ID DB 08923 EPOETIN ZETA C 809 H
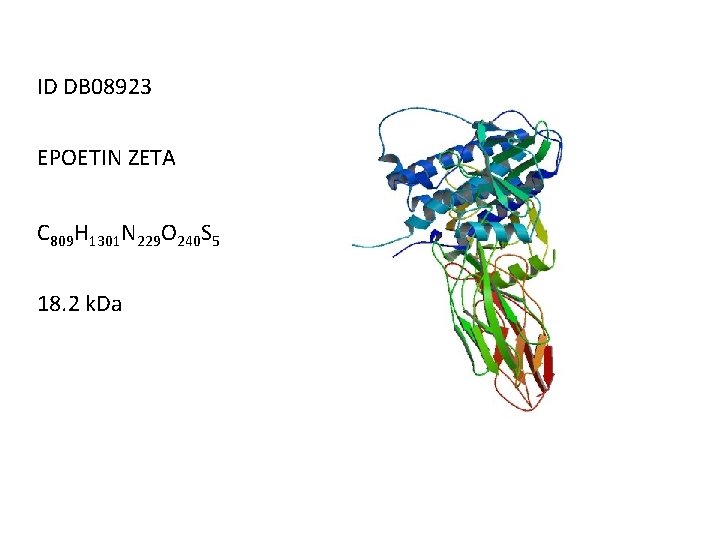
ID DB 08923 EPOETIN ZETA C 809 H 1301 N 229 O 240 S 5 18. 2 k. Da

DESCRIPTION The active substance in Epoetin zeta is a recombinant human erythropoietin (rh. EPO) of identical primary structure produced in Chinese Hamster Ovary (CHO) cells. The molecular weight of the glycosylated protein is 30. 6 k. Da according to the Ph. Eur. monograph, 40% of which are carbohydrate structures. The oligosaccharide chains are subject to posttranslational modifications and display heterogeneity to a certain extent. Epoetin zeta is also identical to Epoetin alfa in terms of its amino acid sequence. INDICATION For use in the treatment of symptomatic anaemia associated with chronic renal failure (CRF) in adult and paediatric patients. Also for use in the treatment of anaemia and reduction of transfusion requirements in adult patients receiving chemotherapy for solid tumours, malignant lymphoma or multiple myeloma, and at risk of transfusion as assessed by the patient's general status (e. g. cardiovascular status, pre existing anaemia at the start of chemotherapy). Also for used to increase the yield of autologous blood from patients in a predonation program. When administered subcutaneously, Epoetin Zeta is equivalent to Epoetin Alfa in terms of clinical effectiveness.

PHARMACODYNAMICS Used in the treatment of anemia. Involved in the regulation of erythrocyte differentiation and the maintenance of a physiological level of circulating erythrocyte mass. MECHANISM OF ACTION Binding of erythropoietin to the erythropoietin receptor leads to receptor dimerization, which facilitates activation of JAK STAT signaling pathways within the cytosol. Activated STAT (signal transducers and activators of transcription) proteins are then translocated to the nucleus where they serve as transcription factors which regulate the activation of specific genes involved in cell division or differentiation. HALF LIFE Toxicokinetic results from rats in a 13 week toxicity study after a single subcutaneous dose: 7. 37 hours (+/ 0. 70) with 500 IU/kg [test 1] 8. 63 hours (+/ 2. 78) with 2500 IU/kg [test 2] 8. 76 hours (+/ 1. 46) with 2500 IU/kg [reference dose]

VOLUME OF DISTRIBUTION = 150 m. L/kg CLEARANCE = 0. 5 0. 7 m. L/minute/kg (SC administration of 3. 45 mcg/kg and 11. 5 mcg/kg in both normal subjects and cancer patients, Neupogen) TARGETS Erythropoietin receptor

BRAND NAMES Epobel® Nobel Ilac Pazarlama ve Sanayii Ltd. STI. , Turkey Eqralys® Hemofarm AD Retacrit® Hospira, Inc. Silapo® Cell Pharm Gmb. H Silapo® Stada Arzneimittel AG

EPOETIN ZETA Epoetin beta was found to differ from epoetin alfa in containing: (a) a greater proportion of more basic isoforms, (b) a greater proportion of EPO binding to Erythrina cristagalli agglutinin (which binds N glycans with nonsialylated outer Gal beta 1 4 Glc. NAc moieties), and (c) isoforms with higher in vivo: in vitro bioactivity ratios. Epoetin beta also contained slightly more than epoetin alfa of EPO binding to Lycopersicon esculentum agglutinin (which binds N glycans containing repeating Gal beta 1 4 Glc. NAc sequences), to the leucoagglutinin of Phaseolus vulgaris (which binds tetraantennary and 2, 6 branched triantennary N glycans) and to Agaricus bisporus agglutinin (which binds Gal beta 1 3 Gal. NAc containing O glycans). No differences were found between the two r. EPOs in their binding to a further five lectins The epoetins are probably glycosylated more completely than endogenous EPO, as the specific in vivo biological activity of r. Hu. EPO is greater (approximately 200, 000 international units (IU)/mg peptide) than that of purified human urinary EPO (70, 000 IU/mg peptide). In one survey in Spain, the epoetin zeta cost analysis reported a total cost saving of nearly 45% with respect to epoetin alfa. Costs per patient per month declined from € 298 for epoetin alfa therapy to € 177 per patient per month for epoetin zeta

After a series of sub cultivations, the cells are seeded into the production fermenter. The production is based on a fed batch process. The drug substance is recovered from the fermentation broth by a conventional protein purification process comprising orthogonal chromatography steps and a viral filtration step. The manufacturing steps are monitored by process controls that include operational parameters, acceptance criteria and specifications. he total glycan pool released from the protein backbone was subjected to fractionation by column chromatography and if necessary further sub fractionation was performed using a different stationary phase in order to yield sub fractions of sufficient purity. HPAEC PAD was used as a major analytical tool to classify the glycans with respect to sialylation and antennarity on each level of sub fractionation. Further analysis of the sub fractions by mass spectroscopy enabled the ability to identify and/or exclude unique or unusual structures. Using NMR techniques, glycan structures were elucidated with respect to their isomeric nature. Positions of N acetyllactosamine repeats on antennae, lack of sialylation of antennae, position of fucosylation (in order to identify/exclude certain Lewis motifs) and presence of O acetylated groups were confirmed. O glycans were mainly characterised by mass spectrometrical methods of the de N glycosylated protein. Applying this analytical strategy, the carbohydrate content of epoetin zeta was confirmed to beessentially free of unusual, potentially immunogenic structures

pre filled syringes contains recombinant human erythropoietin (rhu EPO, epoetin) as drug substance and water for injection, sodium monohydrogen phosphate, sodium dihydrogen phosphate, sodium chloride, calcium chloride, polysorbate 20, glycine, leucine, isoleucine, threonine, glutamic acid and phenylalanine as excipien ts. The syringes (Type I glass) are provided with a fixed steel needle a nd a plunger stopper with PTFE coating for intravenous (IV) or subcutaneous (SC) injection. Eleven different quantitative sizes of pre filled syringes of SB 309 will be available containing defined amounts of epoetin of 1, 000 IU up to 40, 000 IU, respectively. The drug product is presented as a solution for injection in pre filled syringes in the following strengths: 3, 333 IU/ml (presentation of 0. 3, 0. 6 and 0. 9 ml), 10, 000 IU/ml (presentations of 0. 4, 0. 5, 0. 6, 0. 8 and 1 ml) and 40, 000 IU/ml (presentations of 0. 5, 0. 75 and 1 ml), withall strengths exhibiting the same qualitative composition. ADVERSE REACTION: risk of mmunogenicity and the safety profile for serious adverse events raises uncertainties for the long term safety and effectiveness of biosimilars. Future epoetin zeta studies could assess patient outcomes and costs for target Hb levels and further compare results between epoetin zeta and epoetin alfa HALF LIFE: 8. 63 hours

ASSAY: An in vivo comparison of the potency of SB 309 and of Erypo over the dosage range 3. 3 to 90 IU/ml in normocythaemic mice was undertaken using methodology described in the European Pharmacopoeia (01/2005: 1316 corrected, method B). EPO BRP#2 provided by the European Directorate for the Quality of Medicines, European Pharmacopoeia Commission was also tested. The assay quantifies reticulocytes by fluorescence activated cell sorting of blood drawn four days after one subcutaneous administration to female B 6 D 2 F 1 mice. This method allows establishment of a dose response relationship for each test material and includes a check of parallelism of regressions for a valid quantitative comparison. The relative potency should be between 0. 80 and 1. 25.

PROTEIN SEQUENCE APPRLICDSRVLERYLLEAKEAENITTGCAEHCSLNENITVPDTKVNFYAWKRMEVGQ QAVEVWQGLALLSEAVLRGQALLVNSSQPWEPLQLHVDKAVSGLRSLTTLLRALGA QKEAISPPDAASAAPLRTITADTFRKLFRVYSNFLRGKLKLYTGEACRTGDR

REFERENCE • Wizemann V, Rutkowski B, Baldamus C, Scigalla P, Koytchev R: Comparison of the effects of epoetin zeta to epoetin alfa in the maintenance phase of renal anaemia tre Med Res Opin. 2008 Mar; 24(3): 625 37 • Krivoshiev S, Wizemann V, Czekalski S, Schiller A, Pljesa S, Wolf Pflugmann M, Siebert Koytchev R, Bronn A: Therapeutic equivalence of epoetin zeta and alfa, administered subcutaneously, for maintenance treatment of renal anemia. Adv Ther. 2010 Feb; 27( 10. 1007/s 12325 010 0012 y. Epub 2010 Mar 30 • Brinks V, Hawe A, Basmeleh AH, Joachin Rodriguez L, Haselberg R, Somsen GW, Jisko Schellekens H: Quality of original and biosimilar epoetin products. Pharm Res. 2011 F 93. doi: 10. 1007/s 11095 010 0288 2. Epub 2010 Oct 1 • http: //www. ema. europa. eu/docs/en_GB/document_library/EPAR_ _Scientific_Discussion/human/000872/WC 500054374. pdf • http: //www. ncbi. nlm. nih. gov/pubmed/24790409 • http: //www. ncbi. nlm. nih. gov/pubmed/25011615 • http: //www. ncbi. nlm. nih. gov/pubmed/25242760 • http: //www. ncbi. nlm. nih. gov/pubmed/25348203 • http: //www. ncbi. nlm. nih. gov/pubmed/23242745
- Slides: 11