IEEE EMB 11073 WORKING GROUPS STATUS REPORT TOM
IEEE EMB 11073 WORKING GROUPS STATUS REPORT TOM THOMPSON PROGRAM MANAGER 17 January 2020

POINT-OF-CARE DEVICES (POCD) WORKING GROUP (WG) 2
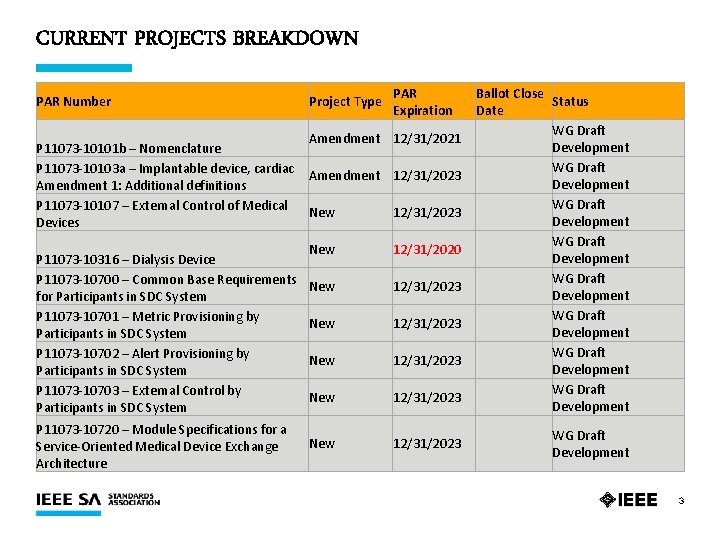
CURRENT PROJECTS BREAKDOWN PAR Number Project Type PAR Expiration Amendment 12/31/2021 P 11073 -10101 b – Nomenclature P 11073 -10103 a – Implantable device, cardiac Amendment 12/31/2023 Amendment 1: Additional definitions P 11073 -10107 – External Control of Medical New 12/31/2023 Devices P 11073 -10316 – Dialysis Device P 11073 -10700 – Common Base Requirements for Participants in SDC System P 11073 -10701 – Metric Provisioning by Participants in SDC System P 11073 -10702 – Alert Provisioning by Participants in SDC System P 11073 -10703 – External Control by Participants in SDC System P 11073 -10720 – Module Specifications for a Service-Oriented Medical Device Exchange Architecture New 12/31/2020 New 12/31/2023 New 12/31/2023 Ballot Close Status Date WG Draft Development WG Draft Development WG Draft Development 3
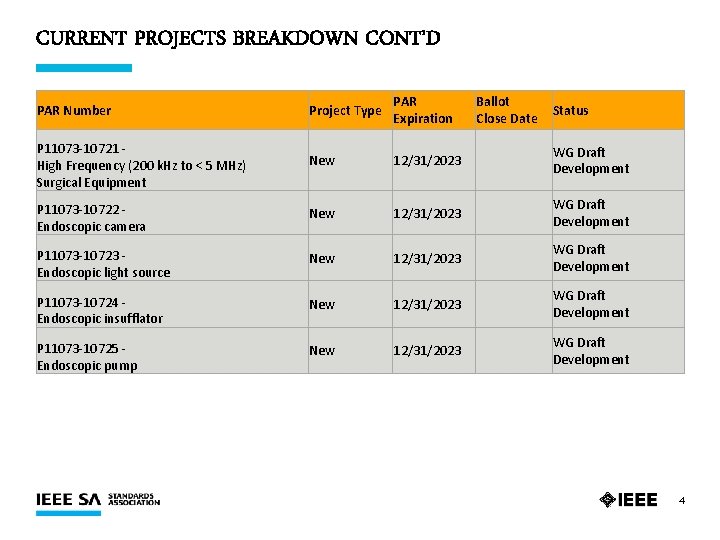
CURRENT PROJECTS BREAKDOWN CONT’D PAR Number Project Type PAR Expiration Ballot Close Date Status P 11073 -10721 - High Frequency (200 k. Hz to < 5 MHz) Surgical Equipment New 12/31/2023 WG Draft Development P 11073 -10722 - Endoscopic camera New 12/31/2023 WG Draft Development P 11073 -10723 - Endoscopic light source New 12/31/2023 WG Draft Development P 11073 -10724 - Endoscopic insufflator New 12/31/2023 WG Draft Development P 11073 -10725 - Endoscopic pump New 12/31/2023 WG Draft Development 4

CURRENT PROJECTS SUMMARY Ø 14 Active PARs Ø 14 Drafts in Development Ø 1 PAR Expiring 31 December 2020 §P 11073 -10316 – Dialysis Device 5
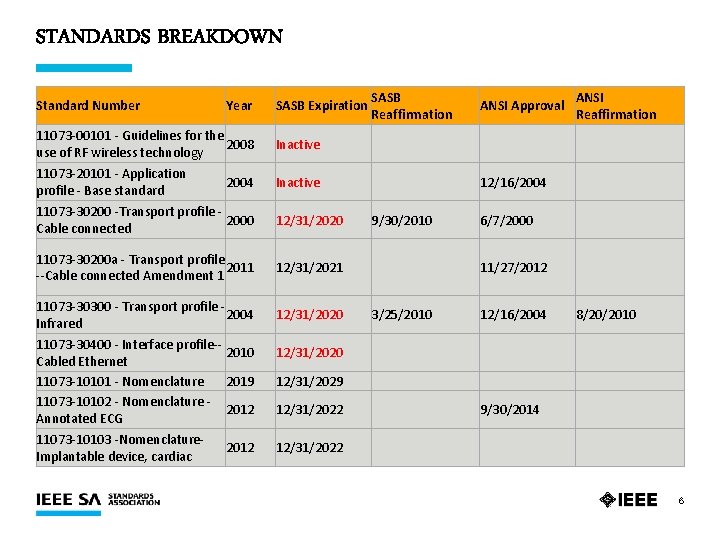
STANDARDS BREAKDOWN SASB Expiration SASB Reaffirmation ANSI Approval ANSI Reaffirmation 11073 -00101 - Guidelines for the 2008 use of RF wireless technology Inactive 11073 -20101 - Application profile - Base standard Inactive 12/16/2004 11073 -30200 -Transport profile - 2000 Cable connected 12/31/2020 9/30/2010 6/7/2000 11073 -30200 a - Transport profile 2011 --Cable connected Amendment 1 12/31/2021 11/27/2012 11073 -30300 - Transport profile - 2004 Infrared 12/31/2020 3/25/2010 12/16/2004 8/20/2010 12/31/2020 12/31/2029 12/31/2022 9/30/2014 12/31/2022 Standard Number Year 2004 11073 -30400 - Interface profile-2010 Cabled Ethernet 11073 -10101 - Nomenclature 2019 11073 -10102 - Nomenclature 2012 Annotated ECG 11073 -10103 -Nomenclature. Implantable device, cardiac 2012 6
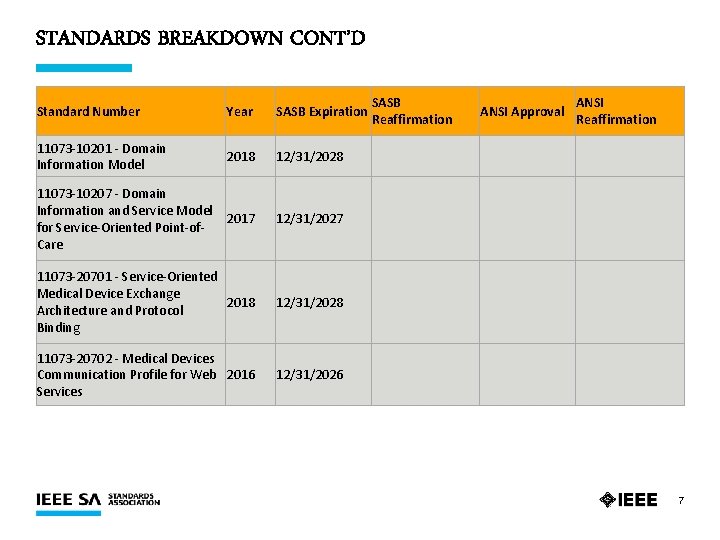
STANDARDS BREAKDOWN CONT’D Standard Number Year SASB Expiration SASB Reaffirmation ANSI Approval ANSI Reaffirmation 11073 -10201 - Domain Information Model 2018 12/31/2028 11073 -10207 - Domain Information and Service Model 2017 for Service-Oriented Point-of. Care 12/31/2027 11073 -20701 - Service-Oriented Medical Device Exchange 2018 Architecture and Protocol Binding 12/31/2028 11073 -20702 - Medical Devices Communication Profile for Web 2016 Services 12/31/2026 7

STANDARDS SUMMARY Ø 11 Active Standards Ø 2 Inactive Standards as of 17 January 2020 §P 11073 -00101 – Guidelines for the use of RF wireless technology §P 11073 -20101 – Application profile - Base standard 8

PERSONAL HEALTH DEVICES (PHD) WORKING GROUP (WG) 9
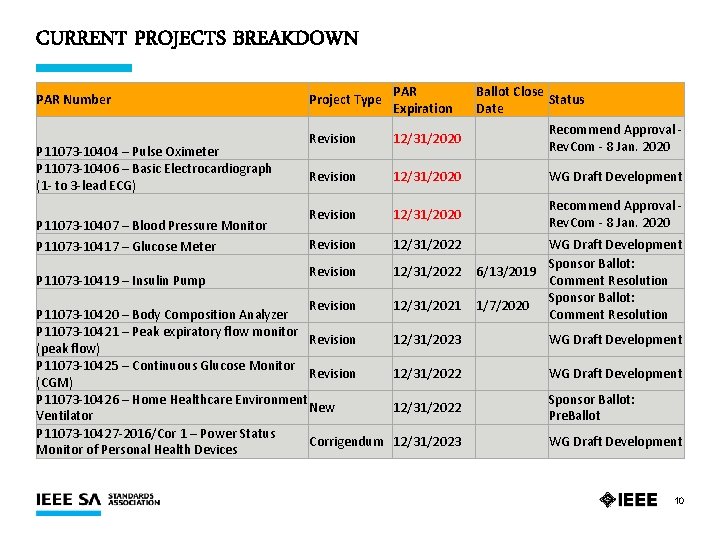
CURRENT PROJECTS BREAKDOWN PAR Number Project Type PAR Expiration Ballot Close Status Date Revision 12/31/2020 Recommend Approval - Rev. Com - 8 Jan. 2020 12/31/2020 WG Draft Development Revision 12/31/2020 Recommend Approval - Rev. Com - 8 Jan. 2020 Revision 12/31/2022 P 11073 -10404 – Pulse Oximeter P 11073 -10406 – Basic Electrocardiograph Revision (1 - to 3 -lead ECG) P 11073 -10407 – Blood Pressure Monitor P 11073 -10417 – Glucose Meter P 11073 -10419 – Insulin Pump Revision P 11073 -10420 – Body Composition Analyzer P 11073 -10421 – Peak expiratory flow monitor Revision (peak flow) P 11073 -10425 – Continuous Glucose Monitor Revision (CGM) P 11073 -10426 – Home Healthcare Environment New Ventilator P 11073 -10427 -2016/Cor 1 – Power Status Corrigendum Monitor of Personal Health Devices 12/31/2021 WG Draft Development Sponsor Ballot: 6/13/2019 Comment Resolution Sponsor Ballot: 1/7/2020 Comment Resolution 12/31/2023 WG Draft Development 12/31/2022 Sponsor Ballot: Pre. Ballot 12/31/2023 WG Draft Development 10
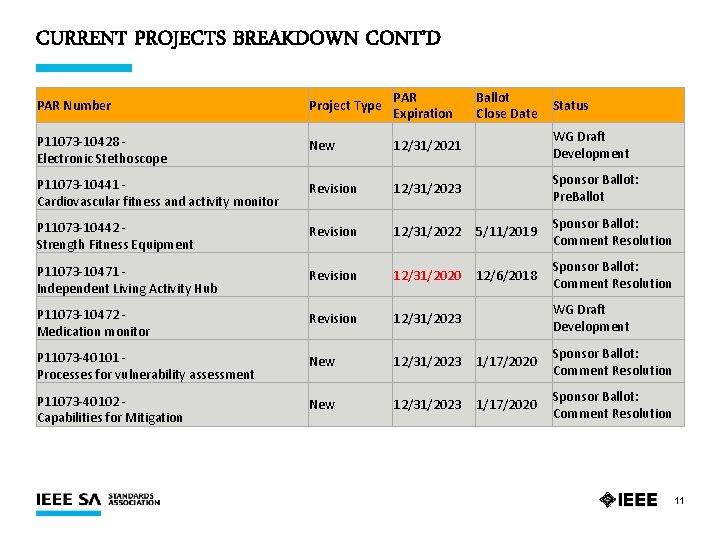
CURRENT PROJECTS BREAKDOWN CONT’D PAR Number Project Type PAR Expiration Ballot Close Date Status P 11073 -10428 - Electronic Stethoscope New 12/31/2021 WG Draft Development P 11073 -10441 - Cardiovascular fitness and activity monitor Revision 12/31/2023 Sponsor Ballot: Pre. Ballot P 11073 -10442 - Strength Fitness Equipment Revision 12/31/2022 5/11/2019 Sponsor Ballot: Comment Resolution P 11073 -10471 - Independent Living Activity Hub Revision 12/31/2020 12/6/2018 Sponsor Ballot: Comment Resolution P 11073 -10472 - Medication monitor Revision 12/31/2023 WG Draft Development P 11073 -40101 - Processes for vulnerability assessment New 12/31/2023 1/17/2020 Sponsor Ballot: Comment Resolution P 11073 -40102 - Capabilities for Mitigation New 12/31/2023 1/17/2020 Sponsor Ballot: Comment Resolution 11

CURRENT PROJECTS SUMMARY Ø 17 Active PARs Ø 7 Drafts in Development Ø 10 Drafts in Ballot or Recirculation Ø 4 PARs Expiring 31 December 2020 §P 11073 -10404 – Pulse Oximeter §P 11073 -10406 – Basic Electrocardiograph (1 -to-3 -lead ECG) §P 11073 -10407 – Blood Pressure Monitor §P 11073 -10471 – Independent Living Activity Hub 12

SA BALLOT HIGHLIGHTS Ø 10404 – Revision – Pulse Oximeter üRecommend Approval - Rev. Com - 8 January 2020 Ø 10407 – Revision – Blood Pressure Monitor üRecommend Approval - Rev. Com - 8 January 2020 Ø 10419 – Revision – Insulin Pump § Comment Resolution from Recirculation 6 Ø 10420 – Revision – Body Composition Analyzer § Comment Resolution from Initial Ballot Ø 10442 – Revision – Strength Fitness Equipment § Comment Resolution from Recirculation 6 Ø 10471 – Revision – Independent Living Activity Hub § Comment Resolution from Initial Ballot 13

SA BALLOT HIGHLIGHTS CONT’D Ø 40101 – New – Cybersecurity/ Processes for Vulnerability Assessment § Comment Resolution from Initial Ballot Ø 40102 – New – Cybersecurity/ Capabilities for Mitigation § Comment Resolution from Initial Ballot Ø 10426 – New – Home Healthcare Environment Ventilator § Pre Ballot Ø 10441 – Revision – Cardiovascular Fitness and Activity Monitor § Pre Ballot 14
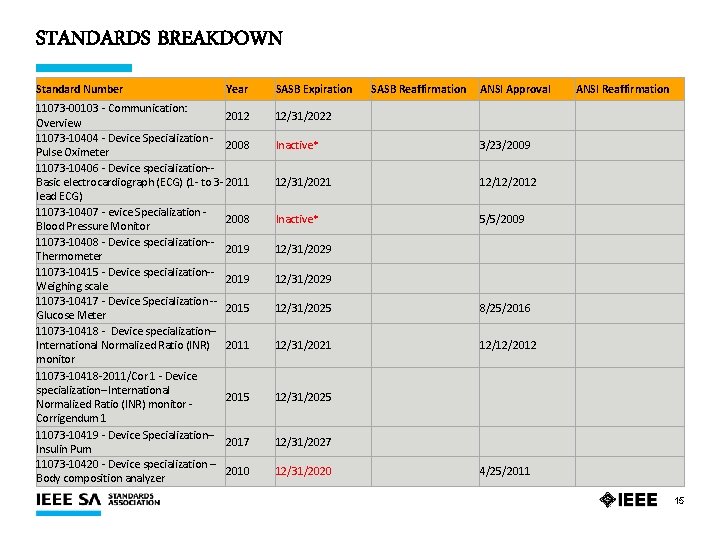
STANDARDS BREAKDOWN Standard Number Year 11073 -00103 - Communication: 2012 Overview 11073 -10404 - Device Specialization - 2008 Pulse Oximeter 11073 -10406 - Device specialization-Basic electrocardiograph (ECG) (1 - to 3 - 2011 lead ECG) 11073 -10407 - evice Specialization - 2008 Blood Pressure Monitor 11073 -10408 - Device specialization-2019 Thermometer 11073 -10415 - Device specialization-2019 Weighing scale 11073 -10417 - Device Specialization -- 2015 Glucose Meter 11073 -10418 - Device specialization-International Normalized Ratio (INR) 2011 monitor 11073 -10418 -2011/Cor 1 - Device specialization--International 2015 Normalized Ratio (INR) monitor - Corrigendum 1 11073 -10419 - Device Specialization-2017 Insulin Pum 11073 -10420 - Device specialization -- 2010 Body composition analyzer SASB Expiration SASB Reaffirmation ANSI Approval ANSI Reaffirmation 12/31/2022 Inactive* 3/23/2009 12/31/2021 12/12/2012 Inactive* 5/5/2009 12/31/2029 12/31/2025 8/25/2016 12/31/2021 12/12/2012 12/31/2025 12/31/2027 12/31/2020 4/25/2011 15
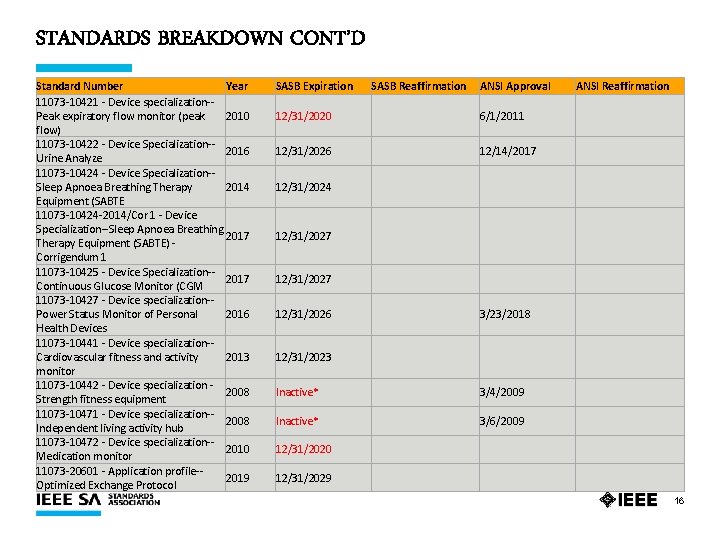
STANDARDS BREAKDOWN CONT’D Standard Number Year 11073 -10421 - Device specialization-Peak expiratory flow monitor (peak 2010 flow) 11073 -10422 - Device Specialization-2016 Urine Analyze 11073 -10424 - Device Specialization-Sleep Apnoea Breathing Therapy 2014 Equipment (SABTE 11073 -10424 -2014/Cor 1 - Device Specialization--Sleep Apnoea Breathing 2017 Therapy Equipment (SABTE) - Corrigendum 1 11073 -10425 - Device Specialization-2017 Continuous Glucose Monitor (CGM 11073 -10427 - Device specialization-Power Status Monitor of Personal 2016 Health Devices 11073 -10441 - Device specialization-Cardiovascular fitness and activity 2013 monitor 11073 -10442 - Device specialization - 2008 Strength fitness equipment 11073 -10471 - Device specialization-2008 Independent living activity hub 11073 -10472 - Device specialization-2010 Medication monitor 11073 -20601 - Application profile-2019 Optimized Exchange Protocol SASB Expiration SASB Reaffirmation ANSI Approval ANSI Reaffirmation 12/31/2020 6/1/2011 12/31/2026 12/14/2017 12/31/2024 12/31/2027 12/31/2026 3/23/2018 12/31/2023 Inactive* 3/4/2009 Inactive* 3/6/2009 12/31/2020 12/31/2029 16

STANDARDS SUMMARY Note: * Denotes an existing PAR associated with this standard and it is in SA Ballot Ø 18 Active Standards Ø 4 Inactive Standards as of 17 January 2020 §P 11073 -10404 – Device Specialization - Pulse Oximeter §P 11073 -10407 – Device Specialization - Blood Pressure Monitor §P 11073 -10442 – Device specialization - Strength fitness equipment §P 11073 -10471 – Device specialization--Independent living activity hub 17

ISO ADOPTION STATUS Balloting Update Ø 11073 -10101 – Nomenclature § FDIS ballot closes 21 May 2020 Ø 11073 -10201 – Domain Information Model § FDIS ballot closes 31 March 2020 Ø 11073 -20701 – Service-Oriented Medical Device Exchange Architecture and Protocol Binding § Awaiting response(s) to comment(s) from FDIS ballot 18

COLLABORATIVE RELATIONSHIPS Memorandums of Understanding (MOUs) § Health Level Seven (HL 7) § Integrating the Healthcare Enterprise (IHE) § Logical Observation Identifiers Names and Codes (LOINC) § National Institute of Standards and Technology (NIST) § Personal Connected Health Alliance (PCHAlliance) Category A Liaisons § ISO/TC 121 § ISO/TC 215 19

2020 STANDARDS ASSOCIATION STANDARDS BOARD (SASB) MEETINGS 20

THANK YOU Tom Thompson Program Manager Operational Program Management (OPM) IEEE Standards Association (IEEE SA) Office: +1 732 562 5564 Mobile: +1 732 540 4053 Email: thomas. thompson@ieee. org Website: http: //standards. ieee. org 21
- Slides: 21