ICBS Master Panels for Kit Evaluation HCV HBV

ICBS Master Panels for Kit Evaluation: HCV, HBV, and HIV (WWW. ICBS-WEB. ORG) Howard A. Fields, Ph. D. Susan Diaz, MPH Division of Viral Hepatitis Centers for Disease Control and Prevention

Objective of Master Panels § Identify, validate, and make available highquality affordable tests for HBV, HCV, and HIV because cost is one of the major impediments to universal screening. § Use as a model for BTEP project to produce Master Panels with Dr. Alexander Kanev, Vector currently in year 3.

HCV Master Panels positive and negative

HCV Master Panel: Diversity • Country of origin • Genotype • Subtype • RIBA 3. 0 banding patterns
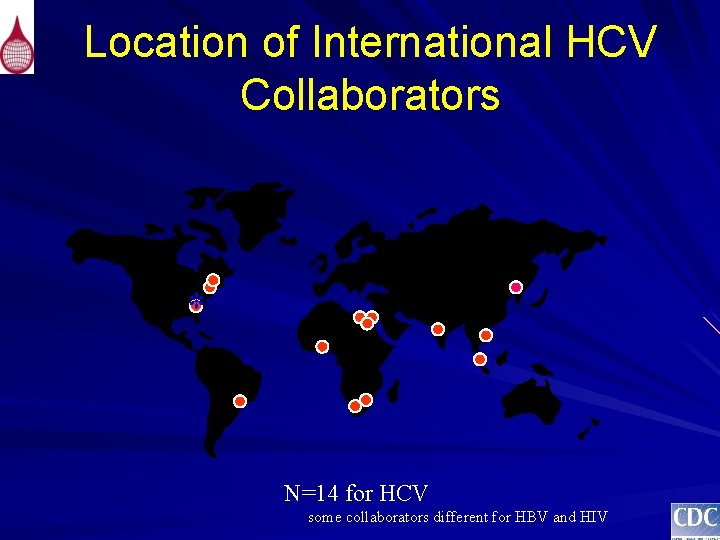
Location of International HCV Collaborators * N=14 for HCV some collaborators different for HBV and HIV

Methods to Characterize Panel Members • Screening Tests Ø Anti-HCV by Ortho 3. 0; positives repeated in duplicate Ø RIBA 3. 0 for HCV by Chiron • Confirmatory Tests • • Quantitative PCR Ø Roche Amplicor for HCV RNA Genotyping / Subtyping by direct DNA Sequencing Ø CDC: ABI Prism 3100 Capillary Sequencer (NS 5 b default to 5’-UTR); phylogenetic analysis Ø Visible Genetics, Inc. Clip Sequencing Reaction for HCV (5’UTR/NS 5 b) percent homology

Collections • HCV: • HBV: • HIV: • COINFECTION: • NEGATIVES: 1007 450 200 62 450 ØTotal Units Received: 2169

HCV Positive Units Description of Selectable Units • • Serologically confirmed as positive (exclude indeterminate specimens) Concordant genotype by direct DNA sequencing from two laboratories Minimum volume 100 ml Additional selection criteria based upon diversity By geographic regions Ø By genotype/subtype Ø By RIBA banding patterns Ø

Diversity by Genotype among Selected Units • Among 1007 HCV IR units received and characterized, 539 units met the definition of selectable (54%) Ø Ø Ø Genotype 1 Genotype 2 Genotype 3 Genotype 4 Genotype 5 Genotype 6 (n=296) (n=26) (n=42) (n=129) (n=3) (n=43)
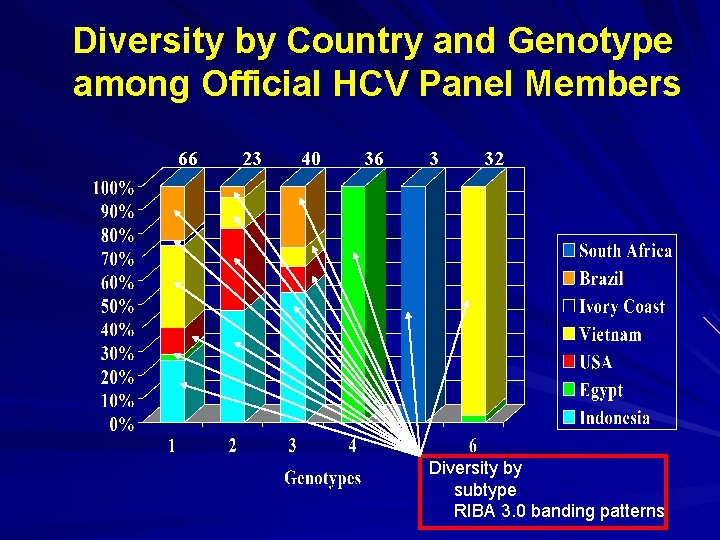
Diversity by Country and Genotype among Official HCV Panel Members 66 23 40 36 3 32 Diversity by subtype RIBA 3. 0 banding patterns
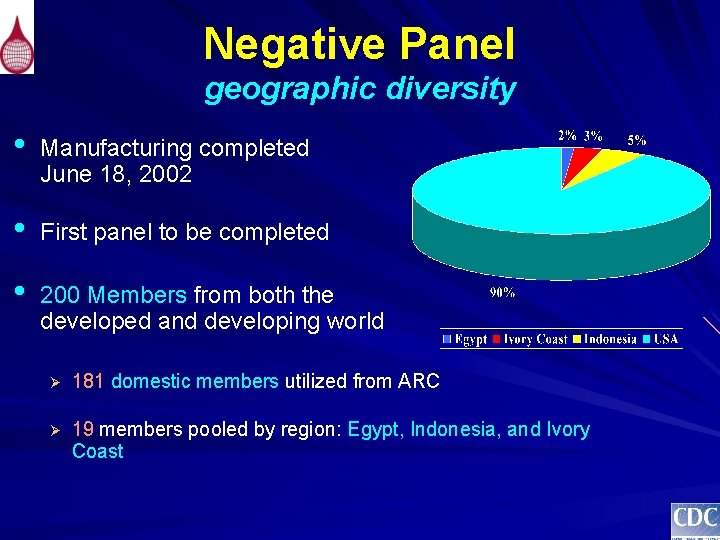
Negative Panel geographic diversity • Manufacturing completed June 18, 2002 • First panel to be completed • 200 Members from both the developed and developing world Ø 181 domestic members utilized from ARC Ø 19 members pooled by region: Egypt, Indonesia, and Ivory Coast

Origin of Kits Sent to PEI China 13 Russia 9 India 7 South Korea 5 EU 4 Costa Rica 1 Indonesia 1 Uzbekistan 1 Brazil 1 Argentina 1 USA (not FDA approved) 1 Total = 44

Russian Kits Sent to PEI for Evaluation Biotechnologitzeskaja Kompanija Bioservis (No. 23, Hepascreen) IMBIO (No. 24, Anti-HCV ELISA) Medizinische Biologische Union (No. 27, HCV-DCM) Union–Diagnostisches-System (No. 25, Anti-HCV ELISA) Vector-Best (No. 29, Recombi. Best anti-HCV-Strip, OPD) Received, but Not Evaluated as of August 23, 2004 Ø Institut Pastera Sankt-Peterburg (Anti-HCV ELISA-G) Ø NPP Akwapast (Immunoenzyme Test-System for Detection of Anti-HCV) Ø Radiopreparat (Anti-HCV) Ø Vector-Best (Rocombi. Best anti-HCV-Strip, TMB)

Specimens ICBS HCV Master Panel (n=200) ICBS negative Master Panel (n=181 and 200) Seroconversion Panel (commercial) Ø core only, genotype 2 b Ø NS 3 only Ø Core & NS 3, genotype 1 a Ø Mean day delay
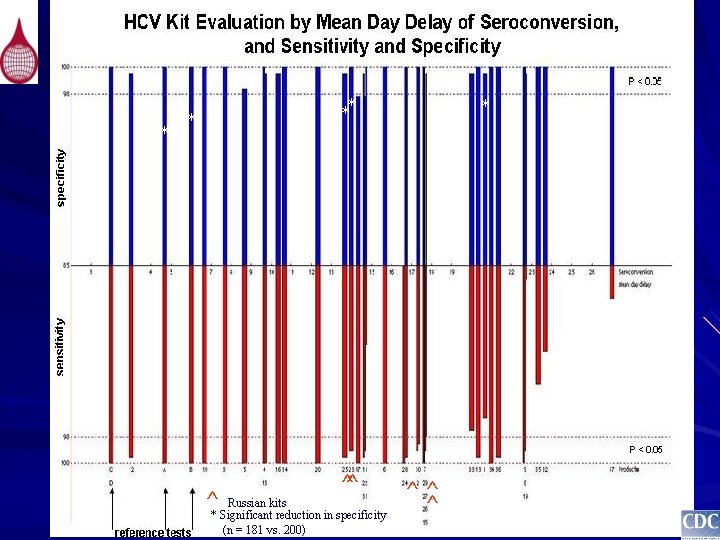
* ** * ^ ^^ Russian kits * Significant reduction in specificity (n = 181 vs. 200) * ^^ ^
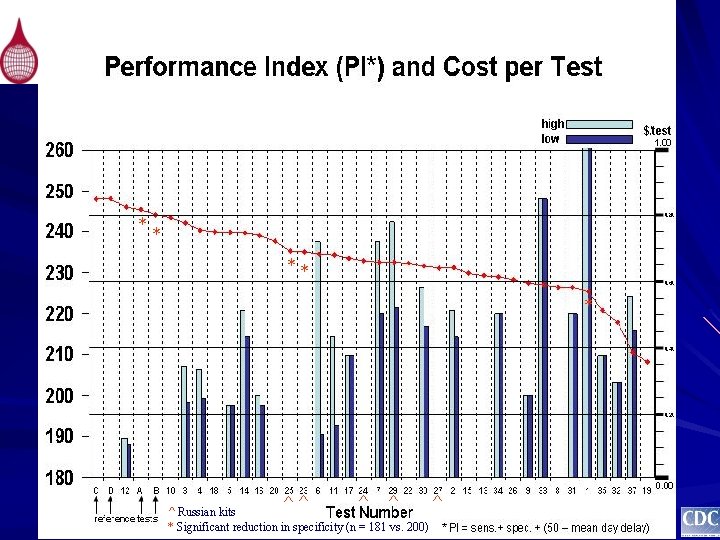
* * ** * ^^ ^ Russian kits * Significant reduction in specificity (n = 181 vs. 200) ^

PI of Russian Kits Evaluated N=181 PI N=200 Union–Diagnostisches-System 235. 28 232. 83* Biotech Kompanija Bioservis IMBIO Vector-Best Medizinische Biolog Union 235. 00 232. 83 231. 78 231. 28 231. 00 232. 33 229. 83 229. 30 * Statistical significant reduction in specificity (n = 181 vs. 200) *

Conclusions (1) ICBS HCV Negative Master Panel manufactured June, 2002. Ø N = 181 US specimens Ø N = 19 specimens from the developing world yielded weakly screening test positive when tested with some assays licensed in the EU. Some specimens also tested positive/indeterminate with some of the EU license supplemental tests. Ø N = 200 specimens confirmed as true negative at CDC (Ortho 3. 0, Chiron RIBA 3. 0, and Roche HCV Amplicor)

Conclusions (2) ICBS HCV Positive Master Panel manufactured September, 2002 Ø Diversity based on geographic origin, genotype, subtype (concordance by DNA sequencing), and RIBA 3. 0 banding patterns Ø All confirmed as true positive by screening (Ortho 3. 0), supplemental testing (RIBA 3. 0), and positive PCR (Roche HCV Amplicor)

Conclusion (3) Russian kits evaluated by PEI 2003 – 2004 Ø PI less than kits licensed in the US and EU and several other kits manufactured in resource-challenged regions of the world Ø Highest PI manufactured by Union–Diagnostisches-System probably equal to the kit manufactured by Biotech Kompanija Bioservis based on 181 US specimens Ø When 19 specimens from the developing world was added (n=200), the specificity of all 5 Russian kits decreased; however, only kits Union–Diagnostisches-System and Biotechnologitzeskaja Kompanija Bioservis decreased to a statistical significant level. Ø Despite this decrease in specificity, the kit manufactured by Union–Diagnostisches-System continued to have the highest PI value among the Russian kits evaluated.
- Slides: 20