IDENTIFYING MACROMOLECULES IN FOOD LAB Introduction Carbohydrates proteins

IDENTIFYING MACROMOLECULES IN FOOD LAB

Introduction Carbohydrates, proteins, and fats are all essential nutrients. We cannot manufacture these nutrients so we must obtain them from our environment.

Introduction In this lab, with the use of indicators as chemical detection tools, you will analyze stomach contents (vomit) for the presence of nutrients. Detection is based upon observing a chemical change that takes place most often a change in color.
Objective Identify the presence of major nutrients such as simple carbohydrates (glucose), complex carbohydrates (starch), protein and fat(lipids) in common foods.

What is an indicator? • Indicators are chemical compounds used to detect the presence of other compounds.
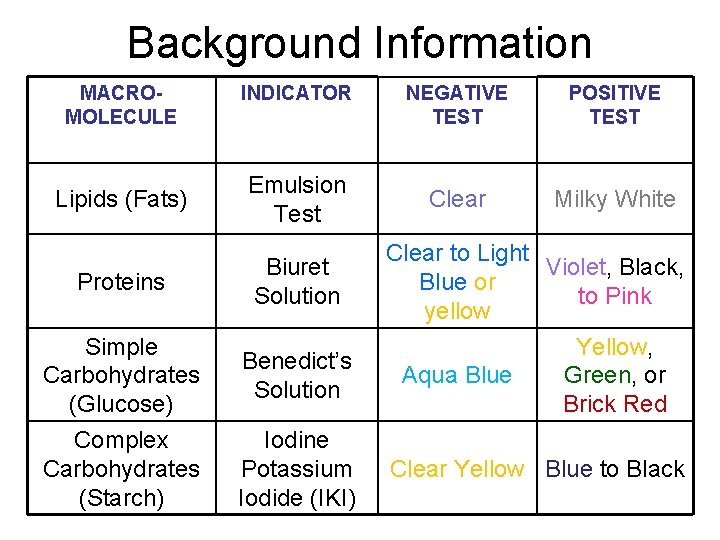
Background Information MACROMOLECULE INDICATOR Lipids (Fats) Emulsion Test Proteins Biuret Solution Simple Carbohydrates (Glucose) Benedict’s Solution Complex Carbohydrates (Starch) Iodine Potassium Iodide (IKI) NEGATIVE TEST POSITIVE TEST Clear Milky White Clear to Light Violet, Black, Blue or to Pink yellow Aqua Blue Yellow, Green, or Brick Red Clear Yellow Blue to Black

What is a Standard? • An acknowledged measure of comparison for quantitative or qualitative value; a criterion.

Test for Simple Carbohydrates Benedict’s solution • Benedict's solution is a chemical indicator for simple sugars such as glucose: C 6 H 12 O 6. • Aqua blue: negative test; yellow/green/brick red, etc. : positive test

Test for Simple Carbohydrates Benedict’s solution • Unlike some other indicators, Benedict’s solution does not work at room temperature - it must be heated first.

Test for Complex Carbohydrates Iodine Potassium Iodide (IKI) solution • IKI solution (Iodine Potassium Iodide) color change = blue to black

Test for Complex Carbohydrates Iodine Solution • Iodine solution is an indicator for a molecule called starch. • Starch is a huge molecule made up of hundreds of simple sugar molecules (such as glucose) connected to each other.

Test for Protein (amino acids) Biuret solution • Biuret solution dark violet blue to pinkish purple

Test for Fats (lipids) Emulsion Test • The emulsion test is a method to determine the presence of lipids by suspending them in ethanol. • The lipids dissolve in the alcohol, but not in water. When mixed together, the lipids will fall out of solution and give a cloudy white appearance.

Procedure Fats (lipids) • Add 5 ml of food sample to test tube. • Add 5 ml of ethanol to the test tube and shake well. • Using a pipette, remove the clear liquid into another test tube with 5 ml of distilled H 2 O. • Record Results:

Procedure Protein (amino acids) 1. Add 5 ml distilled H 2 O using pipette to test tube 2. Add 1 ml of food sample to test tube 3. Add 20 drops of Biuret solution

Procedure Simple carbohydrate 1. Add 5 ml distilled H 2 O using pipette to test tube 2. Add 1 ml of food sample to test tube 3. Add 20 drops of Benedict solution 4. Place test tube in a hot water bath for 10 minutes.

Procedure Complex carbohydrate 1. Add 5 ml distilled H 2 O using pipette to test tube 2. Add 1 ml of food sample to test tube 3. Add 20 drops of IKI solution

LAB SAFETY and CLEAN UP NO EDIBLE WEAR safety THOROUGHLY products in goggles and CLEAN lab area lab apron at all times and equipment
- Slides: 18