Identification of symmetry elements The molecules can be

Identification of symmetry elements The molecules can be classified into groups depending on their symmetry. This helps a chemist to predict their properties. In this presentation the user will learn to identify the symmetry elements and classify simple molecules as per their groups. Learning objective: The learner will learn to identify the point group in molecules that have simple symmetry. Author E Siva Subramaniam Iyer, IIT Bombay
1 Master Layout 2 3 4 5 Nature is symmetric so are molecules. Symmetry simplifies understanding of science

Definitions of the components: 1 2 1. Symmetry operation is an operation which when performed on the molecule leaves the molecule unchanged. 2. Complete set of symmetry operation forms a group. 3. E = Identity operation. Leaving the molecule as such. Doing nothing. 4. Rotation operation = This is an operation of rotating the molecule about an axis. If the rotation be 360 o/ n brings the nuclear framework coincident with original sturucture, the molecule is said to posses Cn axis of rotation. There can be more than one Cn axis of rotation. The axis with highes value of n is called principal axis. 3 5. Reflection Operation = If upon reflection about a plane molecules comes to coincidence with the nuclear framework, then molecule is said to posses a plane of symmetry. It is given a symbol of sigma σ. A subscript of xy, yz, zx may be added to signify the plane or v added as subscript if plane contains the principal axis of rotation or a subscript of h is added if the symmetry plane is perpendicular to principal axis. 4 6. Rotation- Reflection operation. = Represented by Sn. Rotation of molecule by 360/n degrees and reflection about a plane perpendicular to the axis ( or vice versa ) leaves the molecule unchanged then molecule is said to pesses alternating axis of symmetry. 5 7. Inversion operation = A molecule is said to pesses inversaion center i , if about a centre all points are inverted. i. e. if x, y, z are cordinates then they are moved to –x, -y, -z. If such an operation leaves molecule unchanged , then molecule has “i”.
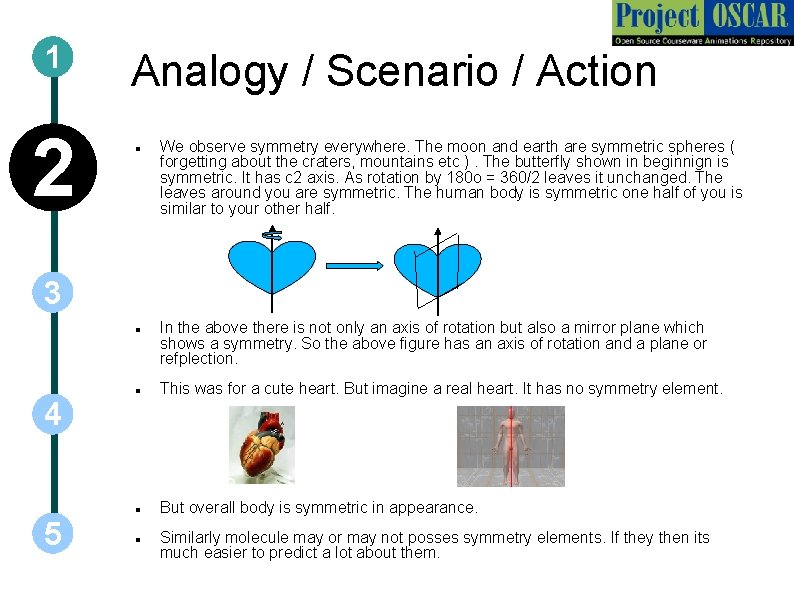
1 2 Analogy / Scenario / Action We observe symmetry everywhere. The moon and earth are symmetric spheres ( forgetting about the craters, mountains etc ). The butterfly shown in beginnign is symmetric. It has c 2 axis. As rotation by 180 o = 360/2 leaves it unchanged. The leaves around you are symmetric. The human body is symmetric one half of you is similar to your other half. 3 This was for a cute heart. But imagine a real heart. It has no symmetry element. But overall body is symmetric in appearance. 4 5 In the above there is not only an axis of rotation but also a mirror plane which shows a symmetry. So the above figure has an axis of rotation and a plane or refplection. Similarly molecule may or may not posses symmetry elements. If they then its much easier to predict a lot about them.

1 2 Symmetry in molecules 3 We shall learn to identify the symmetry elements, one by one in simple molecules. The symmetry elements can be grouped together to give rise to a point group. The rules which apply to the symmetry operations are mathematically studied via the principles of group theory 4 5 Action Description of Action Audio narration Text appears Each statement appears one after another First two statements may be given an audio narration.

1 2 The identity operation : E Doing Nothing ! 3 Figure 1 No Change !!! Figure 2 4 Action 1. 5 The tile appears. Then text in blue appears Description of Action Audio narration The appearance of text The identity operation is accompanied by invovles leaving the audio narration molecule as such. This is like multiplication by 1. E forms part of any
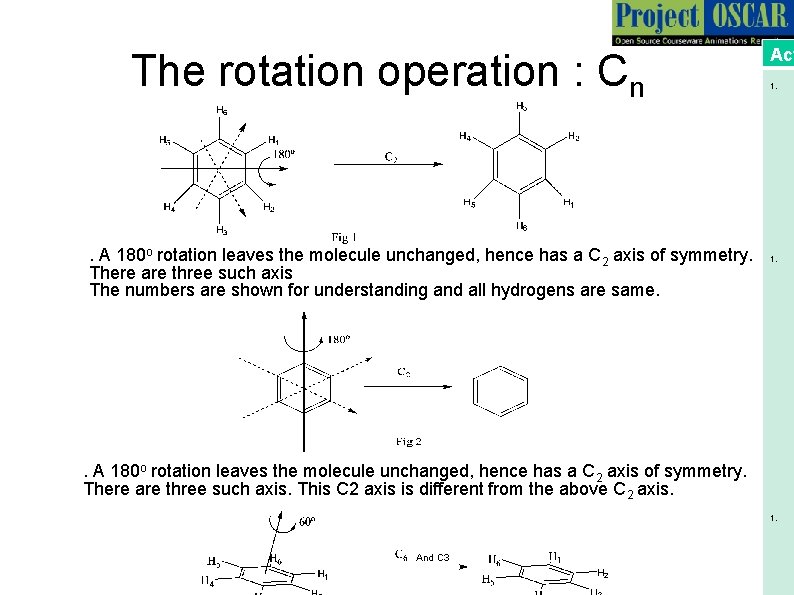
The rotation operation : Cn . A 180 o rotation leaves the molecule unchanged, hence has a C 2 axis of symmetry. There are three such axis The numbers are shown for understanding and all hydrogens are same. Act 1. . A 180 o rotation leaves the molecule unchanged, hence has a C 2 axis of symmetry. There are three such axis. This C 2 axis is different from the above C 2 axis. 1. And C 3

The reflection operation: σ Fig 1 Fig 2 Fig 3 σh = 1 σv = 3 σ’v = 3 total 7 reflection planes The σ’v is called σd plane. A σd plane is a plane that contains the principal axis and bisects the angle formed by two C 2 axis. If this condition is not met, the reflection plane is simply called σ’v Action 1. Fig

The rotation reflection operation: Sn Rotation of molecule by 360/n degrees and reflection about a plane perpendicular to the axis ( or vice versa ) leaves the molecule unchanged then molecule is said to pesses alternating axis of symmetry. Fig 1 Fig 2 Arrow 1 σh Arrow 2 The molecule possess an S 2 axis of rotation reflection Action 1. Title and text appears Description of Action Fig 3 Audio narration let this be understood by an example. Fig 4
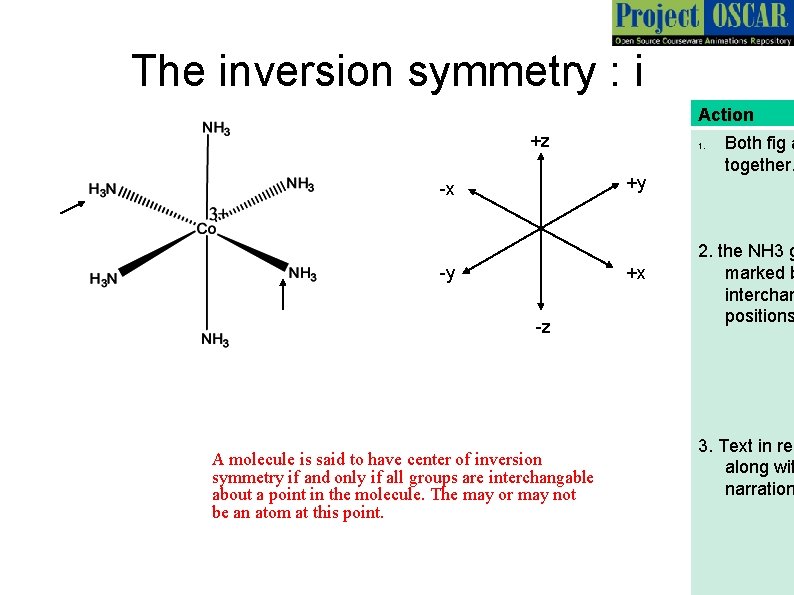
The inversion symmetry : i Action +z 1. +y -x -y +x -z A molecule is said to have center of inversion symmetry if and only if all groups are interchangable about a point in the molecule. The may or may not be an atom at this point. Both fig a together. 2. the NH 3 g marked b interchan positions 3. Text in red along wit narration

Questionnaires Q. What does C∞ mean? a. There are infinite number of C axis. b. there is one axis about which there are infinite orientations. c. Both statements are applicable. d. None of the above. Q. How many planes of symmetry are there in naphthalene? a. 2 b. 3 c. 12 d. 0 Q. Can a molecule have more than one principal axis? a. No b. Yes c. Depends on molecule. d. Yes, but at most two can be there. Q. Do all symmetric molecules have “i” symmetry? a. Yes b. No c. Cant Say d. none of the above Q. Where does point of centre of symmetry lie? a. anywhere Inside the molecule b. outside the molecule c. Centre of molecule, only if there is an atom. D. Centre of molecule, irrespective of an atom being present.

References Reference website http: //www. reciprocalnet. org/edumodules/symmetry/index. html Books: Chemical Application of Group theory, F A Cotton, 3 rd edition, john Wiley and Sons. , 1990 Group Theory and Chemsitry, David Bishop, Dover Publication, New York, 1973. Note to animator: There might be few places whre nomenclature of point groups might have been violated. Please ensure that the format is Axy : first letter caps, rest all small and subscript
- Slides: 12