I nuovi antibiotici per la chemioterapia di precisione

I nuovi antibiotici per la chemioterapia di precisione nella terapia delle infezioni da gramnegativi MDR Francesco Menichetti, MD Professor of Infectious Diseases, University of Pisa Director, Infectious Diseases Department, Cisanello Hospital, Pisa President, Italian Group for Antimicrobial Stewardship (GISA) www. antimicrobialstewardship. net 1

Disclosures 2017 -2018 • Advisory Board: Angelini, MSD, Nordic Pharma • Speaker/chairman: Angelini, Astellas, Basilea, MSD, Pfizer • Events Sponsorship: Astellas, Gilead, MSD, BMS, Jansenn, Vii. V, Bio. Merieux, Biotest, Becton. Dickinson, Nordic Pharma, Pfizer, Shionogi • Ongoing research protocol: Angelini, Astellas, Cidara, MSD, Shionogi, Theravance

MDR gram-negative bacilli, resistance mechanisms & therapeutic options Pop-Vicas A & Opal SM Virulence 2013
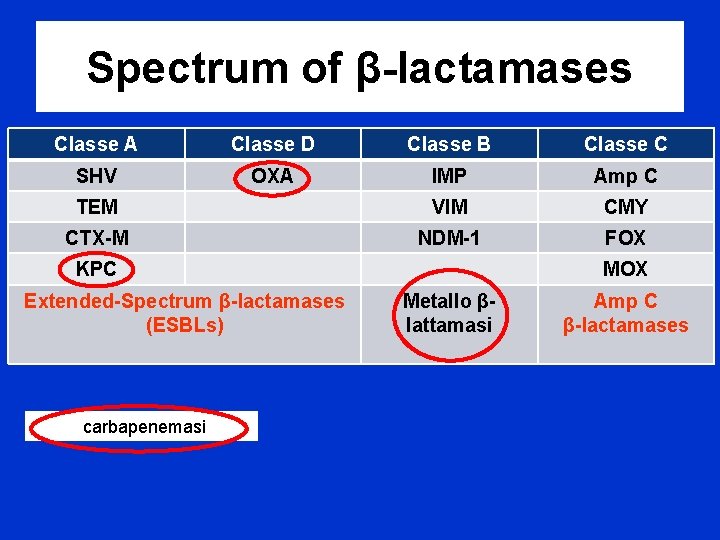
Spectrum of β-lactamases Classe A Classe D Classe B Classe C SHV OXA IMP Amp C TEM VIM CMY CTX-M NDM-1 FOX KPC Extended-Spectrum β-lactamases (ESBLs) carbapenemasi MOX Metallo βlattamasi Amp C β-lactamases
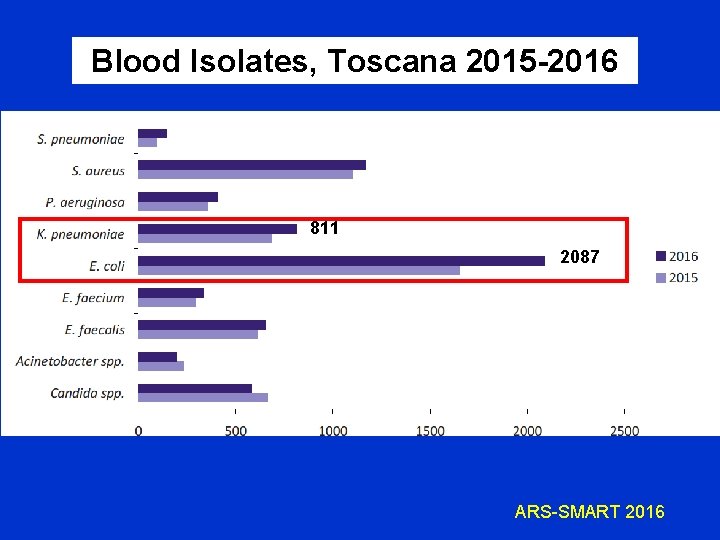
Blood Isolates, Toscana 2015 -2016 811 2087 ARS-SMART 2016

E. coli ESBL, 2016

K. pneumoniae ESBL, 2016

Treatment for ESBL infections The Sanford Guide 2018 ESBL producing strains suspected by their resistance in vitro to Piperacillin, Cefotaxime, Ceftriaxone, Ceftazidime, Aztreonam Life-threatening: Carbapenems Mild/moderate, non bacteremic, low-inoculum, low MIC: Cefepime Piperacillin/tazobactam Limited clinical data: Ceftolozane/tazobactam Ceftazidime/avibactam UTI uncomplicated: Fosfomycin Nitrofurantoin

The MERINO Trial: piperacillin-tazobactam versus meropenem for the definitive treatment of bloodstream infections caused by third-generation cephalosporin non-susceptible Escherichia coli or Klebsiella spp • Adult pts from 32 sites in 9 countries (Singapore, Australia and Turkey). • Pts included 378 (February 2014 to July 2017). • HAI: > 50% of the infections in the study group. • Urosepsis: 60. 9% of the infections • E. coli responsible of 86. 5% of the cases Harris P et al. ECCMID 2018 oral presentation

MERINO trial: preliminary results Harris P et al. ECCMID 2018 oral presentation

The MERINO Trial: piperacillin-tazobactam versus meropenem for the definitive treatment of bloodstream infections caused by third-generation cephalosporin nonsusceptible Escherichia coli or Klebsiella spp • No difference between the two groups regarding subsequent infections of drug-resistant bacteria or C. difficile, but difference in mortality rate was significant. • Twenty-three patients (12. 3%) treated with pip-tazo died by the 30 -day mark compared with seven patients (3. 7%) who had been treated with meropenem. • Pip-tazo was significantly less effective than MPN for potentially fatal BSI caused by ceftriaxone-resistant E. coli and K. pneumoniae and should be avoided Harris P et al. ECCMID 2018 oral presentation

The domino effect of the ESBL pandemic Dissemination of Enterics producing ESBLs Carbapenem-sparing regimens ? Carbapenem Overuse Dissemination of Carbapenem R Gram-negatives Idea: Gianni Rossolini

• 335 pts included; 249 received empiric CPSs and 86 OADs. • Most frequent OADs were AGs (43 pts) and FQs (20 pts). • Empiric therapy with OADs was not associated with increased mortality. • OADs were neither associated with 14 -day clinical failure nor increased length of hospital stay. • This information allows more options to be considered for empiric therapy, at least for some patients, depending on ocal susceptibility patterns of ESBL-E.

• Early administration of OADs for BSI due to ESBL-E does not seem to compromise outcome in comparison with carbapenems, and might be an option for empiric regimens for many patients, depending on local susceptibility patterns. • This may be particularly applied to the use of aminoglycosides in urinary tract sepsis potentially caused by ESBL-E and would justify the design of a randomized trial; • However, until more data are available, we would still recommend considering carbapenems (or BL-BLI) for patients at risk of ESBL-E presenting with septic shock or with a non–urinary tract source of sepsis.

• Ceftolozane demonstrates good activity against Enterobacteriaceae but its activity is limited against ESBLs. • Tazobactam is a potent, irreversible inhibitor of most ESBLs. • The MIC 50/MIC 90 of this agent for ESBL-producing E. coli are 0. 5/4 μg/m. L and for K. pneumoniae 4/>32 μg/m. L • Differences in MIC distributions may be reflective of discrepancies in ESBL genes present. • The bla. CTX-M genes predominate in E. coli, whereas there is often a preponderance of bla. TEM/SHV in K. pneumoniae, with variations in local epidemiology.

• Ceftolozane-tazobactam (in combination with metronidazole) was compared to meropenem for the treatment of complicated intra-abdominal infections in phase 2 and phase 3 trials that included 4 and 50 people, respectively, with ESBL-producing Enterobacteriaceae. • Although the limited number of ESBLs precluded a robust analysis, this compound performed similarly against ESBL-producing and non-ESBL producing isolates.
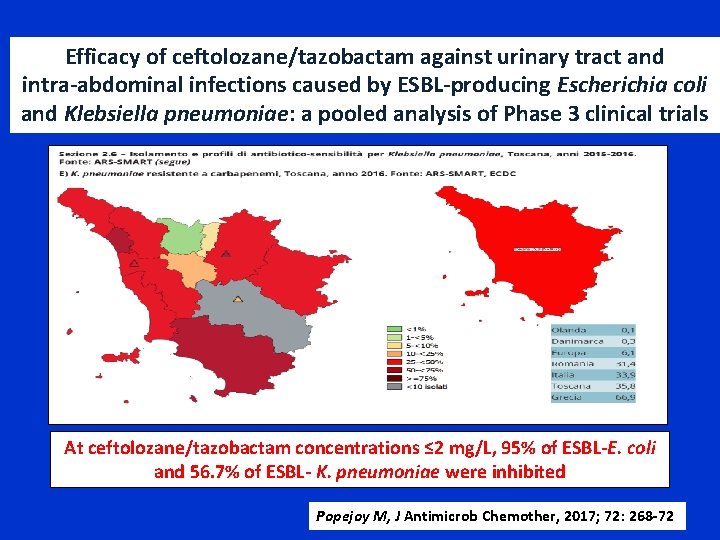
Efficacy of ceftolozane/tazobactam against urinary tract and intra-abdominal infections caused by ESBL-producing Escherichia coli and Klebsiella pneumoniae: a pooled analysis of Phase 3 clinical trials At ceftolozane/tazobactam concentrations ≤ 2 mg/L, 95% of ESBL-E. coli and 56. 7% of ESBL- K. pneumoniae were inhibited Popejoy M, J Antimicrob Chemother, 2017; 72: 268 -72

• Ceftazidime-avibactam is usually more active in vitro against ESBL producers than ceftolozane-tazobactam. • The MIC 50/MIC 90 for ESBL-producing E. coli are 0. 12/0. 25 μg/m. L and for K. pneumoniae 0. 5/1 μg/m. L • Phase 2/3 studies compared ceftazidime-avibactam (plus metronidazole) vs meropenem for intra-abdominal infections, but did not specifically compare outcomes of ESBL-confirmed pathogens. • Data from a phase 3 study comparing ceftazidimeavibactam and doripenem in UTIs showed similar microbiological response for ceftazidime-resistant Enterobacteriaceae, most of which were ESBL producers

Carbapenem-resistant enterobacteriaceae (CRE): definition • CRE was defined as an Enterobacteriaceae isolate demonstrating resistance to any carbapenem (ertapenem, meropenem, imipenem, and/or doripenem), based upon antimicrobial susceptibility testing (AST). • Carbapenem resistance was defined as an Ertapenem MIC ≥ 2 mcg/ml and MPN and/or IP MIC ≥ 4 mcg/ml. Clinical Infectious Diseases Advance Access published November 9, 2016

Meropenem use for CRE: MIC, please • For Meropenem MICs of ≤ 4 mcg/ml: standardinfusion of 2 grams t. i. d. considered active, based on existing p. K/p. D studies. • For Meropenem MICs of 8 mcg/ml: extendedinfusion (2 grams t. i. d. over at least 3 hours) potentially active. • Modeling of extended-infusion strategies suggest that reasonable target attainment can be anticipated with meropenem MICs of up to 8 mcg/ml, and possibly 16 mcg/ml Clinical Infectious Diseases Advance Access published November 9, 2016
K. pneumoniae CR, 2015 - 2016

The double domino effect of the CRE epidemic Dissemination of CRE Further Carbapenem Overuse Colistin overuse Dissemination of Colistin R strains
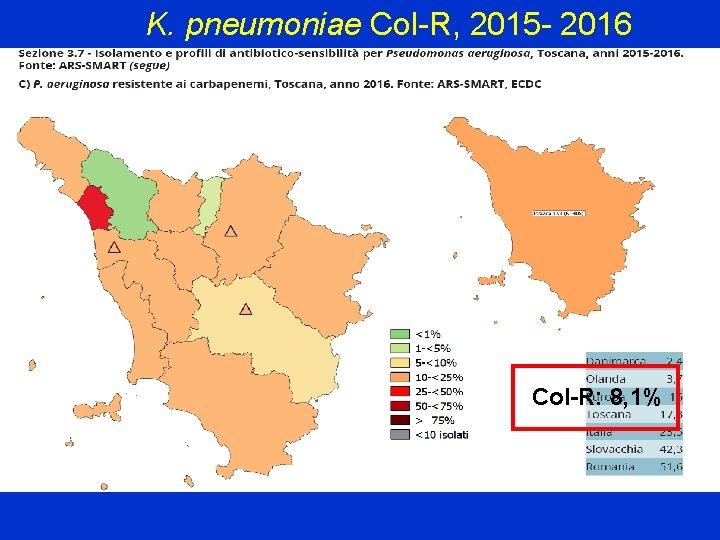
K. pneumoniae Col-R, 2015 - 2016 Col-R: 8, 1%
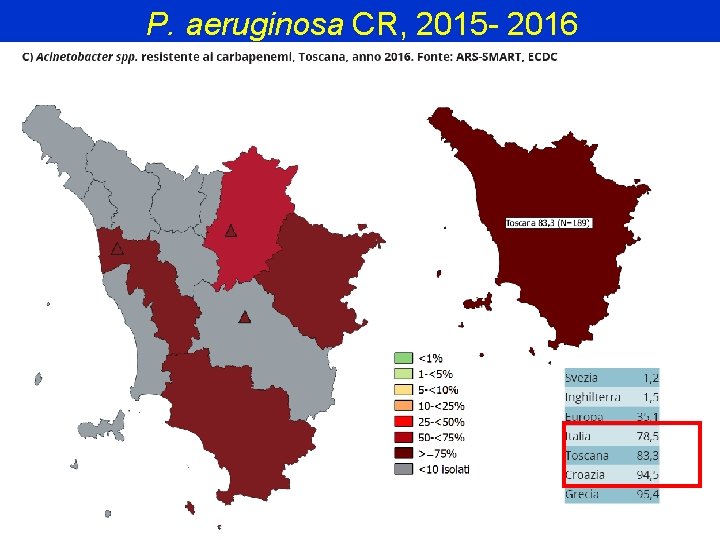
P. aeruginosa CR, 2015 - 2016

Acinetobacter spp. CR, Toscana 2015 -2016

Comparing the Outcomes of Patients with Carbapenemase-Producing and Non-Carbapenemase. Producing Carbapenem-Resistant Enterobacteriaceae Bacteremia • Carbapenem-resistant Enterobacteriaceae (CRE) bacteremia is associated with mortality up to 60%. • Carbapenem resistance can be mediated via: carbapenemase production (CP-CRE) or production of ESBLs and/or Amp. C cephalosporinases combined with altered membrane permeability (non-CP-CRE). • Need to know the outcomes associated with CRE bacteremia according to underlying resistance mechanisms. Clinical Infectious Diseases Advance Access published November 9, 2016

Comparing the Outcomes of Patients with Carbapenemase-Producing and Non. Carbapenemase-Producing Carbapenem. Resistant Enterobacteriaceae Bacteremia • In a cohort of 83 CRE bacteremic patients, the odds of dying within 14 days were four times greater for carbapenemase-producing (CP) CRE compared with non-CP-CRE patients, adjusting for severity of illness, underlying medical conditions, and differences in antibiotic regimens. Clinical Infectious Diseases Advance Access published November 9, 2016

Differential features of acquired carbapenemases KPC-type OXA-48 -like MBLs Amber class A D B CARBAPENEMASE ACTIVITY Strong Weak Strong SPECTRUM Extended (most beta-lactams) Broad (penicillins, NS cephems) Extended, most beta-lactams exc. monobactams PBA Avibactam Relebactam Vaborbactam Avibactam EDTA INHIBITION • •
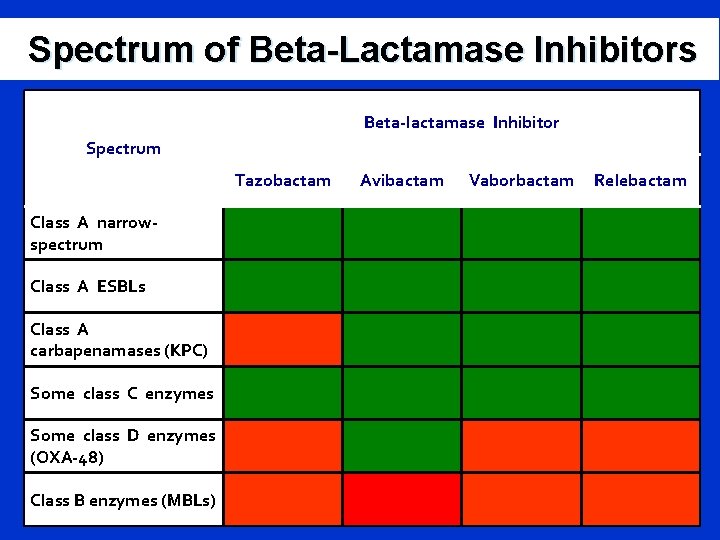
Spectrum of Beta-Lactamase Inhibitors Beta-lactamase Inhibitor Spectrum Tazobactam Class A narrowspectrum Class A ESBLs Class A carbapenamases (KPC) Some class C enzymes Some class D enzymes (OXA-48) Class B enzymes (MBLs) Avibactam Vaborbactam Relebactam
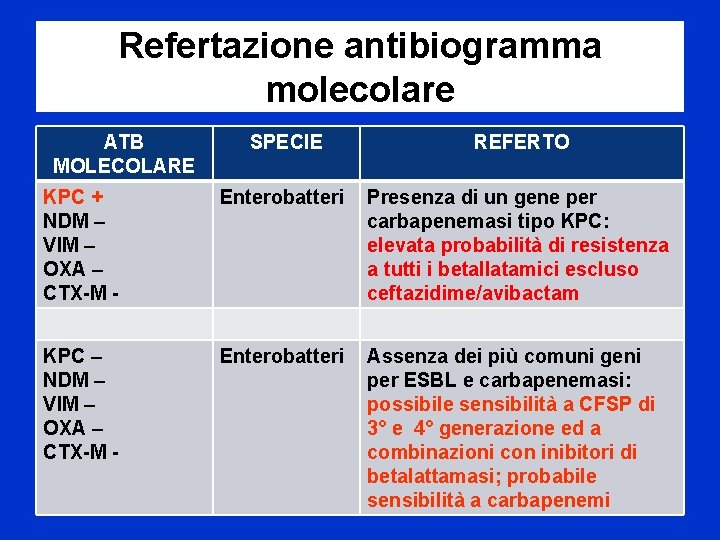
Refertazione antibiogramma molecolare ATB MOLECOLARE SPECIE REFERTO KPC + NDM – VIM – OXA – CTX-M - Enterobatteri Presenza di un gene per carbapenemasi tipo KPC: elevata probabilità di resistenza a tutti i betallatamici escluso ceftazidime/avibactam KPC – NDM – VIM – OXA – CTX-M - Enterobatteri Assenza dei più comuni geni per ESBL e carbapenemasi: possibile sensibilità a CFSP di 3° e 4° generazione ed a combinazioni con inibitori di betalattamasi; probabile sensibilità a carbapenemi
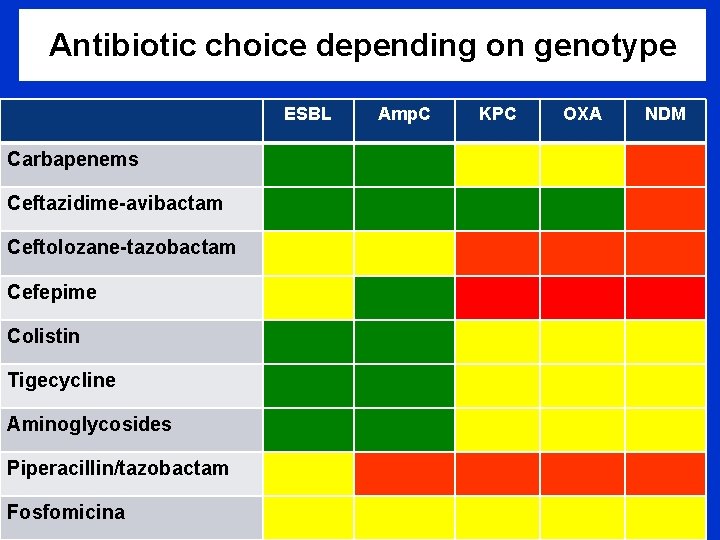
Antibiotic choice depending on genotype ESBL Carbapenems Ceftazidime-avibactam Ceftolozane-tazobactam Cefepime Colistin Tigecycline Aminoglycosides Piperacillin/tazobactam Fosfomicina Amp. C KPC OXA NDM

Expanded spectrum of β-lactamase inhibition J Antimicrob Chemother 2016; 71: 2713 -22

Ceftazidime – Avibactam • Spectrum of activity: Gram-negatives, including MDR P. aeruginosa, ESBL-producing strains, KPCKp • EMA approval in June 2016 Ø c-UTI, including Pyelonephritis Ø C-IAI (plus metronidazole) Ø Nosocomial pneumonia Ø Gram-neg. infection with limited treatment option Ø IV dose: 2. 5 g (2 g ceftazidime; 0. 5 g avibactam) q 8 h (2 -h infusion)

Ceftazidime/avibactam potential drawbacks • The development of resistance to CAZ/AVI (MIC ≥ 16 mg/L) was observed following relatively short courses of therapy. • Reducing the development of resistance by using a combination therapy approach ? • Early development of resistance a unique flaw of CAZ/AVI or might it be seen with other new βL-βLIs combinations ? • Much of the resistance reported seen in K. pneumoniae isolates, how common will this phenotype in non- Klebsiellae spp. harbouring KPCs ? • In geographical areas where MBLs are observed at higher rates, what is the overall utility of ceftazidime/avibactam in clinical practice ?
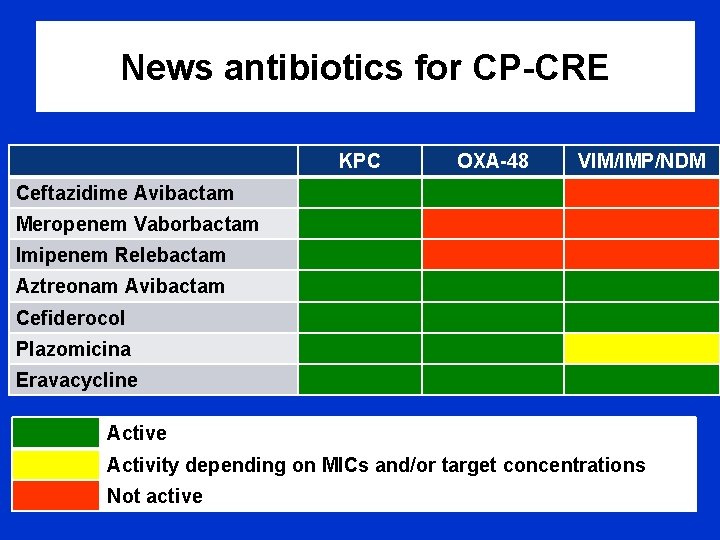
News antibiotics for CP-CRE KPC OXA-48 VIM/IMP/NDM Ceftazidime Avibactam Meropenem Vaborbactam Imipenem Relebactam Aztreonam Avibactam Cefiderocol Plazomicina Eravacycline Activity depending on MICs and/or target concentrations Not active
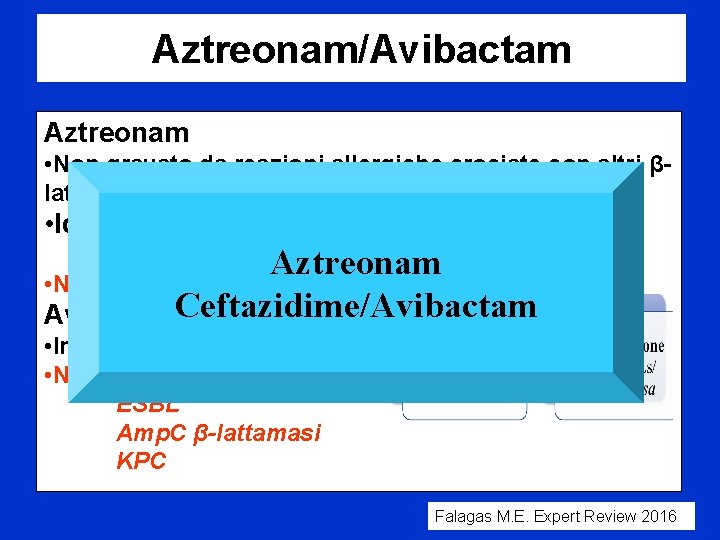
Aztreonam/Avibactam Aztreonam • Non gravato da reazioni allergiche crociate con altri βlattamici (eccetto ceftazidime) • Idrolizzato da ESBL, Amp. C e KPC Aztreonam • NON idrolizzato da MBLs Avibactam. Ceftazidime/Avibactam • Inibitore non β-lattamico • NON Idrolizzato da: ESBL Amp. C β-lattamasi KPC Falagas M. E. Expert Review 2016

Imipenem/Relebactam • Relebactam (MK 7655) is a piperidine analogue diazabicyclooctane beta-lactamase inhibitor designed to have inhibitory activity against class A and class C beta-lactamases. • In KPC-producing K. pneumoniae the addition of relebactam resulted in lowering imipenem MICs by up to 64 -fold. • More modest potentiation was seen with other ESBL- and Amp. C producing strains. • Little or no reduction was seen in OXA-48 -producing K. pneumoniae or OXA-23 -producing A. baumannii, suggesting relebactam, unlike avibactam, does not have significant activity against class D enzymes • Imipenem MICs were reduced from ≥ 16 mg/L to 2 mg/L in the presence of relebactam when imipenem-resistant P. aeruginosa isolates were tested.

Meropenem/Vaborbactam • Vaborbactam is a novel cyclic boronic acid inhibitor of many class A, class C and some class D beta-lactamases. • Vaborbactam acts via the creation of a covalent (dative) bond between its boron moiety and the serine hydroxyl of beta lactamase. • The addition of vaborbactam at 8 μg/ml to meropenem resulted in ≥ 16 -fold reduction in MICs for KPC producing E. coli, K. pneumoniae, and Enterobacter spp. isolates. • In a limited number of strains, the combination appeared to have reduced activity against KPC-producing K. pneumoniae isolates with diminished expression of porin genes omp. K 35 and omp. K 36. • Little effect on A. baumannii containing OXA-type carbapenemases or P. aeruginosa was observed

Cefiderocol • Cefiderocol is a siderophore cephalosporin antibiotic with a catechol moiety on the 3 -position side chain. • The catechol side chain enables ferric iron ion binding, and the resulting complex is actively transported into bacteria via ferric iron transporter systems with subsequent destruction of cell wall. • Cefiderocol has been shown to be potent in vitro against gram-negative organisms, including CRE and MDR P. aeruginosa and A. baumannii. • This activity is considered to be due to the efficient uptake via the active siderophore systems and also to the high stability of cefiderocol against CPS hydrolysis.

Terapia empirica carbapenem-sparing per MDR gram-neg
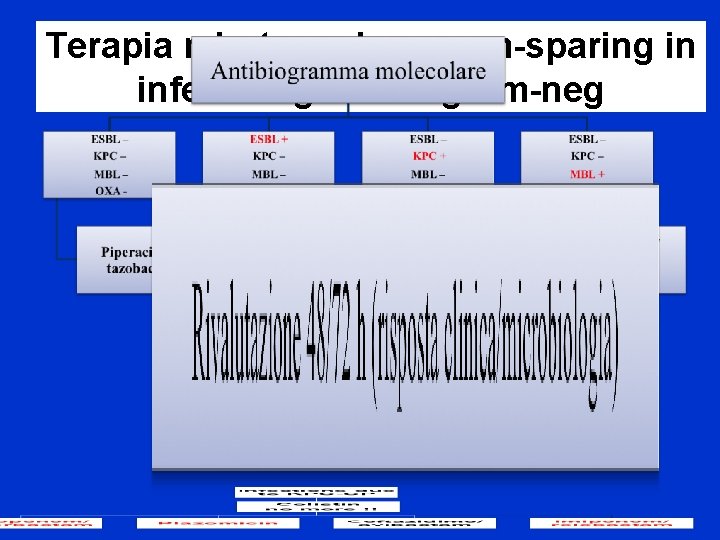
Terapia mirata carbapenem-sparing in infezioni gravi da gram-neg
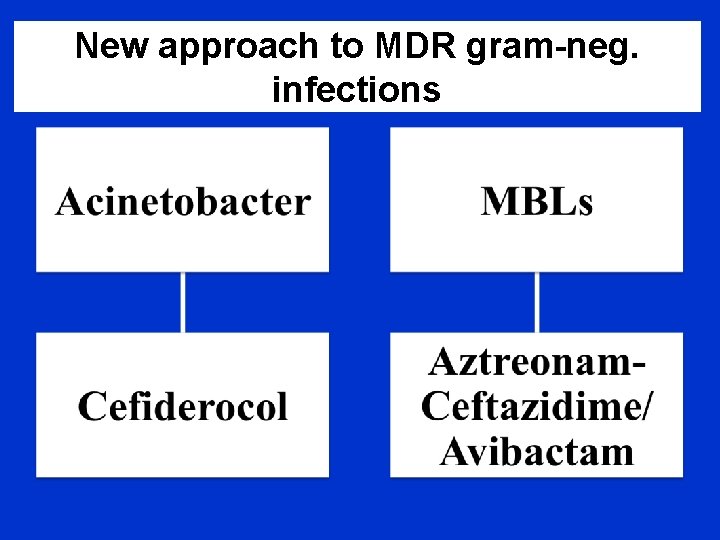
New approach to MDR gram-neg. infections

New approach to MDR gram-neg. infections

Conoscere i nuovi antibiotici: un dovere per il clinico • • …. . ed un’opportunità per i pazienti……. Curiosità, non pigrizia prescrittiva Modello di stewardship condivisa Raccomandazioni terapeutiche condivise Gestione della scheda AIFA Ruolo armonico delle società scientifiche Sistema nazionale delle LG (ISS) www. antimicrobialstewardship. net

www. antimicrobialstewardship. net Presidente Onorario: Prof. Johnatan Cohen Società Scientifica multidisciplinare, promuove la cultura dell’ASP intesa come GOVERNO CLINICO DELLA TERAPIA ANTIMICROBICA attraverso il confronto equo tra esperti e prescrittori.
- Slides: 45