Ideal Bose gas We now study the properties

Ideal Bose gas. We now study the properties of a perfect gas of bosons of non-zero mass. The Pauli principle does not apply in this case, and the low -temperature properties of such a gas are very different from those of a fermion gas discussed in the last lecture. A B-E gas displays most remarkable quantal features. The properties of BE gas follow from the Bose-Einstein (8. 1 ) distribution We must always have nj 0 , as the number of particles in a state cannot be negative. This requires that 1

If the zero of energy is taken at the lowest energy state, state we must (8. 3) have or (8. 4) At absolute zero all the particles will be in the ground state, and we have for no (8. 5) This is satisfied by (8. 6) 2

We now consider the situation at finite temperatures. Let g( )d be the number of states in d at . We have (8. 8) (8. 9) The density of states g( ) can be presented as (8. 10) where (8. 11) 3

The power ½ in is coming from the following consideration: We must be cautious in substituting (8. 10) into (8. 8). At high temperatures there is no problem. But at low temperatures there may be a pile-up of particles in the ground state =0; then we will get an incorrect result for N. This is because g(0)=0 in the approximation we are using, whereas there is actually one state at =0. If this one state is going to be important we should write (8. 12) where ( ) is the Dirac delta function We have then, instead of (8. 8) 4

(8. 13) It is convenient to write (8. 14) where =e / and 0 1 , from (8. 3). If <<1, <<1 the classical Boltzmann distribution is a good approximation. If the distribution is degenerate and most of the particles will be in the ground state. In the treatment of BE gas we are going to need integrals of the form (8. 15) We have 5

(8. 16) The last integral is equal to (8. 17) where (x) is the gamma function. From (8. 16) (8. 18) We have 6

(8. 19) where (8. 20) Further, (8. 21) where (8. 22) Because 1 these series always converge. We note that (8. 23) 7

From (8. 13) (8. 24) or taking the spin to be zero (8. 25) and from (8. 9) (8. 26) 8

Here (8. 27) is the number in the ground state, and (8. 28) is the number of particles in excited states. At high temperatures <<1 we obtain the usual classical result for the energy: (8. 29) 9

Bose-Einstein Condensation Let us consider equation (8. 25) in the quantum region. For =1 we have (8. 30) where is the Riemann zeta function If N 0 is to be a large number (as at low temperatures), then must be very close to 1 and the number of particles in excited states will be given approximately by (8. 28) with F( )=F(1). (8. 28) 10

(8. 31) It should be pointed out that (8. 31) represents an upper limit to the number of particles in states other than the ground state, at the temperature for which is calculated. If N is appreciably greater than N , N 0 must be large and the number of particles in excited states must approach (8. 31) Let us define a temperature T 0 such that (8. 32) where 0 is thermal de Broglie wavelength at T 0. Then, from (8. 31) 11

(8. 33) The number of particles in excited states varies as temperature region for which T 3/2 for T< T 0, in the F( ) F(1)=2. 612. Further, the number of particles in the ground state is given approximately by (8. 34) Thus for T even a little less than T 0 a large number of particles are in the ground state, whereas for T>T 0 there are practically no particles in the ground state. We call T 0 the degeneracy temperature or the condensation temperature. It can be calculated easily from the relation 12

where VM is the molar volume in cm 3 and M is the molecular weight. For liquid helium VM=27. 6 cm 3; M=4, and T 0=3. 1 o. K. It is not correct to treat the atoms in liquid helium as non-interacting, but the approximation is not as bad in some respects as one might think. The rapid increase in population of the ground state below T 0 for a Bose gas is known as the Bose-Einstein condensation. It is illustrated in Figure 8. 1 a condensation in momentum space rather than a condensation in coordinate space such as occurs for liquid-gas phase transformation 13
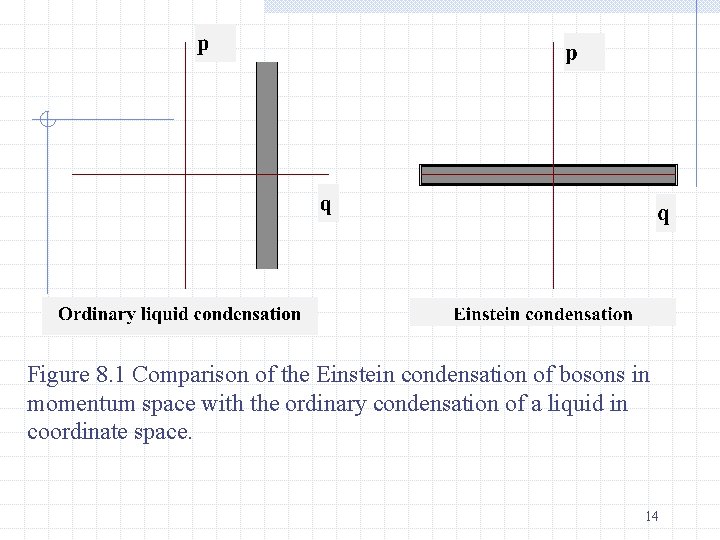
Figure 8. 1 Comparison of the Einstein condensation of bosons in momentum space with the ordinary condensation of a liquid in coordinate space. 14

Normally, gases have no such transition because they all turn into liquids or solids under the conditions required for Bose condensation to occur. However, liquid helium (4 He) has two phases called He I and He II, and He II has anomalous thermal and mechanical properties. It is believed that the lambda-point transition observed in liquid helium at 2. 19 0 K is essentially an Einstein condensation. Remarkable physical properties described as superfluidity are exhibited by the low-temperature phase, which is known as liquid He II. It is generally believed that the superflow properties are related to the Einstein condensation in the ground state. 15

When a material does become a superfluid, superfluid it displays some very strange behaviour; • if it is placed in an open container it will rise up the sides and flow over the top • if the fluid's container is rotated from stationary, the fluid inside will never move, the viscosity of the liquid is zero, so any part of the liquid or it's container can be moving at any speed without affecting any of the surrounding fluid • if a light is shone into a beaker of superfluid and there is an exit at the top the fluid will form a fountain and shoot out of the top exit 16
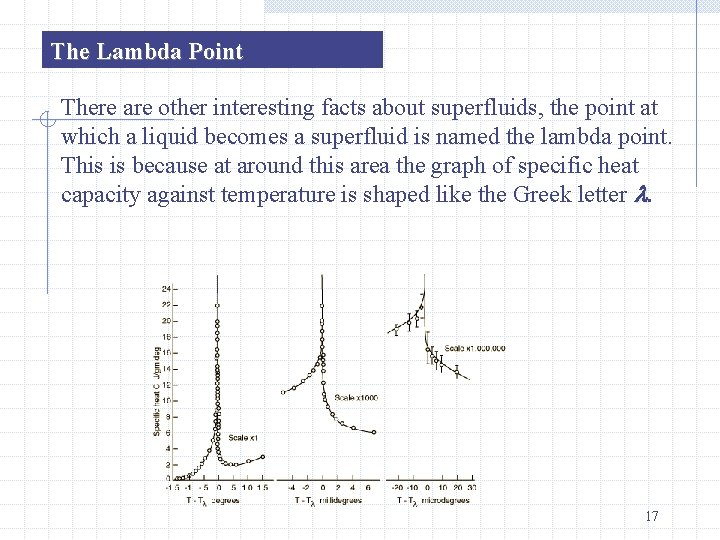
The Lambda Point There are other interesting facts about superfluids, the point at which a liquid becomes a superfluid is named the lambda point. This is because at around this area the graph of specific heat capacity against temperature is shaped like the Greek letter . 17
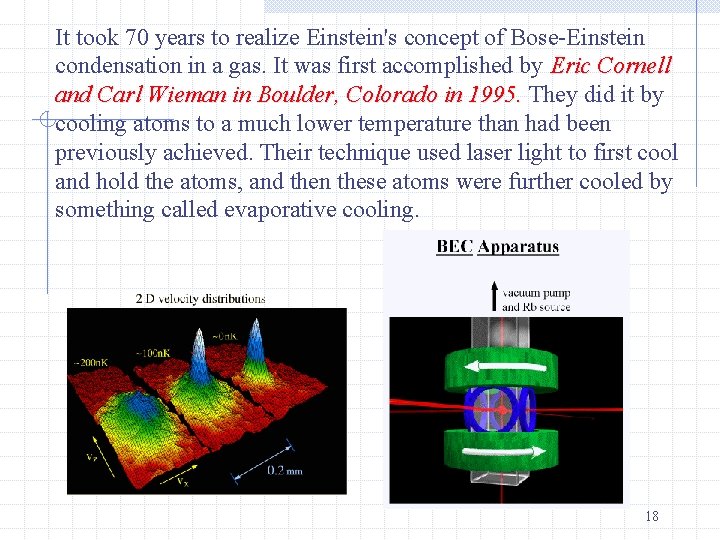
It took 70 years to realize Einstein's concept of Bose-Einstein condensation in a gas. It was first accomplished by Eric Cornell and Carl Wieman in Boulder, Colorado in 1995. They did it by cooling atoms to a much lower temperature than had been previously achieved. Their technique used laser light to first cool and hold the atoms, and then these atoms were further cooled by something called evaporative cooling. 18
- Slides: 18