IABP vs IMPELLA in Cardiogenic Shock Christian Spaulding

IABP vs IMPELLA in Cardiogenic Shock Christian Spaulding, MD, Ph. D Cardiology Department European Hospital Georges Pompidou Paris Descartes University INSERM U 970 Paris, France

Conflicts of interest Grant/Research Support: CERC French Ministry of Health Consulting Fees/Honoraria: Abiomed Medtronic Terumo Medical Company Zoll Stentys Xeltis

Cardiogenic shock 4 to 8% of AMI cases High mortality: 40 to 70% The definition of cardiogenic shock is clinical:
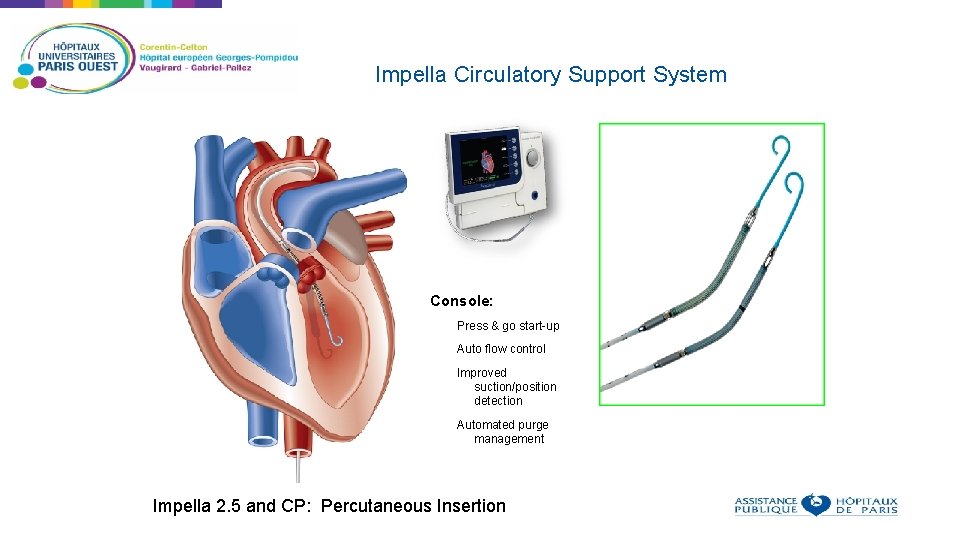
Impella Circulatory Support System Console: Press & go start-up Auto flow control Improved suction/position detection Automated purge management Impella 2. 5 and CP: Percutaneous Insertion
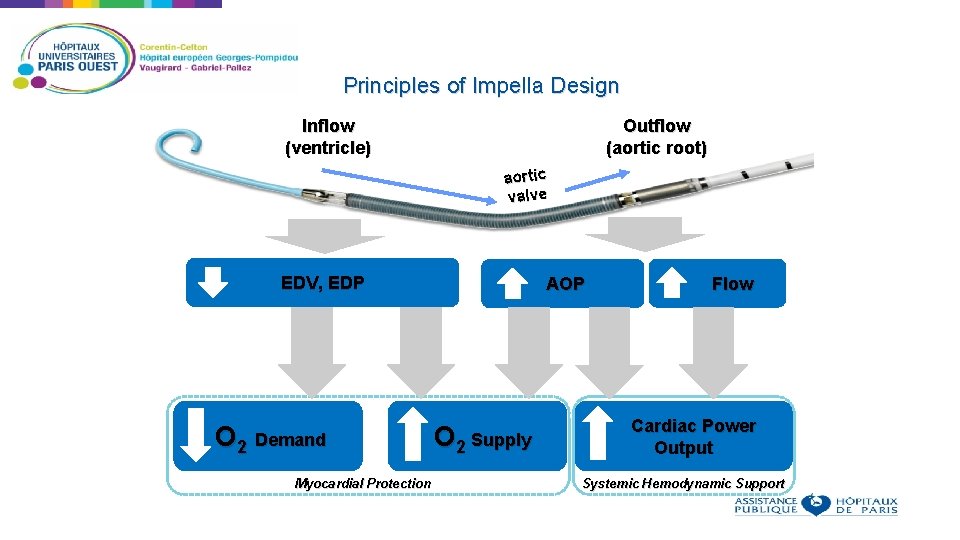
Principles of Impella Design Inflow (ventricle) Outflow (aortic root) aortic valve EDV, EDP O 2 Demand AOP O 2 Supply Myocardial Protection Flow Cardiac Power Output Systemic Hemodynamic Support

Impella: Advantages Easy and fast to implant Immediate hemodynamic support Data ?

Circulation. 2019; 139: 1249– 1258. Circulation. 2019; 139: 1259– 1261

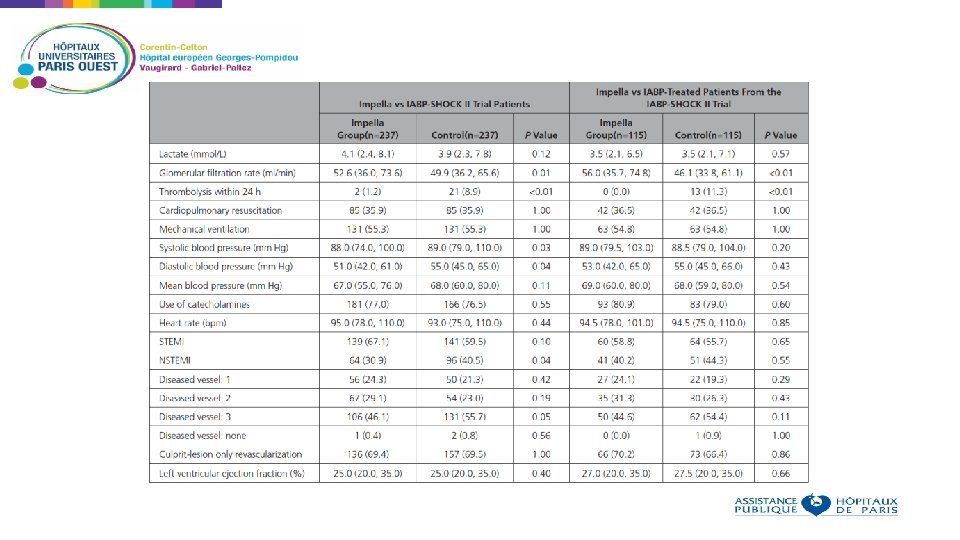
Non randomized analysis, using propensity score Aim: compare outcomes in patients receiving IABP or IMPELLA for cardiogenic shock Patients who received Impella in 13 European centers were compared to patients who received IABP in the IABP SHOCK II trial Using the IABP SHOCK II trial inclusion and exclusion criteria, 372 patients were identified in the registry and matched to the 600 patients of the IABP SHOCK II trial Baseline criteria used as matching parameters: age, sex, mechanical ventilation, ejection fraction, prior cardiopulmonary resuscitation, and lactate 237 patients treated with an Impella matched to 237 patients from the IABP-SHOCK II trial Primary end point: 30 -day all-cause mortality.
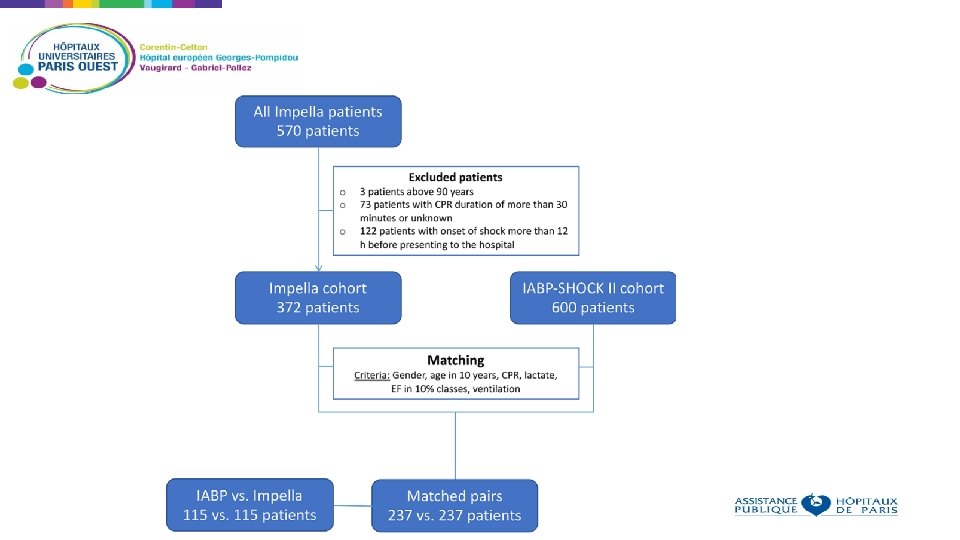
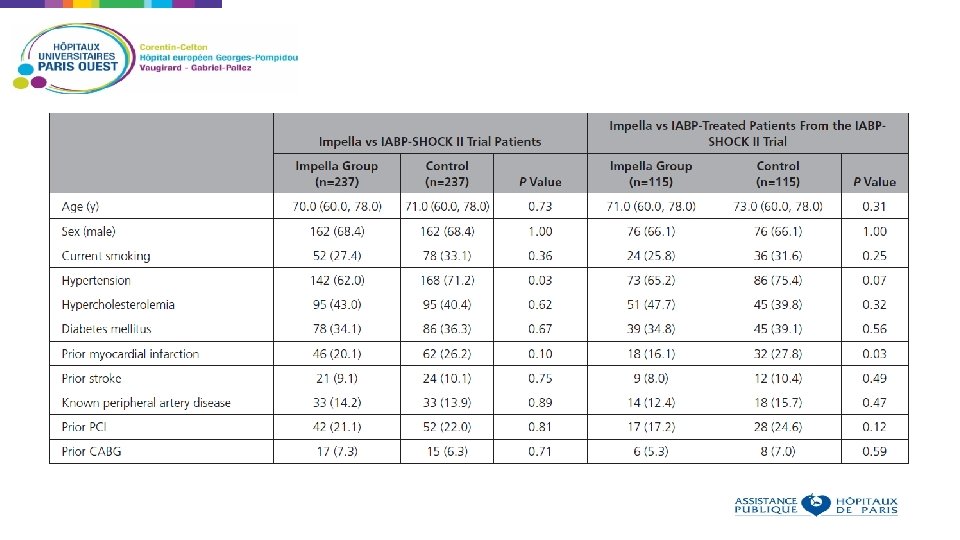
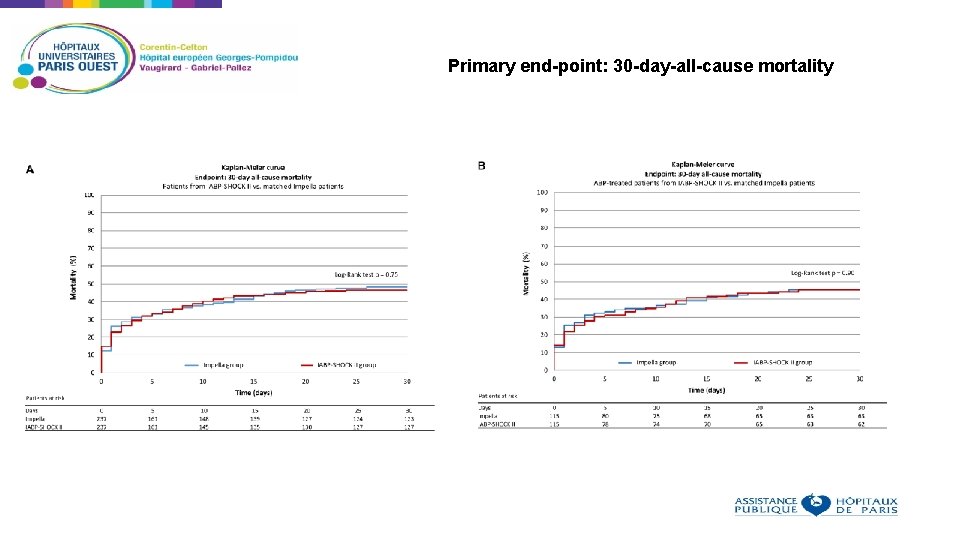
Primary end-point: 30 -day-all-cause mortality
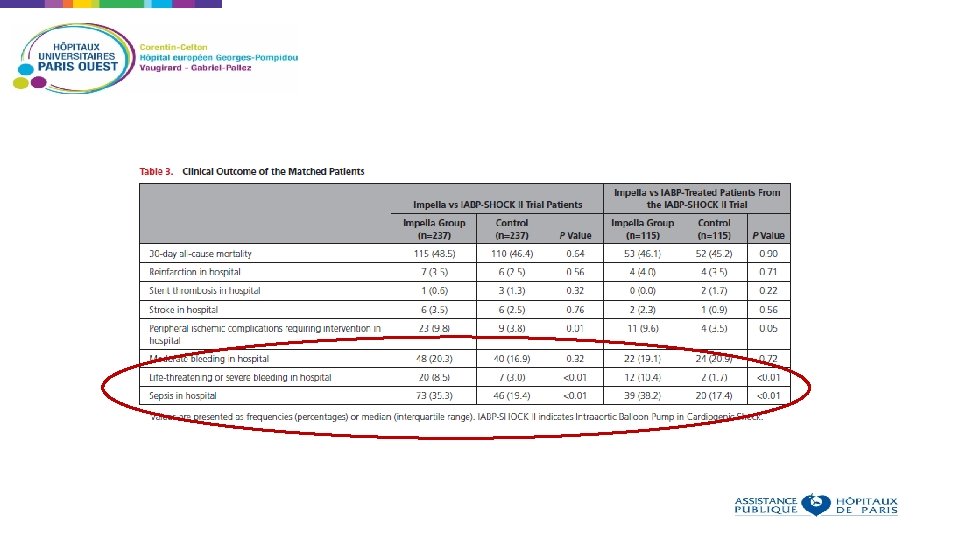

Limitations Non randomized study Insertion of Impella was left to the discretion of the physician « Experienced » centers, however no data on the number of procedures performed before the registry 74 of 237 patients in the Impella group were treated with the Impella 2. 5 However: - Multiple sites with pre-established protocols for Impella insertion - Large study - Similar outcomes compared to previous studies with Impella

Conclusions In this retrospective analysis of patients with AMI related CS, the use of an Impella device was not associated with lower 30 -day mortality compared with matched patients from the IABP-SHOCK II trial treated with an IABP or medical therapy To further evaluate this, a large randomized trial is warranted to determine the effect of the Impella device on outcome in patients with AMI related CS
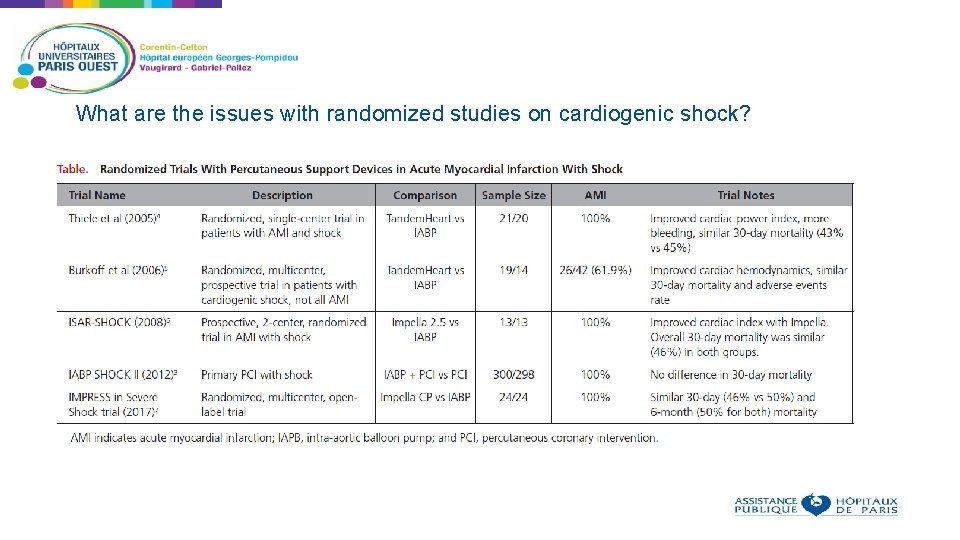
What are the issues with randomized studies on cardiogenic shock?
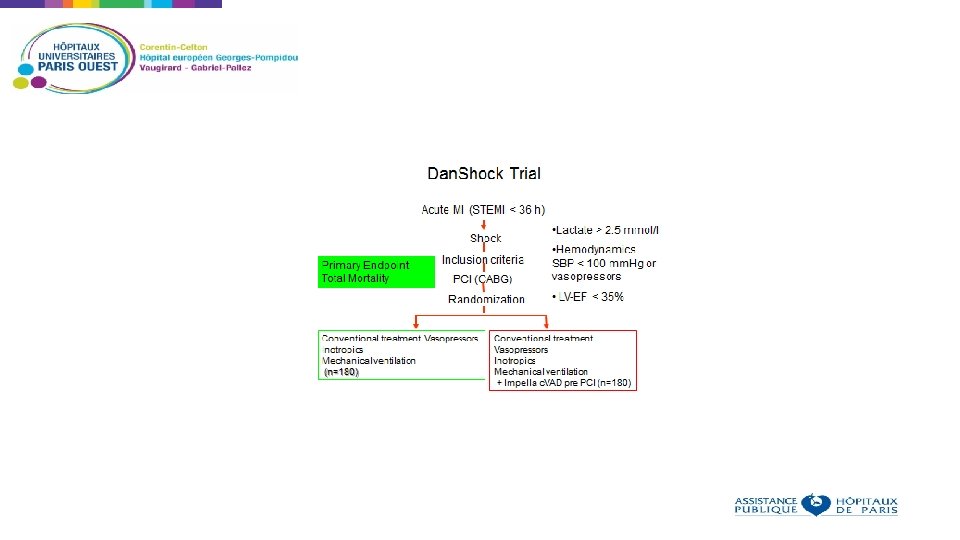

What are the issues with randomized studies on cardiogenic shock? Rate of cardiogenic shock is declining Inclusion of patients in trials on cardiogenic shock and cardiac assist devices is difficult due to the belief of physicians on the effectiveness of these devices and reimbursement issues
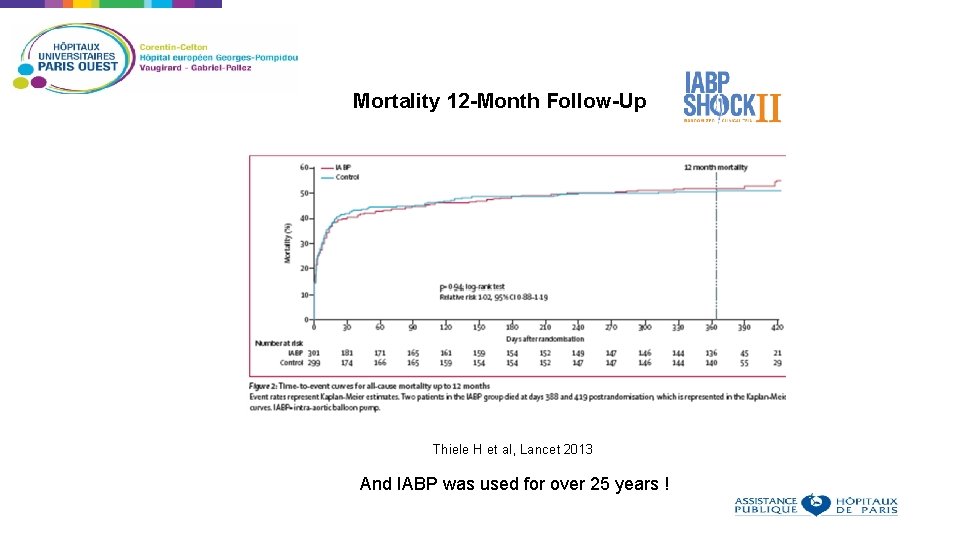
Mortality 12 -Month Follow-Up 45 Mortality (%) 40 35 30 25 20 15 10 5 0 0 30 60 90 120 150 180 Thiele H et al, Lancet 2013 And IABP was used for over 25 years !

A trial on Impella in Cardiogenic Shock ? Large (at least 600 patients) Multi-center Performed in countries where IMPELLA is not reimbursed On « pre-shock » patients With early insertion before PCI
- Slides: 20