IAcids and Bases Prem Sattsangi Copyright 2008 2

I-Acids and Bases Prem Sattsangi Copyright 2008

#2 Introduction to Acids and Bases Chapter - 8 • • • Individual work Section A Facts and Questions. Section C Acid-base titration. M of Unknown HCl U. K. #_____. Section D Calibration of microburet. • Section E Analysis of the egg shell.

#3 Getting Ready • • • Waste Beaker 50 m. L. 24 well tray with solutions. 12 well tray Microstirrer Distilled water bottle.

#4 Stock Solutions • 2. 00 M HCl • 0. 10 M HCl • BTB 0. 1 M Na. OH ~1 M Na. OH • Crushed Egg shell • 95% Ethanol

#5 Acid-Base Titration • Equivalence point = The first blue well • Add 1 drop Na. OH to well #1, stir, wipe the stirrer clean… • SERIAL TITRATION: #Drs Na. OH = well# • Color YY B B • 0. 1 M Na. OH 1 2 3 4 5 6 7 8 9 10 11 12 • 0. 1 M HCl 55555 5 • BTB 11111 1 • Well# 1 2 3 4 5 6 7 8 9 10 11 12
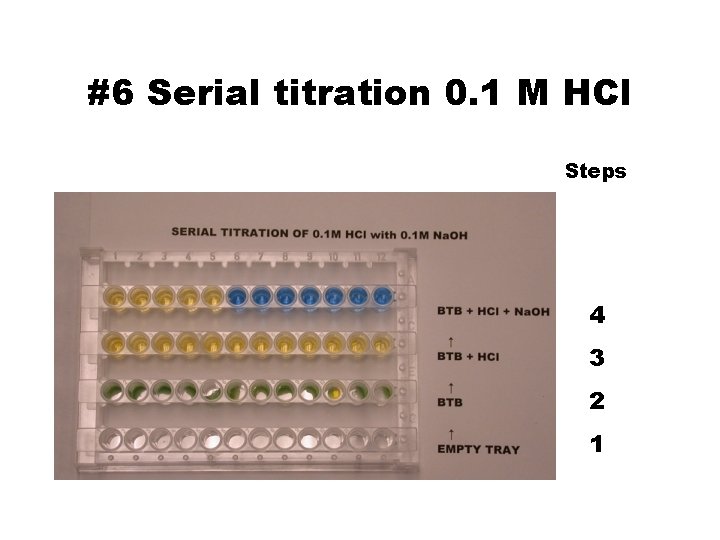
#6 Serial titration 0. 1 M HCl Steps 4 3 2 1

#7 Calculation (drs. In place of m. L) • Na. OH(aq) + HCl(aq) Na. Cl(aq) + H 2 O(l) • ______ drops X 0. 1 M Na. OH • = 5 Drops X 0. 1 M HCl • Discuss with teacher if: • Your end point is not in the fifth well.
![#8 Calibration of the Micropipet [Uniform Size Drops] • • Fill MP with DW, #8 Calibration of the Micropipet [Uniform Size Drops] • • Fill MP with DW,](http://slidetodoc.com/presentation_image_h2/6bc76d3efa1299d1be6f650d4c7700bf/image-8.jpg)
#8 Calibration of the Micropipet [Uniform Size Drops] • • Fill MP with DW, Hold vertical ~1” from the tray. Squeeze gently 1 drop. Record weight up to 0. 0000 g. Tare the balance. Repeat, record 10 readings. Discard the readings that are too much off. You MUST have 10 good readings.

#9 Calculation of Mean and Standard Deviation • Use TI 30 to calculate Mean and Std. Dev. • E. g. if the date is 9, 4, 4, 5, • Mean = 5. 5 Std. Dev. = 2. 38…. • Enter the data as follows. 9, +, 4, +, 5, +, Mean: 2 nd xn-1 = 2. 38; Std. D. 2 nd X(bar) • Write ST. #, Wt of 1 drop (g) on the pipet. • Apply a tape over it.

#10 Getting Ready for U. K. • U. K. #___ Check. It should correspond to your station #. • M ranges from 0. 2 to 0. 4 M. • 12 well tray • 2 drs U. K. + 1 dr BTB • 24 well tray • 20 drs U. K. + 2 drs BTB

#11 Titration of U. K. • • • Empty the U. K. micropipet in its well. Wash 2 -3 times with DW. Rinse 2 -3 times with 0. 1 M Na. OH. Fill ~half way with 0. 1 M Na. OH. Titrate: Keep micro stirrer in tray till the end point is reached.

#12 Weighing of the Egg Shell (Balance drifts) • Check balance reading, 0. 0000 • TARE if needed • Weigh a plastic tray on Analytical balance. • Add slightly less than a level spoon. • (~0. 05 g egg shell) • Record weight up to 4 decimal places.

#13 Weighing Observations OBSERVATIONS: Weight Wt of Plastic Tray = 0. 5153 g • Wt of Tray + egg shell =*10. 5646 After transfer • Weight of empty tray. = *0. 5161 g • Egg shell wt. =*10. 5646 - *0. 5161 Difference in weights: = 0. 0485 g

#14 Reaction: Egg shell + HCl • • Running the reaction: Ca. CO 3 + 2 HCl Ca. Cl 2 + H 2 O + CO 2 [1 mol Ca. CO 3 reacts with 2 mol HCl] Add 1 -2 drops of 95% ethanol to egg shell. • Use calibrated micropipet to add 30 drs of 2. 00 M HCl in the well with egg shell.

#15 Caution: Hot Water • Hold pipet with a folded paper towel. • Take ~1 cm hot water 2 -3 times to condition the glass pipet. • Add hot water in adjoining well. • Leave a microstirrer in the well and stir every other minute. • Allow the reaction to proceed (~10 -15 minutes).

#16. Result Reporting • Molar concentration of Na. OH is to be copied from the Board. • Calculate the result carefully. • Check before submitting.
- Slides: 16