IAS 2017 IASconference Evaluation of tolerability pharmacokinetics and

#IAS 2017 | @IAS_conference Evaluation of tolerability, pharmacokinetics, and anti-retroviral activity of MK-8591 in treatment-naïve HIV-1 -infected adult subjects • MK-8591 (EFd. A) Background – – • Nucleoside reverse transcriptase translocation inhibitor (NRTTI) with a novel mechanism of action. High potency and a high barrier to resistance in vitro. Long half-life in preclinical species and in healthy human subjects. Generally well-tolerated in healthy human subjects to date. MK-8591 Single Dose Monotherapy Study Design – Open-label, single-dose, multiple panel study. – Doses administered: 0. 5 mg, 1 mg, 2 mg, 10 mg, 30 mg. Administered once (orally) per subject. – Subjects strongly encouraged to initiate standard of care therapy after 10 days (30 mg, 10 mg) or 7 days (2 mg, 1 mg, 0. 5 mg). • Study Objectives – Assess tolerability of MK-8591 in HIV-1 -infected subjects. – Assess PK of MK-8591, as parent in plasma and as triphosphate (MK-8591 -TP) in peripheral blood mononuclear cells (PBMCs) in HIV-1 -infected individuals. – Determine the degree of viral load suppression post treatment • Determine the lowest dose at which VL decline is at least 0. 5 log 10 from placebo-corrected baseline. – Examine the PK/PD relationship between MK-8591 -TP and viral load decline.

#IAS 2017 | @IAS_conference MK-8591 generally well-tolerated as a one-time dose in HIV-1 infected adult subjects • AEs in 29/30 subjects, and treatment-emergent (after dosing) AEs in 27/30 subjects. • No SAEs and no discontinuations due to an AE. All AEs were mild or moderate in severity. • No relationship between dose and frequency or severity of AEs. Subjects (#) 30 SAEs None AEs (#) 63 (29 subjects) TEAEs (#) ab Most common MK -8591 TEAEs c 60 (27 subjects) Headache (n=10) Common cold (n=4) Diarrhea (n=3) Vomiting (n=3) TEAE: Treatment-emergent adverse event NCS: Not clinically significant a. All adverse events were mild to moderate in intensity and resolved by study completion. b. TEAEs do not include AEs that occurred prior to dosing. c. TEAEs reported by more than 2 subjects following treatment. d. MK-8591 related TEAEs reported by more than one subject. MK-8591 Related TEAEsd Headache (n=9) Diarrhea (n=2) Eczema (n=2) Vital Signs NCS ECG Parameter Value Changes NCS Lab Safety Findings NCS
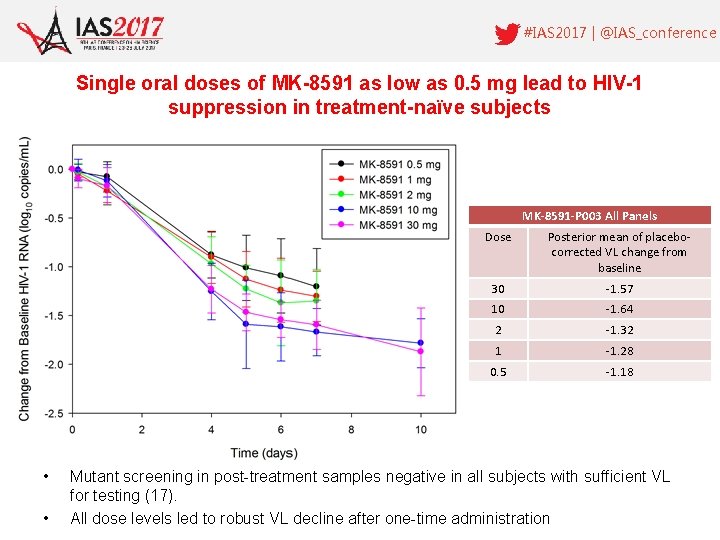
#IAS 2017 | @IAS_conference Single oral doses of MK-8591 as low as 0. 5 mg lead to HIV-1 suppression in treatment-naïve subjects MK-8591 -P 003 All Panels • • Dose Posterior mean of placebocorrected VL change from baseline 30 -1. 57 10 -1. 64 2 -1. 32 1 -1. 28 0. 5 -1. 18 Mutant screening in post-treatment samples negative in all subjects with sufficient VL for testing (17). All dose levels led to robust VL decline after one-time administration
- Slides: 3