I MEMBRANE BIOCHEMISTRY 1 6 Transmembrane Signaling 1

I. MEMBRANE BIOCHEMISTRY § 1. 6 Transmembrane Signaling § 1. 6 a Endocrine Hormones § 1. 6 b Hormone Receptors
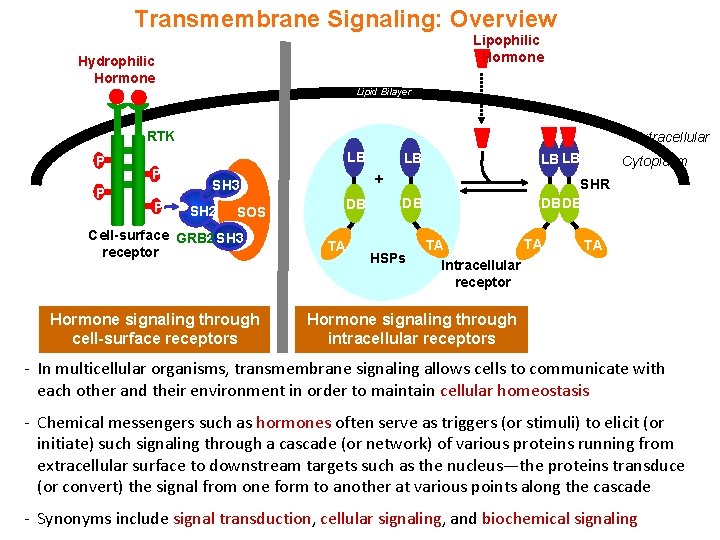
Transmembrane Signaling: Overview Lipophilic Hormone Hydrophilic Hormone Lipid Bilayer RTK P P Extracellular LB P P SOS Cell-surface GRB 2 SH 3 receptor Hormone signaling through cell-surface receptors LB LB DB SHR DB DB + SH 3 SH 2 LB DB TA HSPs TA TA Intracellular receptor Cytoplasm TA Hormone signaling through intracellular receptors - In multicellular organisms, transmembrane signaling allows cells to communicate with each other and their environment in order to maintain cellular homeostasis - Chemical messengers such as hormones often serve as triggers (or stimuli) to elicit (or initiate) such signaling through a cascade (or network) of various proteins running from extracellular surface to downstream targets such as the nucleus—the proteins transduce (or convert) the signal from one form to another at various points along the cascade - Synonyms include signal transduction, cellular signaling, and biochemical signaling

§ 1. 6 a Endocrine Hormones

Synopsis 1. 6 a - Hormones are chemical messengers that play a key role in cellular signaling—they differ from growth factors and cytokines in that they are exclusively produced by endocrine glands and travel in the blood to act at distant sites (systemic action) - On the other hand, growth factors and cytokines are produced throughout the body by numerous cell types and they can act both locally and systemically - Be aware that the terms hormones, growth factors and cytokines are often used interchangeably as there is no rigid boundary separating their classification and their overlapping functions only add to the confusion - The effect of endocrine hormones (as well as growth factors and cytokines) is almost always mediated via a cascade of signaling proteins and receptors, one or more of which are usually enzymes - Such endocrine signaling coupled with endocrine glands and their products (endocrine hormones) are collectively referred to as the “endocrine system”
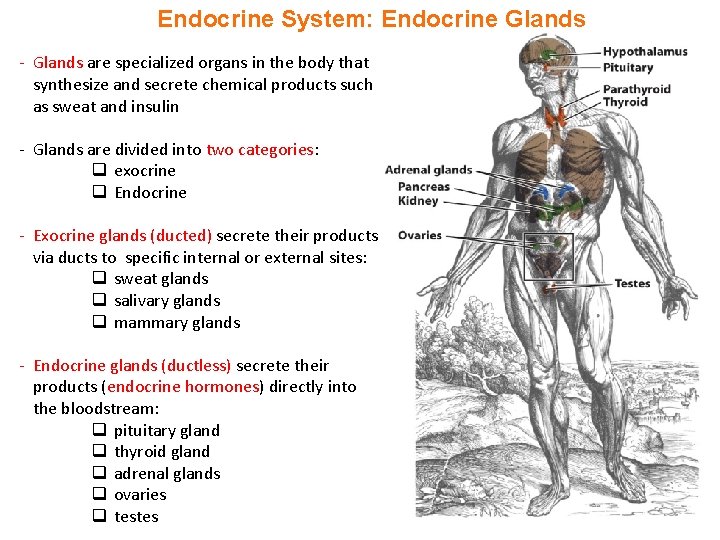
Endocrine System: Endocrine Glands - Glands are specialized organs in the body that synthesize and secrete chemical products such as sweat and insulin - Glands are divided into two categories: q exocrine q Endocrine - Exocrine glands (ducted) secrete their products via ducts to specific internal or external sites: q sweat glands q salivary glands q mammary glands - Endocrine glands (ductless) secrete their products (endocrine hormones) directly into the bloodstream: q pituitary gland q thyroid gland q adrenal glands q ovaries q testes
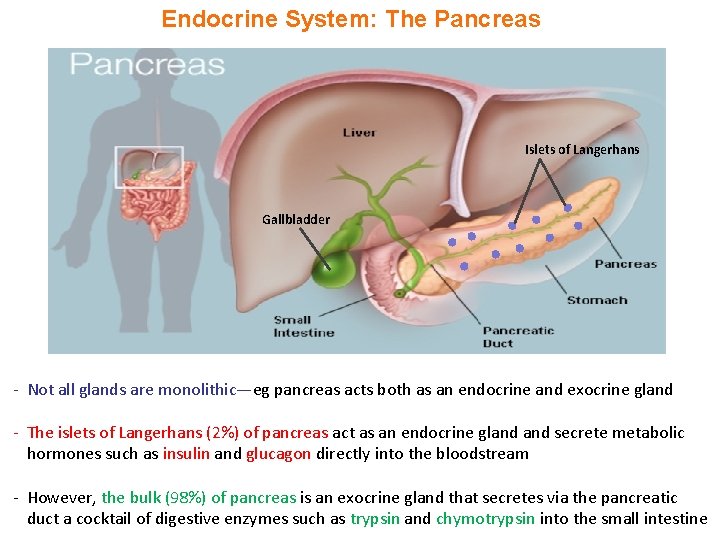
Endocrine System: The Pancreas Islets of Langerhans Gallbladder - Not all glands are monolithic—eg pancreas acts both as an endocrine and exocrine gland - The islets of Langerhans (2%) of pancreas act as an endocrine gland secrete metabolic hormones such as insulin and glucagon directly into the bloodstream - However, the bulk (98%) of pancreas is an exocrine gland that secretes via the pancreatic duct a cocktail of digestive enzymes such as trypsin and chymotrypsin into the small intestine
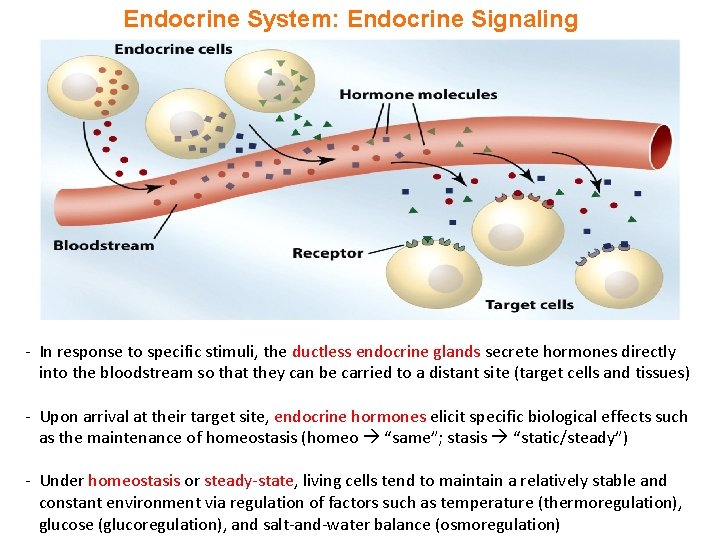
Endocrine System: Endocrine Signaling - In response to specific stimuli, the ductless endocrine glands secrete hormones directly into the bloodstream so that they can be carried to a distant site (target cells and tissues) - Upon arrival at their target site, endocrine hormones elicit specific biological effects such as the maintenance of homeostasis (homeo “same”; stasis “static/steady”) - Under homeostasis or steady-state, living cells tend to maintain a relatively stable and constant environment via regulation of factors such as temperature (thermoregulation), glucose (glucoregulation), and salt-and-water balance (osmoregulation)
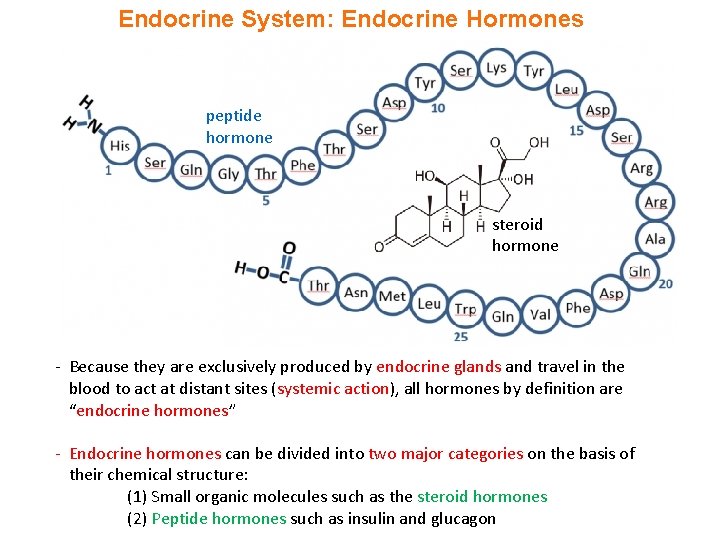
Endocrine System: Endocrine Hormones peptide hormone steroid hormone - Because they are exclusively produced by endocrine glands and travel in the blood to act at distant sites (systemic action), all hormones by definition are “endocrine hormones” - Endocrine hormones can be divided into two major categories on the basis of their chemical structure: (1) Small organic molecules such as the steroid hormones (2) Peptide hormones such as insulin and glucagon
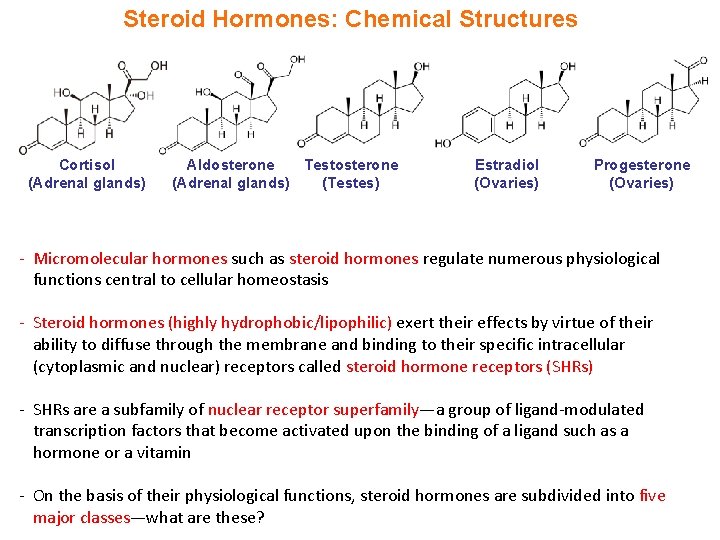
Steroid Hormones: Chemical Structures Cortisol (Adrenal glands) Aldosterone Testosterone (Adrenal glands) (Testes) Estradiol (Ovaries) Progesterone (Ovaries) - Micromolecular hormones such as steroid hormones regulate numerous physiological functions central to cellular homeostasis - Steroid hormones (highly hydrophobic/lipophilic) exert their effects by virtue of their ability to diffuse through the membrane and binding to their specific intracellular (cytoplasmic and nuclear) receptors called steroid hormone receptors (SHRs) - SHRs are a subfamily of nuclear receptor superfamily—a group of ligand-modulated transcription factors that become activated upon the binding of a ligand such as a hormone or a vitamin - On the basis of their physiological functions, steroid hormones are subdivided into five major classes—what are these?
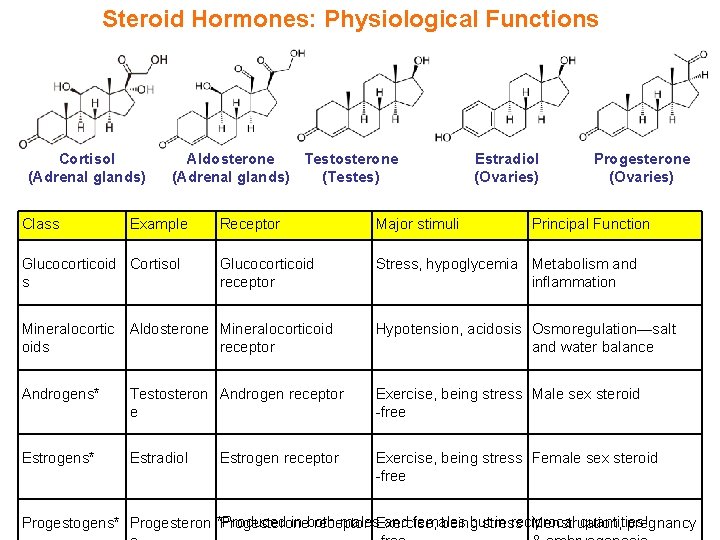
Steroid Hormones: Physiological Functions Cortisol (Adrenal glands) Class Aldosterone Testosterone (Adrenal glands) (Testes) Example Glucocorticoid Cortisol s Estradiol (Ovaries) Progesterone (Ovaries) Receptor Major stimuli Principal Function Glucocorticoid receptor Stress, hypoglycemia Metabolism and inflammation Mineralocortic Aldosterone Mineralocorticoid oids receptor Hypotension, acidosis Osmoregulation—salt and water balance Androgens* Testosteron Androgen receptor e Exercise, being stress Male sex steroid -free Estrogens* Estradiol Exercise, being stress Female sex steroid -free Estrogen receptor in both males. Exercise, and females but in reciprocal quantities! Progestogens* Progesteron *Produced Progesterone receptor being stress Menstruation, pregnancy
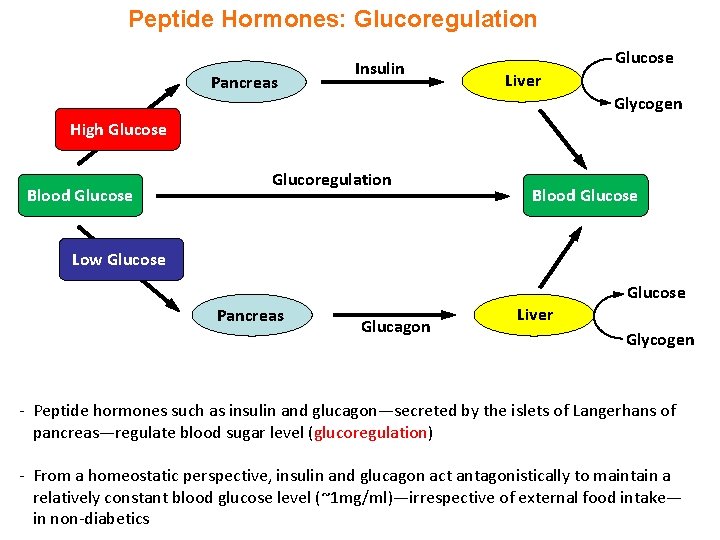
Peptide Hormones: Glucoregulation Pancreas Insulin Glucose Liver Glycogen High Glucose Blood Glucose Glucoregulation Blood Glucose Low Glucose Pancreas Glucagon Liver Glycogen - Peptide hormones such as insulin and glucagon—secreted by the islets of Langerhans of pancreas—regulate blood sugar level (glucoregulation) - From a homeostatic perspective, insulin and glucagon act antagonistically to maintain a relatively constant blood glucose level (~1 mg/ml)—irrespective of external food intake— in non-diabetics
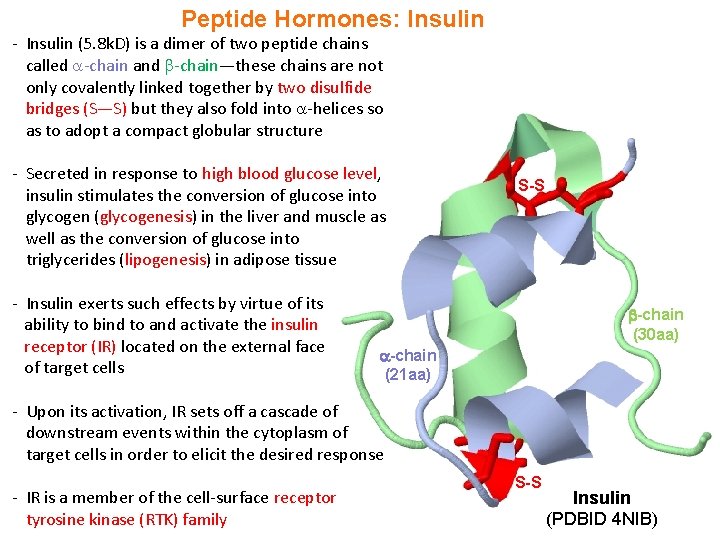
Peptide Hormones: Insulin - Insulin (5. 8 k. D) is a dimer of two peptide chains called -chain and -chain—these chains are not only covalently linked together by two disulfide bridges (S—S) but they also fold into -helices so as to adopt a compact globular structure - Secreted in response to high blood glucose level, insulin stimulates the conversion of glucose into glycogen (glycogenesis) in the liver and muscle as well as the conversion of glucose into triglycerides (lipogenesis) in adipose tissue - Insulin exerts such effects by virtue of its ability to bind to and activate the insulin receptor (IR) located on the external face of target cells S-S -chain (30 aa) -chain (21 aa) - Upon its activation, IR sets off a cascade of downstream events within the cytoplasm of target cells in order to elicit the desired response - IR is a member of the cell-surface receptor tyrosine kinase (RTK) family S-S Insulin (PDBID 4 NIB)

Peptide Hormones: Glucagon (PDBID 1 GCN) - Glucagon (3. 5 k. D) is an -helical peptide comprised of 29 amino acids (29 aa) - Secreted in response to low blood glucose level, glucagon does the opposite of insulin—it stimulates the liver to release glucose through the breakdown of glycogen (glycogenolysis) and the synthesis of glucose from non-carbohydrate precursors such as pyruvate and lactate (gluconeogenesis) - Glucagon exerts such effects by virtue of its ability to bind to and activate the glucagon receptor (GR) located on the external face of target cell - Upon its activation, IR sets off a cascade of downstream events within the cytoplasm of target cells in order to elicit the desired response - GR is a member of the cell-surface G-protein-coupled receptor (GPCR) family

Exercise 1. 6 a - Explain why only certain cells respond to hormones even though all cells in the body are exposed to the hormone - List hormones produced by the pancreas and adrenal glands. What types of molecules are these hormones? - Summarize the biological effects of insulin and glucagon - Summarize the biological effects of steroid hormones
§ 1. 6 b Hormone Receptors

Synopsis 1. 6 b - How do endocrine hormones drive cellular processes as diverse as cell growth and cell proliferation through metabolism to the development and progression of many cancers? Enter hormone receptors! - Hormone receptors mediate extracellular signals in the form of hormones at the cell surface (or after their diffusion into the cytosol) to downstream targets such as transcription factors in the nucleus via what are termed “signaling cascades” - Hormone receptors and many proteins involved in coupling them to downstream cellular targets are “modular”—ie they are functionally subdivided into semi-autonomous regions called “modules” or “domains” - Among a wide plethora of protein modules, Src homology domains such as SH 2 and SH 3 play a central role in directly coupling activated hormone receptors to downstream targets - Hormone receptors can be broadly divided into three major categories: (1) Steroid hormone receptors (SHRs) (2) Receptor tyrosine kinases (RTKs) (3) G-Protein-coupled receptors (GPCRs)

1. Steroid Hormone Receptors (SHRs) Located within the cytoplasm and nucleus, SHRs are ligand-modulated transcription factors that become activated upon binding their cognate ligands (steroid hormones)
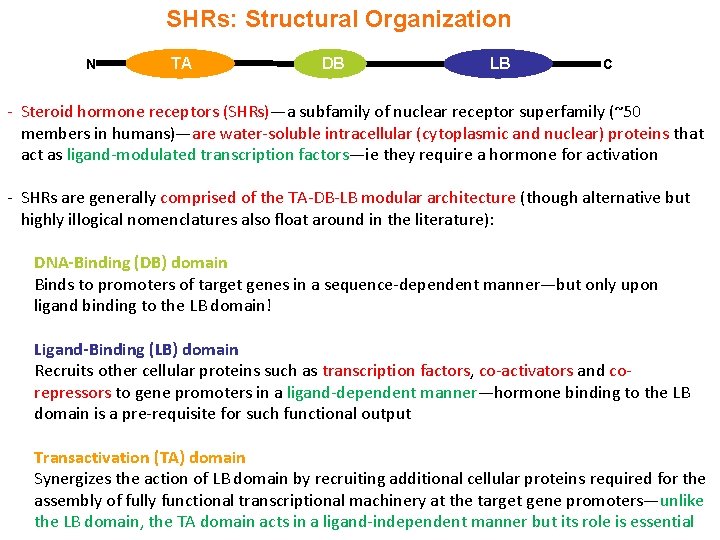
SHRs: Structural Organization N TA DB LB C - Steroid hormone receptors (SHRs)—a subfamily of nuclear receptor superfamily (~50 members in humans)—are water-soluble intracellular (cytoplasmic and nuclear) proteins that act as ligand-modulated transcription factors—ie they require a hormone for activation - SHRs are generally comprised of the TA-DB-LB modular architecture (though alternative but highly illogical nomenclatures also float around in the literature): DNA-Binding (DB) domain Binds to promoters of target genes in a sequence-dependent manner—but only upon ligand binding to the LB domain! Ligand-Binding (LB) domain Recruits other cellular proteins such as transcription factors, co-activators and corepressors to gene promoters in a ligand-dependent manner—hormone binding to the LB domain is a pre-requisite for such functional output Transactivation (TA) domain Synergizes the action of LB domain by recruiting additional cellular proteins required for the assembly of fully functional transcriptional machinery at the target gene promoters—unlike the LB domain, the TA domain acts in a ligand-independent manner but its role is essential
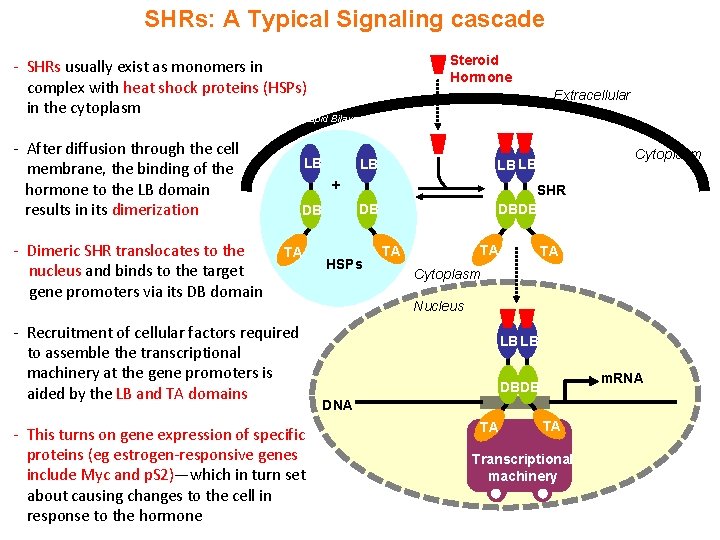
SHRs: A Typical Signaling cascade - SHRs usually exist as monomers in complex with heat shock proteins (HSPs) in the cytoplasm - After diffusion through the cell membrane, the binding of the hormone to the LB domain results in its dimerization - Dimeric SHR translocates to the nucleus and binds to the target gene promoters via its DB domain Steroid Hormone Extracellular Lipid Bilayer LB LB LB DB SHR DB DB + DB TA - Recruitment of cellular factors required to assemble the transcriptional machinery at the gene promoters is aided by the LB and TA domains - This turns on gene expression of specific proteins (eg estrogen-responsive genes include Myc and p. S 2)—which in turn set about causing changes to the cell in response to the hormone Cytoplasm LB HSPs TA TA TA Cytoplasm Nucleus LB LB m. RNA DBDB DNA TA TA Transcriptional machinery

2. Receptor Tyrosine Kinases (RTKs) With a single-transmembrane topology, RTKs become activated upon binding to their cognate ligands (such as hormones) at the cell surface
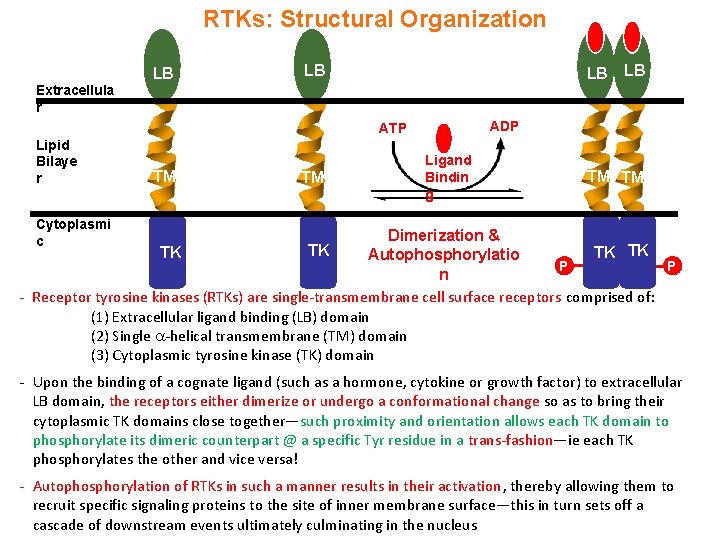
RTKs: Structural Organization Extracellula r LB LB LB ADP ATP Lipid Bilaye r TM TM LB Ligand Bindin g TM TM Cytoplasmi c Dimerization & TK TK Autophosphorylatio P P n - Receptor tyrosine kinases (RTKs) are single-transmembrane cell surface receptors comprised of: (1) Extracellular ligand binding (LB) domain (2) Single -helical transmembrane (TM) domain (3) Cytoplasmic tyrosine kinase (TK) domain - Upon the binding of a cognate ligand (such as a hormone, cytokine or growth factor) to extracellular LB domain, the receptors either dimerize or undergo a conformational change so as to bring their cytoplasmic TK domains close together—such proximity and orientation allows each TK domain to phosphorylate its dimeric counterpart @ a specific Tyr residue in a trans-fashion—ie each TK phosphorylates the other and vice versa! - Autophosphorylation of RTKs in such a manner results in their activation, thereby allowing them to recruit specific signaling proteins to the site of inner membrane surface—this in turn sets off a cascade of downstream events ultimately culminating in the nucleus
RTKs: Autophosphorylation ATP ADP Kinas e Phosphata se - Protein phosphorylation @ one of the hydroxy amino acids (eg Tyr, Thr or Ser) within protein chains is by far the most ubiquitous form of post-translational modification (PTM) observed in proteins—why so? - Protein phosphorylation essentially serves as a “molecular switch” in that it can turn protein function “ON” or “OFF”—courtesy of protein kinases and phosphatases working in tandem to maintain cellular homeostasis - Such molecular switches are a hallmark of the living machinery, and particularly, the cellular signaling pathways that need to be tightly regulated in a highly spatial and temporal manner—ie with respect to both the location of the signaling event (spatial) and the short time that it should be turned on or off (temporal/transient)—for the failure to do so often forms the basis of the development and progression of disease - The fact that the regulation of RTKs is also under the control of phosphorylation offers a fitting tribute to the importance of kinases and phosphatases to the vitality of life at molecular level—the human genome encodes 500 protein kinases and 200 protein phosphatases!
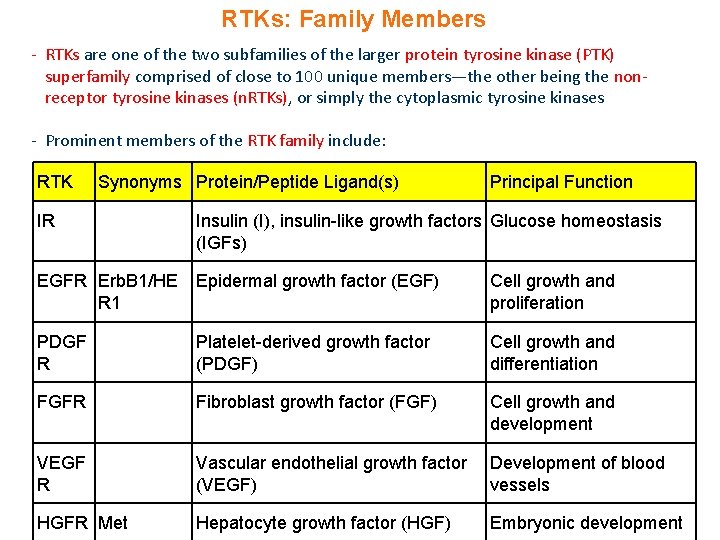
RTKs: Family Members - RTKs are one of the two subfamilies of the larger protein tyrosine kinase (PTK) superfamily comprised of close to 100 unique members—the other being the nonreceptor tyrosine kinases (n. RTKs), or simply the cytoplasmic tyrosine kinases - Prominent members of the RTK family include: RTK Synonyms Protein/Peptide Ligand(s) IR Principal Function Insulin (I), insulin-like growth factors Glucose homeostasis (IGFs) EGFR Erb. B 1/HE Epidermal growth factor (EGF) R 1 Cell growth and proliferation PDGF R Platelet-derived growth factor (PDGF) Cell growth and differentiation FGFR Fibroblast growth factor (FGF) Cell growth and development VEGF R Vascular endothelial growth factor (VEGF) Development of blood vessels HGFR Met Hepatocyte growth factor (HGF) Embryonic development
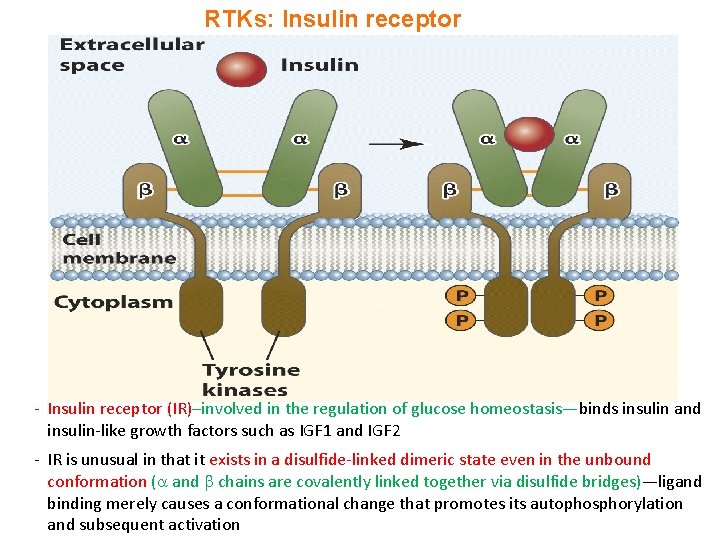
RTKs: Insulin receptor - Insulin receptor (IR)–involved in the regulation of glucose homeostasis—binds insulin and insulin-like growth factors such as IGF 1 and IGF 2 - IR is unusual in that it exists in a disulfide-linked dimeric state even in the unbound conformation ( and chains are covalently linked together via disulfide bridges)—ligand binding merely causes a conformational change that promotes its autophosphorylation and subsequent activation
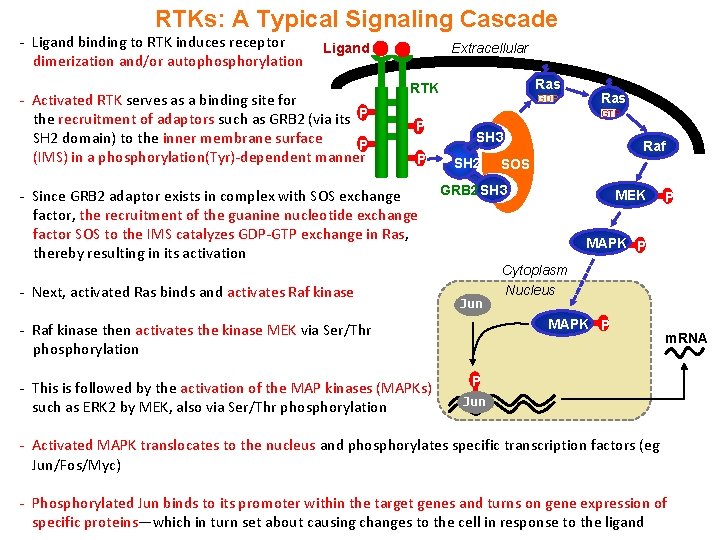
RTKs: A Typical Signaling Cascade - Ligand binding to RTK induces receptor dimerization and/or autophosphorylation Ligand - Activated RTK serves as a binding site for the recruitment of adaptors such as GRB 2 (via its P SH 2 domain) to the inner membrane surface P (IMS) in a phosphorylation(Tyr)-dependent manner Extracellular Ras RTK P P - Since GRB 2 adaptor exists in complex with SOS exchange factor, the recruitment of the guanine nucleotide exchange factor SOS to the IMS catalyzes GDP-GTP exchange in Ras, thereby resulting in its activation - Next, activated Ras binds and activates Raf kinase GDP GTP SH 3 SH 2 Raf SOS GRB 2 SH 3 MEK P MAPK P Jun Cytoplasm Nucleus MAPK P - Raf kinase then activates the kinase MEK via Ser/Thr phosphorylation - This is followed by the activation of the MAP kinases (MAPKs) such as ERK 2 by MEK, also via Ser/Thr phosphorylation Ras m. RNA P Jun - Activated MAPK translocates to the nucleus and phosphorylates specific transcription factors (eg Jun/Fos/Myc) - Phosphorylated Jun binds to its promoter within the target genes and turns on gene expression of specific proteins—which in turn set about causing changes to the cell in response to the ligand

3. G-Protein-Coupled Receptors (GPCRs) With a seven-transmembrane topology, GPCRs become activated upon binding to their cognate ligands (such as hormones) at the cell surface
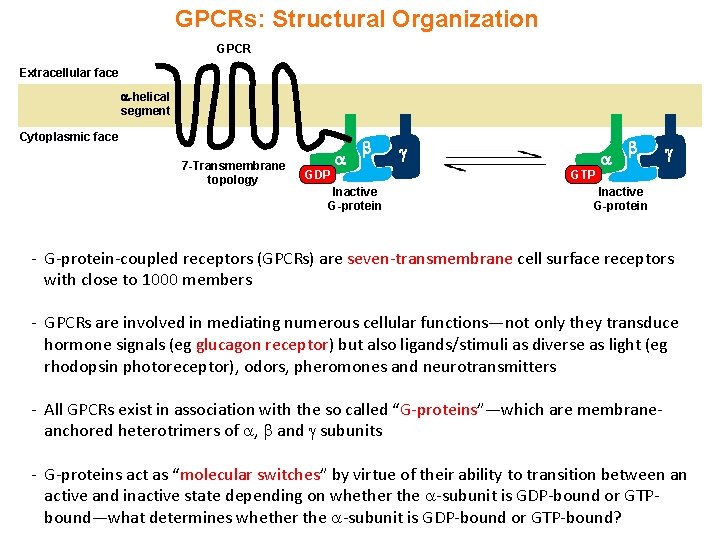
GPCRs: Structural Organization GPCR Extracellular face -helical segment Cytoplasmic face 7 -Transmembrane topology GDP Inactive G-protein GTP Inactive G-protein - G-protein-coupled receptors (GPCRs) are seven-transmembrane cell surface receptors with close to 1000 members - GPCRs are involved in mediating numerous cellular functions—not only they transduce hormone signals (eg glucagon receptor) but also ligands/stimuli as diverse as light (eg rhodopsin photoreceptor), odors, pheromones and neurotransmitters - All GPCRs exist in association with the so called “G-proteins”—which are membraneanchored heterotrimers of , and subunits - G-proteins act as “molecular switches” by virtue of their ability to transition between an active and inactive state depending on whether the -subunit is GDP-bound or GTPbound—what determines whether the -subunit is GDP-bound or GTP-bound?
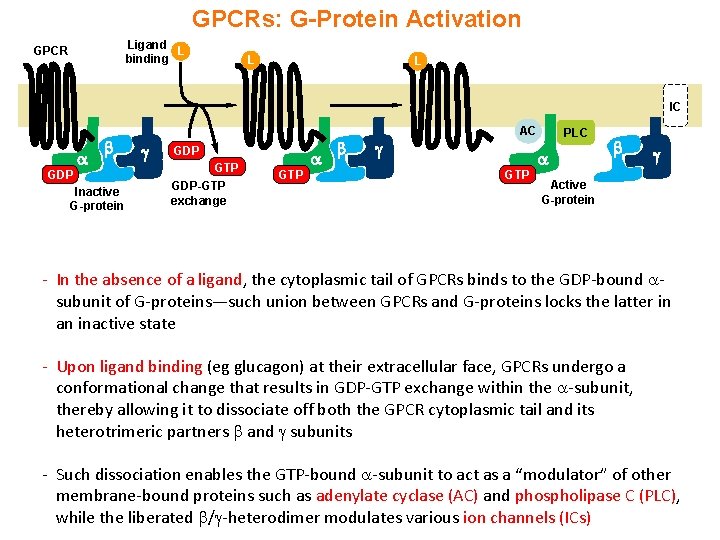
GPCRs: G-Protein Activation Ligand L binding GPCR L L IC GDP Inactive G-protein GDP GTP GDP-GTP exchange GTP AC PLC GTP Active G-protein - In the absence of a ligand, the cytoplasmic tail of GPCRs binds to the GDP-bound subunit of G-proteins—such union between GPCRs and G-proteins locks the latter in an inactive state - Upon ligand binding (eg glucagon) at their extracellular face, GPCRs undergo a conformational change that results in GDP-GTP exchange within the -subunit, thereby allowing it to dissociate off both the GPCR cytoplasmic tail and its heterotrimeric partners and subunits - Such dissociation enables the GTP-bound -subunit to act as a “modulator” of other membrane-bound proteins such as adenylate cyclase (AC) and phospholipase C (PLC), while the liberated / -heterodimer modulates various ion channels (ICs)
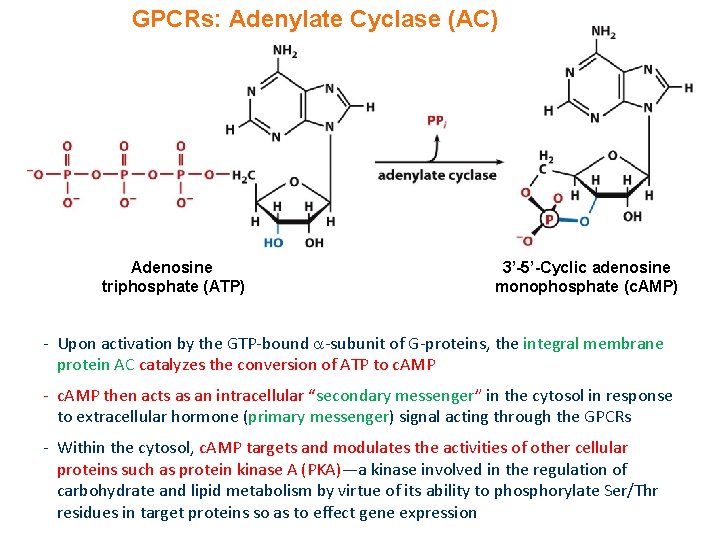
GPCRs: Adenylate Cyclase (AC) Adenosine triphosphate (ATP) 3’-5’-Cyclic adenosine monophosphate (c. AMP) - Upon activation by the GTP-bound -subunit of G-proteins, the integral membrane protein AC catalyzes the conversion of ATP to c. AMP - c. AMP then acts as an intracellular “secondary messenger” in the cytosol in response to extracellular hormone (primary messenger) signal acting through the GPCRs - Within the cytosol, c. AMP targets and modulates the activities of other cellular proteins such as protein kinase A (PKA)—a kinase involved in the regulation of carbohydrate and lipid metabolism by virtue of its ability to phosphorylate Ser/Thr residues in target proteins so as to effect gene expression
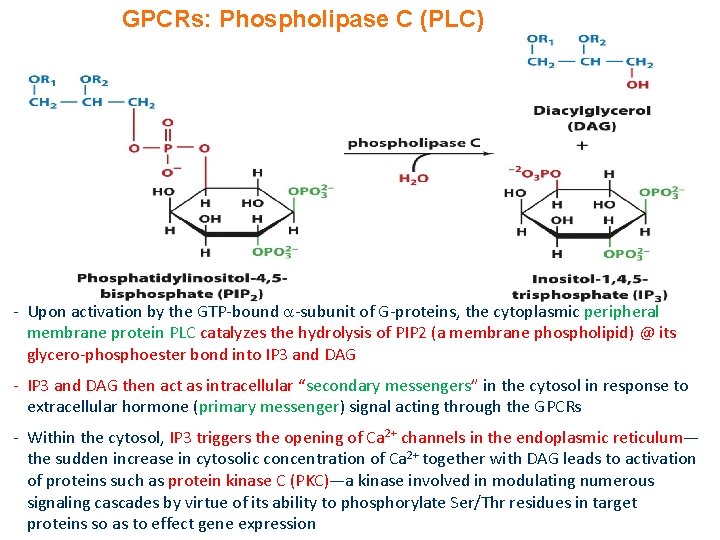
GPCRs: Phospholipase C (PLC) - Upon activation by the GTP-bound -subunit of G-proteins, the cytoplasmic peripheral membrane protein PLC catalyzes the hydrolysis of PIP 2 (a membrane phospholipid) @ its glycero-phosphoester bond into IP 3 and DAG - IP 3 and DAG then act as intracellular “secondary messengers” in the cytosol in response to extracellular hormone (primary messenger) signal acting through the GPCRs - Within the cytosol, IP 3 triggers the opening of Ca 2+ channels in the endoplasmic reticulum— the sudden increase in cytosolic concentration of Ca 2+ together with DAG leads to activation of proteins such as protein kinase C (PKC)—a kinase involved in modulating numerous signaling cascades by virtue of its ability to phosphorylate Ser/Thr residues in target proteins so as to effect gene expression

Exercise 1. 6 b - How does a receptor tyrosine kinase phosphorylate itself? - Summarize various members of RTK family, their ligands and functions - Summarize the roles of proteins such as Grb 2, SOS, Ras, and various protein kinases involved in coupling activated RTKs to downstream cellular targets such as transcription factors in the nucleus - Explain why cells contain an array of protein phosphatases as well as protein kinases - Summarize the steps of signal transduction from a GPCR to phosphorylation of target proteins by PKA - What is the function of a secondary messenger such as c. AMP?
- Slides: 31