ICPH 2019 Kobe Japan December 1 2019 Antioxidant

ICPH 2019, Kobe, Japan December 1, 2019 Antioxidant and Anti-inflammatory Effects of Oat Avenanthramides Li Li Ji, Ph. D, NAK, FACSM Professor and Director, Laboratory of Physiological Hygiene and Exercise Science University of Minnesota at Twin Cities, USA
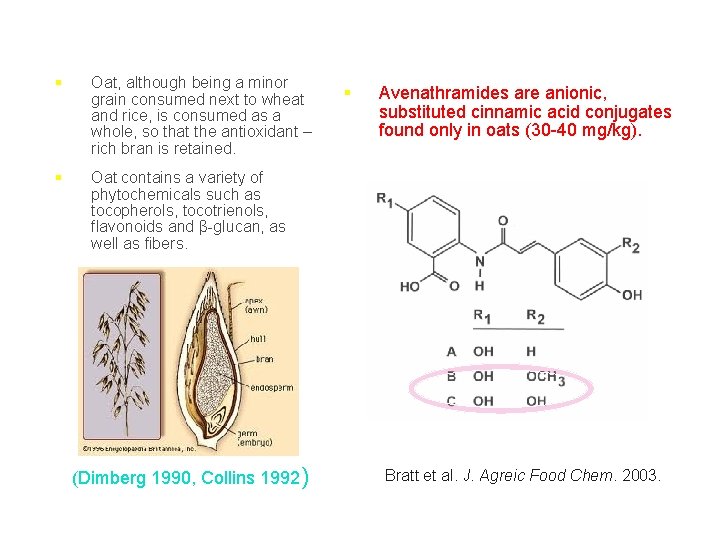
§ Oat, although being a minor grain consumed next to wheat and rice, is consumed as a whole, so that the antioxidant – rich bran is retained. § Oat contains a variety of phytochemicals such as tocopherols, tocotrienols, flavonoids and β-glucan, as well as fibers. (Dimberg 1990, Collins 1992) § Avenathramides are anionic, substituted cinnamic acid conjugates found only in oats (30 -40 mg/kg). Bratt et al. J. Agreic Food Chem. 2003.
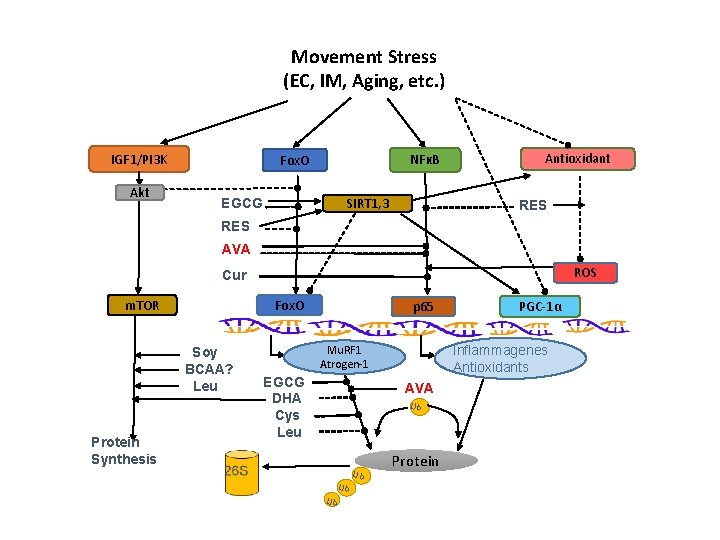
Movement Stress (EC, IM, Aging, etc. ) IGF 1/PI 3 K Akt NFκB Fox. O SIRT 1, 3 EGCG Antioxidant RES AVA ROS Cur Fox. O m. TOR Soy BCAA? Leu Protein Synthesis p 65 Inflammagenes Antioxidants Mu. RF 1 Atrogen-1 EGCG DHA Cys Leu AVA Ub Ub PGC-1α Protein

Oat Avenathramide as Antioxidant and Anti-inflammatory Agent Outlines § In vivo antioxidant and anti-inflammatory properties § Bioavailability of AVA § AVA protection for exercise-induced muscle inflammation § Potential role of AVA in preventing inflammation-induced muscle fiber atropy § Molecular mechanism for antiinflammatory function of AVA
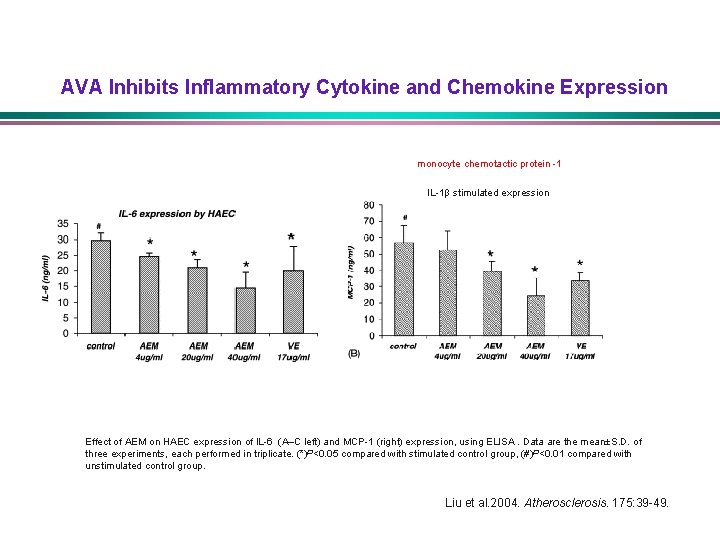
AVA Inhibits Inflammatory Cytokine and Chemokine Expression monocyte chemotactic protein -1 IL-1β stimulated expression Effect of AEM on HAEC expression of IL-6 (A–C left) and MCP-1 (right) expression, using ELISA. Data are the mean±S. D. of three experiments, each performed in triplicate. (*)P<0. 05 compared with stimulated control group, (#)P<0. 01 compared with unstimulated control group. Liu et al. 2004. Atherosclerosis. 175: 39 -49.
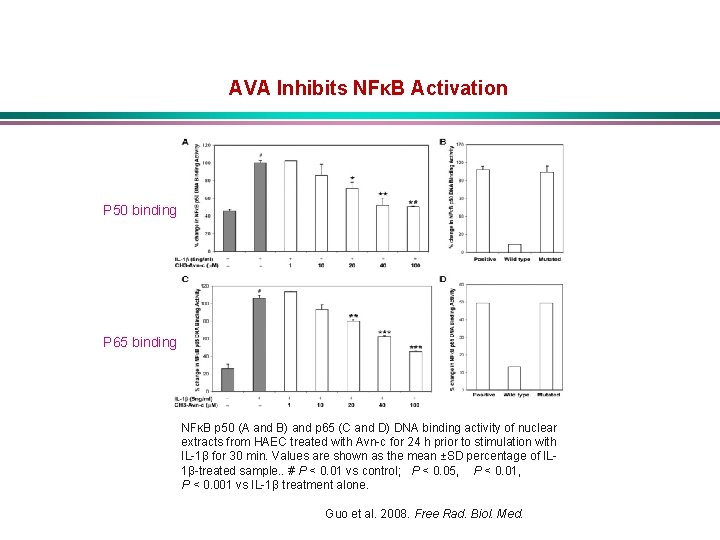
AVA Inhibits NFκB Activation P 50 binding P 65 binding NFκB p 50 (A and B) and p 65 (C and D) DNA binding activity of nuclear extracts from HAEC treated with Avn-c for 24 h prior to stimulation with IL-1β for 30 min. Values are shown as the mean ±SD percentage of IL 1β-treated sample. . # P < 0. 01 vs control; P < 0. 05, P < 0. 01, P < 0. 001 vs IL-1β treatment alone. Guo et al. 2008. Free Rad. Biol. Med.
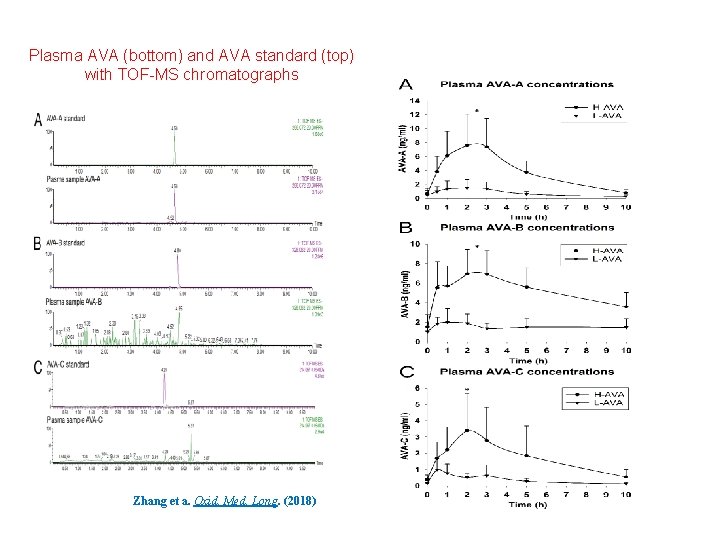
Plasma AVA (bottom) and AVA standard (top) with TOF-MS chromatographs Zhang et a. Oxid. Med. Long. (2018)
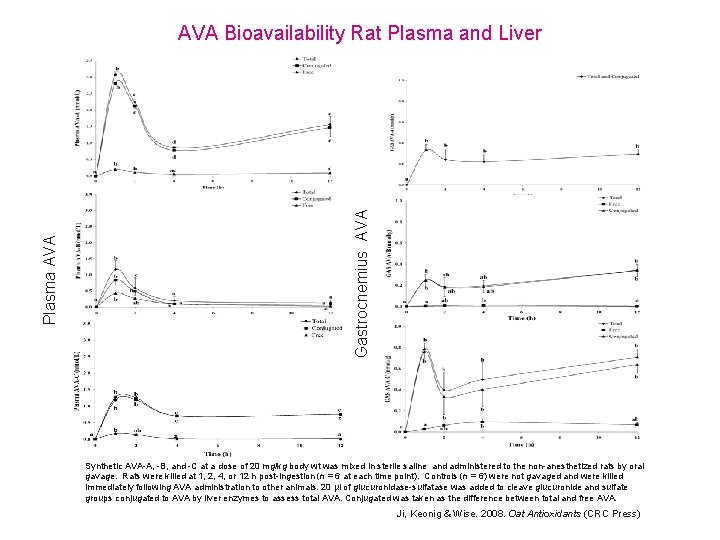
Gastrocnemius AVA Plasma AVA Bioavailability Rat Plasma and Liver Synthetic AVA-A, -B, and -C at a dose of 20 mg/kg body wt was mixed in sterile saline and administered to the non-anesthetized rats by oral gavage. Rats were killed at 1, 2, 4, or 12 h post-ingestion (n = 6 at each time point). Controls (n = 6) were not gavaged and were killed immediately following AVA administration to other animals. 20 µl of glucuronidase-sulfatase was added to cleave glucuronide and sulfate groups conjugated to AVA by liver enzymes to assess total AVA. Conjugated was taken as the difference between total and free AVA. Ji, Keonig & Wise. 2008. Oat Antioxidants (CRC Press)
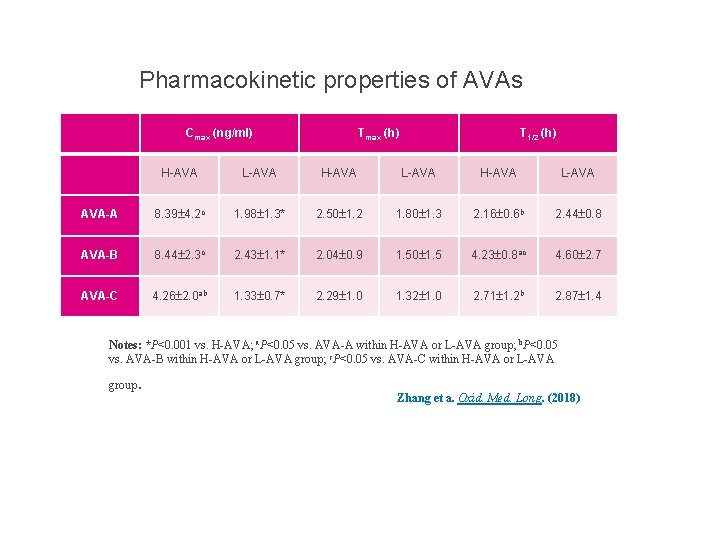
Pharmacokinetic properties of AVAs Cmax (ng/ml) Tmax (h) T 1/2 (h) H-AVA L-AVA AVA-A 8. 39 4. 2 c 1. 98 1. 3* 2. 50 1. 2 1. 80 1. 3 2. 16 0. 6 b 2. 44 0. 8 AVA-B 8. 44 2. 3 c 2. 43 1. 1* 2. 04 0. 9 1. 50 1. 5 4. 23 0. 8 ac 4. 60 2. 7 AVA-C 4. 26 2. 0 ab 1. 33 0. 7* 2. 29 1. 0 1. 32 1. 0 2. 71 1. 2 b 2. 87 1. 4 Notes: *P<0. 001 vs. H-AVA; a. P<0. 05 vs. AVA-A within H-AVA or L-AVA group; b. P<0. 05 vs. AVA-B within H-AVA or L-AVA group; c. P<0. 05 vs. AVA-C within H-AVA or L-AVA group . Zhang et a. Oxid. Med. Long. (2018)

Bioavailibility of AVA in vivo Summary • • AVA can be readily absorbed into the plasma and reaches peak plasma concertation within hours after oral ingestion AVA can be conjugated and absorbed by various organs such as skeletal muscle making it an anti-inflammatory agent in vivo

AVA Supplementation and Muscle Inflammation § Animals: Female Sprague-Dawley rats (n=38, age 6 -7 wk) Control AVA-C Rested 9 10 Downhill 9 10 § Diet: AIN-93 based control diet or a diet containing 0. 2 g/kg AVEN C for 50 days. § Exercise: Treadmill running at 25 m/min, -20% grade for 2 hour. § Tissue Collection: Killed 24 h post-exercise Heart, liver, kidney, vastus lateralis and soleus muscle.
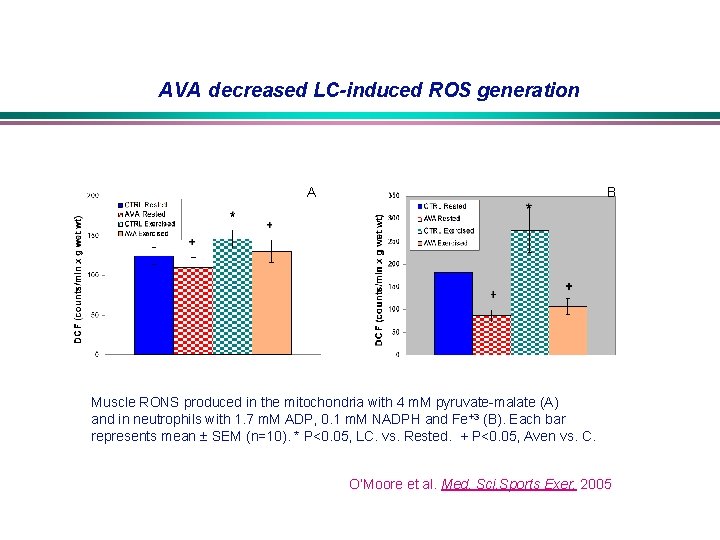
AVA decreased LC-induced ROS generation A B Muscle RONS produced in the mitochondria with 4 m. M pyruvate-malate (A) and in neutrophils with 1. 7 m. M ADP, 0. 1 m. M NADPH and Fe+3 (B). Each bar represents mean SEM (n=10). * P<0. 05, LC. vs. Rested. + P<0. 05, Aven vs. C. O’Moore et al. Med. Sci. Sports Exer. 2005
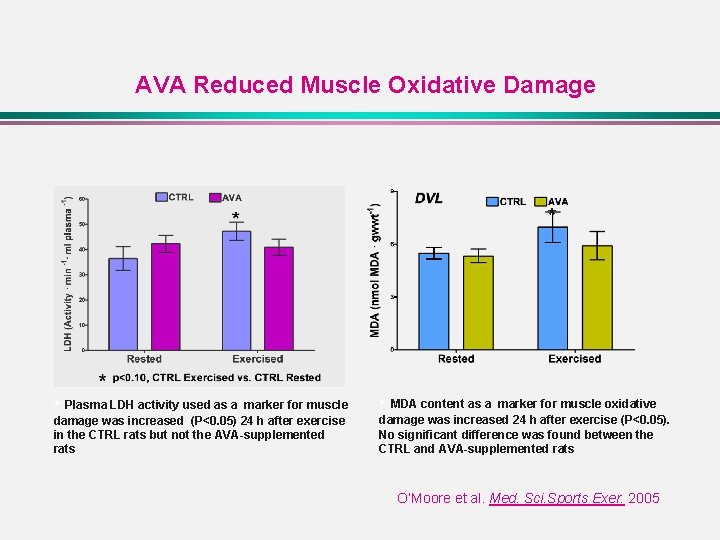
AVA Reduced Muscle Oxidative Damage * • Plasma LDH activity used as a marker for muscle • MDA content as a marker for muscle oxidative damage was increased (P<0. 05) 24 h after exercise in the CTRL rats but not the AVA-supplemented rats. damage was increased 24 h after exercise (P<0. 05). No significant difference was found between the CTRL and AVA-supplemented rats. O’Moore et al. Med. Sci. Sports Exer. 2005
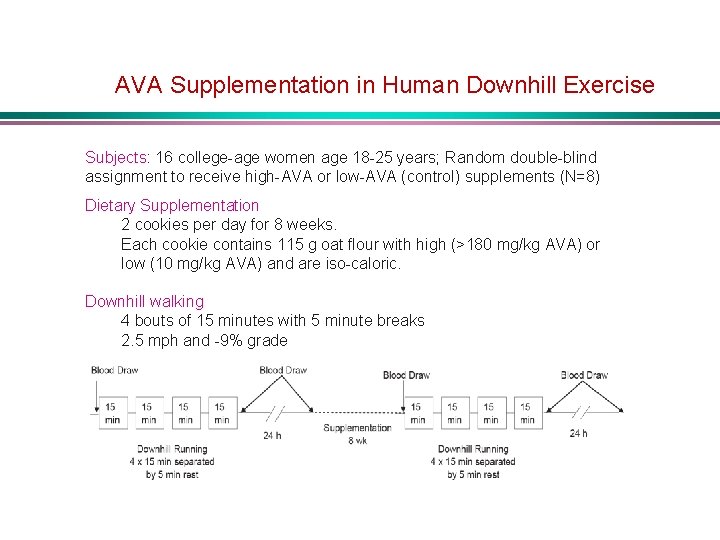
AVA Supplementation in Human Downhill Exercise Subjects: 16 college-age women age 18 -25 years; Random double-blind assignment to receive high-AVA or low-AVA (control) supplements (N=8) Dietary Supplementation 2 cookies per day for 8 weeks. Each cookie contains 115 g oat flour with high (>180 mg/kg AVA) or low (10 mg/kg AVA) and are iso-caloric. Downhill walking 4 bouts of 15 minutes with 5 minute breaks 2. 5 mph and -9% grade
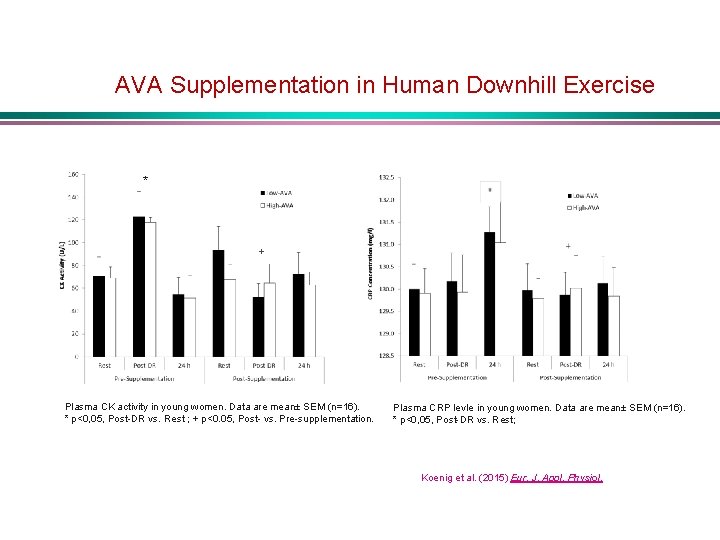
AVA Supplementation in Human Downhill Exercise * + Plasma CK activity in young women. Data are mean± SEM (n=16). * p<0, 05, Post-DR vs. Rest ; + p<0. 05, Post- vs. Pre-supplementation. Plasma CRP levle in young women. Data are mean± SEM (n=16). * p<0, 05, Post-DR vs. Rest; Koenig et al. (2015) Eur. J. Appl. Physiol.
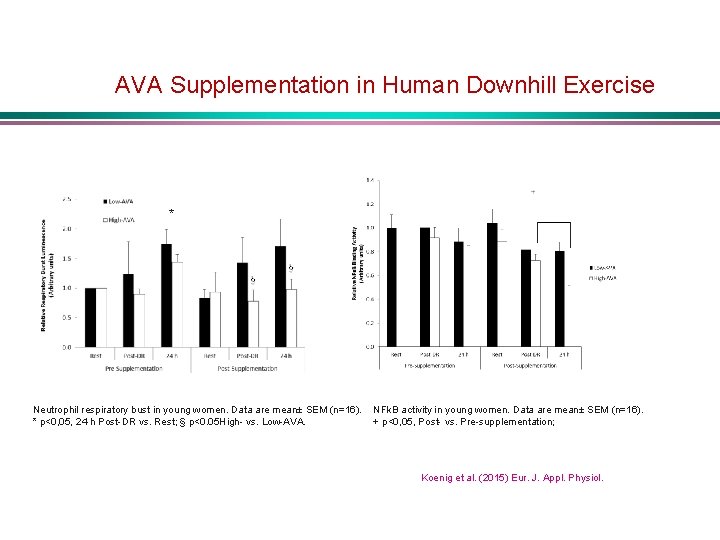
AVA Supplementation in Human Downhill Exercise * Neutrophil respiratory bust in young women. Data are mean± SEM (n=16). * p<0, 05, 24 h Post-DR vs. Rest; § p<0. 05 High- vs. Low-AVA. NFk. B activity in young women. Data are mean± SEM (n=16). + p<0, 05, Post- vs. Pre-supplementation; Koenig et al. (2015) Eur. J. Appl. Physiol.
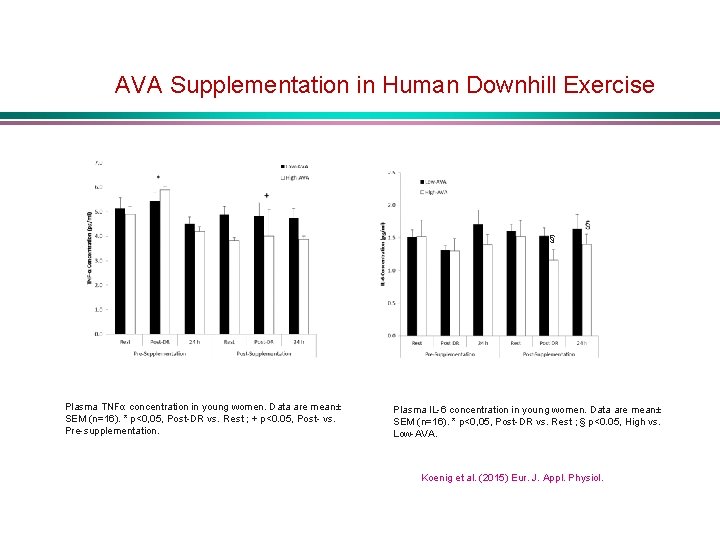
AVA Supplementation in Human Downhill Exercise § Plasma TNFα concentration in young women. Data are mean± SEM (n=16). * p<0, 05, Post-DR vs. Rest ; + p<0. 05, Post- vs. Pre-supplementation. Plasma IL-6 concentration in young women. Data are mean± SEM (n=16). * p<0, 05, Post-DR vs. Rest ; § p<0. 05, High vs. Low-AVA. Koenig et al. (2015) Eur. J. Appl. Physiol.
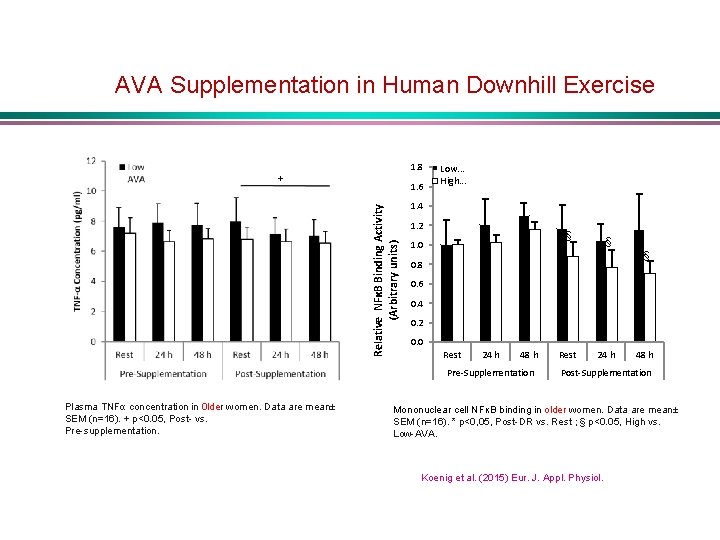
AVA Supplementation in Human Downhill Exercise 1. 8 + Relative NFκB Binding Activity (Arbitrary units) 1. 6 Low. . . High. . . 1. 4 1. 2 § 1. 0 0. 8 0. 6 § 0. 4 0. 2 0. 0 Rest 24 h 48 h Pre-Supplementation Plasma TNFα concentration in 0 lder women. Data are mean± SEM (n=16). + p<0. 05, Post- vs. Pre-supplementation. § Rest 24 h 48 h Post-Supplementation Mononuclear cell NFκB binding in older women. Data are mean± SEM (n=16). * p<0, 05, Post-DR vs. Rest ; § p<0. 05, High vs. Low-AVA. Koenig et al. (2015) Eur. J. Appl. Physiol.

Bioeffacacy of AVA in vivo Summary • • • AVA demonstrated antioxidant function by inhibiting ROS production in animal muscle AVA supplementation in human inhibits production of proinflammatory cytokines and inflammatory myokines There is evidence NFκB activation was inhibited by AVA in response to oxidative challenge
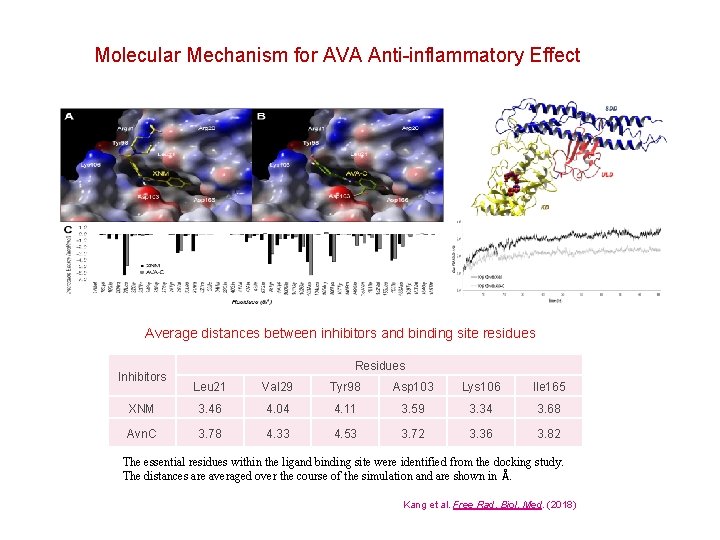
Molecular Mechanism for AVA Anti-inflammatory Effect Average distances between inhibitors and binding site residues Inhibitors Residues Leu 21 Val 29 Tyr 98 Asp 103 Lys 106 Ile 165 XNM 3. 46 4. 04 4. 11 3. 59 3. 34 3. 68 Avn. C 3. 78 4. 33 4. 53 3. 72 3. 36 3. 82 The essential residues within the ligand binding site were identified from the docking study. The distances are averaged over the course of the simulation and are shown in Å. Kang et al. Free Rad. Biol. Med. (2018)
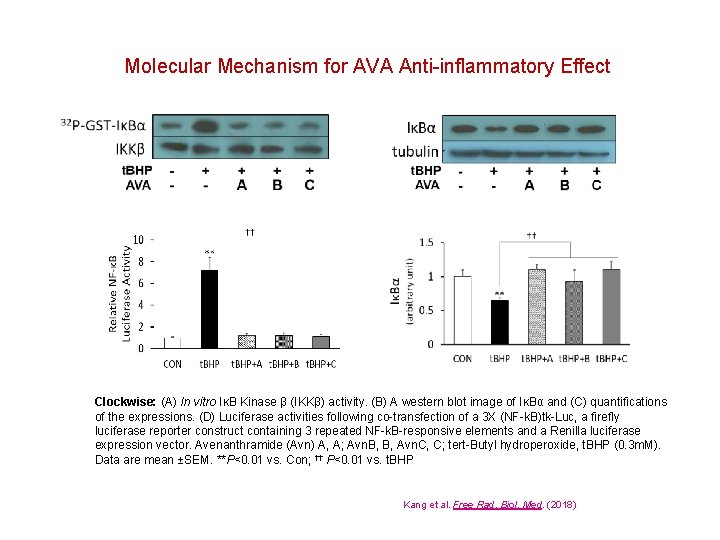
Molecular Mechanism for AVA Anti-inflammatory Effect Clockwise: (A) In vitro IκB Kinase β (IKKβ) activity. (B) A western blot image of IκBα and (C) quantifications of the expressions. (D) Luciferase activities following co-transfection of a 3 X (NF-k. B)tk-Luc, a firefly luciferase reporter construct containing 3 repeated NF-k. B-responsive elements and a Renilla luciferase expression vector. Avenanthramide (Avn) A, A; Avn. B, B, Avn. C, C; tert-Butyl hydroperoxide, t. BHP (0. 3 m. M). Data are mean ±SEM. **P<0. 01 vs. Con; †† P<0. 01 vs. t. BHP Kang et al. Free Rad. Biol. Med. (2018)
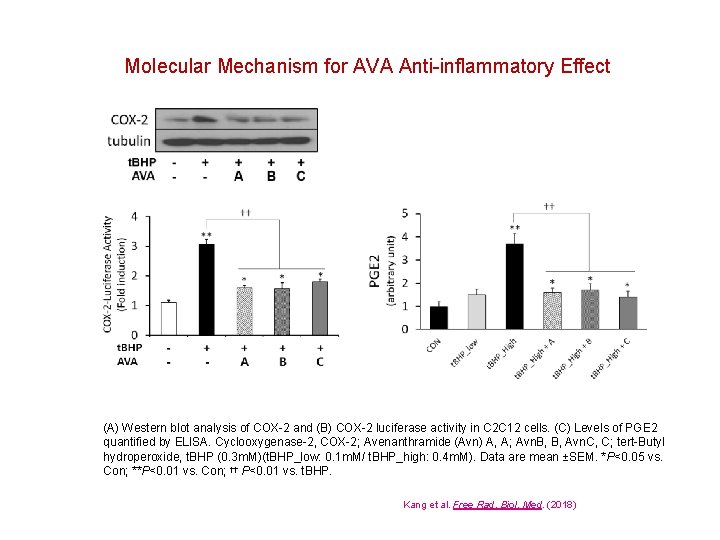
Molecular Mechanism for AVA Anti-inflammatory Effect (A) Western blot analysis of COX-2 and (B) COX-2 luciferase activity in C 2 C 12 cells. (C) Levels of PGE 2 quantified by ELISA. Cyclooxygenase-2, COX-2; Avenanthramide (Avn) A, A; Avn. B, B, Avn. C, C; tert-Butyl hydroperoxide, t. BHP (0. 3 m. M)(t. BHP_low: 0. 1 m. M/ t. BHP_high: 0. 4 m. M). Data are mean ±SEM. *P<0. 05 vs. Con; **P<0. 01 vs. Con; †† P<0. 01 vs. t. BHP. Kang et al. Free Rad. Biol. Med. (2018)
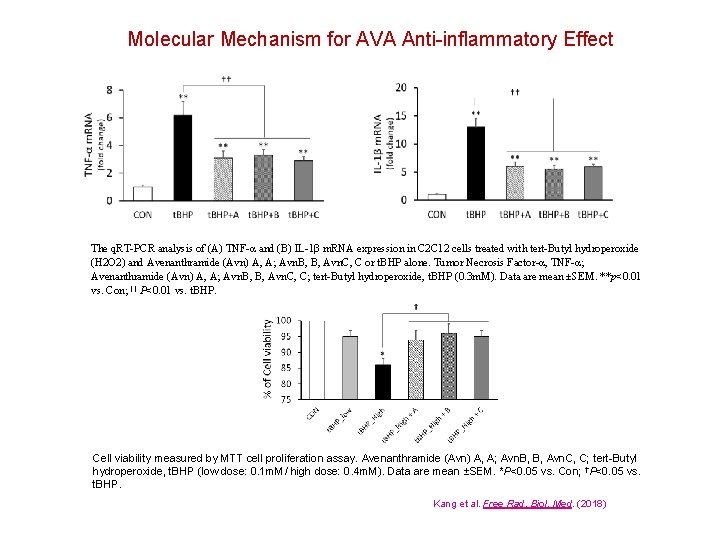
Molecular Mechanism for AVA Anti-inflammatory Effect The q. RT-PCR analysis of (A) TNF-α and (B) IL-1β m. RNA expression in C 2 C 12 cells treated with tert-Butyl hydroperoxide (H 2 O 2) and Avenanthramide (Avn) A, A; Avn. B, B, Avn. C, C or t. BHP alone. Tumor Necrosis Factor-α, TNF-α; Avenanthramide (Avn) A, A; Avn. B, B, Avn. C, C; tert-Butyl hydroperoxide, t. BHP (0. 3 m. M). Data are mean ±SEM. **p<0. 01 vs. Con; †† P<0. 01 vs. t. BHP. Cell viability measured by MTT cell proliferation assay. Avenanthramide (Avn) A, A; Avn. B, B, Avn. C, C; tert-Butyl hydroperoxide, t. BHP (low dose: 0. 1 m. M/ high dose: 0. 4 m. M). Data are mean ±SEM. *P<0. 05 vs. Con; †P<0. 05 vs. t. BHP. Kang et al. Free Rad. Biol. Med. (2018)
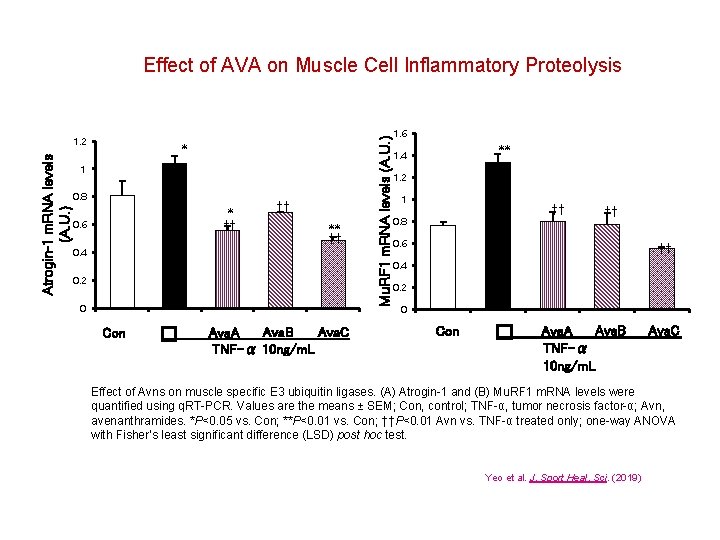
Atrogin-1 m. RNA levels (A. U. ) 1. 2 * 1 0. 8 * †† 0. 6 0. 4 †† ** †† 0. 2 0 Con � Ava. B Ava. C Ava. A TNF-α 10 ng/m. L Mu. RF 1 m. RNA levels (A. U. ) Effect of AVA on Muscle Cell Inflammatory Proteolysis 1. 6 ** 1. 4 1. 2 1 †† 0. 8 †† 0. 6 †† 0. 4 0. 2 0 Con � Ava. B Ava. A TNF-α 10 ng/m. L Ava. C Effect of Avns on muscle specific E 3 ubiquitin ligases. (A) Atrogin-1 and (B) Mu. RF 1 m. RNA levels were quantified using q. RT-PCR. Values are the means ± SEM; Con, control; TNF-α, tumor necrosis factor-α; Avn, avenanthramides. *P<0. 05 vs. Con; **P<0. 01 vs. Con; ††P<0. 01 Avn vs. TNF-α treated only; one-way ANOVA with Fisher’s least significant difference (LSD) post hoc test. Yeo et al. J. Sport Heal. Sci. (2019)
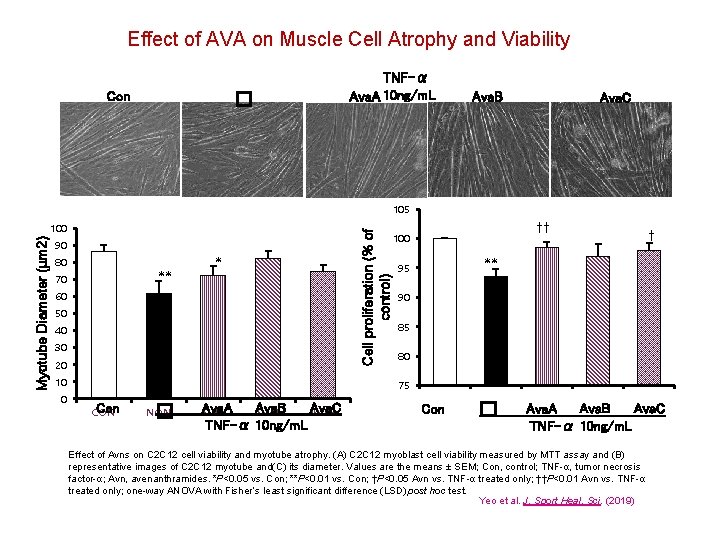
Effect of AVA on Muscle Cell Atrophy and Viability TNF-α Ava. A 10 ng/m. L � Con Ava. B Ava. C 105 Cell proliferation (% of control) Myotube Diameter (µm 2) 100 90 80 ** 70 * 60 50 40 30 20 10 0 †† 100 † ** 95 90 85 80 75 Con CON � NON Ava. A AVAa Ava. B AVAb TNF-α 10 ng/m. L Ava. C AVAc Con � Ava. B Ava. C Ava. A TNF-α 10 ng/m. L Effect of Avns on C 2 C 12 cell viability and myotube atrophy. (A) C 2 C 12 myoblast cell viability measured by MTT assay and (B) representative images of C 2 C 12 myotube and(C) its diameter. Values are the means ± SEM; Con, control; TNF-α, tumor necrosis factor-α; Avn, avenanthramides. *P<0. 05 vs. Con; **P<0. 01 vs. Con; †P<0. 05 Avn vs. TNF-α treated only; ††P<0. 01 Avn vs. TNF-α treated only; one-way ANOVA with Fisher’s least significant difference (LSD) post hoc test. Yeo et al. J. Sport Heal. Sci. (2019)

Antioxidant and Anti-inflammatory Effects Summary • • Oat AVA demonstrate antioxidant and anti-inflammatory effects in vitro and in vivo, both in animal and human research AVA is bioavailable but may be further metabolized to other compounds hours after oral ingestion Inhibition of NFk. B pathway especially allosteric binding of IKK may underline the mechanism of AVA anti-inflammatory effect. Other possibility of AVA bioeffacacy may exist and require further investigation.
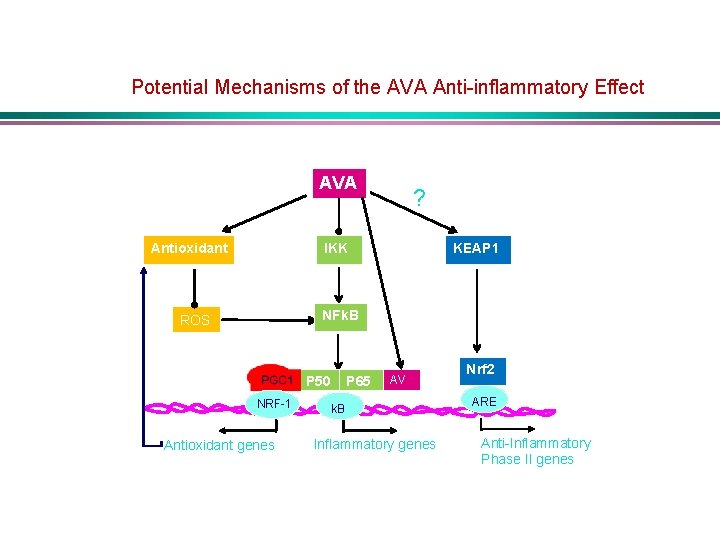
Potential Mechanisms of the AVA Anti-inflammatory Effect AVA Antioxidant ? IKK KEAP 1 NFk. B ROS P 50 NRF-1 Antioxidant genes P 65 AV A k. B Inflammatory genes Nrf 2 ARE Anti-Inflammatory Phase II genes

ICPH 2019, Kobe, Japan December 1, 2019 Acknowledgment Ryan Koenig, Ph. D Jonathan Dickman, Ph. D Kathleen O’Moore, M. S. Chounghun Kang, Ph. D. David Peterson, Ph. D Tianou Zhang, MD, MS Mitch Wise, Ph. D Grants United State Department of Agriculture (USDA) USDA Cereal Research Lab, Madison, Wisconsin Pepsi. Co Nutrition Department (Quaker Oats Co. ) Ceapro Inc. (AB, Canada) Virginia Horn Henry Foundation
- Slides: 28