

Useful tools } TMAs } Many pieces of tissue in a single block } Can be different human tissues or different animal tissues (vet) } Can evaluate and validate antibodies rapidly } Purchase or make yourself } Fixative controls } Tissues in different fixative in a TMA } Tissues fixed different times in a TMA
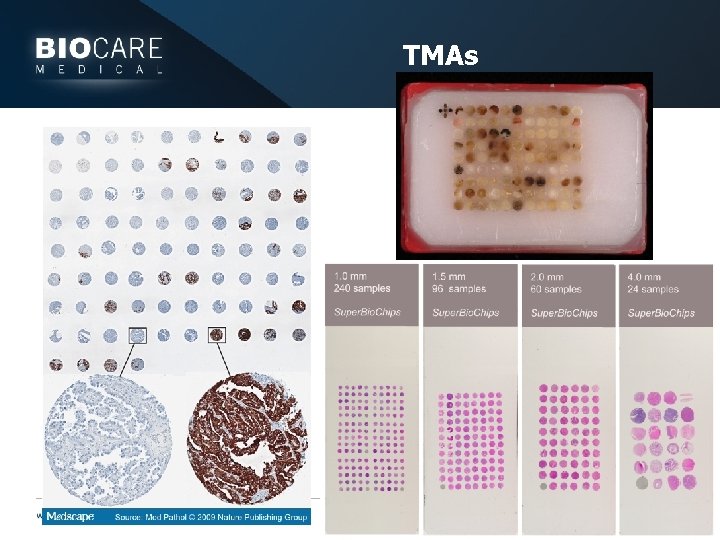
TMAs

Once you’ve solved your problem } Keep track of new protocol } Good quality control will monitor effectiveness of solution } Turn your experience into a learning opportunity } Train others not just on the fix but on the cause and how to recognize the problem


Thank you! Jason A. Ramos, Ph. D. Director of Education and Technical Support Biocare Medical, LLC 925. 768. 2792 jramos@biocare. net
- Slides: 93