ICTR Biostatistics Research Resource Mary Lindstrom Professor Dept

ICTR Biostatistics Research Resource Mary Lindstrom Professor, Dept. of Biostatistics and Medical Informatics Director, ICTR Biostatistics Research Resource Faculty and Staff 20 UW Faculty Biostatisticians 8 UW Staff Biostatisticians 4 Marshfield Clinic Biostatisticians

ICTR Biostatistics Resource Faculty and Staff Clinical Research • UW Faculty: AC Andrei, T Becker, K Buhr, R Chappell, T Cook, M Detry, NM Dowling, M Fisher, P Hutson, R Gangnon, M Lindstrom, M Palta, M Rosenberg, S Wang • UW Staff: J Barnet, M Evans, S Hetzel, Z Li, V Rajamanickam, S Seo • Marshfield Clinic: R Berg, P Chyou, H Liang Statistical Genetics • • • UW Faculty: K Broman, S Keles, C Kendziorski, R Mau, M Newton, S Wang, B Yandell UW Staff: A Andrei, A Broman Marshfield Clinic: Z Ye

Areas of Expertise Traditional Areas • Clinical Trials • Prospective Cohort Studies • Case Control Studies • Retrospective Studies • Pre-clinical Studies Special Areas (growing, unique) • Statistical Genetics • Qualitative Outcomes • Cost and Policy Issues • Pharmacokinetics & Pharmacodynamics

Biostatistics Resource Activities Consulting Collaborative Research Methodology Research Training Protocol & Pilot Project Review

UW - Madison Resource Funding Model 13. 8 FTEs devoted to ICTR Biostatistics

Leveraging UW ICTR Resources Funding Source Activity ICTR Departments Collab. & Centers Grants (3. 7 FTE) (2. 9 FTE) Consulting X X Collaboration X X Methodology Review X Method. Grants (5. 4 FTE) (1. 9 FTE) X X

Marshfield Clinic Funding Model Funding Source FTEs Collaborative Projects 2. 10 Institutional Support 0. 10 ICTR 0. 25 2. 45 FTEs devoted to ICTR Biostatistics

Prioritizing Requests All requests must: • come from ICTR Members • be related to clinical and/or translational research Marshfield Clinic • External, large grant proposals • Internal proposals • Deadlines given priority UW - Madison When we have a waiting list: • New investigators over established • Projects leading to proposals for funding • One active project per investigator • Deadlines given priority • First come, first serve Long term management: • Departments, centers, and investigators with substantial use are asked to provide direct funding.

Units Using Resource Madison Medical School • Anatomy • Anesthesiology • Dermatology • Family Medicine • Medical Physics • Medicine • Neurological Surgery • Neurology • Obstetrics and Gynecology • Ophthalmology & Visual Sciences • Orthopedics and Rehabilitation • Pathology and Laboratory Medicine • Pharmacology • Pediatrics • Population Health Sciences • Psychiatry • Radiology • Surgery Other Schools • Agricultural and Life Sciences • Education • Engineering • Human Ecology • Library and Information Studies • Letters and Science • Nursing • Pharmacy • Veterinary Medicine Other Marshfield • Waisman Center • Cardiology • Center for Health Enhancement Systems Studies • Dermatology • Office of Continuing Professional Development in Medicine and Public Health • Internal Medicine • Department of Veterans Affairs • Division of Education • Neurology • Oncology • Pediatrics • Rheumatology • Sleep Lab • Surgery Red indicates funding for individual statisticians
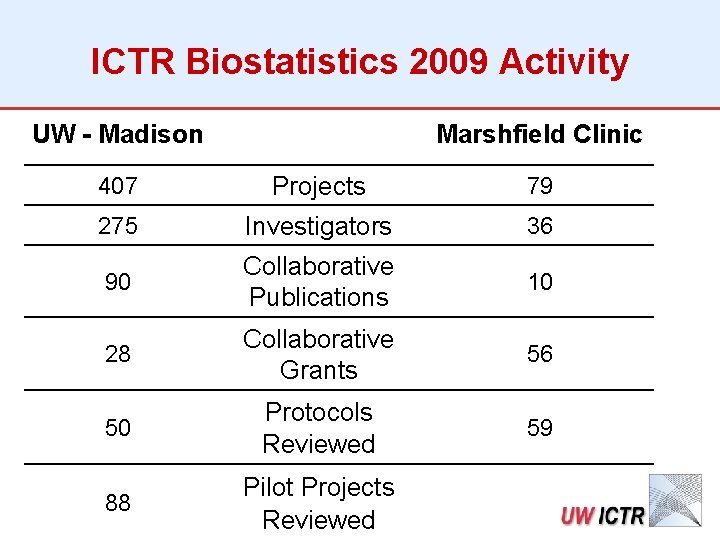
ICTR Biostatistics 2009 Activity UW - Madison Marshfield Clinic 407 Projects 79 275 Investigators 36 90 Collaborative Publications 10 28 Collaborative Grants 56 50 Protocols Reviewed 59 88 Pilot Projects Reviewed

The Future • Upgrade the Activity Tracking System to follow: – the life cycle of grants, protocols, manuscripts, – the effect of training and collaboration on investigator success. • Increase capacity for qualitative endpoints collaboration. • Encourage junior Biostatistics faculty to compete for ICTR pilot grants. • Improve interaction between UW and Marshfield Clinic Biostatistics Research Resources.

Examples of Collaboration Clinical Trials Wisconsin Network for Health Research (Wi. NHR) presented by Mary Lindstrom Stat-Gen Statistical Genetics and Genomics Program presented by Christina Kendziorski

Wisconsin Network for Health Research (Wi. NHR) • A statewide network of clinical sites to collaborate on trials of • Diagnostic, preventative and therapeutic interventions • Comparative effectiveness • Expands capabilities of Wisconsin researchers • More studies done faster • Population of > 3 million • Centralizes some aspects of the research • Data management • IRB approval process • Statistical analysis • Each site has a PI and local research staff
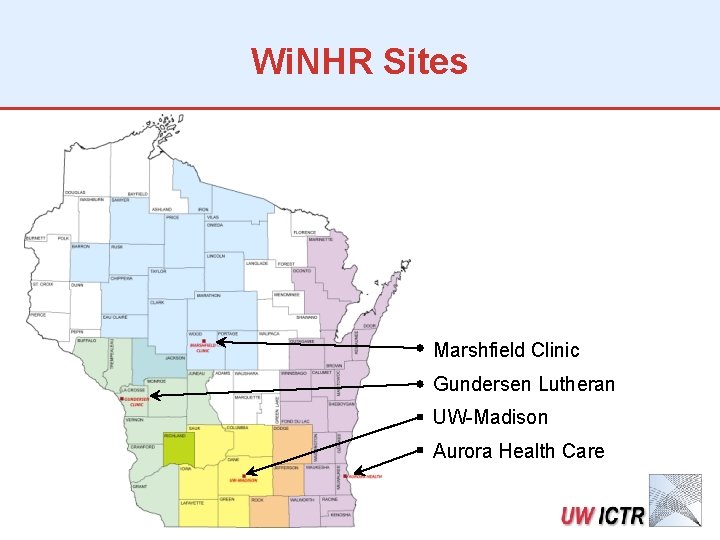
Wi. NHR Sites Marshfield Clinic Gundersen Lutheran UW-Madison Aurora Health Care

Wi. NHR Studies • Protocols • come from all participating sites • are funded independently • Studies • 9 ongoing, 1 completed • 712 patients accrued • 8 different lead investigators • 9/10 studies use 3 or 4 sites.
- Slides: 15